Professional Documents
Culture Documents
Mesenchymal Stem Cell Therapeutic Intervention in Covid19
Mesenchymal Stem Cell Therapeutic Intervention in Covid19
Uploaded by
Bala BhaskarOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Mesenchymal Stem Cell Therapeutic Intervention in Covid19
Mesenchymal Stem Cell Therapeutic Intervention in Covid19
Uploaded by
Bala BhaskarCopyright:
Available Formats
Internal Medicine: Open Access Research Article
Mesenchymal Stem Cell: Therapeutic Intervention in Covid-19
Saurabh Kumar Jha1*, Parma Nand1, Niraj Kumar Jha1, Rashi Srivastava1,2, Ankur Sharma3 ,Harshika
Varshney 3, Anand Prakash 4 , Kumari Swati 4,5, Dhruv Kumar5
1Department of Biotechnology, Sharda University, Greater Noida, India; 2Department of Biotechnology, Dr. A.P.J. Abdul Kalam
Technical University, Lucknow, India; 3Department of Life Science, Sharda University, Greater Noida, India; 4 Department of
Biotechnology, Mahatma Gandhi Central University, Motihari, Bihar, India; 5Department of Molecular Medicine and Stem Cell
Research, Amity University, Uttar Pradesh, Noida, India
ABSTRACT
COVID-19, an ephialtes of mankind in the year 2020, is causing painful demise of many lives due to insufficient
treatment and hence, creating a devastating history in making. This pandemic brought the world together and led
many researches from different biomedical fields to find treatments and potent drugs; ought to help in managing the
pandemic and future loss. However, till date no standard treatment has been discovered for COVID-19. Probably,
preventing the severe respiratory infection cause by SARS-CoV-2 will give time for finding the most potent treatment
for this disease. Easy and manageable methods are there for isolation of Mesenchymal Stem Cells (MSCs) which
attracted the attention for number of therapeutic applications. Considering the present situation, treatment using
Mesenchymal Stem Cells (MSCs) has been suggested as a satisfactory therapeutic approach. A noteworthy population
of cells accumulates in the lungs after intravenous transplantation of MSCs which results in increasing the
immunomodulatory effect that leads the protection of alveolar epithelial cells, refurbishment of the pulmonary
microenvironment, and, prevent lung dysfunction and pulmonary fibrosis. There are several ongoing clinical trials
based on MSCs for exploring out its efficiency in the treatment of COVID-19 patients. However, till date MSCs
based treatment for COVID-19 is still not approved. This review involves the aspects of MSCs in the proposed
treatment of COVID-19 and contains brief discussion on related ongoing clinical trials.
Keywords: Mesenchymal Stem Cell (MSCs); COVID-19; Clinical trials; Immunomodulatory
INTRODUCTION no widespread infections were found in wild or farmed civets
The announced pandemic of [1] COVID-19 (Coronavirus and clinical symptoms of SARS were only overt in the
Disease 2019), broke out with numerous cases at the end of 2019 municipalities sold on the markets, suggesting that civets were
in Wuhan, China exhibiting serious respiratory dysfunctions. It unlikely to have been the primary/natural reservoir host [3,4].
was initially identified for infections, and was supposed to be a Now, the virus has been narrowly called SARS-CoV-2, the name
seasonal flu outbreak by considering those patients who have was designated by the World Health Organization (WHO) [5].
background of work in wholesale market of shrimp and fish. A With the help of several samples, phylogenetic and sequencing
few days back, after the rejection of Seasonal grippe infection, analysis were done which reports that it belongs to coronavirus
avian influenza, adenovirus, SARS, and other pathogens. This family (Orthocoronavirinae subfamily) [6] in Betacoronavirus-2B
novel coronavirus shows 5% genetic correlation with SARS and lineage and shows 96% identity with the bat SARS-CoV (Bat
is a subclass of Sarbecovirus [2]. Since SARS-CoV is zoonotic in Cov RaTG13) [7]. The virus, use its S1 and S2 subunits of
origin, this has resulted in the tentative mass culling of the hetero-trimeric spike proteins to interact with the human cell
masked palm civets. However, further studies later revealed that receptor, Angiotensin-Converting Enzyme 2 (ACE2) [8], to infect
Correspondence to: Saurabh Kumar Jha, Department of Biotechnology, Sharda University, Greater Noida, India, E-mail: saurabh.jha@sharda.ac.in
Received: December 17, 2020; Accepted: December 31, 2020; Published: January 07, 2021
Citation: Varshney H, Srivastava R, Swati K, Sharma A, Jha SK, Kumar D, et al. (2021) Mesenchymal Stem Cell: Therapeutic Intervention in
Covid-19. Intern Med. 11:329.
Copyright: © 2021 Varshney H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Intern Med , Vol.11 Iss.1 No:329 1
Varshney H, et al.
a host cell. SARS-CoV-2 infect lungs (Figure 1) by causing-
angiocentric inflammation, thrombosis and micro-angiopathy,
and angiogenesis [9].
This pandemic influenced all the clinicians and researchers
worldwide to gather from various branches of biomedicines for
seeking a plan or therapy/vaccine to handle the pandemic [10].
Most of the current preclinical and clinical data on antiviral
therapy are available from other viruses, including SARS-CoV-1
(first reported in 2003), Middle East Respiratory Syndrome
Coronavirus (MERS-CoV), (recorded for the first time in 2012)
and non-coronavirus (example- outbreak of the Ebola virus). It is
not obvious about how efficiently these details can be
extrapolated to SARS-CoV-2. In fact, scientific importance of in
vitro antiviral behavior defined as semi-maximal Efficacy
Concentration [EC50] values appears undefined, given in the
absence of pharmacokinetics/pharmacodynamics or surgical
evidence. This equalizes the workable concentrations compared Figure 2: Schematic representation of action of MSCs in SARS-
to these levels to an effect of treatment [11]. Diagnosis is also CoV-2 life cycle.
