Professional Documents
Culture Documents
Pharmaceutical Merits&Demerits of Emulsion
Pharmaceutical Merits&Demerits of Emulsion
Uploaded by
Shemaj GurchumaOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Pharmaceutical Merits&Demerits of Emulsion
Pharmaceutical Merits&Demerits of Emulsion
Uploaded by
Shemaj GurchumaCopyright:
Available Formats
Industrial pharmacy portion 3rd year pharmacy
Emulsions
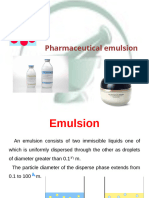
Emulsions are liquid disperse systems consisting of at least two immiscible liquids (or two
liquids that are saturated with each other), one of which is dispersed as small globules (internal
or dispersed phase) within the other liquid phase (external or continuous phase), generally
stabilized by a third substance called emulsifying agent. The process of formation of an emulsion
is termed emulsification.
There are two basic types of emulsions
1. Oil-in-water (O/W) emulsion
2. Water-in-oil (W/O) emulsion
However, depending upon the need, more complex systems (referred to as “double emulsions” or
“multiple emulsions”) in which the oil-in-water or water-in-oil emulsions are dispersed in
another liquid medium can be formulated.
By considering particle size, pharmaceutical emulsions can be
1. Macro emulsions (droplets size usually exceeds 10 mm)
2. Mini emulsions (droplets size usually 0.1–10 µm)
3. Microemulsions (droplets size usually 100-600 nm)
4. Nano Emulsions (droplets size usually below 100 nm)
Emulsions can also be classified based on mode of administration into
1. Oral emulsions e.g., castor oil, liquid paraffin
2. External emulsions e.g., creams
3. Parenteral emulsions e.g., vitamins
4. Rectal emulsions e.g., enema.
This article will focus on oil-in-water (o/w) emulsions, water-in-oil emulsions, multiple
emulsions, and microemulsions.
Oil-in-water emulsion
Oil-in-water (o/w) emulsions contain oil droplets dispersed as globules throughout an aqueous
continuous phase. An oil-in-water emulsion is generally formed if the aqueous phase constitutes
more than 45% of the total weight and a water-loving emulsifier, such as sodium lauryl sulfate,
triethanolamine stearate, sodium oleate, and glyceryl monostearate is used. The emulsifier is
present in the external, continuous phase and helps stabilize the interface with the dispersed
phase globules.
By Shimaji gurchima MTU College of Health Science School of pharmacy Page 1
Industrial pharmacy portion 3rd year pharmacy
Fats or oils for oral administration, either as medicaments in their own right or as vehicles for
oil-soluble drugs, are always formulated as o/w emulsions. Oil-in-water emulsions are non-
greasy and are easily removable from the skin surface. They are used externally to provide
cooling effect and internally to also mask the bitter taste of oil.
Water-soluble drugs are more quickly released from o/w emulsion. O/W emulsions give a
positive conductivity test as water, the external phase is a good conductor of electricity.
Water-in-oil emulsion
In a water-in-oil (w/o) emulsion, the aqueous phase is dispersed as globules in the oil continuous
phase. A lipophilic emulsifier is used for preparing w/o emulsions. The w/o emulsions are used
mainly for external applications and may contain one or several of the following emulsifiers:
calcium palmitate, sorbitan esters (Spans), cholesterol, and wool fats. Thus, the use of a
lipophilic emulsifier enables the formation of w/o emulsions with the oil phase as the external,
continuous phase.
An emulsion is a dispersed system containing at least two immiscible (or partially miscible)
liquid phases. The two phases of emulsions are stabilized by the presence of an emulsifier. The
droplet diameter of the dispersed phase extends from about 0.1 to 10 μM, although particle
diameters as small as 0.01 μM and as large as 100 μM are not uncommon.
This article focuses on the advantages and disadvantages of emulsions.
Advantages of emulsions
The advantages of emulsions as pharmaceutical products include the following:
1. Pharmaceutical emulsions may be used to deliver drugs that are poorly soluble in water but
readily soluble in oils. E.g., in oil-in-water emulsions the drug substance is dissolved in the
discontinuous or internal oil phase. Following oral administration the oil droplets (and hence the
drug) may then be absorbed using the normal absorption mechanism for oils. Some drugs are
more readily absorbed when administered as an emulsion than as other oral comparator
formulations.
