Professional Documents
Culture Documents
CHEN382 - SP17 Problem Set 4 KAO
CHEN382 - SP17 Problem Set 4 KAO
Uploaded by
Eilyza AballaCopyright:
Available Formats
You might also like
- Toshiba Case 3Document4 pagesToshiba Case 3Deta Detade100% (1)
- BS 57 (1951)Document23 pagesBS 57 (1951)dapsiduNo ratings yet
- Part 4 Grade 6 Edumate Introduction On MelodyDocument3 pagesPart 4 Grade 6 Edumate Introduction On MelodyAndrewOribianaNo ratings yet
- BP Intern InterviewDocument5 pagesBP Intern InterviewSergey ParsegovNo ratings yet
- Ic 01 Last Day Test 3Document30 pagesIc 01 Last Day Test 3Sohini0% (1)
- Glass Railing - Structural CalculationDocument36 pagesGlass Railing - Structural CalculationHSY79No ratings yet
- Espectro de AceleracionDocument1 pageEspectro de AceleracionYanely FalcònNo ratings yet
- Sistema: 1-Propanol (A) - Agua (B) : Práctica #9. Equilibrio Líquido - VaporDocument22 pagesSistema: 1-Propanol (A) - Agua (B) : Práctica #9. Equilibrio Líquido - Vaporwaldir marcelinoNo ratings yet
- Simulación Reacción EDocument67 pagesSimulación Reacción EMilagros Morales VergarayNo ratings yet
- Calibration LinearDocument15 pagesCalibration LinearThusith WijayawardenaNo ratings yet
- CostsDocument6 pagesCostsChalaNo ratings yet
- Thermometer Indikator : Penunjukan Alat Standard Koreksi Ketidakpastian (°C) (°C) (°C) (°C)Document20 pagesThermometer Indikator : Penunjukan Alat Standard Koreksi Ketidakpastian (°C) (°C) (°C) (°C)lailiNo ratings yet
- 2023-08-24 Ukur Debit Current Meter DI KarangtalunDocument15 pages2023-08-24 Ukur Debit Current Meter DI KarangtalunrahmawatiNo ratings yet
- Spectru ProiectareDocument27 pagesSpectru ProiectareHoria Ionut MihneaNo ratings yet
- Spectru ProiectareDocument27 pagesSpectru ProiectareHoria Ionut MihneaNo ratings yet
- Loading: Displacement / Micrometer Force / Micro NewtonDocument9 pagesLoading: Displacement / Micrometer Force / Micro NewtonasamNo ratings yet
- Blade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsDocument3 pagesBlade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsBülent KabadayiNo ratings yet
- Evaluacion de La Integral de Duhamel para Cargas SismicasDocument6 pagesEvaluacion de La Integral de Duhamel para Cargas SismicasAngela Odría RubioNo ratings yet
- VLE Using UNIFAC in ExcelDocument55 pagesVLE Using UNIFAC in Exceljdgh1986No ratings yet
- LebensmittelDocument582 pagesLebensmittelmaria budiNo ratings yet
- Blade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsDocument3 pagesBlade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsbadmaNo ratings yet
- Group 7 - Data SheetDocument9 pagesGroup 7 - Data SheetJeremy Kyle Edson AustriaNo ratings yet
- Tablas de TermodinamicaDocument55 pagesTablas de TermodinamicadeymarNo ratings yet
- P' (KN/m2)Document1 pageP' (KN/m2)Andriie DassieNo ratings yet
- Physical Organic 1 Post-LabDocument7 pagesPhysical Organic 1 Post-LabsamNo ratings yet
- Blade Design Spreadsheet Shape: 7 2.40 2 4 0.8 6 Blue CellsDocument2 pagesBlade Design Spreadsheet Shape: 7 2.40 2 4 0.8 6 Blue CellsOptimuz TsNo ratings yet
- Feeding Program FixDocument9 pagesFeeding Program FixLo Christian DjunaidiNo ratings yet