challenging as a number of studies from Singapore [12] and
METHODOLOGY
Germany [13], inferred about the asymptomatic transmission of
SARS-CoV-2 by viral shedding. Morphology of SARS-COV-2
From various published literatures one can infer that the SARS-CoV-2 is a spherical or pleomorphic enveloped particles
Mesenchymal Stem Cells (MSCs) are active against viral with club-shaped glycoprotein projections [15]. SARS-CoV-2 was
infection and these are attributable in the involvement of assessed for its size ranging between 70-90 nm, observed under
specific cytokines improved the efficiency [14]. Therefore, MSCs electron microscope which also suggested its wide variety of
should be presumed to thrive even though they are transplanted intracellular organelles [16]. As it shows high sequence similarity
to a patient reported for COVID-19. As, there is a discrepancy with SARS-CoV, the structure of SARS-CoV-2 is speculated to
in the MSCs therapy for the diagnosis of COVID-19, we have be the same as SARS-CoV [17]. It is found that the envelope,
integrated the clinical literature and trials to convey valuable membrane, and spike protein of coronavirus are embedded in
knowledge in the review. The review provides a deep insight of host membrane derived lipid bilayer which encapsulates the
the role of MSCs in the disease and representation of action of helical nucleocapsid comprising viral RNA [18] illustrated in
MSCs in SARS-CoV-2 life cycle illustrated in (Figure 2). It is (Figure 3). The single stranded RNA is approximately 30 kb long
necessary to alert readers that new data appears approximately having positive polarity with 5’ cap and 3’ Poly-A tail [19].
every hour considering the Medical conditions, care, options to Almost all structures of protease [20] and spike [21] of SARS-
prevent and outcomes of COVID-19. CoV-2 have been resolved which leads an opportunity to
develop or repurpose the drugs for treating COVID-19.
Figure 3: Structure of SARS-CoV-2.
Figure 1: Effect of SARS-CoV-2 on lungs.
Intern Med , Vol.11 Iss.1 No:329 2
Varshney H, et al.
foreseen [29]. MSCs have drawn interests owing to the possible
source, high rate of proliferation, minimal intrusive treatment
and clear of ethical problems. MSC rehabilitation is significantly
better as applied to other therapies [30]. They are widely
available and can be isolated from different tissues, such as bone
marrow and adipose tissues, including the umbilical cord, dental
pulp, menstrual blood, buccal fat pad, fetal liver and others.
MSCs can expand efficiently to clinical volume in adequate time
period and for repeated diagnostic utilization it can be
processed. Moreover, MSC therapy have the potential to prevent
the storm of cytokines and can advance the endogenous repair
by its regenerative and reparative property [31,32] illustrated in
(Figure 4). MSCs therapy has been commonly employed in the
diagnosis and treatment of type 2 diabetes, autoimmune
disorder, spinal cord damage, GVHD and many other disorders,
with strong antibody levels [33-36]. Although MSCs do not have
Figure 4: Proposed role of Mesenchymal stem cells. clear anti-viral effects, studies have shown that they are immune
to viral interaction by the presence of Interferon Gamma
Therapeutic intervention of COVID-19 Stimulated Genes (ISGs) found in them prior to differentiation.
As per 31 August, 2020; neither any drugs nor any biologics MSCs frequently control our body's adaptive and innate
is/are established to be efficient for the prevention/treatment of immune systems by destroying T-cells, maturing dendritic cells,
COVID-19 infection. Many antivirals, parasitic drug decreasing activation and proliferation of B-cells, and hindering
combinations, immunotherapies, and vaccines are under trials NK cell proliferation and cytotoxicity by soluble factors or cell-
and their efficacy against COVID-19 is still dilemmatic. The to-cell pathways (Figure 3) [31,32]. Recently, [37] the use of these
only mode of treatment, widely in use for COVID-19 is cells in the therapeutic diagnosis of H5N1 viral infections with
repurposing of drugs [22]. Drugs like Chloroquine and related consequences on the lung has also been proposed [38].
Hydroxychloroquine [23], Mefloquine [24], certain flavonoid Thus their role in regulating immune malfunctioning is widely
compounds [25], Lopinavir [26], Remdesivir [27], Quercetin [28] reported and can be extrapolated as an eminent tool for
and other reported drugs are currently in use to treat COVID-19 COVID-19 research and treatment.
patients.
Mesenchymal stem cells therapy MSCs related clinical trials for COVID-19
Amidst the COVID-19 rush for various drugs like There are total 3201 clinical studies registered on
Hydroxychloroquine and Remdesivir etc. and various vaccines, ClinicalTrials.gov [39] for treating COIVD-19 out of which only
the US Food and Drug Administration (FDA) have recently 54 studies are related with MSCs. All 54 clinical studies’ title
approved the use of MSCs for coronavirus treatment under the and their status are tabulated in Table 1. In the related trials,
discretion of expanded accesses. At present, cell-based therapy one study is withdrawn and fourteen studies are not recruited
and stem cell therapy in particular has been a remarkable yet. However, one study has been completed in china by using
medical domain in which potential to cure incurable diseases is umbilical cord derived Mesenchymal Stem Cells (MSCs) but
there results are not posted yet.
RESULTS AND DISCUSSION
Sl.No. Trial no Title of the study Place Intervention Status Phase
Clinical research of human
1 NCT04339660 mesenchymal stem cells in the China Biological: UC-MSCs Recruiting I and II
treatment of COVID-19 pneumonia. Other: Placebo
Biological:
Autologous Adipose-derived Stem
2 NCT04428801 - Autologous adipose Not yet recruiting II
Cells (Ad-MSCs) for COVID-19.
derived stem cells
Mesenchymal Stem Cell (MSC)
3 NCT04252118 treatment for pneumonia patients China Biological: MSCs Recruiting I
infected with COVID-19.
Treatment of severe COVID-19
Biological:
pneumonia with Allogeneic
4 NCT04361942 Spain Mesenchymal Stromal Recruiting II
Mesenchymal Stromal Cells
Cells
(COVID-MSC).
Other: Placebo
ACT-20 in patients with severe Biological: ACT-20-
5 NCT04398303 - Not yet recruiting I and II
COVID-19 pneumonia. MSC
Biological: ACT-20-
CM
Biological: Placebo
Intern Med , Vol.11 Iss.1 No:329 3
Varshney H, et al.
Mesenchymal Stem Cell (MSC) Drug: Mesenchymal
6 NCT04444271 Pakistan Recruiting II
infusion for COVID-19 infection. stem cells
Other: Placebo
Efficacy and safety study of allogeneic
Active, not
7 NCT04362189 HB-adMSCs for the treatment of United States Drug: HB-adMSC -
recruiting
COVID-19. Drug: Placebo
Safety and efficacy of Mesenchymal Biological: Umbilical
Stem Cells (MSCs) in the cord derived
8 NCT04429763 - Not yet recruiting II
management of severe COVID-19 mesenchymal stem
pneumonia. cells
Biological: Placebo
Biological:
Mesenchymal Stromal
Cells infusion
Safety and feasibility of allogeneic
9 NCT04467047 - Intravenous 1*10E6 Not yet recruiting -
MSC in the treatment of COVID-19.