2. Pharmaceutical emulsions may be used to mask the bitter taste and odor of drugs, in which the
drug is dissolved in the internal phase of an o/w emulsion. The external phase may then be
formulated to contain the appropriate sweetening and flavoring agents.
3. Drugs that are more stable in an oily phase compared to an aqueous medium can show
improved stability in an emulsion dosage form.
4. Intravenous emulsions of contrast media have been developed to assist in diagnosis.
By Shimaji gurchima MTU College of Health Science School of pharmacy Page 2
Industrial pharmacy portion 3rd year pharmacy
5. Emulsion can be used to prolong the release of the drug (especially semisolid emulsions)
thereby providing sustained release action. The oily phase can serve as a reservoir of the drug,
which slowly partitions into the aqueous phase for absorption.
6. Essential nutrients like carbohydrates, fats, and vitamins can all be emulsified and can be
administered to bedridden patients as sterile intravenous emulsions.
7. Pharmaceutical emulsions may be employed to administer drugs to patients who have
difficulty swallowing solid dosage forms.
8. Emulsions provide protection to drugs which are susceptible to oxidation or hydrolysis.
9. Emulsions are used widely to formulate externally used products like lotions, creams,
liniments etc.
Disadvantages of emulsions.
1. Pharmaceutical emulsions are thermodynamically unstable and therefore must be formulated
to stabilize the emulsion from separation of the two phases. This is by no means straightforward.
2. Pharmaceutical emulsions may be difficult to manufacture.
3. Storage conditions may affect stability.
4. Bulky, difficult to transport, and prone to container breakages.
5. Liable to microbial contamination which can lead to cracking.
6. Uniform and accurate dose my not be achieved.
Pharmaceutical suspension is a liquid dosage form containing finely divided, undissolved drug
particles dispersed throughout a liquid vehicle in which the drug exhibits a minimum degree of
solubility. This dosage form is used for providing a liquid dosage form for insoluble drugs.
In an ideal suspension, particles are uniformly dispersed and remain so even after prolonged
periods of time. Even if sedimentation occurs, particles should readily redisperse upon mild
agitation of the container.
Suspensions have a number of therapeutic applications across different routes of administration.
Most suspensions are available in ready-to-use form from the manufacturer. In cases of physical
or chemical incompatibility, they are marketed as reconstitutable medications in unit-dose sachet
or multidose bottles.
Read Also: How to Reconstitute Oral Suspension
This article focuses on the advantages and disadvantages of pharmaceutical suspensions
By Shimaji gurchima MTU College of Health Science School of pharmacy Page 3
Industrial pharmacy portion 3rd year pharmacy
Advantages of suspensions dosage form
The following advantages can be obtained by formulating API as a suspension dosage form.
1. Suspensions are a useful drug delivery system for therapeutic agents that have a low solubility.
Although low-solubility therapeutic agents may be solubilised and therefore administered as a
solution, the volume of the solvent required to perform this may be large. In addition,
formulations in which the drug has been solubilised using a co-solvent may exhibit precipitation
issues upon storage.
2. Pharmaceutical suspensions may be formulated to mask the unpleasant/ bitter taste of drug.
E.g. Chloramphenicol.
3. Pharmaceutical suspensions may serve as an alternative means used to administer drugs to
children including paediatric and geriatric patients and older patients who have difficulty
swallowing solid dosage forms.
4. Drug in suspension exhibits higher rate of bioavailability than other dosage forms.
Bioavailability is in following order, Solution > Suspension > Capsule > Compressed Tablet >
Coated tablet.
5. Pharmaceutical suspensions may be formulated to provide controlled drug delivery, e.g. as
intramuscular injections.
6. Chemical stability of certain drugs e.g., Procaine penicillin G can be improved when
formulated as suspensions.
7. Pharmaceutical suspension offers resistance to degradation of drugs due to hydrolysis,
oxidation or microbial activity.
8. Duration and onset of action can be controlled e.g., Protamine Zinc-Insulin suspension.
9. When compared to solution dosage forms, relatively higher concentration of drugs can be
incorporated into suspension products.
Disadvantages of suspensions dosage form
The disadvantages of suspensions as pharmaceutical products include the following:
1. Pharmaceutical suspensions are fundamentally unstable and for this reason, it requires
formulation skill to ensure that the physical stability of the formulation is retained over the
period of the shelf-life.
