Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
29 viewsBone Tissue: Osteocytes
Bone Tissue: Osteocytes
Uploaded by
A18- Jessa Mae DayagCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- Pre and Post Natal Development of MandibleDocument14 pagesPre and Post Natal Development of MandibleLiyana G Yaya100% (1)
- HistoPath (Lect #5) TransDocument5 pagesHistoPath (Lect #5) TransSherlyn Giban InditaNo ratings yet
- BONEDocument4 pagesBONEvallecerajamaicaNo ratings yet
- Bones: Defination: Bone Is Highly Vascular, Living, Constantly ChangingDocument21 pagesBones: Defination: Bone Is Highly Vascular, Living, Constantly ChangingMd Ahsanuzzaman PinkuNo ratings yet
- Bone and Bone Graft HealingDocument12 pagesBone and Bone Graft HealingJayanth PerumalNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Bone ReviewerDocument7 pagesBone ReviewerClyde BaltazarNo ratings yet
- Tortora Principles of Anatomy and PhysiologyDocument6 pagesTortora Principles of Anatomy and PhysiologybebeNo ratings yet
- Bone Tissue and The Skeletal System: (Hyaline) CartilageDocument3 pagesBone Tissue and The Skeletal System: (Hyaline) CartilageMae CanlasNo ratings yet
- Alveolar BoneDocument8 pagesAlveolar BoneAsruni Ab GhaniNo ratings yet
- Skeletal SystemDocument14 pagesSkeletal SystemCharlize PalmaNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Histology 8Document6 pagesHistology 8amirhsheikhiNo ratings yet
- CH8 Bone: Medullary CavitiesDocument9 pagesCH8 Bone: Medullary Cavitiesdanny olulanaNo ratings yet
- Chapter 8Document3 pagesChapter 8Ella LagramaNo ratings yet
- Anaphy - Skeletal SystemDocument13 pagesAnaphy - Skeletal SystemYo1No ratings yet
- Tereos - Structure of PresentationDocument72 pagesTereos - Structure of PresentationSumitNo ratings yet
- Bone Is A Mineralized Connective Tissue: Biochemistry DepartmentDocument66 pagesBone Is A Mineralized Connective Tissue: Biochemistry DepartmentYunandhika RizkiNo ratings yet
- BONE UqsDocument6 pagesBONE UqsAroosha JamshaidNo ratings yet
- Biomechanics and Biomaterials in Orthopedic SurgeryDocument11 pagesBiomechanics and Biomaterials in Orthopedic SurgeryManiventhan NachimuthuNo ratings yet
- Human Histology 8 BoneDocument9 pagesHuman Histology 8 BoneBlubby BleuNo ratings yet
- HO Chapter 6 Pharm 123Document5 pagesHO Chapter 6 Pharm 123malfoymidnight394No ratings yet
- Chapter 8-BonesDocument1 pageChapter 8-BonesLynNo ratings yet
- BONE (Histology)Document65 pagesBONE (Histology)fhfebriiNo ratings yet
- Gen Histo Topic 6 BonesDocument4 pagesGen Histo Topic 6 BonesShania CandelarioNo ratings yet
- V. OsteogenesisDocument7 pagesV. OsteogenesisrosalesjirehrNo ratings yet
- Chemistry Project On Estimation of Content of Bone AshDocument18 pagesChemistry Project On Estimation of Content of Bone AshKim TaesiNo ratings yet
- Skeletal SystemDocument14 pagesSkeletal SystemCharlize PalmaNo ratings yet
- AN OVERVIEW OF BONE CELLS AND THEIR REGULATING FACTORS OF DIFFERENTIATION, Mohamed 2008Document9 pagesAN OVERVIEW OF BONE CELLS AND THEIR REGULATING FACTORS OF DIFFERENTIATION, Mohamed 2008Faizal Reza PahleviNo ratings yet
- Skeletal 6Document28 pagesSkeletal 6BSN-2F Neutral Axis SiazonNo ratings yet
- C6-Skeletal SystemDocument5 pagesC6-Skeletal SystemLorrine MagramoNo ratings yet
- 4.2.2. Bone TissueDocument7 pages4.2.2. Bone Tissuealexandra4wineNo ratings yet
- Lec Activity6 Skeletal System CastroDocument8 pagesLec Activity6 Skeletal System CastropelielNo ratings yet
- Bone TissueDocument46 pagesBone TissueFiraol DiribaNo ratings yet
- 6 PATHO 3b Part 1 - Musculoskeletal IIA - Dr. Dy-LedesmaDocument12 pages6 PATHO 3b Part 1 - Musculoskeletal IIA - Dr. Dy-LedesmaBanana BananaNo ratings yet
- Intro To The Skeletal SystemDocument10 pagesIntro To The Skeletal SystemmipeNo ratings yet
- Bone Tissue & Mascular TissueDocument5 pagesBone Tissue & Mascular TissueJanjan GarcesNo ratings yet
- Metabolic Bone DiseaseDocument27 pagesMetabolic Bone DiseaseRifa'atul MahmudahNo ratings yet
- Skeletal System TransesDocument11 pagesSkeletal System Transesadrielvamos28No ratings yet
- BoneDocument1 pageBoneMicah James MaravillasNo ratings yet
- Principles of Bone GraftingDocument6 pagesPrinciples of Bone GraftingJayanth Perumal100% (2)
- 6 Skeletal SystemDocument8 pages6 Skeletal SystemmargaretNo ratings yet
- Skeletal SystemDocument80 pagesSkeletal SystemElaine Victoria ElizanNo ratings yet
- Biología Celular Metabolismo OseoDocument12 pagesBiología Celular Metabolismo Oseoandreaelisa17No ratings yet
- Kalsifikasi, Deposisi Dan Resorpsi TulangDocument40 pagesKalsifikasi, Deposisi Dan Resorpsi TulangrifkaNo ratings yet
- W7 - Bone HistologyDocument10 pagesW7 - Bone HistologynhcsmlmolNo ratings yet
- Alveolar BoneDocument81 pagesAlveolar BoneLavanya KalapalaNo ratings yet
- Histology and Growth of The BoneDocument62 pagesHistology and Growth of The Boneeminbsb01No ratings yet
- SkeletonDocument15 pagesSkeletonPlacido Edgar MagaNo ratings yet
- Skeletal SystemDocument97 pagesSkeletal SystemFardzli MatjakirNo ratings yet
- 6 SkeletalDocument14 pages6 SkeletalprincessstephNo ratings yet
- HISTO LAB Week 1 Bone and CartilageDocument4 pagesHISTO LAB Week 1 Bone and CartilageJennie WatanabeNo ratings yet
- The Main Functions of BonesDocument9 pagesThe Main Functions of BonesabbaslafeNo ratings yet
- Functions of The Musculoskeletal SystemDocument9 pagesFunctions of The Musculoskeletal SystemLinda XueNo ratings yet
- Skeletal System LectureDocument28 pagesSkeletal System LectureLittle CloudNo ratings yet
- Fisiologi TulangDocument33 pagesFisiologi Tulangrizka dwi auliaNo ratings yet
- BONE Healing Uii 24 April 2014Document38 pagesBONE Healing Uii 24 April 2014Taufiq HidayatNo ratings yet