MSCs/kg body weight
Mesenchymal Stromal
Cells infusion
Safety and efficacy study of allogeneic Biological: Allogeneic
human dental pulp mesenchymal human dental pulp
10 NCT04336254 China Recruiting I and II
stem cells to treat severe COVID-19 stem cells (BSH, BTC
patients. & tooth BTC)
Other: Intravenous
saline injection
(Placebo)
Treatment of COVID-19 patients
11 NCT04313322 using Wharton’s jelly-mesenchymal Jordan Biological: WJ-MSCs Recruiting I
stem cells.
A randomized, double blind, placebo
controlled clinical trial to determine
the safety and efficacy of Hope
Enrolling by
12 NCT04348435 Biosciences Allogeneic Mesenchymal United States Drug: HB-adMSCs -
invitation
Stem Cell Therapy (HB-adMSCs) Drug: Placebos
to provide protection against
COVID-19.
Procedure: Placenta
derived MMSCs;
Treatment of coronavirus COVID-19
cryopreserved
pneumonia (pathogen SARS-CoV-2)
13 NCT04461925 Ukraine placenta derived Recruiting -
with cryopreserved allogeneic
multipotent
P-MMSCs and UC-MMSCs.
mesenchymal stromal
cells
Drug: Antibiotics
Drug: Hormones (and
2 more)
NestaCell® Mesenchymal Stem
Biological:
14 NCT04315987 Cell to treat patients with severe Brazil Not yet recruiting II
NestaCell®
COVID-19 pneumonia.
Biological: Placebo
Study of human umbilical cord
15 NCT04273646 mesenchymal stem cells in the China Biological: UC-MSCs Not yet recruiting -
treatment of severe COVID-19. Drug: Placebo
Intern Med , Vol.11 Iss.1 No:329 4
Varshney H, et al.
Bone marrow-derived mesenchymal
stem cell treatment for severe
16 NCT04346368 China Biological: BM-MSCs Not yet recruiting -
patients with coronavirus disease
2019 (COVID-19). Biological: Placebo
Intermediate size Expanded Access
Program (EAP), Mesenchymal
17 NCT04366830 Stromal Cells (MSC) for Acute United states Drug: Remestemcel-L No longer available -
Respiratory Distress Syndrome
(ARDS) due to COVID-19 infection.
Treatment with human umbilical
cord-derived mesenchymal stem cells
18 NCT04288102 China Biological: UC-MSCs Completed II
for severe coronavirus disease 2019
(COVID-19). Biological: Saline
containing 1%
human serum
albumin (solution
without UC-MSCs)
Intermediate size Expanded Access
Program (EAP), Mesenchymal
Stromal Cells (MSC) for Multisystem Biological:
19 NCT04456439 - Available -
Inflammatory Syndrome in Children Remestemcel-L
(MIS-C) associated with coronavirus Drug: Hydrocortisone
disease (COVID-19).
Therapy for pneumonia patients
20 NCT04293692 - Biological: UC-MSCs Withdrawn -
infected by 2019 novel coronavirus.
Other: Placebo
A clinical trial to determine the safety
and efficacy of Hope Biosciences
Enrolling by
21 NCT04349631 Autologous Mesenchymal Stem Cell United States Drug: HB-adMSCs II
invitation
Therapy (HB-adMSCs) to provide
protection against COVID-19.
Mesenchymal Stem Cell for Acute Biological: Infusion
22 NCT04416139 Respiratory Distress Syndrome Mexico IV of Mesenchymal Recruiting II
(ARDS) due for COVID-19. Stem cells
Cellular immuno-therapy for
Biological:
COVID-19 Acute Respiratory
23 NCT04400032 Canada Mesenchymal Stromal Not yet recruiting I
Distress Syndrome (ARDS)-
Cells
Vanguard.
Clinical trial of allogeneic
mesenchymal cells from umbilical Biological:
24 NCT04366271 Spain Recruiting II
cord tissue in patients with Mesenchymal cells
COVID-19. Drug: Standard of
care
Study of the safety of therapeutic
Tx with immunomodulatory MSC Biological: BM-Allo
25 NCT04397796 United States Recruiting I
in adults with COVID-19 infection MSC
requiring mechanical ventilation. Biological: Placebo
Mesenchymal Stem Cells (MSCs) Procedure: Biological
in inflammation-resolution Germany samples specific to Recruiting II
programs of coronavirus disease research
26 NCT04377334
2019 (COVID-19) Induced Acute
Procedure: Clinical
Respiratory Distress Syndrome
examination
Procedure: Telephone
follow-up
Intern Med , Vol.11 Iss.1 No:329 5
Varshney H, et al.
Adipose mesenchymal cells
Biological:
for abatement of SARS-CoV-2
27 NCT04352803 - Autologous Adipose Not yet recruiting I
respiratory compromise in
MSCs
COVID-19 disease.
MSC based therapy in COVID-19 Other: Mesenchymal
28 NCT04525378 associated Acute Respiratory Distress Brazil stromal cell-based Recruiting I
Syndrome (ARDS). therapy
Efficacy and safety evaluation of
mesenchymal stem cells for the Drug: XCEL-UMC-
29 NCT04390139 Spain Recruiting I and II
treatment of patients with respiratory BETA
distress due to COVID-19. Other: Placebo
Biological:
Use of mesenchymal stem cells in Mesenchymal Stem
Active, not
30 NCT04456361 acute respiratory distress syndrome Mexico Cells derived from Early I
recruiting
caused by COVID-19. Wharton jelly of
umbilical cords
Biological: Human
Use of hUC-MSC product umbilical cord
31 NCT04452097 (BX-U001) for the treatment of - mesenchymal stem Not yet recruiting I
COVID-19 with ARDS. cells + best supportive
care
Biological: umbilical
cord mesenchymal
Use of UC-MSCs for COVID-19 Active, not
32 NCT04355728 United States stem cells + heparin I and II
patients. recruiting
along with best
supportive care
Other: Vehicle +
heparin along with
best supportive care
Umbilical Cord Tissue (UCT)
Derived Mesenchymal Stem Cells
33 NCT04490486 (MSCs) versus placebo to treat acute United States Biological: UCMSCs Not yet recruiting I
pulmonary inflammation due to Other: Placebo
COVID-19.