- Calculating-ISO-7886-TOGCDocument9 pagesCalculating-ISO-7886-TOGCﺵﻓﻴﺮﺍ ﺭﺷﻲ ﺍﻧﺎNo ratings yet
- 001 AggregateDocument4 pages001 Aggregatevanessa ImlerNo ratings yet
- Laporan Modul 5Document4 pagesLaporan Modul 5Adryan Fathur RahmanNo ratings yet
- Cut Off PointDocument8 pagesCut Off Pointfiruzz1No ratings yet
- Factor XP 02Document2 pagesFactor XP 02Javier FcNo ratings yet
- Chart TitleDocument11 pagesChart Titlemuhammad alfatihNo ratings yet
- Member Name Member # Steel N Total Bar (#) HA Doux Bar L (CM) Total L (CM) Poids/mLDocument269 pagesMember Name Member # Steel N Total Bar (#) HA Doux Bar L (CM) Total L (CM) Poids/mLYoumna ShatilaNo ratings yet
- Ejercicio 1 Metodos NumericosDocument17 pagesEjercicio 1 Metodos Numericosjader florezNo ratings yet
- Assignment 2 Final Version1Document78 pagesAssignment 2 Final Version1Christopher Bayo DaramolaNo ratings yet
- Armaduras en 2D PerfectDocument48 pagesArmaduras en 2D Perfectjosluis fNo ratings yet
- N FourierDocument6 pagesN FourierZokiNo ratings yet
- Επιφάνεια Βαφής Και Βάρος Δοκών Μεταλλικής ΚατασκευήςDocument27 pagesΕπιφάνεια Βαφής Και Βάρος Δοκών Μεταλλικής ΚατασκευήςTheodoros AtheridisNo ratings yet
- EstadisticaDocument2 pagesEstadisticaJuan BonifazNo ratings yet
- Spun 500 Load Settlement CurveDocument2 pagesSpun 500 Load Settlement Curveyin hoe ongNo ratings yet
- Dow Engineering InformationDocument29 pagesDow Engineering InformationbenakiaNo ratings yet
- Loading: Displacement / Micrometer Force / Micro NewtonDocument11 pagesLoading: Displacement / Micrometer Force / Micro NewtonasamNo ratings yet
- Loading: Displacement / Micrometer Force / Micro NewtonDocument7 pagesLoading: Displacement / Micrometer Force / Micro NewtonasamNo ratings yet
- Vrednosti Odgovora U Desetinama Pojedinih Delova SpektraDocument4 pagesVrednosti Odgovora U Desetinama Pojedinih Delova SpektraNiko NištićNo ratings yet
- Plan Is Acceptable: Sample Size 200 Acceptable Quality Level 1 % Alpha 5 % Unacceptable Quality Level 5 % Beta 5 %Document2 pagesPlan Is Acceptable: Sample Size 200 Acceptable Quality Level 1 % Alpha 5 % Unacceptable Quality Level 5 % Beta 5 %Cellulose NitroNo ratings yet
- Curva Fe FabianDocument11 pagesCurva Fe FabianFabian RodriguezNo ratings yet
- Hasil Regresi Soal No.2 Fix Tinggal (P)Document3 pagesHasil Regresi Soal No.2 Fix Tinggal (P)Neiscya Dhitya AmritaNo ratings yet
- Dow Water Solutions DOWEX™ Ion Exchange Resins and FILMTEC™ Membranes Engineering InformationDocument29 pagesDow Water Solutions DOWEX™ Ion Exchange Resins and FILMTEC™ Membranes Engineering Informationirshad sondeNo ratings yet
- Specifikacija paraDocument5 pagesSpecifikacija paraLjiljana Radović BukaNo ratings yet
- Reserve Estimation Unit: Ooip Recov Oil (MMSTB) (MMSTB)Document8 pagesReserve Estimation Unit: Ooip Recov Oil (MMSTB) (MMSTB)FreddyCorredorNo ratings yet
- Variograma Regular: DistanciasDocument3 pagesVariograma Regular: DistanciasyulianNo ratings yet
- Experiment 1: Physical Properties of Water ObjectivesDocument12 pagesExperiment 1: Physical Properties of Water ObjectivesWan XïnNo ratings yet
- Datos: T/TP Q/QP TDocument9 pagesDatos: T/TP Q/QP TManuel CerveraNo ratings yet
- 5.1 Holding Period ReturnDocument48 pages5.1 Holding Period ReturnSrilekha BasavojuNo ratings yet
