Professional Documents
Culture Documents
Lesson 1 Alkenes and Alyknes
Lesson 1 Alkenes and Alyknes
Uploaded by
MARY JANE ANGELICA SEVAOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Lesson 1 Alkenes and Alyknes
Lesson 1 Alkenes and Alyknes
Uploaded by
MARY JANE ANGELICA SEVACopyright:
Available Formats
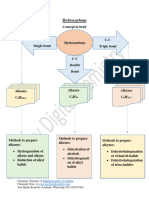
ORGANIC CHEMISTRY
Lesson 1/midterms/08-03-2021
Alkenes and Alkynes - The parent name is that of the longest chain that
- Alkene: a hydrocarbon that contains one or more contains the C=C.
carbon-carbon double bonds. - Number the chain from the end that gives the lower
o ethylene is the simplest alkene. numbers to the carbons of the C=C.
- Alkyne: a hydrocarbon that contains one or more - Locate the C=C by the number of its first carbon.
carbon-carbon triple bonds. - Use the ending -ene to show the presence of the
o acetylene is the simplest alkyne. C=C
- Branched-chain alkenes are named in a manner
H H
C C H-C C-H
similar to alkanes; substituted groups are located and
H H named.
Ethylene
(an alkene)
Acetylene
(an alkyne) Alkenes - IUPAC Names
Structure of Alkenes • Examples
- Valence Shell Electron Repulsion (VSEPR) model 5
4
3
2
1
predicts bond angles of 120° about each carbon of a 6 4 2 6 4 2
5 3 1 5 3 1
double bond. CH3 CH2 CH3
- In ethylene, the actual angles are close to 120°. CH3 CH2 CH2 CH2 CH=CH2 CH3 CH2 CHCH2 CH=CH2 CH3 CH2 CHC=CH2
CH2 CH3
- In substituted alkenes, angles about each carbon of 1-Hexene 4-Methyl-1-hexene 2,3-Diethyl-1-pentene
the double bond may be greater than 120° because of Alkynes - IUPAC Nomenclature
repulsion between groups bonded to the double • follow the same rules as for alkenes, but use the
bond. ending -yne to show the presence of the triple bond.
1
124.7°
1 CH3 3 4
121.7° 4
2 2 5
H H H3 C H CH3 CHC CH 3 CH3 CH2 C CCH2 CCH3 6 7
C C C C
CH3 CH3
H H H H 3-Methyl-1-butyne 6,6-Dimethyl-3-heptyne
Ethylene Propene
Common Names
• VSEPR theory assumes that each atom in a • Common names are still used for some alkenes and
molecule will achieve a geometry that alkynes, particularly those of low molecular weight.
minimizes the repulsion between electrons in the CH3
valence shell of that atom. CH2 =CH2 CH3 CH= CH2 CH3 C=CH2
• The five compounds shown in the figure below IUPAC name: Ethene Propene 2-Methylpropene
can be used to demonstrate how the VSEPR Common name: Ethylene Propylene Isobutylene
theory can be applied to simple molecules. HC CH CH3 C CH CH3 C CCH3
IUPAC name: Ethyne Propyne 2-Butyne
Common name: Acetylene Methylacetylene Dimethylacetylene
Cycloalkenes
• To name a cycloalkene:
- number the carbon atoms of the ring double
bond 1 and 2 in the direction that gives the
lower number to the substituent encountered
first.
- number and list substituents in alphabetical
order.
Alkenes 4
6
1
3 5
Cis-trans isomerism 5
4
- because of restricted rotation about a carbon-carbon 1 2 3
2
double bond, an alkene with two different groups on 3-Methylcyclopentene 4-Ethyl-1-methylcyclohexene
each carbon of the double bond shows cis-trans (not 5-methylcyclopentene) (not 5-ethyl-2-methylcyclohexene)
isomerism. Dienes, Trienes, Polyenes
H H H CH3 • Alkenes that contain more than one double bond
C C
H3 C CH3 H3 C
C C
H
are named as alkadienes, alkatrienes, and so on.
cis-2-Butene trans-2-Butene • Those that contain several double bonds are
mp -139°C, bp 4°C mp -106°C, bp 1°C referred to more generally as polyenes (Greek:
Alkenes - IUPAC Nomenclature poly, many).