- Alveolar Bone in Health: Part - BDocument37 pagesAlveolar Bone in Health: Part - BrayaimNo ratings yet
- MSK NotesDocument83 pagesMSK NotesabdulNo ratings yet
- Dr. Ishita Singhal Mds First YearDocument189 pagesDr. Ishita Singhal Mds First YearDR. ISHITA SINGHALNo ratings yet
- MicroLec GroupWork1Document10 pagesMicroLec GroupWork1A18- Jessa Mae DayagNo ratings yet
- Adipose Tissue: Dr. Bernal September 2018Document3 pagesAdipose Tissue: Dr. Bernal September 2018A18- Jessa Mae DayagNo ratings yet
- A. Cells of Connective TissueDocument4 pagesA. Cells of Connective TissueA18- Jessa Mae Dayag100% (1)
- Histology Nervous Tissue and The Nervous SystemDocument7 pagesHistology Nervous Tissue and The Nervous SystemA18- Jessa Mae DayagNo ratings yet
- Cartilage: Histology Dr. BernalDocument4 pagesCartilage: Histology Dr. BernalA18- Jessa Mae DayagNo ratings yet
- Histology Dr. Noelyn Bernal Cytoplasm-September 7,2018: Page - PEREZDocument13 pagesHistology Dr. Noelyn Bernal Cytoplasm-September 7,2018: Page - PEREZA18- Jessa Mae DayagNo ratings yet
- Forms and Characteristics of Epithelial Cells: Basement MembranesDocument5 pagesForms and Characteristics of Epithelial Cells: Basement MembranesA18- Jessa Mae DayagNo ratings yet
- 105844-BondocDocument130 pages105844-BondocA18- Jessa Mae DayagNo ratings yet
- Histology THE NUCLEUSDocument6 pagesHistology THE NUCLEUSA18- Jessa Mae DayagNo ratings yet
- Histology Its Methods of StudyDocument6 pagesHistology Its Methods of StudyA18- Jessa Mae DayagNo ratings yet
- Modul Ajar 1 Sistem RangkaDocument21 pagesModul Ajar 1 Sistem RangkaYuanita AndrianiNo ratings yet
- Legal Medicine: K.-S. Saternus, H. Maxeiner, G. Kernbach-Wighton, J. KoebkeDocument6 pagesLegal Medicine: K.-S. Saternus, H. Maxeiner, G. Kernbach-Wighton, J. KoebkeEolia EffendiNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Anatomy - Physiology (Chapter 6 - Skeletal System)Document43 pagesAnatomy - Physiology (Chapter 6 - Skeletal System)Kyla DuenaNo ratings yet
- CH 05 Lecture Presentation Skeletal-SystemDocument166 pagesCH 05 Lecture Presentation Skeletal-SystemDenver SorilNo ratings yet
- Chapter 7 - CartilageDocument17 pagesChapter 7 - CartilageREMAN ALINGASANo ratings yet
- Biology Chapter 13 Concepts 01 35 Human Biology Answers PDFDocument145 pagesBiology Chapter 13 Concepts 01 35 Human Biology Answers PDFKamryn MontgomeryNo ratings yet
- AO Surgery ReferenceDocument8 pagesAO Surgery ReferenceHurulaeniNo ratings yet
- Bone and Joints 2Document38 pagesBone and Joints 2Eryn HooverNo ratings yet
- Chapter 3Document12 pagesChapter 3Deolita BadiangNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Instructional PlanDocument17 pagesInstructional PlanJamal P. AlawiyaNo ratings yet
- Classification of BonesDocument4 pagesClassification of BoneshyyjNo ratings yet
- 1.04 - The Skeletal SystemDocument7 pages1.04 - The Skeletal Systemelio pascualNo ratings yet
- Musculoskeletal SystemDocument210 pagesMusculoskeletal SystemDr. Serin KuriakoseNo ratings yet
- The Skeletal System The Skeletal System: © 2018 Pearson Education, Ltd. 1Document16 pagesThe Skeletal System The Skeletal System: © 2018 Pearson Education, Ltd. 1lourd nabNo ratings yet
- Chapter 6 Skeletal System 1Document18 pagesChapter 6 Skeletal System 1M GarciaNo ratings yet
- 1 The Development and Growth of The FaceDocument15 pages1 The Development and Growth of The FacebarbiemeNo ratings yet
- Maders Understanding Human Anatomy and Physiology 9th Edition Longenbaker Test BankDocument59 pagesMaders Understanding Human Anatomy and Physiology 9th Edition Longenbaker Test Bankjohnnavarronpisaoyzcx100% (30)
- Postions Movement Bones JointsDocument8 pagesPostions Movement Bones JointsNini BakradzeNo ratings yet
- Question Bank General AnatomyDocument2 pagesQuestion Bank General AnatomyRohit GNo ratings yet
- Skeletal SystemDocument12 pagesSkeletal SystemRojan V Min KookNo ratings yet
- Development of MandibleDocument60 pagesDevelopment of MandibleAlisha Aranha100% (1)
- Worksheet-5 Musculoskeletal-System Group3 BFFDocument79 pagesWorksheet-5 Musculoskeletal-System Group3 BFFAldrin LimcuandoNo ratings yet
- Healing of FracturesDocument19 pagesHealing of FracturesTracy100% (4)
- Anatomy and Physiology CHAP 6Document30 pagesAnatomy and Physiology CHAP 6Jaica Mangurali Tumulak100% (1)
- G&DDocument20 pagesG&Dahead1234No ratings yet
- Bone Basic NDocument98 pagesBone Basic NSuraj kumar GuptaNo ratings yet
- Zooarchaeology: Teacher's ManualDocument21 pagesZooarchaeology: Teacher's ManualRamos CarolinaNo ratings yet
Bone Tissue: Osteocytes
Bone Tissue: Osteocytes
Uploaded by
A18- Jessa Mae Dayag0 ratings0% found this document useful (0 votes)
29 views11 pagesOriginal Title
HISTOLOGY-BONES
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
29 views11 pagesBone Tissue: Osteocytes
Bone Tissue: Osteocytes
Uploaded by
A18- Jessa Mae DayagCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 11
- When their synthesizing activity declines:
flatten and basophilia is reduced
TOPIC OUTLINE:
Osteoid - layer of new (but not yet
I. BONE CELLS calcified) material
Osteoblasts
Osteocytes
Osteoclasts
Osteocalcin - prominent noncollagen
II. BONE MATRIX proteins secreted by osteoblasts, vitamin K–
III. PERIOSTEUM & ENDOSTEUM dependent polypeptide
IV. TYPES OF BONE Matrix vesicles - rich in alkaline
Lamellar Bone phosphatase, raises the local concentration
Woven Bone of PO4− ions. With high concentrations of
V. OSTEOGENESIS both calcium and phosphate ions, these
Intramembranous Ossification vesicles serve as foci for the formation of
Endochondral Ossification
hydroxyapatite [Ca10(PO4)6(OH)2] crystals,
VI. BONE REMODELING & REPAIR
( the first visible step in calcification) .
