Professional Documents
Culture Documents
Co-Infection of Dengue and COVID-19: A Case: Neglected Tropical Diseases
Co-Infection of Dengue and COVID-19: A Case: Neglected Tropical Diseases
Uploaded by
FihzanCopyright:
Available Formats
You might also like
- A2 UNIT 4 Test Answer Key HigherDocument2 pagesA2 UNIT 4 Test Answer Key HigherMaxi Comas60% (5)
- Teste Ingles TecnologiaDocument5 pagesTeste Ingles TecnologiaMeninaDuMarNo ratings yet
- Wiens N Priebe - Occlusal StabilityDocument25 pagesWiens N Priebe - Occlusal Stabilitymoji_puiNo ratings yet
- Bilco WellboreDocument27 pagesBilco WellboreJhonathan Miranda100% (4)
- Faggionvinholo2020 Q1Document5 pagesFaggionvinholo2020 Q1NasriNo ratings yet
- Herpes Zoster in COVID19Document2 pagesHerpes Zoster in COVID19Vienesha Aress ShaNo ratings yet
- Varicella DengueDocument3 pagesVaricella DengueDian Fajar FebrianiNo ratings yet
- Measles During Arbovirus Outbreak: A Diagnostic Challenge: Case ReportDocument4 pagesMeasles During Arbovirus Outbreak: A Diagnostic Challenge: Case ReportnisceNo ratings yet
- DengueDocument2 pagesDengueElsa YuliusNo ratings yet
- Dengue Shock Syndrome in An Infant: Linda Aurpibul, Punyawee Khumlue, Satja Issaranggoon Na Ayuthaya, Peninnah OberdorferDocument4 pagesDengue Shock Syndrome in An Infant: Linda Aurpibul, Punyawee Khumlue, Satja Issaranggoon Na Ayuthaya, Peninnah OberdorferLIdya Goprani UmarNo ratings yet
- Dengue Haemorrhagic Fever/dengue Shock Syndrome: Lessons From The Cuban Epidemic, 1981Document6 pagesDengue Haemorrhagic Fever/dengue Shock Syndrome: Lessons From The Cuban Epidemic, 1981WahyuNo ratings yet
- Variants of Erythema Multiforme: A Case Report and Literature ReviewDocument8 pagesVariants of Erythema Multiforme: A Case Report and Literature Reviewranz ibonkNo ratings yet
- E042398 FullDocument10 pagesE042398 Fullcomfica.formacion1.2023No ratings yet
- 1 s2.0 S0399077X20300688 MainDocument4 pages1 s2.0 S0399077X20300688 MainAnastasija LevcenkoNo ratings yet
- Tammaro2020 Covid 19Document7 pagesTammaro2020 Covid 19eva yustianaNo ratings yet
- Art:10.1007/s00431-009-0986-0 222Document6 pagesArt:10.1007/s00431-009-0986-0 222Sybelle CostaNo ratings yet
- High Mortality Associated With An Outbreak of Hepatitis E Among Displaced Persons in Darfur, SudanDocument6 pagesHigh Mortality Associated With An Outbreak of Hepatitis E Among Displaced Persons in Darfur, SudanKatrina ManurungNo ratings yet
- Journal Pre-Proof: M Edecine Et Maladies InfectieusesDocument12 pagesJournal Pre-Proof: M Edecine Et Maladies Infectieusessameer sahaanNo ratings yet
- Covid19 PDFDocument6 pagesCovid19 PDFJeremy Kartika SoeryonoNo ratings yet
- Prevention of Nosocomial Covid 19 Another Challenge of The PandemicDocument2 pagesPrevention of Nosocomial Covid 19 Another Challenge of The PandemicZied FehriNo ratings yet
- 51 Uysal EtalDocument4 pages51 Uysal EtaleditorijmrhsNo ratings yet
- Article - N - Clinical Manifestations of Imported Cases of Dengue FeverDocument3 pagesArticle - N - Clinical Manifestations of Imported Cases of Dengue FeverIm jungkook JUSTIN SEAGULL A.K.A jungshookNo ratings yet
- TPMD 200176Document2 pagesTPMD 200176Marcelita DuwiriNo ratings yet
- Tetanus: Clostridium Tetani. The Disease Was First Described in The 14th Century by John of ArderneDocument5 pagesTetanus: Clostridium Tetani. The Disease Was First Described in The 14th Century by John of Ardernedefitri sariningtyasNo ratings yet
- Jurnal Inggris 4Document6 pagesJurnal Inggris 4pikNo ratings yet
- 2019 Novel Coronavirus Disease (COVID-19) in Taiwan: Reports of Two Cases From Wuhan, ChinaDocument4 pages2019 Novel Coronavirus Disease (COVID-19) in Taiwan: Reports of Two Cases From Wuhan, ChinaClaav HugNo ratings yet
- HellowDocument6 pagesHellowafina rasaki asriNo ratings yet
- 220 FullDocument4 pages220 FullOttofianus Alvedo Hewick KalangiNo ratings yet
- Dengue in Renal Transplant Patients A ReDocument3 pagesDengue in Renal Transplant Patients A ReMaira EscobarNo ratings yet
- Primary Varicella Zooster and SARS-CoV-2 Coinfection: Case ReportDocument7 pagesPrimary Varicella Zooster and SARS-CoV-2 Coinfection: Case ReportIrfan CorovicNo ratings yet
- Dengue_activity_in_Puerto_Rico_during_an_interepidDocument10 pagesDengue_activity_in_Puerto_Rico_during_an_interepidCrónicos Jurisdicción J5No ratings yet
- Case Report Severe Covid-19Document3 pagesCase Report Severe Covid-1911TH-01 Lâm Nguyễn Ngọc AnhNo ratings yet
- The First Case of 2019 Novel Coronavirus Pneumonia Imported Into Korea From Wuhan, China: Implication For Infection Prevention and Control MeasuresDocument4 pagesThe First Case of 2019 Novel Coronavirus Pneumonia Imported Into Korea From Wuhan, China: Implication For Infection Prevention and Control MeasuresTian KurniawanNo ratings yet
- 1 s2.0 S0399077X20301104 MainDocument4 pages1 s2.0 S0399077X20301104 MainNaira ValentinaNo ratings yet
