Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
24 viewsBrownian Movement
Brownian Movement
Uploaded by
cherokeebrownian movementr
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5825)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1093)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (852)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (590)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (903)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (541)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (349)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (823)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (403)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Feel Cool With Arcticool ! Kills 99.9 % of Germs Helps Prevent Cavities Gives Long Lasting Fresh BreathDocument1 pageFeel Cool With Arcticool ! Kills 99.9 % of Germs Helps Prevent Cavities Gives Long Lasting Fresh BreathcherokeeNo ratings yet
- Clinical Electives MedicineDocument2 pagesClinical Electives MedicinecherokeeNo ratings yet
- Cabinet: Title NameDocument3 pagesCabinet: Title NamecherokeeNo ratings yet
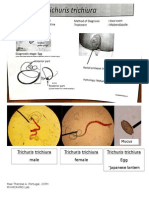
- Trichuris Trichiura Female Trichuris Trichiura Male Trichuris Trichiura EggDocument1 pageTrichuris Trichiura Female Trichuris Trichiura Male Trichuris Trichiura EggcherokeeNo ratings yet
- Reflection Paper On Entrepreneurs Assign 1Document1 pageReflection Paper On Entrepreneurs Assign 1cherokeeNo ratings yet
- Package InsertDocument2 pagesPackage InsertcherokeeNo ratings yet
- NSTP ReflectionDocument1 pageNSTP ReflectioncherokeeNo ratings yet
- Grades For Hospi PharDocument1 pageGrades For Hospi PharcherokeeNo ratings yet
- Nelson MethodDocument1 pageNelson MethodcherokeeNo ratings yet
- Experiment 9Document3 pagesExperiment 9cherokeeNo ratings yet
- Frog PithingDocument14 pagesFrog PithingcherokeeNo ratings yet
- Financial Aspects of Inventory Management: Learning ObjectivesDocument24 pagesFinancial Aspects of Inventory Management: Learning ObjectivescherokeeNo ratings yet
- Classification of Fruits: Part of A FruitDocument7 pagesClassification of Fruits: Part of A FruitcherokeeNo ratings yet
Brownian Movement
Brownian Movement
Uploaded by
cherokee0 ratings0% found this document useful (0 votes)
24 views1 pagebrownian movementr
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documentbrownian movementr
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
24 views1 pageBrownian Movement
Brownian Movement
Uploaded by
cherokeebrownian movementr
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
Brownian Movement
Brownian movement is the random motion of particles suspended in a fluid (a
liquid or a gas) resulting from their collision with the fast-moving atoms or
molecules in the gas or liquid.
The Tyndall effect is the scattering of light as a light beam passes through a colloid. The
individual suspension particles scatter and reflect light, making the beam visible. ... The size of
the particles is what distinguishes a colloid from a true solution.
Tyndall Effect
An easy way of determining whether a mixture is colloidal or not is through use of
the Tyndall Effect. When light is shined through a true solution, the light passes
cleanly through the solution, however when light is passed through a colloidal
solution, the substance in the dispersed phases scatters the light in all directions,
making it readily seen. An example of this is shining a flashlight into fog. The beam
of light can be easily seen because the fog is a colloid.
What are protective colloids give an example?
The protective colloid is a type of lyophilic (water loving) colloid which is used to protect
the lyophobic colloids from precipitating in an electrolytic solution. The common
example of protective colloids are : gelatin, casein, hemoglobin, egg albumin etc
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5825)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1093)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (852)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (590)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (903)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (541)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (349)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (823)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (403)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Feel Cool With Arcticool ! Kills 99.9 % of Germs Helps Prevent Cavities Gives Long Lasting Fresh BreathDocument1 pageFeel Cool With Arcticool ! Kills 99.9 % of Germs Helps Prevent Cavities Gives Long Lasting Fresh BreathcherokeeNo ratings yet
- Clinical Electives MedicineDocument2 pagesClinical Electives MedicinecherokeeNo ratings yet
- Cabinet: Title NameDocument3 pagesCabinet: Title NamecherokeeNo ratings yet
- Trichuris Trichiura Female Trichuris Trichiura Male Trichuris Trichiura EggDocument1 pageTrichuris Trichiura Female Trichuris Trichiura Male Trichuris Trichiura EggcherokeeNo ratings yet
- Reflection Paper On Entrepreneurs Assign 1Document1 pageReflection Paper On Entrepreneurs Assign 1cherokeeNo ratings yet
- Package InsertDocument2 pagesPackage InsertcherokeeNo ratings yet
- NSTP ReflectionDocument1 pageNSTP ReflectioncherokeeNo ratings yet
- Grades For Hospi PharDocument1 pageGrades For Hospi PharcherokeeNo ratings yet
- Nelson MethodDocument1 pageNelson MethodcherokeeNo ratings yet
- Experiment 9Document3 pagesExperiment 9cherokeeNo ratings yet
- Frog PithingDocument14 pagesFrog PithingcherokeeNo ratings yet
- Financial Aspects of Inventory Management: Learning ObjectivesDocument24 pagesFinancial Aspects of Inventory Management: Learning ObjectivescherokeeNo ratings yet
- Classification of Fruits: Part of A FruitDocument7 pagesClassification of Fruits: Part of A FruitcherokeeNo ratings yet