VII. METABOLIC ROLE OF BONE
VIII. JOINTS
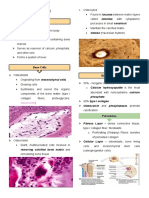
Osteocytes
- differentiated osteoblasts
- When compared with osteoblasts, the flat,
BONE TISSUE almond shaped osteocytes exhibit
significantly less RER, smaller Golgi
- main constituent of the adult skeleton complexes, and more condensed nuclear
- provides solid support for the body chromatin.
- protects vital organs such as those in the - express a different array of genes compared
cranial and thoracic cavities to osteoblasts
- harbors cavities containing bone marrow - products such as the protein sclerostin and
certain cytokines help regulate bone
where blood cells are formed
remodelling.
- serves as a reservoir of calcium, phosphate,
and other ions
Osteoclasts
- very large (due to their origin from the fusion
of bone marrow–derived cells), motile cells
Osteoblasts
with multiple nuclei and play a major role in
- synthesize and secrete the organic
matrix resorption during bone growth and
components of bone matrix (type I collagen
remodelling.
fibers, proteoglycans, and several
- development requires two polypeptides
BONE CELLS produced by osteoblasts:macrophage-
colony–stimulating factor (M-CSF) and the
glycoproteins such as osteonectin)
- Mature osteoblasts are located exclusively receptor activator of nuclear factor-κB ligand
(RANKL).
at the surfaces of bone matrix ( resembling
a simple epithelium) Resorption cavities (also called
- When actively engaged in matrix synthesis, Howship lacunae) - enzymatically
osteoblasts: cuboidal to columnar shape etched depressions or cavities In
and basophilic cytoplasm. areas of bone undergoing
resorption.
LIBAN, MAPALO Page 1
- In active osteoclasts, the surface against The organic matter embedded in the calcified
the bone matrix is folded into irregular matrix ---- 90% type I collagen, but also includes
projections, forming a ruffled border mostly small proteoglycans and multiadhesive
surrounded by a cytoplasmic zone (where glycoproteins (osteonectin).
bone resorption occurs) rich in actin
Calcium binding proteins, notably osteocalcin, and
filaments, which is the site of adhesion to
the phosphatases released from cells in matrix
the matrix.
- Osteoclast activity is controlled by local
signalling factors and hormones and have
receptors for calcitonin.
PERIOSTEUM & ENDOSTEUM
Osteoblasts activated by parathyroid vesicles promote calcification of the matrix.
hormone (PTH) produce M-CSF, The association of minerals with collagen fibers
RANKL, and other factors that during calcification provides the hardness and
regulate the formation and activity of resistance required for bone function.
osteoclasts.
Periosteum - is organized much like the
perichondrium of cartilage, with an outer fibrous
layer of dense connective tissue, containing mostly
bundled type I collagen, but also fibroblasts and
blood vessels.
Perforating (or Sharpey) fibers -
Bundles of periosteal collagen,
penetrate the bone matrix and bind
the periosteum to the bone.
Osteoprogenitor cells - periosteum’s
inner layer, more cellular and
includes osteoblasts, bone lining
cells, and mesenchymal stem cells.
About 50% of the dry weight of bone matrix is TYPES OF BONE
inorganic materials. Endosteum - covers small trabeculae of bony
matrix that project into the marrow cavities.
- Calcium hydroxyapatite is most abundant - also contains osteoprogenitor cells,
- bicarbonate, citrate, magnesium, potassium, osteoblasts, and bone lining cells, but within
and sodium ions are also found. a sparse, delicate matrix of collagen fibers.
BONE MATRIX
Compact (cortical) bone which represents 80% of
- Significant quantities of noncrystalline
the total bone mass, and deeper areas with
calcium phosphate are also present.
numerous interconnecting cavities, called
The surface of hydroxyapatite
cancellous (trabecular) bone, constituting about
crystals are hydrated, facilitating the
20% of total bone mass.
exchange of ions between the
mineral and body fluids.
Terms:
LIBAN, MAPALO Page 2
Epiphyses - the bulbous ends that are composed is laid down around areas with
of cancellous bone covered by a thin layer of preexisting blood vessels.
compact cortical bone.
Diaphysis - cylindrical part, almost totally dense Interstitial lamellae - scattered among the intact
compact bone, with a thin region of cancellous osteons are numerous irregularly shaped groups of
bone on the inner surface around the central parallel lamellae.These structures are lamellae
marrow cavity. remaining from osteons partially destroyed by
Plates - two layers of compact bone in which osteoclasts during growth and remodeling of bone.
calvaria are form.
Diploë - separates the thicker layer of cancellous Compact bone (eg, in the diaphysis
bone in plates. of long bones) also includes parallel
lamellae organized as multiple
external circumferential lamellae
Lamellar Bone immediately beneath the periosteum
and fewer inner circumferential
lamellae around the marrow cavity.