- (14761645 - The American Journal of Tropical Medicine and Hygiene) Case Report - Severe COVID-19 and Dengue in An Indonesian InfantDocument5 pages(14761645 - The American Journal of Tropical Medicine and Hygiene) Case Report - Severe COVID-19 and Dengue in An Indonesian InfantAnggrainiAlamNo ratings yet
- Chronic Meningococcemia in A 16-Year-Old Boy: A Case ReportDocument3 pagesChronic Meningococcemia in A 16-Year-Old Boy: A Case ReportUjean Santos SagaralNo ratings yet
- Art 04Document7 pagesArt 04Florentino HerreraNo ratings yet
- New-Onset Myasthenia Gravis After Novel Coronavirus 2019 InfectionDocument3 pagesNew-Onset Myasthenia Gravis After Novel Coronavirus 2019 Infectionarenasgrecia1No ratings yet
- Anthrax of The Gastrointestinal Tract: Thira Sirisanthana and Arthur E. BrownDocument3 pagesAnthrax of The Gastrointestinal Tract: Thira Sirisanthana and Arthur E. BrownJuli SatriaNo ratings yet
- Al Muthanna International Trauma Conference MAY 9 - 11, 2020 Samawa, IraqDocument5 pagesAl Muthanna International Trauma Conference MAY 9 - 11, 2020 Samawa, IraqKhaliel AlsarafNo ratings yet
- 2020 Case 23-2020, A 76-Year-Old Woman Who Died From Covid-19Document8 pages2020 Case 23-2020, A 76-Year-Old Woman Who Died From Covid-19alejandromfunes1749No ratings yet
- Tiapride Associated Toxic Epidermal Necrolysis in An Hivinfected Patient. 2165 7920.1000323Document3 pagesTiapride Associated Toxic Epidermal Necrolysis in An Hivinfected Patient. 2165 7920.1000323Mega RafikaNo ratings yet
- Spectrum - of - Clinicopathologic - Findings - In.1 (1Document11 pagesSpectrum - of - Clinicopathologic - Findings - In.1 (1tzuskyNo ratings yet
- LepsDocument4 pagesLepslynharee100% (1)
- Difficulties in Diagnosis of Psittacosis or Ornithosis: A Case ReportDocument4 pagesDifficulties in Diagnosis of Psittacosis or Ornithosis: A Case ReportYusrinabillaNo ratings yet
- Corynebacterium Ulcerans Cutaneous Diphtheria: Grand RoundDocument8 pagesCorynebacterium Ulcerans Cutaneous Diphtheria: Grand RoundRafa NaufalinNo ratings yet
- 1287-Article Text-3033-1-10-20170221Document15 pages1287-Article Text-3033-1-10-20170221andi ramdhanNo ratings yet
- Dr. DEEPAK.N.S (PG), Dr. PAGADPALLY SRINIVAS (Professor & Head) Dept. of Paediatrics, VMMC, KKLDocument2 pagesDr. DEEPAK.N.S (PG), Dr. PAGADPALLY SRINIVAS (Professor & Head) Dept. of Paediatrics, VMMC, KKLSumanth KumarNo ratings yet
- Follow Up of Skin Lesions During The Evolution of COVID 19 A CaseDocument4 pagesFollow Up of Skin Lesions During The Evolution of COVID 19 A CaseZihan ZetiraNo ratings yet
- Clinical Characteristics of Covid-19 Cases in Guayaquil, EcuadorDocument5 pagesClinical Characteristics of Covid-19 Cases in Guayaquil, EcuadorFarida MurtianiNo ratings yet
- 493 FullDocument4 pages493 FullDevy KurniawatiNo ratings yet
- Chikungunya in Children 2015Document3 pagesChikungunya in Children 2015Douglas UmbriaNo ratings yet
- JMV 25815Document2 pagesJMV 25815fadil ahmadiNo ratings yet
- Wabah Hepatititis EDocument7 pagesWabah Hepatititis EKatrina ManurungNo ratings yet
- Epidemiological Description of Chikungunya Virus Outbreak in Dire Dawa Administrative City, Eastern Ethiopia, 2019Document5 pagesEpidemiological Description of Chikungunya Virus Outbreak in Dire Dawa Administrative City, Eastern Ethiopia, 2019Nigatu AdmasuNo ratings yet
- PIIS0163445320302176Document2 pagesPIIS0163445320302176Hanan ArielNo ratings yet
- Nosocomial Infections in A Brazilian Burn Unit: Jefferson Lessa Soares de Macedo, Joa o Barberino SantosDocument5 pagesNosocomial Infections in A Brazilian Burn Unit: Jefferson Lessa Soares de Macedo, Joa o Barberino SantossriyuswantiNo ratings yet
- Clinical Findings h7n9Document5 pagesClinical Findings h7n9Martin HaynesNo ratings yet
- Three Cases of COVID-19 Patients Presenting With Erythema: Concise CommunicationDocument4 pagesThree Cases of COVID-19 Patients Presenting With Erythema: Concise CommunicationNur Alfia KusumaningsihNo ratings yet
- Spectrum of Clinicopathologic Findings in COVID-19-induced Skin LesionsDocument9 pagesSpectrum of Clinicopathologic Findings in COVID-19-induced Skin LesionsggenesisNo ratings yet
- Covid 19: Dennis N. Muñoz, RN, RM, LPTDocument103 pagesCovid 19: Dennis N. Muñoz, RN, RM, LPTDennis Nabor Muñoz, RN,RMNo ratings yet
- Envenimations ViperinesDocument6 pagesEnvenimations ViperinesIJAR JOURNALNo ratings yet
- Corona Virus Disease 2019, DHF and The Clinical SimilarityDocument3 pagesCorona Virus Disease 2019, DHF and The Clinical Similarityketut suryanaNo ratings yet
- Facing COVID-19 with the benefits of medical cannabisFrom EverandFacing COVID-19 with the benefits of medical cannabisNo ratings yet
- DR NLR 6-8-2021Document2 pagesDR NLR 6-8-2021FihzanNo ratings yet
- Nama/Usia/Bb/Tb Diagnosa Laboratorium Terapi Rencana 16/8: TC Apheresis 1 Unit (Masih Cari Donor)Document2 pagesNama/Usia/Bb/Tb Diagnosa Laboratorium Terapi Rencana 16/8: TC Apheresis 1 Unit (Masih Cari Donor)FihzanNo ratings yet