• To name an alkene:
ORGANIC CHEMISTRY
Lesson 1/midterms/08-03-2021
OH
CH3 OH
CH2 = CHCH2 CH=CH2 CH2 =CCH=CH2
1,4-Pentadiene 2-Methyl-1,3-butadiene 1,3-Cyclopentadiene Farnesol Vitamin A (retinol)
(Isoprene) (Lily-of-the valley)
Physical Properties Reactions of Alkenes
- Alkenes and alkynes are nonpolar compounds. • The most common reaction is addition
- The only attractive forces between their molecules Reaction Descriptive Name(s )
are London dispersion forces. H Cl
- Their physical properties are similar to those of C C + HCl C C hydrochlorination
alkanes with the same carbon skeletons. H OH
- Alkenes and alkynes are insoluble in water but C C + H2 O C C hydration
soluble in one another and in nonpolar organic Br Br
liquids. C C + Br2 C C bromination
- Alkenes and alkynes that are liquid or solid at room H H
temperature have densities less than 1.0 g/mL; they C C + H2 C C hydrogenation
(reduction)
float on water.
Most alkene addition reactions are exothermic.
Ethene, an alkene, is a gas with sweetish odor and the • The products are more stable (lower in energy)
most important raw material in organic chemical than the reactants.
industry. • Just because they are exothermic doesn’t mean that
• It is obtained from the refining of petroleum alkene addition reactions occur rapidly.
• It is used in making plastic polyethylene • Many alkene addition reactions require a catalyst.
• Also produced by plants which causes the H H H H
are replaced by
change in fruit color that occurs as fruits ripen C C + H H H C C H + heat
• Used by producers to produce proper ripe color H H H H
Ethyne, an alkyne, is better known by its common name one double bond three single bonds
acetylene. and one single bond
• Acetylene burns with oxygen in the Addition of HX
oxyacetylene torch to produce a very hot flame Addition of HX (HCl, HBr, or HI) to an alkene gives a
(about 3000 to 5400 OC that is used for cutting haloalkane.
and welding metals • H adds to one carbon of the C=C and X to the
• A very reactive gas that is also an important other.
starting material in the production of plastics and H Cl
synthetic rubber CH2 =CH2 + HCl CH2 -CH2
Terpenes Ethylene Chloroethane
(Ethyl chloride)
• Terpene: a compound whose carbon skeleton
• reaction is regioselective.
can be divided into five-carbon units identical
• Markovnikov’s rule: H adds to the less
with the carbon skeleton of isoprene.
CH3 head C tail substituted carbon and X to the more substituted
CH2 =C-CH=CH2
1 2 3 4
C-C-C-C
carbon.
Cl H H Cl
2-Methyl-1,3-butadiene Isoprene unit
(Isoprene) CH3 CH=CH2 + HCl CH3 CH-CH2 CH3 CH-CH2
•Terpenes illustrate an important principle of the Propene 2-Chloropropane 1-Chloropropane
(not formed)
molecular logic of living systems; Markovnikov's Rule: describes the outcome of some
in building large molecules, small subunits are
Alkenes ‘addition reactions
bonded together by a series of enzyme-catalyzed
• the acid hydrogen (H) becomes attached to
reactions and then chemically modified by additional
the carbon with more hydrogen substituents,
enzyme-catalyzed reactions.
forming this and the halide (X) group becomes attached to
head bond makes the carbon with more alkyl substituents.
OH the ring
• Alternatively, the rule can be stated that the
tail
OH hydrogen atom is added to the carbon with
the greatest number of hydrogen atoms while
Myrcene Geraniol Limonene
(Bay oil) (Rose and (Lemon
Menthol
(Peppermint)
the X component is added to the carbon with
other flowers) and orange) the least number of hydrogen atoms
ORGANIC CHEMISTRY
Lesson 1/midterms/08-03-2021
• Chemists account for the addition of HX to an – reaction is generally carried out using pure
alkene by a two-step reaction mechanism. reagents, or mixing them in a nonreactive organic
• We use curved arrows to show the solvent
Br Br
repositioning of electron pairs during a CH3 CH=CHCH3 + Br2 CH3 CH-CHCH3
chemical reaction. 2-Butene
CH2 Cl2
2,3-Dibromobutane
• The tail of an arrow shows the origin of Br
the electron pair (either on an atom or in + Br2
CH2 Cl2
the double bond). Br
Cyclohexene 1,2-Dibromocyclohexane
• The head of the arrow shows its new
position. – addition of Br2 is a useful qualitative test for the
• Curved arrows show us which bonds presence of a carbon-carbon double bond
break and which new ones form. – Br2 has a deep red color; dibromoalkanes are
colorless.