The lamellae of these outer and
- characterized by multiple layers or lamellae
innermost areas of compact bone
of calcified matrix, each 3-7 μm thick.
enclose and strengthen the middle
region containing vascularized
Lamellae - are organized as parallel sheets or
osteons.
concentrically around a central canal. In each
Bone remodeling occurs
lamella, type I collagen fibers are aligned (causes
continuously throughout life. In
birefringence with polarizing light microscopy),
compact bone, remodeling resorbs
with the pitch of the fibers’ orientation shifted
orthogonally (by about 90 degrees) in successive
lamellae. Woven Bone
parts of old osteons and produces
Osteon (or Haversian system) - refers to the new ones.
complex of concentric lamellae, typically 100-250
μm in diameter, surrounding a central canal that - nonlamellar and characterized by random
contains small blood vessels, nerves, and disposition of type I collagen fibers and is
endosteum. the first bone tissue to appear in embryonic
Each osteon is a long, sometimes development and in fracture repair.
bifurcated, cylinder generally parallel - usually temporary and is replaced in adults
to the long axis of the diaphysis. by lamellar bone, except in a very few
Each has 5-20 concentric lamellae places in the body, for example, near the
around the central canal which sutures of the calvaria and in the insertions
communicates with the marrow of some tendons.
cavity and the periosteum. Canals In addition to the irregular, interwoven
also communicate with one another array of collagen fibers, woven bone
through transverse perforating typically has a lower mineral content (it
canals (or Volkmann canals) which is more easily penetrated by x-rays) and
have few, if any, concentric lamellae. a higher proportion of osteocytes than
All central osteonic canals and mature lamellar bone. These features
perforating canals form when matrix reflect the facts that immature woven
LIBAN, MAPALO Page 3
bone forms more quickly but has less Process:
strength than lamellar bone.
Osteoid secreted by osteoblasts
calcifies forming small irregular areas of woven
bone with osteocytes in lacunae and canaliculi.
Continued matrix secretion and calcification
enlarges these areas and leads to the fusion of
neighboring ossification centers.
Anatomical bone forms as woven bone
matrix is replaced by compact bone that
encloses a region of cancellous bone with
marrow and larger blood vessels.
Mesenchymal regions that do not
undergo ossification give rise to the endosteum
OSTEOGENESIS (BONE DEVELOPMENT) and periosteum of the new bone.
Two processes:
1. Intramembranous ossification = Cranial flat bones= lamellar bone formation
osteoblasts differentiate directly from predominates over bone resorption at the
mesenchyme and begin secreting osteoid. internal and external surfaces.
2. Endochondral ossification = pre-existing Internal and external plates of compact bone
matrix of hyaline cartilage is eroded and arise. Central portion maintain its cancellous
invaded by osteoblast, which the begin nature.
osteoid production
Fontanelles (“soft spots”) on heads of
Names refer to the mechanisms by which the bone
newborns= membranous tissue is not yet
forms initially. In both processes woven bone is
ossified.
INTRAMEMBRANOUS OSSIFICATION
produced first and is soon replaced by stronger ENDOCHONDRAL OSSIFICATION
lamellar bone.
Takes place within the hyaline cartilage shaped
as a small version, or model, of the bone to be
By which most flat bones begin to form, takes
formed.
place within condensed sheets (“membranes”)
of embryonic mesenchymal tissue.
Forms most bones of the body and is
e.g. scapula, clavicle
especially well studied in developing long
bones.
Ossification centers = areas in which
osteoprogenitor cells arise, proliferate, and
form incomplete layers of osteoblasts around a Process:
network of developing capillaries. Ossification first occurs within a bone
collar produced by osteoblasts that
differentiate within the perichondrium
LIBAN, MAPALO Page 4
(transitioning to periosteum) around the the epiphyses of the cartilage model and
cartilage model diaphysis. develop in a similar manner. During expansion
and remodelling, the primary and secondary
Bone collar now impedes diffusion of ossification centers produce cavities that are
oxygen and nutrients into the underlying gradually filled with bone marrow.
cartilage, promoting degenerative changes.
Two regions of cartilage in primary and
Chondrocytes then produce alkaline secondary ossification centers:
phosphatase and swell up (hypertrophy),
enlarging their lacunae. These changes Articular cartilage within joints which
compresses the matrix into narrower persists throughout adult life and does
trabeculae and lead to calcification in these not contribute to bone growth in length,
structures and
Death of the chondrocytes results in a
porous 3D structure formed by the remnants of Epiphyseal cartilage
the calcified cartilage matrix. (epiphyseal/growth plate), which
connects each epiphysis to the
Blood vessels from periosteum diaphysis. It is responsible for the
penetrate through the bone collar bringing growth in length of the bone and
osteoprogenitor cells to the porous central disappears in adults, which is why bone
region. Osteoblasts then adhere to the calcified growth ceases in adulthood.
cartilage matrix and produce continuous layers
of primary bone that surround the cartilaginous 5 zones of a plate of epiphyseal cartilage:
matrix remnants. 1. RESTING ZONE (zone of reserve) -
(calcified cartilage=basophilic, hyaline cartilage with typical
primary bone=eosinophilic) chondrocytes.
2. PROLIFERATIVE ZONE= chondrocytes
begin to divide rapidly and form columns
of stacked cells parallel to the long axis
of the bone. Cartilage cells secrete more
type II collagen and proteoglycans.