- 1 IRIS in ChildrenDocument6 pages1 IRIS in ChildrenFihzanNo ratings yet
- NIH Public Access: Author ManuscriptDocument15 pagesNIH Public Access: Author ManuscriptFihzanNo ratings yet
- Vitamin D Deficiency in Children With Newly Diagnosed Idiopathic EpilepsyDocument5 pagesVitamin D Deficiency in Children With Newly Diagnosed Idiopathic EpilepsyFihzanNo ratings yet
- 2525-Main Article Text (Blinded Article File) - 5250-2!10!20201009Document3 pages2525-Main Article Text (Blinded Article File) - 5250-2!10!20201009FihzanNo ratings yet
- Arif 2009Document2 pagesArif 2009FihzanNo ratings yet
- Funda Finals ReviewerDocument23 pagesFunda Finals Reviewerchloepaxton030No ratings yet
- Nubank: A Brazilian FinTech Worth $10 Billion - MEDICIDocument25 pagesNubank: A Brazilian FinTech Worth $10 Billion - MEDICIKay BarnesNo ratings yet
- Final 4 For Theoriees of LearningDocument2 pagesFinal 4 For Theoriees of LearningRose B Unlad67% (6)
- Bapi Sarkar CV 3Document2 pagesBapi Sarkar CV 3BapiNo ratings yet
- Pasteurization and Heat Sterilization - 2Document33 pagesPasteurization and Heat Sterilization - 2Taf Du Plessis Ngara50% (2)
- Tentative Awards ListDocument121 pagesTentative Awards ListA.R MaheshNo ratings yet
- Lab 1Document11 pagesLab 1Saimun MahumdNo ratings yet
- Organic Chemistry Lab Caffeine ExtractionDocument8 pagesOrganic Chemistry Lab Caffeine Extractionneuronerd50% (2)
- Manual - HSIDocument23 pagesManual - HSIrichardfloyd100% (2)
- Journal Pmed 1003501Document28 pagesJournal Pmed 1003501Abi HinojosaNo ratings yet
- Sicoma Twin Shaft Mixer Brochure JECDocument8 pagesSicoma Twin Shaft Mixer Brochure JECShabrina Meitha Nadhila RamadhanNo ratings yet
- The Effects of Drinking Alcoholic Beverages in The 4 Year Nursing Students of Brent Hospital and Colleges Incorporated School Year 2010 - 2011Document17 pagesThe Effects of Drinking Alcoholic Beverages in The 4 Year Nursing Students of Brent Hospital and Colleges Incorporated School Year 2010 - 2011Chauz Undo100% (2)
- Acting Shakespeare Scansion Assignment WorksheetDocument7 pagesActing Shakespeare Scansion Assignment WorksheetShalomNo ratings yet
- K8V-X Se: User GuideDocument82 pagesK8V-X Se: User GuidethegamersNo ratings yet
- May 2016 Computer Science Exam Paper, Paper 2 in English Time Zone 0Document15 pagesMay 2016 Computer Science Exam Paper, Paper 2 in English Time Zone 0Vidya DubeyNo ratings yet
- AN1671 MC145170 PSpice Modeling KitDocument14 pagesAN1671 MC145170 PSpice Modeling KitfahkingmoronNo ratings yet
- HP ProLiant ML370 G6 ServerDocument4 pagesHP ProLiant ML370 G6 ServerTrenell Steffan IsraelNo ratings yet
- Bloom Ks2-Maths-Blooms-Taxonomy-Meet-The-Bloom-Superheroes-Desk-Mat - Ver - 3Document1 pageBloom Ks2-Maths-Blooms-Taxonomy-Meet-The-Bloom-Superheroes-Desk-Mat - Ver - 3hongbongNo ratings yet
- Total Equivalength Length For Pipes - Valves - FittingsDocument6 pagesTotal Equivalength Length For Pipes - Valves - Fittingsvanchai sapaNo ratings yet
- Aurora of The Philosophers by ParacelsusDocument23 pagesAurora of The Philosophers by ParacelsusWesley MartusewiczNo ratings yet
- Impact of Working Capital Management On PDFDocument113 pagesImpact of Working Capital Management On PDFAhmed Afridi Bin FerdousNo ratings yet
- 07363769410070872Document15 pages07363769410070872Palak AgarwalNo ratings yet
- Sunpower 405, 410 y 415 WDocument2 pagesSunpower 405, 410 y 415 WArmandoVelazcoRangelNo ratings yet
- Hap Heenot ModuleDocument11 pagesHap Heenot Modulejohn kyaniaNo ratings yet
- Commercial Book ListDocument6 pagesCommercial Book Listoto saviourNo ratings yet
- Practical TestDocument3 pagesPractical TestsunoothecutestNo ratings yet
Co-Infection of Dengue and COVID-19: A Case: Neglected Tropical Diseases
Co-Infection of Dengue and COVID-19: A Case: Neglected Tropical Diseases
Uploaded by
FihzanOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Co-Infection of Dengue and COVID-19: A Case: Neglected Tropical Diseases
Co-Infection of Dengue and COVID-19: A Case: Neglected Tropical Diseases
Uploaded by
FihzanCopyright:
Available Formats
PLOS NEGLECTED TROPICAL DISEASES
SYMPOSIUM
Co-infection of dengue and COVID-19: A case
report
Morgane Verduyn ID1*, Nathalie Allou1, Virgile Gazaille ID1, Michel Andre ID1,
Tannvir Desroche ID2, Marie-Christine Jaffar ID3, Nicolas Traversier3, Cecile Levin ID4,
Marie Lagrange-Xelot4, Marie-Pierre Moiton4, Stella Hoang ID4
1 Department of Respiratory Medicine, University Hospital Centre of Réunion, Saint-Denis, France,
2 Department of Dermatology, University Hospital Centre of Réunion, Saint-Denis, France, 3 Department of
Microbiology, University Hospital Centre of Réunion, Saint-Denis, France, 4 Department of Infectious
Diseases, University Hospital Centre of Réunion, Saint-Pierre, France
* morgane.verduyn@chu-reunion.fr
Dengue and coronavirus disease 2019 (COVID-19) may share clinical and laboratory features.