Mesenchymal Stromal Cells (MSCs)
Biological:
for the treatment of SARS-CoV-2
34 NCT04345601 United States Mesenchymal stromal Not yet recruiting Early I
induced acute respiratory failure
cells
(COVID-19 disease).
Other: Supportive
care
Clinical trial, multicenter,
randomized and controlled, to assess Drug: Allogeneic
the safety and efficacy of intravenous and expanded
35 NCT04366323 administration of allogeneic adult Spain adipose tissue-derived Recruiting I and II
mesenchymal stem cells of expanded mesenchymal stem
adipose tissue in patients with severe cells
pneumonia due to COVID-19.
Administration of allogenic UC- Drug: Oseltamivir Recruiting I
36 NCT04457609 MSCs as adjuvant therapy for Indonesia
Drug: Azithromycin
critically-ill COVID-19 patients.
Biological: Umbilical
cord mesenchymal
stem cells
Biological: Human
cord tissue
37 NCT04399889 hCT-MSCs for COVID19 ARDS. United States Recruiting I and II
mesenchymal stromal
cells
Intern Med , Vol.11 Iss.1 No:329 6
Varshney H, et al.
Drug: Allogeneic
and expanded
Battle against COVID-19 using
38 NCT04348461 - adipose tissue derived Not yet recruiting II
Mesenchymal Stromal Cells (MSCs).
mesenchymal stromal
cells
Mesenchymal stem cell therapy for
acute respiratory distress syndrome Biological: Cell
39 NCT04366063 Iran Recruiting II and III
in coronavirus infection: a phase 2-3 therapy protocol 1
clinical trial. Biological: Cell
therapy protocol 2
Multiple dosing of mesenchymal Biological:
40 NCT04466098 stromal cells in patients with ARDS United States Mesenchymal stromal Recruiting II
(COVID-19). cells
Other: Placebo
Biological:
41 NCT04371393 MSCs in COVID-19 ARDS. United States Recruiting III
Remestemcel-L
Drug: Placebo
Novel coronavirus induced severe Biological: Dental
42 NCT04302519 pneumonia treated by dental pulp - pulp mesenchymal Not yet Recruiting Early I
mesenchymal stem cells. stem cells
Safety and effectiveness of Active, Not
Drug: Oseltamivir Early I
mesenchymal stem cells in the Recruiting
43 NCT04371601 China Drug: Hormones
treatment of pneumonia of
coronavirus disease 2019. Device: Oxygen
therapy
Procedure:
Mesenchymal stem
cells
An exploratory study of adr-001 in Biological:
44 NCT04522986 patients with severe pneumonia Japan Mesenchymal stem Not yet recruiting I
caused by SARS-CoV-2 infection. cell
Biological: Allogenic
Treatment of Covid-19 associated
pooled olfactory
pneumonia with allogenic Enrolling by
45 NCT04382547 Belarus mucosa derived I and II
pooled olfactory mucosa-derived invitation
mesenchymal stem
mesenchymal stem cells.
cells
Other: Standard
treatment according
to the Clinical
protocols
Cell therapy using umbilical cord- Biological: Umbilical
46 NCT04333368 derived mesenchymal stromal cells in France cord Wharton's jelly- Recruiting I and II
SARS-CoV-2-related ARDS. derived human
Other: NaCl 0.9%
Repair of acute respiratory Biological: Human
distress syndrome by stromal umbilical cord derived
47 NCT03042143 United Kingdom Recruiting I and II
cell administration (REALIST) CD362 enriched
(COVID-19). MSCs
Biological: Placebo
(Plasma-Lyte 148)
Clinical use of stem cells for the Biological: MSC
48 NCT04392778 Turkey Recruiting I and II
treatment of Covid-19. treatment
Biological: Saline
control
Intern Med , Vol.11 Iss.1 No:329 7
Varshney H, et al.
A study to collect bone marrow for Procedure: Bone
process development and production marrow harvest
49 NCT04397471 United Kingdom Not yet recruiting -
of BM-MSC to treat severe healthy volunteer
COVID-19 pneumonitis. bone marrow harvest
Umbilical Cord (UC) derived
Mesenchymal Stem Cells (MSCs)
50 NCT04269525 China Biological: UC-MSCs Recruiting II
treatment for the 2019 Novel
Coronavirus (nCOV) pneumonia.
Drug: Wharton's jelly
Safety and efficacy of intravenous derived mesenchymal Not yet recruiting I and II
Wharton's jelly derived mesenchymal stem cells.
51 NCT04390152 Colombia Drug:
stem cells in acute respiratory distress
syndrome due to COVID-19. Hydroxychloroquine,
Lopinavir/Ritonavir
or Azithromycin and
Placebo (standard
therapy)
Biological:
Mesenchymal stromal cell therapy for
52 NCT04445454 Belgium Mesenchymal stromal Recruiting I and II
severe Covid-19 infection.
cells
Mesenchymal stromal cell therapy Drug: Mesenchymal
53 NCT04447833 for the treatment of acute respiratory Sweden stromal stem cells - KI- Recruiting I
distress syndrome. MSC-PL-205
A pilot clinical study on inhalation
of mesenchymal stem cells exosomes Biological: MSCs-
54 NCT04276987 - Not yet recruiting I
treating severe novel coronavirus derived exosomes
pneumonia.
Table 1: MSCs based clinical trials for treating COVID-19.
Being free from vital ethical concerns with low risk of teratoma 3. Hilgenfeld R. From SARS to MERS: Crystallographic studies on
coronaviral proteases enable antiviral drug design. FEBS J.
formation, MSCs gains the status of most commonly used stem 2014;281(18):4085-4096.
cells in cell therapy. Moreover, their regenerative capacity
immunomodulatory effect, anti-inflammatory effect as well as 4. Cui J, Li F, Shi ZL. Origin and evolution of pathogenic coronaviruses.
handling the cytokines storm have made MSCs a potent Nat Rev Microbiol. 2019;17(3):181-192.
candidate for preclinical and human clinical trials for viral 5. Naming the coronavirus disease (COVID-19) and the virus that causes
treatment. However, certain limitations resides in MSC-based it, World Health Organization, 2020.
therapies should never be neglected. The heterogeneity nature 6. Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, et al. A novel
coronavirus from patients with pneumonia in China, 2019. N Engl J
of MSCs might bring the inconsistency in research results. With
Med. 2020;382(8):727-733.
recent literature it is found that MSCs may not be
immunologically silent as assumed previously [40]. 7. Aylward B, Liang W (PRC). Report of the WHO-China Joint Mission
on Coronavirus Disease 2019 (COVID-19). 2020;16-24.