Addition of HCl to 2 Butene
Step 1: Addition of H2 – Reduction
– reaction of the carbon-carbon double bond with • Virtually all alkenes add H2 in the presence of a
H+ gives a secondary (2°) carbocation transition metal catalyst, commonly Pd, Pt, or Ni.
H3 C H
intermediate. C C + H2
Pd
CH3 CH2 CH2 CH3
+
H 25°C, 3 atm
CH3 CH=CHCH3 + H+ CH3 CH-CHCH3
H CH3 Butane
A 2° carbocation intermediate
trans-2-Butene
Step 2: Pd
+ H2
– reaction of the carbocation intermediate with 25°C, 3 atm
chloride ion completes the addition. Cyclohexene Cyclohexane
:
: Cl :
- +
Polymerization
: :
: Cl : + CH3 CHCH2 CH3 CH3 CHCH2 CH3
Chloride 2° Carbocation 2-Chlorobutane • From the perspective of the organic chemical
ion intermediate industry, the single most important reaction of
Addition of H2O alkenes is polymerization:
• Addition of water is called hydration – polymer: Greek: poly, many and meros, part
– hydration is acid catalyzed, most commonly by – monomer: Greek: mono, single and meros,
H2SO4. part
– hydration follows Markovnikov’s rule; H adds to initiator
nCH2 =CH2 CH2 CH2 n
the less substituted carbon and OH adds to the (polymerization)
more substituted carbon. Ethylene Polyethylene
OH H • Show the structure of a polymer by placing
H2 SO4
CH3 CH=CH2 + H2 O CH3 CH-CH2
Propene 2-Propanol
parentheses around the repeating monomer unit.
CH3 CH3 • Place a subscript, n, outside the parentheses to
H2 SO4
CH3 C=CH2 + H2 O CH3 C-CH2 indicate that this unit repeats n times.
HO H
2-Methylpropene 2-Methyl-2-propanol • The structure of a polymer chain can be reproduced
by repeating the enclosed structure in both
• Step 1: directions.
+ H • following a section of polypropene (polypropylene)
CH3 CH=CH2 + H+ CH3 CHCH2 monomer units shown in red
A 2° carbocation
intermediate
• Step 2: n
H H CH3
CH3 CH3 CH3 CH3
:
+ O+
CH2 CH-CH2 CH-CH2 CH-CH2 CH CH2 CH n
:
CH3 CHCH3 + :O-H CH3 CHCH3
H An oxonium ion Part of an extended polymer chain The repeating unit
• Step 3:
H H
:
O+ : OH
CH3 CHCH3 CH3 CHCH3 + H+
2-Propanol
Addition of Cl2 and Br2
• Addition takes place readily at room temp.
ORGANIC CHEMISTRY
Lesson 1/midterms/08-03-2021
Monomer Common Polymer Name(s) and
Formula Name Common Uses
CH2 =CH2 ethylene polyethylene, Polythene;
break-resistant containers
CH2 =CHCH3 propylene polypropylene, Herculon;
textile and carpet fibers
CH2 =CHCl vinyl chloride poly(vinyl chloride), PVC;
construction tubing
CH2 =CCl2 1,1-dichloro- poly(1,1-dichloroethylene);
ethylene Saran Wrap is a copolymer
with vinyl chloride
CH2 =CHCN acrylonitrile polyacrylonitrile, Orlon;
acrylics and acrylates
CF2 = CF2 tetrafluoro- polytetrafluoroethylene, PTFE;
ethylene Teflon, nonstick coatings
CH2 =CHC6 H5 styrene polystyrene, Styrofoam; insulation
CH2 =CHCOOC2 H5 ethyl acrylate poly(ethyl acrylate); latex paints
CH2 =CCOOCH3 methyl poly(methyl methacrylate), Lucite,
CH3 methacrylate Plexiglas; glass substitutes
Polyethylene
Low-density polyethylene (LDPE):
• a highly branched polymer; polymer chains do not
pack well and London dispersion forces between
them are weak.