Reunion Island is a French overseas department located in the Indian Ocean with a popula-
tion of more than 850,000 inhabitants. Due to its tropical climate, Reunion Island is at risk of
arbovirus outbreaks. An increase in the number of dengue cases has been reported on the
island since the beginning of 2018, with 3 different serotypes circulating mostly in austral sum-
mer. According to the last epidemiological report of March 30, 2020 from Santé Publique
France, 3,144 new cases of dengue have been diagnosed since the beginning of 2020 in
a1111111111
a1111111111 Reunion Island [1]. On March 2020, the first COVID-19 cases were imported to the island
a1111111111 from metropolitan France by airplane.
a1111111111 We report the case of an 18-year-old male living in Reunion Island, with no relevant past
a1111111111 medical history except occasional migraines. Our patient travelled back from Strasbourg (ini-
tial French epicenter of COVID-19) to Reunion Island on March 18, 2020. After his arrival, he
returned to his parents’ home, respected national confinement guidelines, and only went shop-
ping once.
The onset of symptoms occurred on April 3, with fever (39˚C), asthenia, anorexia, and
OPEN ACCESS
headache. On April 4, he tested positive in the emergency department for severe acute respira-
Citation: Verduyn M, Allou N, Gazaille V, Andre M, tory syndrome coronavirus 2 (SARS-CoV-2) infection by reverse transcription (RT)-PCR (E
Desroche T, Jaffar M-C, et al. (2020) Co-infection
gene, RdRP gene, and N gene positive), the causative virus of COVID-19. He was discharged
of dengue and COVID-19: A case report. PLoS Negl
Trop Dis 14(8): e0008476. https://doi.org/10.1371/ from the emergency room after diagnosis.
journal.pntd.0008476 On April 5, an itchy erythema rash appeared. He came back to the hospital on April 7 for
persistent fever (38.7˚C), arthromyalgia, dyspnea with polypnea (respiratory rate of 24 breaths
Editor: Laith Yakob, London School of Hygiene &
Tropical Medicine, UNITED KINGDOM per minute), and itchy maculopapular rash. The dengue rapid test was positive (NS1 antigen+)
in the emergency department. Therefore, he was hospitalized the same day in the COVID-19
Published: August 3, 2020
unit of St Denis University Hospital Center.
Copyright: © 2020 Verduyn et al. This is an open The physical examination revealed a body temperature of 38˚C, blood pressure of 112/63
access article distributed under the terms of the mmHg, pulse of 63 beats per minute, and oxygen saturation of 99% in ambient air. He had dry
Creative Commons Attribution License, which
cough (since February) and no chest pain. Pulmonary auscultation was normal. He had no
permits unrestricted use, distribution, and
reproduction in any medium, provided the original hematuria. He described retro-orbital eye pain and mild photophobia, with anorexia, nausea,
author and source are credited. and vomiting. He had infracentimetric cervical lymphadenopathies. Skin examination showed
a roseoliform maculopapular exanthema of the trunk, limbs, and face, which rapidly evolved
Funding: The authors received no specific funding
for this work. into a scarlatiniform-like rash. There were no real intervals of healthy skin but rounded islands
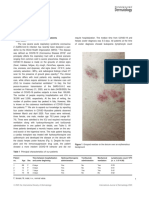
of sparing (“white islands in a sea of red”) (Figs 1 and 2). There was no mucosal involvement
Competing interests: The authors have declared
nor hand and feet affection involvement. The itching had stopped, and there was no scratching
that no competing interests exist.
PLOS Neglected Tropical Diseases | https://doi.org/10.1371/journal.pntd.0008476 August 3, 2020 1/5
PLOS NEGLECTED TROPICAL DISEASES Co-infection of dengue and COVID-19: A case report
Fig 1. Photograph at hospital admission: roseoliform maculopapular exanthema with healthy skin intervals on left
arm.
https://doi.org/10.1371/journal.pntd.0008476.g001
lesion. At admission, he had thrombocytopenia (platelet count 106 × 109/mL), leucopenia
(1.7 × 109/mL), lymphopenia (0.6 × 109/mL), and neutropenia (1. × 109/mL). Liver function
test were subnormal (aspartate aminotransferase 51 U/L and alanine aminotransferase 23 U/
L). C-reactive protein was normal (4.7 mg/L). Serotype 1 dengue was confirmed by RT-PCR
and positive serology (immunoglobulin M [IgM]: 3.9 and IgG: 2.1) on day 6 after the onset of
symptoms. The computed tomography (CT) scan performed at admission was normal without
any ground glass opacities nor consolidations (Fig 3).
Fever above 39˚C lasted 10 days, and the patient’s symptoms gradually improved. He
returned home after 7 days of hospitalization. After initial worsening of thrombocytopenia
(41 × 109/mL) and cytolysis (alanine aminotransferase: 545 U/L, aspartate aminotransferase:
621 U/L), the biological balance had started to improve at the end of hospitalization.
The patient’s parents tested negative for SARS-CoV-2 and declared a dengue at the same
time.Informed consent was given orally by the individual to participate in this local retrospec-
tive observational study, which was approved by the local ethics committee and was declared
to the Commission Nationale de l’Informatique et des Libertés (French Data Protection
Agency or CNIL MR004). Informed consent was also obtained for publication of this case
report with photographs.
To our knowledge, this is the first case of confirmed co-infection of dengue and COVID-
19. In Singapore, 2 patients initially tested positive with a dengue rapid test. Ultimately,
RT-PCR for dengue was negative, and both patients tested positive for SARS-CoV-2 infection
by RT-PCR [2].