3. HYPERTROPHIC CARTILAGE
ZONE=contains swollen chondrocytes
whose cytoplasm has accumulated
glycogen. Hypertrophy compresses the
matrix into thin septa between the
chondrocytes
4. CALCIFIED CARTILAGE ZONE= loss
This process in the diaphysis forms the of chondrocytes by apoptosis
primary ossification center (embryonic accompanied by calcification of the
development-1st trimester). Secondary septa of cartilage matrix by the
ossification centers appear slightly later at formation of hydroxyapatite crystals
LIBAN, MAPALO Page 5
5. OSSIFICATION ZONE= bone tissue BONE MODELING/OSTEOGENESIS
first appears. Capillaries and -the sum of osteoblasts and osteoclasts
osteoprogenitor cells from periosteum activities in a growing bone
invade the cavities left by the -maintains each bone’s general shape
chondrocytes. Cavities will be merged while increasing its mass
and become the marrow cavity. -bone turnover rate is very active in
Osteoprogenitor cells form osteoblasts, young children; 200 times faster than in adults
which settle in a discontinuous layer
over the septa of calcified cartilage BONE REMODELING
matrix. Osteoblasts deposit osteoid over -process of continually renewing the
the spicules of calcified cartilage matrix, skeleton in adults
forming woven bone. -involves coordinated, localized cellular
activities for bone resorption and bone
formation
-constant remodelling ensures that
despite its hardness, the tissue remains plastic
and capable of adapting its internal structures
in the face of changing stresses (ex. Ability of
positions of teeth in the jawbone to be modified
by the lateral pressures produced by
orthopaedic appliances)
Appositional growth= growth in the
circumference of long bones occurs through
the activity of osteoblasts developing from
osteoprogenitor cells in the periosteum. This
BONE REPAIR
begins with the formation of bone collar on the
-bone has excellent capacity for repair
cartilaginous diaphysis. Increase in bone
because it contains osteoprogenitor stem cells
circumference is accompanied by enlargement
in the periosteum, endosteum and marrow, and
of central marrow cavity by the activity of the
is very well vascularized
osteoclasts in the endosteum.
-in fracture or other damage, uses cells,
signalling molecules and processes already
active in bone remodelling
BONE REMODELING AND REPAIR
LIBAN, MAPALO Page 6
-surgically created gaps in bone can be polypeptide hormones also target bone cells to
filled with new bone, especially when influence calcium homeostasis:
periosteum is left in place
Parathyroid hormone (PTH) = raises low
BONE FRACTURE REPAIR blood calcium levels by stimulating osteoclasts
Major phases are: and osteocytes to resorb bone matrix and
-initial formation of fibrocartilage release Ca2+. Effect on osteoclasts is indirect;
-replacement with a temporary callus of PTH receptors occur on osteoblasts, which
woven bone respond by secreting RANKL and other
paracrine factors that stimulate osteoclast
formation and activity.
Calcitonin= produced within the thyroid gland,
can reduce elevated blood calcium levels by
METABOLIC ROLE OF BONE opposing the effect of PTH in the bone. Directly
targets osteoclasts to slow matrix resorption
and bone turnover.
Calcium
-required for activity of many enzymes
and many proteins mediating cell adhesion, JOINTS
cytoskeletal movements, exocytosis,
membrane permeability and other cellular -regions where adjacent bones are capped and
functions held together firmly by other connective tissues
-concentration in blood = 9-10 mg/dL -type of bone determines the degree of
movement
-concentration is stable due to
continuous interchange between blood calcium -classified as synarthroses = allow very
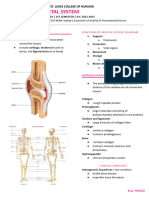
and bone calcium limited movement or no movement, or
Diarthroses= permits free bone movement
-skeleton serves as calcium reservoir
(99% of the body’s total calcium in -subdivided into fibrous and cartilaginous joints
hydroxyapatite crystals) depending on the type of tissue joining the
bones
-principal mechanism for increasing
blood calcium levels= mobilization of ions from 1. SYNOSTOSES
hydroxyapatite to interstitial fluid (primarily in - bone linked to other bones and allow
the cancellous bone) essentially no movement
- unite the skull bones in older adults
-mobilization is regulated by paracrine which in children and young adults are
interactions among bone cells. Two held together by sutures, or thin layers
LIBAN, MAPALO Page 7
of dense connective tissue with derived from embryonic notochord, the only
osteogenic cells cells of that structure to persist postnatally. It is
large in children, but gradually becomes
2. SYNDESMOSES smaller with age and partially replaced by
-join bones by dense connective tissue fibrocartilage.
only
-e.g. interosseous ligament of the
inferior tibiofibular joint and the posterior DIARTHROSES
region of the sacroiliac joints
-e.g. knee and elbow joints
3. SYMPHYSES -unite long bones and allow great mobility
-have a thick pad of fibrocartilage -ligaments and a capsule of connective tissue
between the thin articular cartilage maintain proper alignment of the bones
covering the ends of the bones
-all occur in the midline of the body -capsule encloses a sealed joint cavity
(intervertebral discs and pubic containing a clear, viscous liquid called
symphysis) synovial fluid
Intervertebral discs= large symphyses -joint cavity is lined by a specialized connective
between the articular surfaces of tissue called the synovial membrane, which
successive bony vertebral bodies. extend folds and villi into the joint cavity and
These discoid components of the produces the lubricant synovial fluid.
intervertebral joints cushion the bones -synovial membrane may have prominent
and facilitate limited movements of the regions with dense CT or fat. These regions
vertebral column. are usually well vascularized, with many
porous (fenestrated) capillaries. Characterized
Parts of the intervertebral disc: by having typical cells of Ct proper and a
changing population of leukocytes and two
Annulus fibrosus (outer portion) specialized cells:
-consists of concentric fibrocartilage
laminae in which collagen bundles are Macrophage-like synovial cells (type
arranged orthogonally in adjacent layers. The A cells) – derived from monocytes and
multiple lamellae of fibrocartilage produce a remove wear-and-tear debris from the
disc with unusual toughness able to withstand synovial fluid. Represent approx. 25% of
pressures and tortions the cells lining the synovium. These are
-center portion, nucleus pulposus, gel- important in regulating inflammatory
like body allows each disc to function as a events within diarthrotic joints.
shock absorber. It consists of a viscous fluid
Fibroblastic synovial cells (type B
MAJOR TYPES OF SYNARTHROSES
cells) – produce abundant hyaluronan
matrix rich in hyaluronan and type II collagen and smaller amounts of proteoglycans.
fibers. Also contains scattered vacuolated cells Much of the material is transported by
LIBAN, MAPALO Page 8
water from the capillaries into the joint ion levels and maintain the adjacent bone
cavity to form the synovial fluid, which matrix accordingly. Lack of exercise (or the
lubricates the joint, reducing friction on weightlessness experienced by astronauts)
all internal surfaces, and supplies leads to decreased bone density, due in
nutrients and oxygen to the articular part to the lack of mechanical stimulation of
cartilage. these cells.