2. Aesthetic pharmaceutical suspension is difficult to formulate that is they lack elegance.
By Shimaji gurchima MTU College of Health Science School of pharmacy Page 4
Industrial pharmacy portion 3rd year pharmacy
3. Accuracy of dosage is less reliable than with solution unless suspension is packed in unit
dosage form.
4. Suspension formulations may be bulky and therefore difficult for a patient to carry.
5. Preparation must be shaken prior to measuring a dose.
6. Pourability and syringeability issues (e.g., injectable suspensions)
7. Crystal formation (Ostwald ripening) and breaking of suspension.
Multiple emulsions
Multiple emulsions are emulsions whose dispersed phase contains droplets of another emulsion.
They can be considered as emulsions of emulsions.
Multiple emulsions can be either
1. Water-in-oil-in-water (w1/o/w2) emulsion
2. Oil-in-water-in-oil (o1/w/o2) emulsion.
Emulsifying a w/o emulsion using water-soluble surfactants (which stabilize an oily dispersed
phase) can produce w/o/w emulsions with an external aqueous phase, which generally has a
lower viscosity than the primary w/o emulsion. Oil-in-water-in-oil (o/w/o) type multiple
emulsions on the other hand consist of very small droplets of oil dispersed in the water globules
of a water-in-oil emulsion.
Both water-in-oil-in-water (w/o/w) and oil-in-water-in-oil (o/w/o) multiple emulsions are of
interest as delayed- and/or sustained-action drug delivery systems. They also have applications in
cosmetics. Multiple emulsions can also be used for the encapsulation of peptides/proteins and
hydrophilic drugs.
Microemulsions
Microemulsions are visually homogeneous, transparent/isotropic systems of low viscosity. In
their simplest form, microemulsions are small droplets (diameter 5–140 nm) of one liquid
dispersed throughout another by virtue of the presence of a fairly large amount of surfactant(s)
and cosolvent(s). Microemulsions have a very finely subdivided dispersed phase, and often
contain a high concentration of the emulsifier(s) and a cosolvent (such as ethanol).
Microemulsions are thermodynamically stable for prolonged periods of time. They can be
dispersions of o/w or w/o. The type of microemulsion (w/o or o/w) formed is determined largely
by the nature of the surfactants. Microemulsions can be used to increase the bioavailability of
poorly water-soluble drugs by incorporating them into the oily phase. Incorporation of etoposide
and methotrexate diester derivative into w/o microemulsion has been suggested as a potential
carrier for cancer therapy.
By Shimaji gurchima MTU College of Health Science School of pharmacy Page 5
Industrial pharmacy portion 3rd year pharmacy
References sources
Dash, A., Singh, S. and Tolman, J. (2014). Pharmaceutics: Basic Principles and Application to
Pharmacy Practice.USA: Elsevier Inc.
Mahato, R. and Narang, A. (2018). Pharmaceutical Dosage Forms and Drug Delivery (3rd ).New
York: Taylor & Francis Group, LLC.
By Shimaji gurchima MTU College of Health Science School of pharmacy Page 6
You might also like
- Formulation of EmulsionDocument8 pagesFormulation of EmulsionSyafiqah Zainoddin100% (1)
- Pharmacokinetic Model Questions by Shimaji GDDocument22 pagesPharmacokinetic Model Questions by Shimaji GDShemaj Gurchuma100% (1)
- Water Lab PDFDocument4 pagesWater Lab PDFjosafdi askdfhNo ratings yet
- Intake Manifold DesignDocument13 pagesIntake Manifold Designshubhaastro2827100% (1)
- Pharmaceutical Merits&Demerits of EmulsionDocument6 pagesPharmaceutical Merits&Demerits of EmulsionShemaj GurchumaNo ratings yet