- Other Numerical Integration ExamplesDocument19 pagesOther Numerical Integration Examplessiswanto.micoNo ratings yet
- FractionatorsDocument9 pagesFractionatorsjoker princeNo ratings yet
- 2 Absorvs ConcentrationDocument2 pages2 Absorvs ConcentrationKristin SheafferNo ratings yet
- Movimeinto de Tierras Ramon CastillaDocument19 pagesMovimeinto de Tierras Ramon CastillaGiancarlos AguinagaNo ratings yet
- Consol Grades Per BlankDocument2 pagesConsol Grades Per BlankSunddee S RomagosNo ratings yet
- LillieforsDocument2 pagesLillieforsGebz Ai BianNo ratings yet
- Polar Fineza: Axis Title Axis TitleDocument4 pagesPolar Fineza: Axis Title Axis TitleUriel Gallegos PérezNo ratings yet
- Grupo Hogares Ingreso % Acumulado de Hogares % Acumulado Del IngresoDocument6 pagesGrupo Hogares Ingreso % Acumulado de Hogares % Acumulado Del IngresoLIZETH KATHERIN MONROY MACIASNo ratings yet
- Class Customer Count Unit Consumed Basic Tariff Service Charge Street Light LevyDocument3 pagesClass Customer Count Unit Consumed Basic Tariff Service Charge Street Light LevySankalp TiwariNo ratings yet
- O Tec Reference 4Document5 pagesO Tec Reference 4Eilyza AballaNo ratings yet
- Soap Detergents and Related Compounds 6Document10 pagesSoap Detergents and Related Compounds 6Eilyza AballaNo ratings yet
- Industrial Acids: I. Properties of AcidsDocument4 pagesIndustrial Acids: I. Properties of AcidsEilyza AballaNo ratings yet
- CHE 410 4 Petroleum ProductsDocument23 pagesCHE 410 4 Petroleum ProductsEilyza AballaNo ratings yet
- Chea 432 Paints Pigmwents and Industrial CoatingsDocument63 pagesChea 432 Paints Pigmwents and Industrial CoatingsEilyza AballaNo ratings yet
- Che 410 Glass Industry 7Document11 pagesChe 410 Glass Industry 7Eilyza AballaNo ratings yet
- NFLX 2019 10-KDocument93 pagesNFLX 2019 10-KEilyza AballaNo ratings yet
- CHE 410 Unit Process and Operations Part2Document20 pagesCHE 410 Unit Process and Operations Part2Eilyza AballaNo ratings yet
- CHE 410 Fundamentals in Plants Equipment InstrumentationDocument34 pagesCHE 410 Fundamentals in Plants Equipment InstrumentationEilyza AballaNo ratings yet
- Industrial Acids and Bases NotesDocument4 pagesIndustrial Acids and Bases NotesEilyza AballaNo ratings yet
- Otec 1Document44 pagesOtec 1Eilyza AballaNo ratings yet
- Food Expo Choconana Ice Cream DiscussionDocument3 pagesFood Expo Choconana Ice Cream DiscussionEilyza AballaNo ratings yet
- Sample/practice Exam 2010, Questions and Answers Sample/practice Exam 2010, Questions and AnswersDocument14 pagesSample/practice Exam 2010, Questions and Answers Sample/practice Exam 2010, Questions and AnswersEilyza Aballa100% (1)
- NFLX 4M 2020-6-16Document23 pagesNFLX 4M 2020-6-16Eilyza AballaNo ratings yet
- 363 P.Casal ST., Quiapo, Manila: Technological Institute of The PhilippinesDocument53 pages363 P.Casal ST., Quiapo, Manila: Technological Institute of The PhilippinesEilyza AballaNo ratings yet
- Perry-TABS - Tabs For Perry Chemical Engineer's Handbook Perry-TABS - Tabs For Perry Chemical Engineer's HandbookDocument9 pagesPerry-TABS - Tabs For Perry Chemical Engineer's Handbook Perry-TABS - Tabs For Perry Chemical Engineer's HandbookEilyza Aballa100% (1)