C. In the genetic disease osteopetrosis,
Collagen fibers of the hyaline articular cartilage which is characterized by dense, heavy
are disposed as arches with their tops near the bones (“marble bones”), the osteoclasts
exposed surface which, is not covered by lack ruffled borders and bone resorption is
perichondrium. This arrangement helps defective. This disorder results in
distribute the forces generated by pressure on overgrowth and thickening of bones, often
joints more evenly. with obliteration of the marrow cavities,
depressing blood cell formation and
CLINICAL SIGNIFICANCE causing anemia and the loss of white blood
cells. The defective osteoclasts in most
patients with osteopetrosis have mutations
in genes for the cells’ proton-ATPase
pumps or chloride channels.
Articular cartilage absorbs the intermittent
mechanical pressures to which joints may be D. Osteoporosis, frequently found in
subjected. immobilized patients and in
postmenopausal women, is an imbalance
A. Cancer originating directly from bone cells in skeletal turnover so that bone resorption
(a primary bone tumor) is fairly uncommon exceeds bone formation. This leads to
(0.5% of all cancer deaths), although a calcium loss from bones and reduced bone
cancer called osteosarcoma can arise in mineral density (BMD). Individuals at risk
osteoprogenitor cells. The skeleton is often for osteoporosis are routinely tested for
the site of secondary, metastatic tumors, BMD by dual-energy x-ray absorptiometry
however, arising when cancer cells move (DEXA scans).
into bones via small blood or lymphatic
vessels from malignancies in other organs, E. The antibiotic tetracycline is a fluorescent
most commonly the breast, lung, prostate molecule that binds newly deposited
gland, kidney, or thyroid gland osteoid matrix during mineralization with
high affinity and specifically labels new
bone under the UV microscope. This
B. The network of dendritic processes discovery led to methods for measuring the
extending from osteocytes has been called rate of bone growth, an important
a “mechanostat,” monitoring areas within parameter in the diagnosis of certain bone
bones where loading has been increased disorders. In one technique tetracycline is
or decreased, and signaling cells to adjust administered twice to patients, with an
LIBAN, MAPALO Page 9
intervening interval of 11-14 days. A bone can give rise to osteomalacia (osteon + Gr.
biopsy is then performed, sectioned malakia, softness), characterized by
without decalcification, and examined. deficient calcification of recently formed
Bone formed while tetracycline was bone and partial decalcification of already
present appears as fluorescent lamellae calcified matrix.
and the distance between the labeled
layers is proportional to the rate of bone H. Bone fractures are repaired by a
appositional growth. This procedure is of developmental process involving
diagnostic importance in such diseases as fibrocartilage formation and osteogenic
osteomalacia, in which mineralization is activity of the major bone cells. Bone
impaired, and osteitis fibrosa cystica, in fractures disrupt blood vessels, causing
which increased osteoclast activity results bone cells near the break to die. The
in removal of bone matrix and fibrous damaged blood vessels produce a
degeneration. localized hemorrhage or hematoma.
Clotted blood is removed along with tissue
F. Osteogenesis imperfecta, or “brittle debris by macrophages and the matrix of
bone disease,” refers to a group of related damaged, cell-free bone is resorbed by
congenital disorders in which the osteoclasts. The periosteum and the
osteoblasts produce deficient amounts of endosteum at the fracture site respond with
type I collagen or defective type I collagen intense proliferation and produce a soft
due to genetic mutations. Such defects callus of fibrocartilage-like tissue that
lead to a spectrum of disorders, all surrounds the fracture and covers the
characterized by significant fragility of the extremities of the fractured bone. The
bones. The fragility reflects the deficit in fibrocartilaginous callus is gradually
normal collagen, which normally reinforces replaced in a process that resembles a
and adds a degree of resiliency to the combination of endochondral and
mineralized bone matrix. intramembranous ossification. This
produces a hard callus of woven bone
G. Calcium deficiency in children can lead to around the fractured ends of bone.
rickets, a disease in which the bone matrix Stresses imposed on the bone during
does not calcify normally and the repair and during the patient’s gradual
epiphyseal plate can become distorted by return to activity serve to remodel the bone
the normal strains of body weight and callus. The immature, woven bone of the
muscular activity. Ossification processes callus is gradually resorbed and replaced
are consequently impeded, which causes by lamellar bone, remodeling and restoring
bones to grow more slowly and often the original bone structure.
become deformed. The deficiency can be
due either to insufficient calcium in the diet I. In addition to PTH and calcitonin, several
or a failure to produce the steroid other hormones act on bone. The anterior
prohormone vitamin D, which is important lobe of the pituitary synthesizes growth
for the absorption of Ca2+ by cells of the hormone (GH or somatotropin), which
small intestine. In adults calcium deficiency stimulates the liver to produce insulin-like
LIBAN, MAPALO Page 10
growth factor-1 (IGF-1 or somatomedin). compressed nerve fibers—usually the
IGF has an overall growth-promoting lower lumbar region.
effect, especially on the epiphyseal
cartilage. Consequently, lack of growth
hormone during the growing years causes
pituitary dwarfism; an excess of growth
hormone causes excessive growth of the
long bones, resulting in gigantism. Adult
bones cannot increase in length even with
excess IGF because they lack epiphyseal
cartilage, but they do increase in width by
periosteal growth. In adults, an increase in
GH causes acromegaly, a disease in which
the bones—mainly the long ones—become
very thick.
J. In rheumatoid arthritis chronic
inflammation of the synovial membrane
causes thickening of this connective tissue
and stimulates the macrophages to release
collagenases and other hydrolytic
enzymes. Such enzymes eventually cause
destruction of the articular cartilage,
allowing direct contact of the bones
projecting into the joint.
K. Within an intervertebral disc, collagen loss
or other degenerative changes in the
annulus fibrosus are often accompanied by
displacement of the nucleus pulposus, a
condition variously called a slipped or
herniated disc. This occurs most
frequently on the posterior region of the
intervertebral disc where there are fewer
collagen bundles. The affected disc
frequently dislocates or shifts slightly from
its normal position. If it moves toward
nerve plexuses, it can compress the
nerves and result in severe pain and other
neurologic disturbances. The pain
accompanying a slipped disc may be
perceived in areas innervated by the HAPPY READING!