CONCLUSION 8. Wong SK, Li W, Moore MJ, Choe H, Farzan M. A 193-amino acid
fragment of the SARS coronavirus S protein efficiently binds
To overcome the residing limitations, use of cell-free therapeutic angiotensin-converting enzyme 2. J Biol Chem. 2004;279(5):
strategies such as MSC-exosomes should be focused more. There 3197-3201.
are limited clinical trials and study of MSCs on virus-associated 9. Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T,
diseases and in the case of SARS CoV-2 these investigations are Laenger F, et al. Pulmonary vascular endothelialitis, thrombosis, and
angiogenesis in Covid-19. N Engl J Med. 2020;383:120-128.
still not completed. Hence, the efficacy and potency of MSCs
the treatment of COVID-19 cannot be concluded at this time. 10. Metcalfe SM. Mesenchymal stem cells and management of COVID-19
pneumonia. Medicine in Drug Discovery.2020;100019.
This review highlighted the MSCs therapy application and
clinical trials for COVID-19. 11. Stevens MP, Patel PK, Nori P. Involving antimicrobial stewardship
programs in COVID-19 response efforts: All hands on deck. Infect
REFERENCES Control Hosp Epidemiol. 2020;41(6):744-745.
12. Wei WE, Li Z, Chiew CJ, Yong SE, Toh MP, Lee VJ. Presymptomatic
1. World Health Organization W. WHO Director-General’s opening
Transmission of SARS-CoV-2-Singapore, January 23-March 16, 2020.
remarks at the media briefing on COVID-19-11 March 2020. WHO
Morb Mortal Wkly Rep. 2020;69(14):411.
Director General’s speeches. 2020.
13. Rothe C, Schunk M, Sothmann P, Bretzel G, Froeschl G, Wallrauch
2. Wu F, Zhao S, Yu B, Chen YM, Wang W, Song ZG, et al. A new
C, et al. Transmission of 2019-nCoV infection from an asymptomatic
coronavirus associated with human respiratory disease in China.
contact in Germany. N Engl J Med. 2020;382(10):970-971.
Nature. 2020;579(7798):265-269.
Intern Med , Vol.11 Iss.1 No:329 8
Varshney H, et al.
14. Blau HM, Daley GQ. Stem cells in the treatment of disease. N Engl J 28. Colunga Biancatelli RM, Berrill M, Catravas JD, Marik PE. Quercetin
Med. 2019;380(18):1748-1760. and vitamin C: An experimental, synergistic therapy for the
prevention and treatment of SARS-CoV-2 related disease (COVID-19).
15. Tyrrell DAJ, Myint SH. Coronaviruses. Medical Microbiology. 4th ed. Front Immunol. 2020;11:1451.
1996.
29. Golchin A, Farahany TZ. Biological products: Cellular therapy and
16. Park WB, Kwon NJ, Choi SJ, Kang CK, Choe PG, Kim JY, et al. Virus FDA approved products. Stem Cell Rev. 2019;15(2):166-175.
isolation from the first patient with SARS-CoV-2 in Korea. J Korean 30. Golchin A, Farahany TZ, Khojasteh A, Soleimanifar F,
Med Sci. 2019;35(7):e84. Ardeshirylajimi A. The clinical trials of mesenchymal stem cell therapy
17. Kumar S, Maurya VK, Prasad AK, Bhatt MLB, Saxena SK. Structural, in skin diseases: An update and concise review. Curr Stem Cell Resear
glycosylation and antigenic variation between 2019 novel coronavirus Ther. 2019;14(1):22-33.
(2019-nCoV) and SARS coronavirus (SARS-CoV) Virus Dis. 31. Weiss AR, Dahlke MH. Immunomodulation by mesenchymal stem
2020;31(1):13-21. cells (MSCs): Mechanisms of action of living, apoptotic, and dead
MSCs. Front Immunol. 2019;10:1191.
18. Finlay BB, See RH, Brunham RC. Rapid response research to
emerging infectious diseases: Lessons from SARS. Nat Rev Microbiol. 32. Glenn JD, Whartenby KA. Mesenchymal stem cells: Emerging
2004;2(7):602-607. mechanisms of immunomodulation and therapy. World J Stem Cells.
2014;6(5):526-539.
19. Woo PC, Huang Y, Lau SK, Yuen KY. Coronavirus genomics and
bioinformatics analysis. Viruses. 2010;2(8):1804-1820. 33. Liang B, Chen J, Li T, Wu H, Yang W, Li Y, et al. Clinical remission of
a critically ill COVID-19 patient treated by human umbilical cord
20. Zhang L, Lin D, Sun X, Curth U, Drosten C, Sauerhering L, Becker S,
mesenchymal stem cells: A case report. Medicine. 2020;99(31):e21429.
Rox K, Hilgenfeld R. Crystal structure of SARS-CoV-2 main protease
provides a basis for design of improved α-ketoamide inhibitors. 34. Golchin A, Shams F, Karami F. Advancing Mesenchymal stem cell
Science. 2020;368(6489):409-412. therapy with CRISPR/Cas9 for clinical trial studies. Cell Biol
Translational Med. 2019;8:89-100.
21. Yan R, Zhang Y, Li Y, Xia L, Guo Y, Zhou Q. Structural basis for the
recognition of SARS-CoV-2 by full-length human ACE2. Science. 35. Zhao K, Liu Q. The clinical application of mesenchymal stromal cells
2020;367(6485):1444-1448. in hematopoietic stem cell transplantation. J Hematol Oncol.
2016;9(1):46.
22. Sun W, Sanderson PE, Zheng W. Drug combination therapy increases
successful drug repositioning. Drug Discov Today. 2016;21(7): 36. Novello S, Debouche A, Philippe M, Naudet F, Jeanne S. Clinical
1189-1195. application of mesenchymal stem cells in periodontal regeneration: A
systematic review and meta‐analysis. J Periodontal Res. 2020;55(1):
23. Colson P, Rolain JM, Lagier JC, Brouqui P, Raoult D. Chloroquine
1-12.
and hydroxychloroquine as available weapons to fight COVID-19. Int J
Antimicrob Agents. 2020;105932. 37. Chen J, Hu C, Chen L, Tang L, Zhu Y, Xu X, et al. Clinical study of
mesenchymal stem cell treating acute respiratory distress syndrome
24. Teslova E. Russia claims it produces drug to treat COVID-19. 2020.
induced by epidemic Influenza A (H7N9) infection, a hint for
25. Jo S, Kim S, Shin DH, Kim MS. Inhibition of SARS-CoV 3CL COVID-19 treatment. Engineering. 2020;6(10):1153-1161.
protease by flavonoids. J Enzyme Inhib Med Chem. 2020;35(1): 38. Leng Z, Zhu R, Hou W, Feng Y, Yang Y, Han Q, et al. Transplantation
145-151. of ACE2-mesenchymal stem cells improves the outcome of patients
26. Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A trial of with COVID-19 pneumonia. Aging Dis. 2020;11(2):216–228.
lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl 39. US National Library of Medicine.ClinicalTrials.gov. 2020.