Distinction between dengue fever and COVID-19’s clinical features may be difficult. Our
patient’s symptoms are consistent with dengue, including prolonged fever, facial flushing skin
PLOS Neglected Tropical Diseases | https://doi.org/10.1371/journal.pntd.0008476 August 3, 2020 2/5
PLOS NEGLECTED TROPICAL DISEASES Co-infection of dengue and COVID-19: A case report
Fig 2. Photograph during hospitalization: diffuse exanthema with rounded island of sparing (“white island in a
sea of red”).
https://doi.org/10.1371/journal.pntd.0008476.g002
Fig 3. CT scan at admission was normal. CT, computed tomography.
https://doi.org/10.1371/journal.pntd.0008476.g003
PLOS Neglected Tropical Diseases | https://doi.org/10.1371/journal.pntd.0008476 August 3, 2020 3/5
PLOS NEGLECTED TROPICAL DISEASES Co-infection of dengue and COVID-19: A case report
erythema, generalized body ache, myalgia, arthralgia, retro-orbital eye pain, photophobia,
rubeoliform exanthema, and headache [3–4]. But some of them may be also consistent with
clinical symptoms of COVID-19 [5]. Thrombocytopenia and elevated liver enzymes are also
reported in both diseases. Thrombocytopenia and cytolysis were reported, respectively, in
36.2% and 21.3% of the patients with COVID-19 [5]. As in dengue fever [6], immune-medi-
ated damage or direct cytotoxicity due to active viral replication in hepatic cells may be
involved in hepatic injuries in COVID-19, but it could be also related to hypoxic hepatitis due
to anoxia, reactivation of preexisting liver disease, or drug-induced liver injury (such as para-
cetamol, antiviral agents, etc.) [7].
In our case, making the hypothesis of a COVID-19 contamination during the flight on
March 18 (where a confirmed COVID-19 passenger had been identified), an incubation
period until symptoms on April 3 would have been longer than what has been described so far
[8]. It is more likely that our patient was asymptomatic for SARS-CoV-2 infection but that
RT-PCR was still positive on day 17, as previously described [9], and that most of his symp-
toms were related to dengue fever. In that case, it would have been interesting to use SARS--
CoV-2 serology to identify a real and active co-infection from a case of dengue fever occurring
in a SARS-CoV-2 cured patient.
Nonetheless, our patient presented a quite severe dengue infection with no previous epi-
sodes to his knowledge. Dengue serology on day 6 was positive for IgG (2.1) and IgM (3.9).
One hypothesis could be that SARS-CoV-2 infection is more likely to give more severe symp-
toms in the case of co-infection.
Recently, skin damage has been described in COVID-19, but none of it seems to be specific
to COVID-19. In Italy, 14 of 18 patients with cutaneous manifestations developed an erythem-
atous rash, and 3 patients developed widespread urticarial [10]. The main region involved was
trunk, and itching was low or absent. In Thailand, dermatologists also reported the case of a
patient with an exanthema with fever initially diagnosed as dengue; finally, the patient was
diagnosed for COVID-19 infection [11]. In April 2020, a French dermatologist reported the
appearance of pseudo-frostbite of the extremities, sudden appearance of persistent redness,
and sometimes painful, temporary, hive-like lesions [12]. In tropical areas where COVID-19
and arboviruses coexist, clinical distinction between different skin symptoms may be difficult.
In our case, rounded islands of sparing “white islands in a sea of red” seem to be more specific
of dengue virus [13].
We described here the first confirmed case of co-infection of dengue fever and COVID-19.
In tropical areas where arboviruses and COVID-19 may coexist, clinical diagnosis is difficult,
and patients should be tested for both viruses. Larger studies are needed to evaluate increased
morbidity of these co-infections.
References
1. Dengue à la Réunion Augmentation du nombre de cas hebdomadaire [cited 2020 January 20] Available
from: https://www.santepubliquefrance.fr/regions/ocean-indien/documents/bulletin-regional/2020/
surveillance-de-la-dengue-a-la-reunion.-point-au-20-janvier-2020.
2. Yan G, Lee CK, Lam LTM, Yan B, Chua YX, Lim AYN, et al. Covert COVID-19 and false-positive den-
gue serology in Singapore. Lancet Infect Dis. 2020; 20(5): 536. https://doi.org/10.1016/S1473-3099
(20)30158-4 PMID: 32145189
3. Muller DA, Depelsenaire ACI, Young PR. Clinical and laboratory diagnosis of dengue virus infection. J
Infect Dis. 2017; 215: S89–S95. https://doi.org/10.1093/infdis/jiw649 PMID: 28403441
4. Ligon BL. Dengue fever and Dengue Hemorrhagic fever: A review of history, transmission, treatment
and prevention. Semin Pediatr Infect Dis. 2004; 16:60–5.
5. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan Lei CL, et al. Clinical Characteristics of
Coronavirus Disease 2019 in China. N Engl J Med. 2020;30; 382(18):1708–1720. https://doi.org/10.
1056/NEJMoa2002032 PMID: 32109013
PLOS Neglected Tropical Diseases | https://doi.org/10.1371/journal.pntd.0008476 August 3, 2020 4/5
PLOS NEGLECTED TROPICAL DISEASES Co-infection of dengue and COVID-19: A case report
6. Tan SS, Bujang MA. The Clinical Features and Outcomes of Acute Liver Failure Associated With Den-
gue Infection in Adults: A Case Series. Braz J Infect Dis. 2013; 17(2):164–90 https://doi.org/10.1016/j.
bjid.2012.09.007 PMID: 23453417
7. Sun J, Aghemo A, Forner A, Valenti L. COVID-19 and liver disease. Liver Int. 2020; 40(6):1278–1281.
https://doi.org/10.1111/liv.14470 PMID: 32251539
8. Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y,Ren R et al. Early Transmissions Dynamics in Wuhan,
China, of Novel Coronavirus–Infected Pneumonia. N Engl J Med. 2020; 26; 382(13): 1199–1207.
https://doi.org/10.1056/NEJMoa2001316 PMID: 31995857
9. https://pubmed.ncbi.nlm.nih.gov/32200654/?from_term=covid+19+kinetics&from_pos=4 To KK, Tsang
OT, Leung WS, Tam AR, Wu TC, Lung DC, Yip CC, Cai JP, et al. Temporal profiles of viral load in pos-
terior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2:
an observational cohort study. Lancet Infect Dis. 2020; 23:S1473-3099(20)30196-1.