LIBAN, MAPALO Page 11
You might also like
- Pre and Post Natal Development of MandibleDocument14 pagesPre and Post Natal Development of MandibleLiyana G Yaya100% (1)
- HistoPath (Lect #5) TransDocument5 pagesHistoPath (Lect #5) TransSherlyn Giban InditaNo ratings yet
- BONEDocument4 pagesBONEvallecerajamaicaNo ratings yet
- Bones: Defination: Bone Is Highly Vascular, Living, Constantly ChangingDocument21 pagesBones: Defination: Bone Is Highly Vascular, Living, Constantly ChangingMd Ahsanuzzaman PinkuNo ratings yet
- Bone and Bone Graft HealingDocument12 pagesBone and Bone Graft HealingJayanth PerumalNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Bone ReviewerDocument7 pagesBone ReviewerClyde BaltazarNo ratings yet
- Tortora Principles of Anatomy and PhysiologyDocument6 pagesTortora Principles of Anatomy and PhysiologybebeNo ratings yet
- Bone Tissue and The Skeletal System: (Hyaline) CartilageDocument3 pagesBone Tissue and The Skeletal System: (Hyaline) CartilageMae CanlasNo ratings yet
- Alveolar BoneDocument8 pagesAlveolar BoneAsruni Ab GhaniNo ratings yet
- Skeletal SystemDocument14 pagesSkeletal SystemCharlize PalmaNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Histology 8Document6 pagesHistology 8amirhsheikhiNo ratings yet
- CH8 Bone: Medullary CavitiesDocument9 pagesCH8 Bone: Medullary Cavitiesdanny olulanaNo ratings yet
- Chapter 8Document3 pagesChapter 8Ella LagramaNo ratings yet
- Anaphy - Skeletal SystemDocument13 pagesAnaphy - Skeletal SystemYo1No ratings yet
- Tereos - Structure of PresentationDocument72 pagesTereos - Structure of PresentationSumitNo ratings yet
- Bone Is A Mineralized Connective Tissue: Biochemistry DepartmentDocument66 pagesBone Is A Mineralized Connective Tissue: Biochemistry DepartmentYunandhika RizkiNo ratings yet
- BONE UqsDocument6 pagesBONE UqsAroosha JamshaidNo ratings yet
- Biomechanics and Biomaterials in Orthopedic SurgeryDocument11 pagesBiomechanics and Biomaterials in Orthopedic SurgeryManiventhan NachimuthuNo ratings yet
- Human Histology 8 BoneDocument9 pagesHuman Histology 8 BoneBlubby BleuNo ratings yet
- HO Chapter 6 Pharm 123Document5 pagesHO Chapter 6 Pharm 123malfoymidnight394No ratings yet
- Chapter 8-BonesDocument1 pageChapter 8-BonesLynNo ratings yet
- BONE (Histology)Document65 pagesBONE (Histology)fhfebriiNo ratings yet
- Gen Histo Topic 6 BonesDocument4 pagesGen Histo Topic 6 BonesShania CandelarioNo ratings yet
- V. OsteogenesisDocument7 pagesV. OsteogenesisrosalesjirehrNo ratings yet
- Chemistry Project On Estimation of Content of Bone AshDocument18 pagesChemistry Project On Estimation of Content of Bone AshKim TaesiNo ratings yet
- Skeletal SystemDocument14 pagesSkeletal SystemCharlize PalmaNo ratings yet
- AN OVERVIEW OF BONE CELLS AND THEIR REGULATING FACTORS OF DIFFERENTIATION, Mohamed 2008Document9 pagesAN OVERVIEW OF BONE CELLS AND THEIR REGULATING FACTORS OF DIFFERENTIATION, Mohamed 2008Faizal Reza PahleviNo ratings yet
- Skeletal 6Document28 pagesSkeletal 6BSN-2F Neutral Axis SiazonNo ratings yet
- C6-Skeletal SystemDocument5 pagesC6-Skeletal SystemLorrine MagramoNo ratings yet
- 4.2.2. Bone TissueDocument7 pages4.2.2. Bone Tissuealexandra4wineNo ratings yet
- Lec Activity6 Skeletal System CastroDocument8 pagesLec Activity6 Skeletal System CastropelielNo ratings yet
- Bone TissueDocument46 pagesBone TissueFiraol DiribaNo ratings yet
- 6 PATHO 3b Part 1 - Musculoskeletal IIA - Dr. Dy-LedesmaDocument12 pages6 PATHO 3b Part 1 - Musculoskeletal IIA - Dr. Dy-LedesmaBanana BananaNo ratings yet
- Intro To The Skeletal SystemDocument10 pagesIntro To The Skeletal SystemmipeNo ratings yet
- Bone Tissue & Mascular TissueDocument5 pagesBone Tissue & Mascular TissueJanjan GarcesNo ratings yet
- Metabolic Bone DiseaseDocument27 pagesMetabolic Bone DiseaseRifa'atul MahmudahNo ratings yet
- Skeletal System TransesDocument11 pagesSkeletal System Transesadrielvamos28No ratings yet
- BoneDocument1 pageBoneMicah James MaravillasNo ratings yet
- Principles of Bone GraftingDocument6 pagesPrinciples of Bone GraftingJayanth Perumal100% (2)
- 6 Skeletal SystemDocument8 pages6 Skeletal SystemmargaretNo ratings yet
- Skeletal SystemDocument80 pagesSkeletal SystemElaine Victoria ElizanNo ratings yet
- Biología Celular Metabolismo OseoDocument12 pagesBiología Celular Metabolismo Oseoandreaelisa17No ratings yet
- Kalsifikasi, Deposisi Dan Resorpsi TulangDocument40 pagesKalsifikasi, Deposisi Dan Resorpsi TulangrifkaNo ratings yet
- W7 - Bone HistologyDocument10 pagesW7 - Bone HistologynhcsmlmolNo ratings yet
- Alveolar BoneDocument81 pagesAlveolar BoneLavanya KalapalaNo ratings yet
- Histology and Growth of The BoneDocument62 pagesHistology and Growth of The Boneeminbsb01No ratings yet
- SkeletonDocument15 pagesSkeletonPlacido Edgar MagaNo ratings yet
- Skeletal SystemDocument97 pagesSkeletal SystemFardzli MatjakirNo ratings yet
- 6 SkeletalDocument14 pages6 SkeletalprincessstephNo ratings yet
- HISTO LAB Week 1 Bone and CartilageDocument4 pagesHISTO LAB Week 1 Bone and CartilageJennie WatanabeNo ratings yet