J Med 2020;382:1787-1799.
40. Joswig AJ, Mitchell A, Cummings KJ, Levine GJ, Gregory CA, Smith
27. Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in R, et al. Repeated intra-articular injection of allogeneic mesenchymal
adults with severe COVID-19: A randomised, double-blind, placebo-
stem cells causes an adverse response compared to autologous cells in
controlled, multicentre trial. The Lancet. 2020;395(10236):1569-1578.
the equine model. Stem Cell Resear Ther. 2017;8(1):1-11.
Intern Med , Vol.11 Iss.1 No:329 9
You might also like
- IV Drip Calculation Practice ProblemsDocument3 pagesIV Drip Calculation Practice ProblemsNursingNow100% (4)
- Best of Five MCQs For The Endocrinology and Diabetes SCE 2022Document353 pagesBest of Five MCQs For The Endocrinology and Diabetes SCE 2022azmy2000100% (2)
- Ccsap 2019 Infection Critical Care PDFDocument170 pagesCcsap 2019 Infection Critical Care PDFAlaa Darwish100% (3)
- Case Presentation On Neonatal Sepsis: BY-Anisha ManeDocument45 pagesCase Presentation On Neonatal Sepsis: BY-Anisha ManeAnisha Mane100% (5)
- Pediatric Department Book Part 2 (2019-2020)Document236 pagesPediatric Department Book Part 2 (2019-2020)Mohammed ArarNo ratings yet
- John PFTDocument231 pagesJohn PFTAlexander Santiago ParelNo ratings yet
- Breast Cancer Key Facts PDFDocument2 pagesBreast Cancer Key Facts PDFIsaac Ekow Mensah100% (1)
- Raza2021 Article PrimedMesenchymalStemCellsAPotDocument10 pagesRaza2021 Article PrimedMesenchymalStemCellsAPotBala BhaskarNo ratings yet
- Computational Chemistry To Repurposing Drugs For The Control of COVID-19Document18 pagesComputational Chemistry To Repurposing Drugs For The Control of COVID-19Mai Mohamed HasanNo ratings yet
- Mesenchymal Stem Cell Infusion Shows Promise For Combating Coronavirus (COVID-19) - Induced PneumoniaDocument3 pagesMesenchymal Stem Cell Infusion Shows Promise For Combating Coronavirus (COVID-19) - Induced PneumoniadinaNo ratings yet
- Review Article Mesenchymal Stem Cells and COVID-19: Cure, Prevention, and VaccinationDocument12 pagesReview Article Mesenchymal Stem Cells and COVID-19: Cure, Prevention, and VaccinationBala BhaskarNo ratings yet
- Covid19 An Overview of Diagnostic Approach and Novel Directions For Treatment StrategiesDocument4 pagesCovid19 An Overview of Diagnostic Approach and Novel Directions For Treatment StrategiesMarie Kath LeenNo ratings yet
- Pharmacological Therapeutics Targeting RNA-Dependent RNA Polymerase, Proteinase and Spike ProteinDocument23 pagesPharmacological Therapeutics Targeting RNA-Dependent RNA Polymerase, Proteinase and Spike ProteinAndrei OanesNo ratings yet
- Respiratory MedicineDocument8 pagesRespiratory MedicineSky AngelNo ratings yet
- Preprints ChloroquineDocument24 pagesPreprints ChloroquineJorgelina BernetNo ratings yet
- Bigger Problems From Smaller Colonies Emergence of Antibiotic-Tolerant Small Colony Variants of Mycobacterium Avium Complex in MAC-pulmonary Disease PatientsDocument16 pagesBigger Problems From Smaller Colonies Emergence of Antibiotic-Tolerant Small Colony Variants of Mycobacterium Avium Complex in MAC-pulmonary Disease Patientsd4rkgr455No ratings yet
- s41231 021 00093 2Document8 pagess41231 021 00093 2Julián ZilliNo ratings yet
- COVID para Cardiologistas JACC 2020Document19 pagesCOVID para Cardiologistas JACC 2020Maria Do SocorroNo ratings yet
- In Vitro and Animal Models For Sars-Cov-2 Research: ForumDocument5 pagesIn Vitro and Animal Models For Sars-Cov-2 Research: ForumJack SupramaniamNo ratings yet
- Jurnal 2 InggrisDocument14 pagesJurnal 2 InggrisM RIDWAN MIRO LUBISNo ratings yet
- Combing Through Traditional Texts To Prevent Covid-19 - A Scientific ApproachDocument14 pagesCombing Through Traditional Texts To Prevent Covid-19 - A Scientific ApproachVinayNo ratings yet
- Covid-19: Characteristics and TherapeuticsDocument29 pagesCovid-19: Characteristics and TherapeuticsHelenaNo ratings yet
- Mesenchymal Stem Cell Immunomodulation and Regeneration TherapeuticsDocument13 pagesMesenchymal Stem Cell Immunomodulation and Regeneration TherapeuticswiwiNo ratings yet
- Paper Jbuon Nov 21 (26-5-1719)Document4 pagesPaper Jbuon Nov 21 (26-5-1719)dimitriosroukasNo ratings yet
- A Short Review On Antibody Therapy For COVID-19Document4 pagesA Short Review On Antibody Therapy For COVID-19Master Littérature et CinémaNo ratings yet
- Probiotic-Based Vaccines May Provide Effective Protection Against COVID-19 Acute Respiratory DiseaseDocument21 pagesProbiotic-Based Vaccines May Provide Effective Protection Against COVID-19 Acute Respiratory DiseaseOppi TidjaniNo ratings yet
- Reviews: A Materials-Science Perspective On Tackling COVID-19Document14 pagesReviews: A Materials-Science Perspective On Tackling COVID-19Abdul jeleelNo ratings yet
- Covid 19Document12 pagesCovid 19Philippus TrismegistoNo ratings yet
- Nanobodies - Covid19 RockDocument6 pagesNanobodies - Covid19 Rockrock1311No ratings yet
- Revision de Sdra en CovidDocument14 pagesRevision de Sdra en CovidRociio HernandezNo ratings yet