• softens and melts above 115°C.
• approximately 65% used for the production of films
for packaging and for trash bags.
High-density polyethylene (HDPE):
• only minimal chain branching; chains pack well and
London dispersion forces between them are strong.
• has higher melting point than LDPE and is stronger
• can be blow molded to squeezable jugs and bottles.
Codes for Plastics
Code Polymer Common Uses
1 PET poly(ethylene soft drink bottles, household
terephthalate) chemical bottles, films, textile fibers
2 HDPE high-density milk and water jugs, grocery bags,
polyethylene squeezable bottles
3 V poly(vinyl shampoo bottles, pipes, shower curtains,
chloride), PVC vinyl siding, wire insulation, floor tiles
4 LDPE low-density shrink wrap, trash and grocery bags,
polyethylene sandwich bags, squeeze bottles
5 PP polypropylene plastic lids, clothing fibers, bottle caps,
toys, diaper linings
6 PS polystyrene styrofoam cups, egg cartons, disposable
utensils, packaging materials, appliances
7 all other plastics various
You might also like
- Schaum's Easy Outline of Organic Chemistry, Second EditionFrom EverandSchaum's Easy Outline of Organic Chemistry, Second EditionRating: 3.5 out of 5 stars3.5/5 (2)
- Lesson 1 EyesDocument8 pagesLesson 1 EyesMARY JANE ANGELICA SEVANo ratings yet
- Major Aquatic Chemical ProcessesDocument4 pagesMajor Aquatic Chemical ProcessesGea DizonNo ratings yet
- Road Construction TechnologyDocument98 pagesRoad Construction Technologysuman subedi100% (4)
- Tartarus Magistery Manfred JuniusDocument10 pagesTartarus Magistery Manfred JuniusJanWillNo ratings yet
- Naming Alkenes and Alkynes Rules PDFDocument2 pagesNaming Alkenes and Alkynes Rules PDFMiguel RuizNo ratings yet
- Hydrocarbons: C.P.Garcia, PHDDocument27 pagesHydrocarbons: C.P.Garcia, PHDVictoria Gaña PangilinanNo ratings yet
- Chapter 5 NewDocument35 pagesChapter 5 NewNoor farahin bainNo ratings yet
- Chapter 5: Structure and Preparation of Alkenes: Elimination ReactionsDocument14 pagesChapter 5: Structure and Preparation of Alkenes: Elimination ReactionsKumar LinkanNo ratings yet
- AlkenesDocument65 pagesAlkenesIntresting FactsNo ratings yet
- Naming of Alkynes Based On IUPAC Rules: CH H C CH CHDocument3 pagesNaming of Alkynes Based On IUPAC Rules: CH H C CH CHikfisaini yuniarNo ratings yet
- 1 HydrocarbonsDocument38 pages1 HydrocarbonsGURPARABJOT KAURNo ratings yet
- Chapter 3 AlkenesDocument63 pagesChapter 3 AlkenesKonoli NuingNo ratings yet
- Hydrocarbon 2-Jeemain - Guru PDFDocument29 pagesHydrocarbon 2-Jeemain - Guru PDFMamta TripathiNo ratings yet
- Lecture FourDocument20 pagesLecture FourNadia JibriilNo ratings yet
- Chapter 6. Alkenes: Structure and Stability: or Add 1H To The Molecular Formula of Each HalogenDocument19 pagesChapter 6. Alkenes: Structure and Stability: or Add 1H To The Molecular Formula of Each HalogenMarilyn Castro LaquindanumNo ratings yet
- Alkenes and Alcohols Slide 2Document31 pagesAlkenes and Alcohols Slide 2Solangé HolbyNo ratings yet
- Chapter 2 AlkanesDocument77 pagesChapter 2 AlkanesFaizal ZedyNo ratings yet