- P HARMACOLOGYDocument3 pagesP HARMACOLOGYOsorio JerichoNo ratings yet
- In Laboratory How Can Prepare Prescription As Emulsion Dose FromDocument7 pagesIn Laboratory How Can Prepare Prescription As Emulsion Dose Fromمحمد حميد عطيةNo ratings yet
- Emulsion and ApplicationDocument26 pagesEmulsion and ApplicationSamay Sharma100% (1)
- Emulsion 1-1-4Document4 pagesEmulsion 1-1-4saleem seharNo ratings yet
- (?) ????????Document21 pages(?) ????????marcolivinstNo ratings yet
- Emulsions and MicroemulsionsDocument20 pagesEmulsions and MicroemulsionsRIDER1993666No ratings yet
- Suspension and EmulsionDocument7 pagesSuspension and EmulsionNaacsriNo ratings yet
- Pharmaceutical EmulsionsDocument55 pagesPharmaceutical EmulsionsMAHALAKSHMI S100% (1)
- Pharmaceutical EmulsionsDocument55 pagesPharmaceutical EmulsionschandramohanNo ratings yet
- Industrial Pharmaceutics Semester 8 Sir. Talib HussainDocument9 pagesIndustrial Pharmaceutics Semester 8 Sir. Talib HussainMuhammad KafeelNo ratings yet
- 14 Disperse Systems With AnswersDocument5 pages14 Disperse Systems With AnswersPatricia Camryne Ambida100% (2)
- Lec. 1 EmulsionDocument13 pagesLec. 1 EmulsionSujitha DineshNo ratings yet
- EMULSIONSDocument10 pagesEMULSIONSsahilarrain0011No ratings yet
- Current Drug Delivery Volume 3 Issue 4 2006 (Doi 10.2174/156720106778559056) Yaqoob Khan, Azhar Talegaonkar, Sushama Iqbal, Zeenat Jalees - Multiple Emulsions - An OverviewDocument16 pagesCurrent Drug Delivery Volume 3 Issue 4 2006 (Doi 10.2174/156720106778559056) Yaqoob Khan, Azhar Talegaonkar, Sushama Iqbal, Zeenat Jalees - Multiple Emulsions - An Overviewagus nugrahaNo ratings yet
- Disperse SystemsDocument4 pagesDisperse SystemsAbby CruzNo ratings yet
- Pharmaceutical Dosage Preparations Dosage Forms Are Essentially Pharmaceutical Products in The Form Which Involves A Mixture of ActiveDocument13 pagesPharmaceutical Dosage Preparations Dosage Forms Are Essentially Pharmaceutical Products in The Form Which Involves A Mixture of ActiveKhara TeanoTanNo ratings yet
- EPT Page 1568Document19 pagesEPT Page 1568lirisNo ratings yet
- Liquid Dosage Form 1-6Document14 pagesLiquid Dosage Form 1-6mohammedNo ratings yet
- Final Assignment of TechnologyDocument15 pagesFinal Assignment of TechnologyKazi Mehnaz Subrine 1821385049No ratings yet
- Emulsion Dosage FormDocument11 pagesEmulsion Dosage FormKinza WaqasNo ratings yet
- EmulsionDocument10 pagesEmulsionfdt11No ratings yet
- N.K. Sarathchandraprakash, Chandrika Mahendra, S.J. Prashanth, Krishan ManralDocument16 pagesN.K. Sarathchandraprakash, Chandrika Mahendra, S.J. Prashanth, Krishan Manralsusi susiNo ratings yet
- Pharmaceutical Ointments and Pastes: General DescriptionDocument6 pagesPharmaceutical Ointments and Pastes: General DescriptionToukir AhmedNo ratings yet
- EMULSIONSDocument86 pagesEMULSIONSMusfira KhalidNo ratings yet
- Chapter 14:DISPERSE SY StemsDocument49 pagesChapter 14:DISPERSE SY StemsAnaliza Kitongan LantayanNo ratings yet
- pdf 20221120 193206 ٠٠٠٠Document68 pagespdf 20221120 193206 ٠٠٠٠asma2002mhNo ratings yet
- Flavour Emulsion PDFDocument36 pagesFlavour Emulsion PDFVishrut DhadwalNo ratings yet
- Publication 11 16756 6264 PDFDocument8 pagesPublication 11 16756 6264 PDFVikram VermaNo ratings yet
- Hawler Medical University College of Nursing Academic Year 2019-2020 Second Semester/First Trial Scientific Report GuidelineDocument7 pagesHawler Medical University College of Nursing Academic Year 2019-2020 Second Semester/First Trial Scientific Report Guidelineshnya sdiqNo ratings yet