- 30 39Document10 pages30 39Helena Putri SiraitNo ratings yet
- Advances and Challenges in The Prevention and Treatment of COVID-19Document8 pagesAdvances and Challenges in The Prevention and Treatment of COVID-19RoyNo ratings yet
- Breve Reseña Sobre Medicamentos y Vacunas Reutilizados para El Posible Tratamiento de COVID-19Document15 pagesBreve Reseña Sobre Medicamentos y Vacunas Reutilizados para El Posible Tratamiento de COVID-19Cristal Jewels DiamondNo ratings yet
- An Evidence-Based Guide To Sars-Cov-2 Vaccination of Patients On Immunotherapies in DermatologyDocument15 pagesAn Evidence-Based Guide To Sars-Cov-2 Vaccination of Patients On Immunotherapies in DermatologyMelchizedek M. SantanderNo ratings yet
- s41423 021 00813 6Document12 pagess41423 021 00813 6Josué Cristhian Del Valle HornaNo ratings yet
- Toward Nanotechnology-Enabled Approaches Against The COVID-19 PandemicDocument24 pagesToward Nanotechnology-Enabled Approaches Against The COVID-19 Pandemicmadcraft9832No ratings yet
- Updates On Clinical Trials Evaluating The Regenerative Potential of Allogenic Mesenchymal Stem Cells in COVID-19Document11 pagesUpdates On Clinical Trials Evaluating The Regenerative Potential of Allogenic Mesenchymal Stem Cells in COVID-19diana.alyNo ratings yet
- Ijms 22 01308Document64 pagesIjms 22 01308NADIA VICUÑANo ratings yet
- Stem Cell Based Therapy Option in COVID-19 - Is It Really Promising?Document17 pagesStem Cell Based Therapy Option in COVID-19 - Is It Really Promising?diana.alyNo ratings yet
- Baden NEJM 2020 Moderna PH IIIDocument14 pagesBaden NEJM 2020 Moderna PH IIIantonioNo ratings yet
- Cei 202 162Document31 pagesCei 202 162chaterinaNo ratings yet
- Recombinant Vaccines For COVID 19Document9 pagesRecombinant Vaccines For COVID 19Salvador Emmanuel Sanchez CuapioNo ratings yet
- COVID-19 Vaccination Efficacy and Safety Literature Review: ArticleDocument19 pagesCOVID-19 Vaccination Efficacy and Safety Literature Review: ArticlePromiseNo ratings yet
- (2020) Ozone (O3) and SARS-CoV-2 - Physiological Bases and Their Therapeutic Possibilities According To COVID-19 Evolutionary StageDocument9 pages(2020) Ozone (O3) and SARS-CoV-2 - Physiological Bases and Their Therapeutic Possibilities According To COVID-19 Evolutionary StageVu TranNo ratings yet
- PRL CovidDocument8 pagesPRL Covidzainy zehraNo ratings yet
- Sars-Cov-2: Immune Response Elicited by Infection and Development of Vaccines and TreatmentsDocument19 pagesSars-Cov-2: Immune Response Elicited by Infection and Development of Vaccines and TreatmentschaterinaNo ratings yet
- COVID-19 Vaccination Efficacy and Safety Literature Review: ArticleDocument19 pagesCOVID-19 Vaccination Efficacy and Safety Literature Review: ArticleBasma AboalezzNo ratings yet
- Pandemic SARS Coronavirus-2 Infections in Humans-COVID-19: Nasir Mustafa, Hina Zahoor, Fuzail M. MAJOODocument17 pagesPandemic SARS Coronavirus-2 Infections in Humans-COVID-19: Nasir Mustafa, Hina Zahoor, Fuzail M. MAJOOmaito2270No ratings yet
- Potential of Algal Metabolites For The Development of Broad Spectrum Antiviral Therapeutics Possible Implications inCOVID-19 TherapyDocument21 pagesPotential of Algal Metabolites For The Development of Broad Spectrum Antiviral Therapeutics Possible Implications inCOVID-19 TherapyNica AuliaNo ratings yet
- Autoimmunity Reviews: SciencedirectDocument9 pagesAutoimmunity Reviews: SciencedirectetienNo ratings yet
- Viral Diseases Attachment and Genome Replication Cell KillingDocument3 pagesViral Diseases Attachment and Genome Replication Cell KillingAnonymous AERDSoPBQ3No ratings yet
- COVID-19 Vaccination Efficacy and Safety Literature Review: Journal of Clinical and Medical Research February 2021Document19 pagesCOVID-19 Vaccination Efficacy and Safety Literature Review: Journal of Clinical and Medical Research February 2021guanellaNo ratings yet
- Mesenchymal Stromal Cell Therapy For Coronavirus Disease 2019 - Which? When? and How Much?Document14 pagesMesenchymal Stromal Cell Therapy For Coronavirus Disease 2019 - Which? When? and How Much?diana.alyNo ratings yet
- Vaccines Saving LivesDocument12 pagesVaccines Saving Livesmashke82No ratings yet
- SARS CoV 2 Variants, VaccinesDocument21 pagesSARS CoV 2 Variants, VaccinesPAOLA ALEJANDRA GONZALEZ RODRIGUEZNo ratings yet
- Excellent Paper On Covid 19Document20 pagesExcellent Paper On Covid 19GalenNo ratings yet
- 1 s2.0 S0753332223007874 MainDocument18 pages1 s2.0 S0753332223007874 MainFernando del BlancoNo ratings yet
- Can We Cure The Coronavirus Disease 19 (COVID19) by Using Rhus Coriaria (Sumac) ExtractDocument7 pagesCan We Cure The Coronavirus Disease 19 (COVID19) by Using Rhus Coriaria (Sumac) ExtractInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Biomedicine & Pharmacotherapy: ReviewDocument18 pagesBiomedicine & Pharmacotherapy: ReviewRahmanda LintangNo ratings yet
- 1-S2.0-S1044532320300385-Sars Cov VaccineDocument16 pages1-S2.0-S1044532320300385-Sars Cov VaccineSIUSANTO HadiNo ratings yet