- AlkenaDocument32 pagesAlkenaAdi Kurniawan EffendiNo ratings yet
- Hydrocarbon Compounds: AlkeneDocument59 pagesHydrocarbon Compounds: AlkeneUMMU MARDHIAH ABDUL HALIMNo ratings yet
- MafudDocument20 pagesMafudKesandung Duwek OINo ratings yet
- Hydrocarbon NomenclatureDocument20 pagesHydrocarbon NomenclatureAngeline CuriosoNo ratings yet
- Notes - Organic Chemistry KeyDocument21 pagesNotes - Organic Chemistry KeyJm AshiiNo ratings yet
- Class 10 Chemistry Chapter Hydrocarbons NotesDocument28 pagesClass 10 Chemistry Chapter Hydrocarbons Notesnaveedhafiz78612No ratings yet
- Chapter 3-AlkenesDocument87 pagesChapter 3-AlkenesNURUL BALQIS DZULKIFLINo ratings yet
- Sim Finished at Last!Document23 pagesSim Finished at Last!michaelNo ratings yet
- Hydrocarbon Part 2 - Nomenclature of AlkaneDocument19 pagesHydrocarbon Part 2 - Nomenclature of AlkaneShofwa AnnisaNo ratings yet
- Zumdahl Chemprin 6e CSM ch21 PDFDocument63 pagesZumdahl Chemprin 6e CSM ch21 PDFMohit ShirpurkarNo ratings yet
- Naming Organic CompoundsDocument33 pagesNaming Organic CompoundsCorpus, Irene Zen P.No ratings yet
- Naming HydrocarbonsDocument28 pagesNaming HydrocarbonsavreljeaneboncalesNo ratings yet
- Organic Chemistry Organic ChemistryDocument37 pagesOrganic Chemistry Organic ChemistryEr Ishan SharmaNo ratings yet
- DieneDocument11 pagesDieneJen EscosesNo ratings yet
- Alkenes: Organic Chemistry CHM 207Document62 pagesAlkenes: Organic Chemistry CHM 207Nur Ayu Nadhirah Bt YahyaNo ratings yet
- Chapters 7 & 8Document66 pagesChapters 7 & 8Amirabbas SaffariNo ratings yet
- Chapter 3 - Alkenes PDFDocument63 pagesChapter 3 - Alkenes PDFSITI NUR ALISSA BINTI AHMAD RASMANNo ratings yet
- Reaction and Syntesis of Alkynes: Pertemuan 13Document37 pagesReaction and Syntesis of Alkynes: Pertemuan 13Amelia ulfaNo ratings yet
- 4.2 Nomenclature of Alkenes: ProblemDocument8 pages4.2 Nomenclature of Alkenes: ProblemHao TranNo ratings yet
- 1.1 - 1.3 Alkanes, Enes, Ynes, AromaticsDocument44 pages1.1 - 1.3 Alkanes, Enes, Ynes, AromaticsTiwanka MadugalleNo ratings yet
- Consumer Chemistry: Quarter 1 - Week 5Document5 pagesConsumer Chemistry: Quarter 1 - Week 5Princess Loraine DuyagNo ratings yet
- PH 122 (PH106) Organic Chemistry I: Instructor: D. Parmena, (Document43 pagesPH 122 (PH106) Organic Chemistry I: Instructor: D. Parmena, (Hamis KaloloNo ratings yet
- Alkenes 1Document42 pagesAlkenes 1Suhaila Hanim ShaariNo ratings yet
- Al KanesDocument12 pagesAl KanesHarsh TyagiNo ratings yet
- Hydrocarbon Compounds: AlkaneDocument47 pagesHydrocarbon Compounds: AlkaneUMMU MARDHIAH ABDUL HALIMNo ratings yet
- Alkane and AlkeneDocument40 pagesAlkane and AlkenePawankumar Gupta91% (11)
- 有机化学英文命名Document113 pages有机化学英文命名HAIRIN LeeNo ratings yet
- Lectures 4-6, Term 3, AY 22-23Document53 pagesLectures 4-6, Term 3, AY 22-23LujainNo ratings yet
- Organic Compound NomenclatureDocument31 pagesOrganic Compound Nomenclaturetasneem100% (1)
- 19a Organic Chemistry 1 DetailedDocument55 pages19a Organic Chemistry 1 DetailedKarel BrionesNo ratings yet
- 5.2 Alkene 2Document6 pages5.2 Alkene 2Yusra IqbalNo ratings yet