10. Recalcati S. Cutaneous manifestations in COVID-19: A first perspective. J Eur Acad Dermatol Venereol
2020; 34(5):e212–e213. https://doi.org/10.1111/jdv.16387 PMID: 32215952
11. Joob B, Wiwanitkit V. COVID-19 can present with a rash and be mistaken for Dengue. J Am Acad Der-
matol. 2020; 82(5):e177. https://doi.org/10.1016/j.jaad.2020.03.036 PMID: 32213305
12. Le syndicat National des Dermatologues-Vénéréologues (SNDV) alerte sur les manifestations cuta-
nées du COVID 19. [cited 2020 April 4] Available from: https://www.syndicatdermatos.org/
communique/communique-de-presse-06-04-20-covid19-et-peau.
13. Waterman SH, Gubler DJ. Dengue fever. Clin Dermatol. 1989; 7:117–22. https://doi.org/10.1016/0738-
081x(89)90034-5 PMID: 2647259
PLOS Neglected Tropical Diseases | https://doi.org/10.1371/journal.pntd.0008476 August 3, 2020 5/5
You might also like
- A2 UNIT 4 Test Answer Key HigherDocument2 pagesA2 UNIT 4 Test Answer Key HigherMaxi Comas60% (5)
- Teste Ingles TecnologiaDocument5 pagesTeste Ingles TecnologiaMeninaDuMarNo ratings yet
- Wiens N Priebe - Occlusal StabilityDocument25 pagesWiens N Priebe - Occlusal Stabilitymoji_puiNo ratings yet
- Bilco WellboreDocument27 pagesBilco WellboreJhonathan Miranda100% (4)
- Faggionvinholo2020 Q1Document5 pagesFaggionvinholo2020 Q1NasriNo ratings yet
- Herpes Zoster in COVID19Document2 pagesHerpes Zoster in COVID19Vienesha Aress ShaNo ratings yet
- Varicella DengueDocument3 pagesVaricella DengueDian Fajar FebrianiNo ratings yet
- Measles During Arbovirus Outbreak: A Diagnostic Challenge: Case ReportDocument4 pagesMeasles During Arbovirus Outbreak: A Diagnostic Challenge: Case ReportnisceNo ratings yet
- DengueDocument2 pagesDengueElsa YuliusNo ratings yet
- Dengue Shock Syndrome in An Infant: Linda Aurpibul, Punyawee Khumlue, Satja Issaranggoon Na Ayuthaya, Peninnah OberdorferDocument4 pagesDengue Shock Syndrome in An Infant: Linda Aurpibul, Punyawee Khumlue, Satja Issaranggoon Na Ayuthaya, Peninnah OberdorferLIdya Goprani UmarNo ratings yet
- Dengue Haemorrhagic Fever/dengue Shock Syndrome: Lessons From The Cuban Epidemic, 1981Document6 pagesDengue Haemorrhagic Fever/dengue Shock Syndrome: Lessons From The Cuban Epidemic, 1981WahyuNo ratings yet
- Variants of Erythema Multiforme: A Case Report and Literature ReviewDocument8 pagesVariants of Erythema Multiforme: A Case Report and Literature Reviewranz ibonkNo ratings yet
- E042398 FullDocument10 pagesE042398 Fullcomfica.formacion1.2023No ratings yet
- 1 s2.0 S0399077X20300688 MainDocument4 pages1 s2.0 S0399077X20300688 MainAnastasija LevcenkoNo ratings yet
- Tammaro2020 Covid 19Document7 pagesTammaro2020 Covid 19eva yustianaNo ratings yet
- Art:10.1007/s00431-009-0986-0 222Document6 pagesArt:10.1007/s00431-009-0986-0 222Sybelle CostaNo ratings yet
- High Mortality Associated With An Outbreak of Hepatitis E Among Displaced Persons in Darfur, SudanDocument6 pagesHigh Mortality Associated With An Outbreak of Hepatitis E Among Displaced Persons in Darfur, SudanKatrina ManurungNo ratings yet
- Journal Pre-Proof: M Edecine Et Maladies InfectieusesDocument12 pagesJournal Pre-Proof: M Edecine Et Maladies Infectieusessameer sahaanNo ratings yet
- Covid19 PDFDocument6 pagesCovid19 PDFJeremy Kartika SoeryonoNo ratings yet
- Prevention of Nosocomial Covid 19 Another Challenge of The PandemicDocument2 pagesPrevention of Nosocomial Covid 19 Another Challenge of The PandemicZied FehriNo ratings yet
- 51 Uysal EtalDocument4 pages51 Uysal EtaleditorijmrhsNo ratings yet
- Article - N - Clinical Manifestations of Imported Cases of Dengue FeverDocument3 pagesArticle - N - Clinical Manifestations of Imported Cases of Dengue FeverIm jungkook JUSTIN SEAGULL A.K.A jungshookNo ratings yet
- TPMD 200176Document2 pagesTPMD 200176Marcelita DuwiriNo ratings yet
- Tetanus: Clostridium Tetani. The Disease Was First Described in The 14th Century by John of ArderneDocument5 pagesTetanus: Clostridium Tetani. The Disease Was First Described in The 14th Century by John of Ardernedefitri sariningtyasNo ratings yet
- Jurnal Inggris 4Document6 pagesJurnal Inggris 4pikNo ratings yet
- 2019 Novel Coronavirus Disease (COVID-19) in Taiwan: Reports of Two Cases From Wuhan, ChinaDocument4 pages2019 Novel Coronavirus Disease (COVID-19) in Taiwan: Reports of Two Cases From Wuhan, ChinaClaav HugNo ratings yet
- HellowDocument6 pagesHellowafina rasaki asriNo ratings yet
- 220 FullDocument4 pages220 FullOttofianus Alvedo Hewick KalangiNo ratings yet
- Dengue in Renal Transplant Patients A ReDocument3 pagesDengue in Renal Transplant Patients A ReMaira EscobarNo ratings yet
- Primary Varicella Zooster and SARS-CoV-2 Coinfection: Case ReportDocument7 pagesPrimary Varicella Zooster and SARS-CoV-2 Coinfection: Case ReportIrfan CorovicNo ratings yet