- Orange3 Text PDFDocument53 pagesOrange3 Text PDFfajrina rinaNo ratings yet
- A Remote Home Security System Based On Wireless Sensor Network Using GSM TechnologyDocument3 pagesA Remote Home Security System Based On Wireless Sensor Network Using GSM TechnologyAjith Kumar GSNo ratings yet
- Ibot vs. TaycoDocument2 pagesIbot vs. TaycoMishal Oisin100% (1)
- HI FOG For BuildingsDocument32 pagesHI FOG For BuildingsKaustubh BidkarNo ratings yet
- DLL in Solar SystemDocument19 pagesDLL in Solar SystemginalynNo ratings yet
- Programa FinalDocument63 pagesPrograma Finalpatmos666No ratings yet
- PoonamDocument54 pagesPoonamCH Rajan GujjarNo ratings yet
- NMKV - WikipediaDocument17 pagesNMKV - WikipediaNUTHI SIVA SANTHANNo ratings yet
- 0RBIAR0B4E1ADocument321 pages0RBIAR0B4E1AAnkur VermaNo ratings yet
- WH2009 WaterHorseCatalogDocument132 pagesWH2009 WaterHorseCatalogAiko FeroNo ratings yet
- Ib Phys DC ESQ ADocument30 pagesIb Phys DC ESQ AkjsaccsNo ratings yet
- PFW - Vol. 23, Issue 08 (August 18, 2008) Escape To New YorkDocument0 pagesPFW - Vol. 23, Issue 08 (August 18, 2008) Escape To New YorkskanzeniNo ratings yet
- Assessing The Feasibility of A New Venture 1.1 Assessment and Evaluation of Entrepreneurial OpportunitiesDocument13 pagesAssessing The Feasibility of A New Venture 1.1 Assessment and Evaluation of Entrepreneurial OpportunitiesBereket Desalegn100% (1)
- Hydrogen EconomyDocument19 pagesHydrogen EconomyEdgar Gabriel Sanchez DominguezNo ratings yet
- Product Risk Management Report For Spine ProductsDocument25 pagesProduct Risk Management Report For Spine ProductsAlejandro Landinez100% (1)
- Basic Bible SeminarDocument45 pagesBasic Bible SeminarjovinerNo ratings yet
- TCL022150e Theory Past Paper 2019 May A - Grade 3Document8 pagesTCL022150e Theory Past Paper 2019 May A - Grade 3Andrea Price100% (1)
- ObjectivesDocument3 pagesObjectivesSameer IrfanNo ratings yet
- VRF-SLB013-EN - 0805115 - Catalogo Ingles 2015 PDFDocument50 pagesVRF-SLB013-EN - 0805115 - Catalogo Ingles 2015 PDFJhon Lewis PinoNo ratings yet
- Mage Wars: Variant With No Dice: Basic RulesDocument8 pagesMage Wars: Variant With No Dice: Basic RulesMáté BoaNo ratings yet
- Lesson Plan PatrickDocument4 pagesLesson Plan PatrickPatrick Jerome SilvanoNo ratings yet
- Biostat IntroductionDocument31 pagesBiostat IntroductionteklayNo ratings yet
- Uponor Control System Radio Installation Manual Controller and Thermostats, UK, IRLDocument34 pagesUponor Control System Radio Installation Manual Controller and Thermostats, UK, IRLjamppajoo2No ratings yet
- Podman Part4Document5 pagesPodman Part4anbuchennai82No ratings yet
- Botech. IDocument37 pagesBotech. IKevin Rose BarnuevoNo ratings yet
CHEN382 - SP17 Problem Set 4 KAO
CHEN382 - SP17 Problem Set 4 KAO
Uploaded by
Eilyza AballaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
CHEN382 - SP17 Problem Set 4 KAO
CHEN382 - SP17 Problem Set 4 KAO
Uploaded by
Eilyza AballaCopyright:
Available Formats
CHEN382_SP17 Problem set 4 KAO
1. Decarboxylation0of0glyoxylate0(S)0that0occurs0in0the0mitochondria0is0inhibited0by0
malonate0(I).00Using0the0following0initial0rate0data0obtained0in0batch0experiments,0