- AlkenesDocument52 pagesAlkeneszaharanuraaNo ratings yet
- Topic 3 - AlkenesDocument16 pagesTopic 3 - AlkenesRichard WalkerNo ratings yet
- Alkene Preparation and ReactionsDocument46 pagesAlkene Preparation and Reactionsnlprofessional07No ratings yet
- Organic Chemistry: Raven Xavier N. Nuyles, RPHDocument34 pagesOrganic Chemistry: Raven Xavier N. Nuyles, RPHAIRAH SALALIMANo ratings yet
- Chapter 3.1-ALKENE PDFDocument51 pagesChapter 3.1-ALKENE PDFYasserAbyNo ratings yet
- Alkanes, Alkenes and AlkynesDocument85 pagesAlkanes, Alkenes and AlkynesYoichi Kho100% (1)
- Organic Chemistry: Topic 10.1.1 - 10.1.8Document40 pagesOrganic Chemistry: Topic 10.1.1 - 10.1.8Agustín CastroNo ratings yet
- Full Organic Chemistry Flow Charts and Brief TheoryDocument214 pagesFull Organic Chemistry Flow Charts and Brief Theorykoradasirisha2007No ratings yet
- Alkanes and CycloalkanesDocument63 pagesAlkanes and Cycloalkanesayunna ayunniNo ratings yet
- Organic ChemistryDocument87 pagesOrganic ChemistryPhil De GuzmanNo ratings yet
- Chapter 2-AlkanesDocument63 pagesChapter 2-AlkanesNURUL BALQIS DZULKIFLINo ratings yet
- 2intro Org PDFDocument87 pages2intro Org PDFSarah SarahNo ratings yet
- AlkyneDocument25 pagesAlkyneaditya.sachanNo ratings yet
- 2018 H2 Alkenes - Lecture - TR - FinalDocument26 pages2018 H2 Alkenes - Lecture - TR - FinalJoshua LeongNo ratings yet
- Strained Organic Molecules: Organic Chemistry: A Series of Monographs, Vol. 38From EverandStrained Organic Molecules: Organic Chemistry: A Series of Monographs, Vol. 38No ratings yet
- Lesson 2 ScalpDocument8 pagesLesson 2 ScalpMARY JANE ANGELICA SEVANo ratings yet
- Chess: Setting Up The Chess Board Check and CheckmateDocument2 pagesChess: Setting Up The Chess Board Check and CheckmateMARY JANE ANGELICA SEVANo ratings yet
- Lesson 2 Different Types of SportsDocument2 pagesLesson 2 Different Types of SportsMARY JANE ANGELICA SEVANo ratings yet
- Lesson 1 Definition & History of Sports and Components of Physical FitnessDocument3 pagesLesson 1 Definition & History of Sports and Components of Physical FitnessMARY JANE ANGELICA SEVANo ratings yet
- Lesson 1234 - EthicsDocument3 pagesLesson 1234 - EthicsMARY JANE ANGELICA SEVANo ratings yet
- Lesson 4 Aldehydes and KetoneDocument4 pagesLesson 4 Aldehydes and KetoneMARY JANE ANGELICA SEVANo ratings yet
- Lesson 2 Upper ExtremitiesDocument23 pagesLesson 2 Upper ExtremitiesMARY JANE ANGELICA SEVANo ratings yet
- Lesson 1 Carboxylic AcidsDocument5 pagesLesson 1 Carboxylic AcidsMARY JANE ANGELICA SEVANo ratings yet
- Lesson 3 Nervous SystemDocument11 pagesLesson 3 Nervous SystemMARY JANE ANGELICA SEVANo ratings yet
- Lesson 2 Endocrine SystemDocument18 pagesLesson 2 Endocrine SystemMARY JANE ANGELICA SEVANo ratings yet
- Vpi DatasheetDocument1 pageVpi DatasheetMihir RanaNo ratings yet
- Creation of Mesostructured Hollow Y Zeolite by Selective.2013Document13 pagesCreation of Mesostructured Hollow Y Zeolite by Selective.2013Cecilia ManriqueNo ratings yet
- Agua para EspressoDocument5 pagesAgua para EspressoManuel MoraNo ratings yet
- ManualDocument278 pagesManualAnnn SoooNo ratings yet