- Dengue_activity_in_Puerto_Rico_during_an_interepidDocument10 pagesDengue_activity_in_Puerto_Rico_during_an_interepidCrónicos Jurisdicción J5No ratings yet
- Case Report Severe Covid-19Document3 pagesCase Report Severe Covid-1911TH-01 Lâm Nguyễn Ngọc AnhNo ratings yet
- The First Case of 2019 Novel Coronavirus Pneumonia Imported Into Korea From Wuhan, China: Implication For Infection Prevention and Control MeasuresDocument4 pagesThe First Case of 2019 Novel Coronavirus Pneumonia Imported Into Korea From Wuhan, China: Implication For Infection Prevention and Control MeasuresTian KurniawanNo ratings yet
- 1 s2.0 S0399077X20301104 MainDocument4 pages1 s2.0 S0399077X20301104 MainNaira ValentinaNo ratings yet
- (14761645 - The American Journal of Tropical Medicine and Hygiene) Case Report - Severe COVID-19 and Dengue in An Indonesian InfantDocument5 pages(14761645 - The American Journal of Tropical Medicine and Hygiene) Case Report - Severe COVID-19 and Dengue in An Indonesian InfantAnggrainiAlamNo ratings yet
- Chronic Meningococcemia in A 16-Year-Old Boy: A Case ReportDocument3 pagesChronic Meningococcemia in A 16-Year-Old Boy: A Case ReportUjean Santos SagaralNo ratings yet
- Art 04Document7 pagesArt 04Florentino HerreraNo ratings yet
- New-Onset Myasthenia Gravis After Novel Coronavirus 2019 InfectionDocument3 pagesNew-Onset Myasthenia Gravis After Novel Coronavirus 2019 Infectionarenasgrecia1No ratings yet
- Anthrax of The Gastrointestinal Tract: Thira Sirisanthana and Arthur E. BrownDocument3 pagesAnthrax of The Gastrointestinal Tract: Thira Sirisanthana and Arthur E. BrownJuli SatriaNo ratings yet
- Al Muthanna International Trauma Conference MAY 9 - 11, 2020 Samawa, IraqDocument5 pagesAl Muthanna International Trauma Conference MAY 9 - 11, 2020 Samawa, IraqKhaliel AlsarafNo ratings yet
- 2020 Case 23-2020, A 76-Year-Old Woman Who Died From Covid-19Document8 pages2020 Case 23-2020, A 76-Year-Old Woman Who Died From Covid-19alejandromfunes1749No ratings yet
- Tiapride Associated Toxic Epidermal Necrolysis in An Hivinfected Patient. 2165 7920.1000323Document3 pagesTiapride Associated Toxic Epidermal Necrolysis in An Hivinfected Patient. 2165 7920.1000323Mega RafikaNo ratings yet
- Spectrum - of - Clinicopathologic - Findings - In.1 (1Document11 pagesSpectrum - of - Clinicopathologic - Findings - In.1 (1tzuskyNo ratings yet
- LepsDocument4 pagesLepslynharee100% (1)
- Difficulties in Diagnosis of Psittacosis or Ornithosis: A Case ReportDocument4 pagesDifficulties in Diagnosis of Psittacosis or Ornithosis: A Case ReportYusrinabillaNo ratings yet
- Corynebacterium Ulcerans Cutaneous Diphtheria: Grand RoundDocument8 pagesCorynebacterium Ulcerans Cutaneous Diphtheria: Grand RoundRafa NaufalinNo ratings yet
- 1287-Article Text-3033-1-10-20170221Document15 pages1287-Article Text-3033-1-10-20170221andi ramdhanNo ratings yet
- Dr. DEEPAK.N.S (PG), Dr. PAGADPALLY SRINIVAS (Professor & Head) Dept. of Paediatrics, VMMC, KKLDocument2 pagesDr. DEEPAK.N.S (PG), Dr. PAGADPALLY SRINIVAS (Professor & Head) Dept. of Paediatrics, VMMC, KKLSumanth KumarNo ratings yet
- Follow Up of Skin Lesions During The Evolution of COVID 19 A CaseDocument4 pagesFollow Up of Skin Lesions During The Evolution of COVID 19 A CaseZihan ZetiraNo ratings yet
- Clinical Characteristics of Covid-19 Cases in Guayaquil, EcuadorDocument5 pagesClinical Characteristics of Covid-19 Cases in Guayaquil, EcuadorFarida MurtianiNo ratings yet
- 493 FullDocument4 pages493 FullDevy KurniawatiNo ratings yet
- Chikungunya in Children 2015Document3 pagesChikungunya in Children 2015Douglas UmbriaNo ratings yet
- JMV 25815Document2 pagesJMV 25815fadil ahmadiNo ratings yet
- Wabah Hepatititis EDocument7 pagesWabah Hepatititis EKatrina ManurungNo ratings yet
- Epidemiological Description of Chikungunya Virus Outbreak in Dire Dawa Administrative City, Eastern Ethiopia, 2019Document5 pagesEpidemiological Description of Chikungunya Virus Outbreak in Dire Dawa Administrative City, Eastern Ethiopia, 2019Nigatu AdmasuNo ratings yet
- PIIS0163445320302176Document2 pagesPIIS0163445320302176Hanan ArielNo ratings yet
- Nosocomial Infections in A Brazilian Burn Unit: Jefferson Lessa Soares de Macedo, Joa o Barberino SantosDocument5 pagesNosocomial Infections in A Brazilian Burn Unit: Jefferson Lessa Soares de Macedo, Joa o Barberino SantossriyuswantiNo ratings yet
- Clinical Findings h7n9Document5 pagesClinical Findings h7n9Martin HaynesNo ratings yet
- Three Cases of COVID-19 Patients Presenting With Erythema: Concise CommunicationDocument4 pagesThree Cases of COVID-19 Patients Presenting With Erythema: Concise CommunicationNur Alfia KusumaningsihNo ratings yet