- JOHNSON OLAMIDE-WPS OfficeDocument14 pagesJOHNSON OLAMIDE-WPS OfficeMark GalantyNo ratings yet
- EmulsionsDocument11 pagesEmulsionsMatías León100% (1)
- Dosage - Chapter 14Document57 pagesDosage - Chapter 14kaukau4ever100% (1)
- Basics of Pharmaceutical Emulsions: A Review: African Journal of Pharmacy and Pharmacology December 2011Document12 pagesBasics of Pharmaceutical Emulsions: A Review: African Journal of Pharmacy and Pharmacology December 2011Lilia AndromedaNo ratings yet
- Dispensing (Solution)Document11 pagesDispensing (Solution)Sami AbubakerNo ratings yet
- EmulsionDocument4 pagesEmulsionAh Rep BeanNo ratings yet
- Lect.1, Emulsion (1) As PDFDocument18 pagesLect.1, Emulsion (1) As PDFMariam BurhanNo ratings yet
- Mr. Saran Kumar Das Ms. Deepika U M.Pharm Department of PharmaceuticsDocument36 pagesMr. Saran Kumar Das Ms. Deepika U M.Pharm Department of PharmaceuticsChilaNo ratings yet
- Emulsifiers in The Dairy IndustryDocument9 pagesEmulsifiers in The Dairy IndustryVasudha Sharma100% (1)
- 9. Pharmaceutical EMULSIONSDocument81 pages9. Pharmaceutical EMULSIONSKaro MotiNo ratings yet
- Multiple Emulsion: AssignmentDocument10 pagesMultiple Emulsion: AssignmentMD REFATNo ratings yet
- Journal Emulgel 2Document4 pagesJournal Emulgel 2Atikah ArifahNo ratings yet
- Emulsifying AgentsDocument8 pagesEmulsifying Agentsromnay100% (2)
- Ijcpr 2011Document5 pagesIjcpr 2011Adriansyah AdriNo ratings yet
- An Overviewon Multiple EmulsionsDocument6 pagesAn Overviewon Multiple EmulsionsAmina BADAOUINo ratings yet
- Pharmaceutical ExcipientsDocument19 pagesPharmaceutical ExcipientsTikaUlamNo ratings yet
- Multiple Emulsions and Its Stabilization: A Review: International Journal of Chemistry StudiesDocument5 pagesMultiple Emulsions and Its Stabilization: A Review: International Journal of Chemistry StudiesKaio PimentelNo ratings yet
- Basics of Pharmaceutical EmulsionsDocument12 pagesBasics of Pharmaceutical Emulsionsxxtryxx48No ratings yet
- Lipid Emulsions - Margaret M RobinsDocument9 pagesLipid Emulsions - Margaret M Robinskagaku09No ratings yet
- Women Institute of Learning AbbottabadDocument15 pagesWomen Institute of Learning AbbottabadRida syedNo ratings yet
- PHT 312 EmulsionDocument45 pagesPHT 312 EmulsionChristopher GambleNo ratings yet
- EmulsionDocument83 pagesEmulsionVaishnavi BendaleNo ratings yet
- Emulsions: CompositionsDocument2 pagesEmulsions: CompositionsOula HatahetNo ratings yet
- Emulsions: Lec 1 Ansel's Pharmaceutical Dosage Forms and Drug Delivery Systems DR Hala SadeqDocument26 pagesEmulsions: Lec 1 Ansel's Pharmaceutical Dosage Forms and Drug Delivery Systems DR Hala SadeqHM A92No ratings yet
- All Questions of D.FDocument18 pagesAll Questions of D.FYamaan Yamaan100% (1)
- Suspension & EmulsionsDocument57 pagesSuspension & Emulsionsimam mahdi tv officialNo ratings yet
- A Review On Comparative Study Between Emulsion, Microemulsion and NanoemulsionDocument8 pagesA Review On Comparative Study Between Emulsion, Microemulsion and NanoemulsionHoàngNo ratings yet
- EmulsionDocument42 pagesEmulsionShakeel IjazNo ratings yet
- Pharmaceutical Merits&Demerits of EmulsionDocument6 pagesPharmaceutical Merits&Demerits of EmulsionShemaj GurchumaNo ratings yet
- Practice ProblemsDocument2 pagesPractice ProblemsShemaj GurchumaNo ratings yet
- Model Question From Website by SGDocument11 pagesModel Question From Website by SGShemaj GurchumaNo ratings yet
- Analysis Model QuestionDocument9 pagesAnalysis Model QuestionShemaj GurchumaNo ratings yet