- Erythro VLPsDocument19 pagesErythro VLPsLucas HeNo ratings yet
- Gao Et Al. 2020 - Chemistry and Biology of SARS-CoV-2Document13 pagesGao Et Al. 2020 - Chemistry and Biology of SARS-CoV-2TSA KishoreNo ratings yet
- An Insight Into The Pathogenesis, Diagnosis and Treatment of SARS-Cov-2 With A Synopsis of Situation in Malaysia During The 2020 OutbreakDocument12 pagesAn Insight Into The Pathogenesis, Diagnosis and Treatment of SARS-Cov-2 With A Synopsis of Situation in Malaysia During The 2020 OutbreakUMYU Journal of Microbiology Research (UJMR)No ratings yet
- Repurposing Ivermectin For COVID-19: Molecular Aspects and Therapeutic PossibilitiesDocument12 pagesRepurposing Ivermectin For COVID-19: Molecular Aspects and Therapeutic PossibilitiesVicko SuswidiantoroNo ratings yet
- European Journal of Pharmacology: Gayathri Krishna, Vinod Soman Pillai, Mohanan Valiya VeettilDocument11 pagesEuropean Journal of Pharmacology: Gayathri Krishna, Vinod Soman Pillai, Mohanan Valiya VeettilautomationenggNo ratings yet
- Golchin2020 Article MesenchymalStemCellTherapyForCDocument7 pagesGolchin2020 Article MesenchymalStemCellTherapyForCJuan Pablo Rodríguez CoronaNo ratings yet
- COVID-19 LEGACY: SARS-CoV-2 clinical trials, vaccines trials and bioethicsFrom EverandCOVID-19 LEGACY: SARS-CoV-2 clinical trials, vaccines trials and bioethicsNo ratings yet
- Review Article Mesenchymal Stem Cells and COVID-19: Cure, Prevention, and VaccinationDocument12 pagesReview Article Mesenchymal Stem Cells and COVID-19: Cure, Prevention, and VaccinationBala BhaskarNo ratings yet
- Mesenchymal Stem Cells (MSCS) As A Novel Therapeutic Option For Ncovid-19-A ReviewDocument16 pagesMesenchymal Stem Cells (MSCS) As A Novel Therapeutic Option For Ncovid-19-A ReviewBala BhaskarNo ratings yet
- Raza2021 Article PrimedMesenchymalStemCellsAPotDocument10 pagesRaza2021 Article PrimedMesenchymalStemCellsAPotBala BhaskarNo ratings yet
- Effectiveness of Whatsapp For Blood Donor Mobilization Campaigns During Covid-19 PandemicDocument5 pagesEffectiveness of Whatsapp For Blood Donor Mobilization Campaigns During Covid-19 PandemicBala BhaskarNo ratings yet
- Hemasphere: Uncontrolled Freezing of Peripheral Blood Stem Cell at - 80°C: Our ExperienceDocument6 pagesHemasphere: Uncontrolled Freezing of Peripheral Blood Stem Cell at - 80°C: Our ExperienceBala BhaskarNo ratings yet
- Sciencedirect: Letter To The EditorDocument2 pagesSciencedirect: Letter To The EditorBala BhaskarNo ratings yet
- Sciencedirect: Letter To The EditorDocument2 pagesSciencedirect: Letter To The EditorBala BhaskarNo ratings yet
- Then and Now: Chemistry & Society: Chronic Traumatic EncephalopathyDocument4 pagesThen and Now: Chemistry & Society: Chronic Traumatic Encephalopathyapi-269382043No ratings yet
- Case Pre Ovarian CystDocument56 pagesCase Pre Ovarian Cystthesa1201No ratings yet
- Bali Spa Overseas Prices Update1Document5 pagesBali Spa Overseas Prices Update1Dian WijayantiNo ratings yet
- Tailor PciDocument11 pagesTailor PciMathew McCarthyNo ratings yet
- List Morgan StoeltingDocument2 pagesList Morgan StoeltingzuzuNo ratings yet
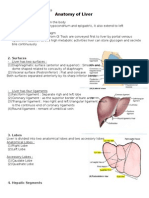
- Anatomy of LiverDocument2 pagesAnatomy of LiverelsafirdaaaNo ratings yet
- Nursing Care PlanDocument1 pageNursing Care PlanTam Tam FreeNo ratings yet
- 为什么你总在吃,还一直饿?Document5 pages为什么你总在吃,还一直饿?Victoria TangNo ratings yet
- Apolonia Nr292Document158 pagesApolonia Nr292Nikola DerebanNo ratings yet
- Comprehensive Exam 1Document19 pagesComprehensive Exam 1karenkaren09100% (1)
- 547792129c17bec4c106ead404e0c4ddDocument52 pages547792129c17bec4c106ead404e0c4ddHugoRoCkstarNo ratings yet
- Consumer World Amr Awareness Week 2023 Power Point PresentationDocument29 pagesConsumer World Amr Awareness Week 2023 Power Point PresentationSoha GalalNo ratings yet
- 1700 Subjectwise 2017 p197 232Document36 pages1700 Subjectwise 2017 p197 232Pravin MaratheNo ratings yet
- Atlas Medical BacteriologyDocument104 pagesAtlas Medical Bacteriologyradulus100% (1)
- Notre Dame-Siena College of General Santos City: Physical Education & Health 11Document7 pagesNotre Dame-Siena College of General Santos City: Physical Education & Health 11Adrian IntrinaNo ratings yet
- Cleft Lip & Cleft Palate: Presentor Ms. Jeewan JyotiDocument25 pagesCleft Lip & Cleft Palate: Presentor Ms. Jeewan JyotiHamss AhmedNo ratings yet
- 4-Microbiology & ParasitologyDocument2 pages4-Microbiology & ParasitologyIbrahimFikry100% (1)
- RETDEM 3 ScriptDocument3 pagesRETDEM 3 ScriptMyangel LoiseNo ratings yet
- Ect ReportDocument50 pagesEct ReportRhea Andrea Uy100% (1)
- TB Guidelines 2017Document7 pagesTB Guidelines 2017Galih DibiraNo ratings yet
- List of Instruments For Part II ExaminationDocument32 pagesList of Instruments For Part II ExaminationananthNo ratings yet
- FlushingDocument10 pagesFlushing林昌恩No ratings yet
- Case-Presentation Final Nagd Ni ShetDocument7 pagesCase-Presentation Final Nagd Ni ShetMary Beth AbelidoNo ratings yet