- Spectrum of Clinicopathologic Findings in COVID-19-induced Skin LesionsDocument9 pagesSpectrum of Clinicopathologic Findings in COVID-19-induced Skin LesionsggenesisNo ratings yet
- Covid 19: Dennis N. Muñoz, RN, RM, LPTDocument103 pagesCovid 19: Dennis N. Muñoz, RN, RM, LPTDennis Nabor Muñoz, RN,RMNo ratings yet
- Envenimations ViperinesDocument6 pagesEnvenimations ViperinesIJAR JOURNALNo ratings yet
- Corona Virus Disease 2019, DHF and The Clinical SimilarityDocument3 pagesCorona Virus Disease 2019, DHF and The Clinical Similarityketut suryanaNo ratings yet
- Facing COVID-19 with the benefits of medical cannabisFrom EverandFacing COVID-19 with the benefits of medical cannabisNo ratings yet
- DR NLR 6-8-2021Document2 pagesDR NLR 6-8-2021FihzanNo ratings yet
- Nama/Usia/Bb/Tb Diagnosa Laboratorium Terapi Rencana 16/8: TC Apheresis 1 Unit (Masih Cari Donor)Document2 pagesNama/Usia/Bb/Tb Diagnosa Laboratorium Terapi Rencana 16/8: TC Apheresis 1 Unit (Masih Cari Donor)FihzanNo ratings yet
- 1 IRIS in ChildrenDocument6 pages1 IRIS in ChildrenFihzanNo ratings yet
- NIH Public Access: Author ManuscriptDocument15 pagesNIH Public Access: Author ManuscriptFihzanNo ratings yet
- Vitamin D Deficiency in Children With Newly Diagnosed Idiopathic EpilepsyDocument5 pagesVitamin D Deficiency in Children With Newly Diagnosed Idiopathic EpilepsyFihzanNo ratings yet
- 2525-Main Article Text (Blinded Article File) - 5250-2!10!20201009Document3 pages2525-Main Article Text (Blinded Article File) - 5250-2!10!20201009FihzanNo ratings yet
- Arif 2009Document2 pagesArif 2009FihzanNo ratings yet
- Funda Finals ReviewerDocument23 pagesFunda Finals Reviewerchloepaxton030No ratings yet
- Nubank: A Brazilian FinTech Worth $10 Billion - MEDICIDocument25 pagesNubank: A Brazilian FinTech Worth $10 Billion - MEDICIKay BarnesNo ratings yet
- Final 4 For Theoriees of LearningDocument2 pagesFinal 4 For Theoriees of LearningRose B Unlad67% (6)
- Bapi Sarkar CV 3Document2 pagesBapi Sarkar CV 3BapiNo ratings yet
- Pasteurization and Heat Sterilization - 2Document33 pagesPasteurization and Heat Sterilization - 2Taf Du Plessis Ngara50% (2)
- Tentative Awards ListDocument121 pagesTentative Awards ListA.R MaheshNo ratings yet
- Lab 1Document11 pagesLab 1Saimun MahumdNo ratings yet
- Organic Chemistry Lab Caffeine ExtractionDocument8 pagesOrganic Chemistry Lab Caffeine Extractionneuronerd50% (2)
- Manual - HSIDocument23 pagesManual - HSIrichardfloyd100% (2)
- Journal Pmed 1003501Document28 pagesJournal Pmed 1003501Abi HinojosaNo ratings yet
- Sicoma Twin Shaft Mixer Brochure JECDocument8 pagesSicoma Twin Shaft Mixer Brochure JECShabrina Meitha Nadhila RamadhanNo ratings yet
- The Effects of Drinking Alcoholic Beverages in The 4 Year Nursing Students of Brent Hospital and Colleges Incorporated School Year 2010 - 2011Document17 pagesThe Effects of Drinking Alcoholic Beverages in The 4 Year Nursing Students of Brent Hospital and Colleges Incorporated School Year 2010 - 2011Chauz Undo100% (2)
- Acting Shakespeare Scansion Assignment WorksheetDocument7 pagesActing Shakespeare Scansion Assignment WorksheetShalomNo ratings yet
- K8V-X Se: User GuideDocument82 pagesK8V-X Se: User GuidethegamersNo ratings yet
- May 2016 Computer Science Exam Paper, Paper 2 in English Time Zone 0Document15 pagesMay 2016 Computer Science Exam Paper, Paper 2 in English Time Zone 0Vidya DubeyNo ratings yet
- AN1671 MC145170 PSpice Modeling KitDocument14 pagesAN1671 MC145170 PSpice Modeling KitfahkingmoronNo ratings yet
- HP ProLiant ML370 G6 ServerDocument4 pagesHP ProLiant ML370 G6 ServerTrenell Steffan IsraelNo ratings yet
- Bloom Ks2-Maths-Blooms-Taxonomy-Meet-The-Bloom-Superheroes-Desk-Mat - Ver - 3Document1 pageBloom Ks2-Maths-Blooms-Taxonomy-Meet-The-Bloom-Superheroes-Desk-Mat - Ver - 3hongbongNo ratings yet
- Total Equivalength Length For Pipes - Valves - FittingsDocument6 pagesTotal Equivalength Length For Pipes - Valves - Fittingsvanchai sapaNo ratings yet
- Aurora of The Philosophers by ParacelsusDocument23 pagesAurora of The Philosophers by ParacelsusWesley MartusewiczNo ratings yet
- Impact of Working Capital Management On PDFDocument113 pagesImpact of Working Capital Management On PDFAhmed Afridi Bin FerdousNo ratings yet
- 07363769410070872Document15 pages07363769410070872Palak AgarwalNo ratings yet
- Sunpower 405, 410 y 415 WDocument2 pagesSunpower 405, 410 y 415 WArmandoVelazcoRangelNo ratings yet
- Hap Heenot ModuleDocument11 pagesHap Heenot Modulejohn kyaniaNo ratings yet
- Commercial Book ListDocument6 pagesCommercial Book Listoto saviourNo ratings yet
- Practical TestDocument3 pagesPractical TestsunoothecutestNo ratings yet