- Gr2 Wk14 Solids Liquids and GasesDocument2 pagesGr2 Wk14 Solids Liquids and GasesAlison Mc Namara100% (1)
- PSE 9e CH 29Document21 pagesPSE 9e CH 29M ANo ratings yet
- Lab Report Exp 3 Electrochemical CellDocument5 pagesLab Report Exp 3 Electrochemical CellYe Woon LimNo ratings yet
- Deflection1 PDFDocument17 pagesDeflection1 PDFKandee Marie ApiNo ratings yet
- COLLOIDSDocument8 pagesCOLLOIDSJanna Kate SajullaNo ratings yet
- Empirical Equations For Deformation Capacity of Conventional RC Shear WallsDocument12 pagesEmpirical Equations For Deformation Capacity of Conventional RC Shear WallsCarsonBakerNo ratings yet
- B.tech. Ct2 Autumn Semester 2020 2021 Qps Ami AP Pps Egd Pce Ac Bee E&e IwtDocument23 pagesB.tech. Ct2 Autumn Semester 2020 2021 Qps Ami AP Pps Egd Pce Ac Bee E&e Iwtsantanu janaNo ratings yet
- Fibre Reinforced Xoncrete - A State of The Art ReviewDocument10 pagesFibre Reinforced Xoncrete - A State of The Art ReviewGhina Fatikah SalimNo ratings yet
- Chapter (3) Turbulence Kinetic Energy: 3.1 The TKE Budget DerivationDocument11 pagesChapter (3) Turbulence Kinetic Energy: 3.1 The TKE Budget DerivationMaitraNo ratings yet
- Calculation of Von Mises Stress at Plastic DeformationDocument4 pagesCalculation of Von Mises Stress at Plastic DeformationMaurizio FalconieriNo ratings yet
- Uns 31803 SHPDocument1 pageUns 31803 SHPuttamkboseNo ratings yet
- The Einstein SolidDocument12 pagesThe Einstein SolidFederico Alejandro GeserNo ratings yet
- Nano Technology (OE) - Unit 2Document94 pagesNano Technology (OE) - Unit 2Department of Chemical EngineeringNo ratings yet
- Lumped Parameter Leakoff ModelDocument13 pagesLumped Parameter Leakoff Modelamramazon88No ratings yet
- .Analytical and Experimental Analysis of Cantilever Beam Under Forced Vibration PDFDocument4 pages.Analytical and Experimental Analysis of Cantilever Beam Under Forced Vibration PDFtesfayeNo ratings yet
- 3C43 Lasers & Modern Optics Problem Sheet 2 - Lasers: G I DX DiDocument2 pages3C43 Lasers & Modern Optics Problem Sheet 2 - Lasers: G I DX DiShootingStarPhotonsNo ratings yet
- Surface TensionDocument18 pagesSurface TensionMD CHHIMPANo ratings yet
- NA To Sls en 1993-6Document12 pagesNA To Sls en 1993-6Shan Sandaruwan AbeywardeneNo ratings yet
- Laboratory Quality Management CertificateDocument2 pagesLaboratory Quality Management CertificateMuhammad Daud AliNo ratings yet
- Analiza PiesaDocument7 pagesAnaliza PiesaRauLasH26No ratings yet
- Introduction On CreepDocument24 pagesIntroduction On CreepAbdul RahimNo ratings yet
- Natural Sciences. 2nd of ESO: Heat & TemperatureDocument15 pagesNatural Sciences. 2nd of ESO: Heat & TemperatureAlberto DíazNo ratings yet
- Fluid Mech Orifice 3Document9 pagesFluid Mech Orifice 3TatendaNo ratings yet
- Bragg VS LaueDocument7 pagesBragg VS LaueSHIVAM GUPTANo ratings yet
- Glass Ceramics QuizDocument3 pagesGlass Ceramics QuizRon Pascual100% (1)
- Mechanical SealDocument64 pagesMechanical SealKhoh Kai ShengNo ratings yet
- Metallic Material Specification: Title: 2.25 Cr-1 Mo (ASTM A182, Grade F22 Mod), 85 KSI Min Yield Strength - Unified SpecDocument6 pagesMetallic Material Specification: Title: 2.25 Cr-1 Mo (ASTM A182, Grade F22 Mod), 85 KSI Min Yield Strength - Unified SpecReginaldo SantosNo ratings yet
- Purlin Data:-: 10-Sep-2014 1 OF 2 VGC SMA Design of Intermediate Purlin 0 IS: 801Document2 pagesPurlin Data:-: 10-Sep-2014 1 OF 2 VGC SMA Design of Intermediate Purlin 0 IS: 801keerthivasan_mrvNo ratings yet