- The Main Functions of BonesDocument9 pagesThe Main Functions of BonesabbaslafeNo ratings yet
- Functions of The Musculoskeletal SystemDocument9 pagesFunctions of The Musculoskeletal SystemLinda XueNo ratings yet
- Skeletal System LectureDocument28 pagesSkeletal System LectureLittle CloudNo ratings yet
- Fisiologi TulangDocument33 pagesFisiologi Tulangrizka dwi auliaNo ratings yet
- BONE Healing Uii 24 April 2014Document38 pagesBONE Healing Uii 24 April 2014Taufiq HidayatNo ratings yet
- Alveolar Bone in Health: Part - BDocument37 pagesAlveolar Bone in Health: Part - BrayaimNo ratings yet
- MSK NotesDocument83 pagesMSK NotesabdulNo ratings yet
- Dr. Ishita Singhal Mds First YearDocument189 pagesDr. Ishita Singhal Mds First YearDR. ISHITA SINGHALNo ratings yet
- MicroLec GroupWork1Document10 pagesMicroLec GroupWork1A18- Jessa Mae DayagNo ratings yet
- Adipose Tissue: Dr. Bernal September 2018Document3 pagesAdipose Tissue: Dr. Bernal September 2018A18- Jessa Mae DayagNo ratings yet
- A. Cells of Connective TissueDocument4 pagesA. Cells of Connective TissueA18- Jessa Mae Dayag100% (1)
- Histology Nervous Tissue and The Nervous SystemDocument7 pagesHistology Nervous Tissue and The Nervous SystemA18- Jessa Mae DayagNo ratings yet
- Cartilage: Histology Dr. BernalDocument4 pagesCartilage: Histology Dr. BernalA18- Jessa Mae DayagNo ratings yet
- Histology Dr. Noelyn Bernal Cytoplasm-September 7,2018: Page - PEREZDocument13 pagesHistology Dr. Noelyn Bernal Cytoplasm-September 7,2018: Page - PEREZA18- Jessa Mae DayagNo ratings yet
- Forms and Characteristics of Epithelial Cells: Basement MembranesDocument5 pagesForms and Characteristics of Epithelial Cells: Basement MembranesA18- Jessa Mae DayagNo ratings yet
- 105844-BondocDocument130 pages105844-BondocA18- Jessa Mae DayagNo ratings yet
- Histology THE NUCLEUSDocument6 pagesHistology THE NUCLEUSA18- Jessa Mae DayagNo ratings yet
- Histology Its Methods of StudyDocument6 pagesHistology Its Methods of StudyA18- Jessa Mae DayagNo ratings yet
- Modul Ajar 1 Sistem RangkaDocument21 pagesModul Ajar 1 Sistem RangkaYuanita AndrianiNo ratings yet
- Legal Medicine: K.-S. Saternus, H. Maxeiner, G. Kernbach-Wighton, J. KoebkeDocument6 pagesLegal Medicine: K.-S. Saternus, H. Maxeiner, G. Kernbach-Wighton, J. KoebkeEolia EffendiNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Anatomy - Physiology (Chapter 6 - Skeletal System)Document43 pagesAnatomy - Physiology (Chapter 6 - Skeletal System)Kyla DuenaNo ratings yet
- CH 05 Lecture Presentation Skeletal-SystemDocument166 pagesCH 05 Lecture Presentation Skeletal-SystemDenver SorilNo ratings yet
- Chapter 7 - CartilageDocument17 pagesChapter 7 - CartilageREMAN ALINGASANo ratings yet
- Biology Chapter 13 Concepts 01 35 Human Biology Answers PDFDocument145 pagesBiology Chapter 13 Concepts 01 35 Human Biology Answers PDFKamryn MontgomeryNo ratings yet
- AO Surgery ReferenceDocument8 pagesAO Surgery ReferenceHurulaeniNo ratings yet
- Bone and Joints 2Document38 pagesBone and Joints 2Eryn HooverNo ratings yet
- Chapter 3Document12 pagesChapter 3Deolita BadiangNo ratings yet
- CHAPTER 6 ANAPHY TransesDocument13 pagesCHAPTER 6 ANAPHY TransesHoly HaeinNo ratings yet
- Instructional PlanDocument17 pagesInstructional PlanJamal P. AlawiyaNo ratings yet
- Classification of BonesDocument4 pagesClassification of BoneshyyjNo ratings yet
- 1.04 - The Skeletal SystemDocument7 pages1.04 - The Skeletal Systemelio pascualNo ratings yet
- Musculoskeletal SystemDocument210 pagesMusculoskeletal SystemDr. Serin KuriakoseNo ratings yet
- The Skeletal System The Skeletal System: © 2018 Pearson Education, Ltd. 1Document16 pagesThe Skeletal System The Skeletal System: © 2018 Pearson Education, Ltd. 1lourd nabNo ratings yet
- Chapter 6 Skeletal System 1Document18 pagesChapter 6 Skeletal System 1M GarciaNo ratings yet
- 1 The Development and Growth of The FaceDocument15 pages1 The Development and Growth of The FacebarbiemeNo ratings yet
- Maders Understanding Human Anatomy and Physiology 9th Edition Longenbaker Test BankDocument59 pagesMaders Understanding Human Anatomy and Physiology 9th Edition Longenbaker Test Bankjohnnavarronpisaoyzcx100% (30)
- Postions Movement Bones JointsDocument8 pagesPostions Movement Bones JointsNini BakradzeNo ratings yet
- Question Bank General AnatomyDocument2 pagesQuestion Bank General AnatomyRohit GNo ratings yet
- Skeletal SystemDocument12 pagesSkeletal SystemRojan V Min KookNo ratings yet
- Development of MandibleDocument60 pagesDevelopment of MandibleAlisha Aranha100% (1)
- Worksheet-5 Musculoskeletal-System Group3 BFFDocument79 pagesWorksheet-5 Musculoskeletal-System Group3 BFFAldrin LimcuandoNo ratings yet
- Healing of FracturesDocument19 pagesHealing of FracturesTracy100% (4)
- Anatomy and Physiology CHAP 6Document30 pagesAnatomy and Physiology CHAP 6Jaica Mangurali Tumulak100% (1)
- G&DDocument20 pagesG&Dahead1234No ratings yet
- Bone Basic NDocument98 pagesBone Basic NSuraj kumar GuptaNo ratings yet
- Zooarchaeology: Teacher's ManualDocument21 pagesZooarchaeology: Teacher's ManualRamos CarolinaNo ratings yet