- Alfaflex - Alfaflat MDocument1 pageAlfaflex - Alfaflat MMoussa BANo ratings yet
- Finger Print Region in IR SpectrosDocument3 pagesFinger Print Region in IR SpectrosRinku KunduNo ratings yet
- 8.T-MEET315 Fundamentals MatScie Engg Module 8Document26 pages8.T-MEET315 Fundamentals MatScie Engg Module 8Cj TilamNo ratings yet
- ChECal SolutionsDocument39 pagesChECal SolutionsMARGARET FLORESNo ratings yet
- The First Experiment I Will Do Is Evaporative Cooling. The Supplies Needed AreDocument3 pagesThe First Experiment I Will Do Is Evaporative Cooling. The Supplies Needed AreMarinelle R. EumagueNo ratings yet
- Tipical Solvent For Cementing ThermoplasticsDocument1 pageTipical Solvent For Cementing ThermoplasticsDamiano CrestanNo ratings yet
- JEE Lecture Plan - 12th PackageDocument3 pagesJEE Lecture Plan - 12th PackageAman KumarNo ratings yet
- Hci 2013 P1Document7 pagesHci 2013 P120Y3G HONG JIN KAINo ratings yet
- USP-NF AlcoholDocument4 pagesUSP-NF AlcoholOIL TEST INTERNACIONAL DE COLOMBIA S.A.SNo ratings yet
- Ndoro Corrected DissertationDocument78 pagesNdoro Corrected DissertationOnesime Muteba100% (1)
- Fun Nemonics in ChemDocument3 pagesFun Nemonics in ChemSuresh BabuNo ratings yet
- Phan111lec FinalsDocument34 pagesPhan111lec FinalsANDREA ROSELLE FULLONANo ratings yet
- Evaluation of The Effectiveness of Local Clay From Ebonyi State Nigeria As A Substitute For Bentonite in Drilling FluidsDocument10 pagesEvaluation of The Effectiveness of Local Clay From Ebonyi State Nigeria As A Substitute For Bentonite in Drilling FluidsShadrachNo ratings yet
- Antidiabetic and Hypolipidemic Effects of MahanimbDocument10 pagesAntidiabetic and Hypolipidemic Effects of MahanimbTri WiyonoNo ratings yet
- 18-F1927.29667-1 OtrDocument6 pages18-F1927.29667-1 OtrBruna Elias100% (1)
- Chem 16 Finals SamplexDocument3 pagesChem 16 Finals SamplexKayeNo ratings yet
- CT - Isomerism - Isomerism - 03042021 - Isomerism - Practice Sheet 1 To 4Document15 pagesCT - Isomerism - Isomerism - 03042021 - Isomerism - Practice Sheet 1 To 4Anita Akhilesh YadavNo ratings yet
- Physioex Lab Report: Pre-Lab Quiz ResultsDocument4 pagesPhysioex Lab Report: Pre-Lab Quiz Results-No ratings yet
- Chem Lab - Student - Week 1 - Chemical Reactions - v2Document34 pagesChem Lab - Student - Week 1 - Chemical Reactions - v2Đặng Quỳnh NhưNo ratings yet
- Permeation Enhancer Strategies in Transdermal Drug DeliveryDocument16 pagesPermeation Enhancer Strategies in Transdermal Drug DeliveryADVOCATE ASHUTOSH SHARMANo ratings yet
- 1 s2.0 S0039914023007051 MainDocument12 pages1 s2.0 S0039914023007051 MainAARON BENJAMIN QUISPE ROMERONo ratings yet
- Evaluating The Effect of Varying Drying Air Temperatures On Quality Attributes of Avocado (Persea Americana) PeelsDocument7 pagesEvaluating The Effect of Varying Drying Air Temperatures On Quality Attributes of Avocado (Persea Americana) PeelsMamta AgarwalNo ratings yet
- UG100 Single Seals: Standard Mechanical Seals - Elastomer Bellows SealsDocument2 pagesUG100 Single Seals: Standard Mechanical Seals - Elastomer Bellows SealsLuis BassoNo ratings yet