determine0the0following:0
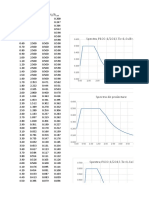
Glyox,0S0(mM)0 Rate0of0CO20evolution,0v0(mmoles/LJhour)0
0 [I]0=000 [I]0=01.260mM0 [I]0=01.950mM0
0.250 1.020 0.730 0.560
0.330 1.390 0.870 0.750
0.400 1.670 1.090 0.850
0.500 1.890 1.300 1.000
0.600 2.080 1.410 1.280
0.750 2.440 1.820 1.390
1.000 2.500 2.170 1.820
a. What0type0of0inhibition0is0this?0
b. Determine0the0constants0Vm,0Km’,0and0KI.0
0
Answer:0
0 0 0 0
4.000 0.980 1.370 1.790
3.030 0.720 1.150 1.330
2.500 0.600 0.920 1.180
2.000 0.530 0.770 1.000
1.670 0.480 0.710 0.780
1.330 0.410 0.550 0.720
1.000 0.400 0.460 0.550
0
$"!#
0
("'#
0
0
("&#
0
0
("%#
0
("$#
0
!"#$
("!#
0
0
!"'#
0
!"&#
0
0
!"%#
0
!"$#
0
!"!#
)*"!# )%"!# )+"!# )$"!# )("!# !"!# ("!# $"!# +"!# %"!# *"!#
!"%&'$
(,-#./0!1# (,-#./0("$&1# (,-#./0("2*1#
CHEN382_SP17 Problem set 4 KAO
0
2. Amyloglucosidase0from0Endomycopsis+bispora0is0immobilized0in0a0very0small0
polyacrylamide0gel0beads.00The0activities0of0immobilized0and0soluble0enzyme0are0
compared0at080ºC.00Rate0data0are0measured0at0a0fixed0substrate0concentration0with0
the0following0results.00Determine0the0halfJlife0of0each0form0of0enzyme.00Assume0
υ ~ Vm .000
0 Enzyme0Activity0(µmol0mlJ10minJ1)0
Time0(min)0 Soluble0enzyme0 Immobilized0enzyme0
00 0.860 0.450
30 0.790 0.440
60 0.700 0.430
90 0.650 0.430
150 0.580 0.410
200 0.460 0.400
250 0.410 0.390
300 J0 0.380
400 J0 0.370
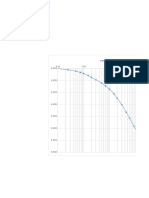
3. αJAmylase0from0malt0is0used0to0hydrolyze0starch.00The0dependence0of0the0initial0
reaction0rate0on0temperature0is0determined0experimentally.00Results0measured0at0
fixed0starch0and0enzyme0concentrations0are0listed0in0the0following0table.00R0=01.990cal0
gmolJ10KJ1.00Assume0 υ ~ Vm .0
0
Temperature)(ºC)) Rate)of)glucose)production)(mmol)m83)s81))
) )
200 0.310
300 0.660
400 1.200
600 6.330
a. Determine0the0activation0energy0for0this0reaction.0
b. αJAmylase0is0used0to0break0down0starch0in0baby0food.00It0is0proposed0to0
conduct0the0reaction0at0a0relatively0high0temperature0so0that0the0viscosity0is0
reduced.00What0is0the0reaction0rate0at055ºC0compared0with025ºC?0
c. Thermal0deactivation0of0this0enzyme0is0described0by0the0equation:0
kd = 2.25 ×10 27 e−41,630/RT
0
where0kd0is0the0deactivation0rate0constant0in0hJ1,0R0is0the0ideal0gas0constant0in0
cal0gmolJ10KJ1,0and0T0is0temperature0in0K.00What0is0the0halfJlife0of0the0enzyme0
at055ºC0compared0with025ºC?00Which0of0these0two0operating0temperatures0is0
more0practical0for0processing0baby0food?00Explain.0
0
0
CHEN382_SP17 Problem set 4 KAO
4. Enzyme0E0catalyzes0the0reaction0S0!0P0and0has0a0Km’0of050x010J50M0and0a0Vmax0of0250
µM/min.00Compound0I0is0an0inhibitor0for0enzyme0E.00There0is0only0one0binding0pocket0
on0 the0 enzyme0 that0 can0 bind0 both0 the0 substrate0 and0 inhibitor.0 0 A0 reaction0 was0
started0with010x010J70M0of0E0and010x010J40M0of0S.00What0is0the0percent0reduction0in0the0
initial0velocity0of0the0reaction0(S0!0P)0if040x010J40M0of0I0were0added0to0the0reaction?00
The0KI0is030x010J40M.000
5. An0inhibitor0(I)0is0added0to0the0enzymatic0reaction0at0a0concentration0of01.00g/L.00The0
following0data0were0obtained.00The0Km’0=09.20g0S/L0in0the0absence0of0inhibitor.0
0
v0(g0LJ10hJ1)0 [S]0(g/L)0
0.9090 200
0.6580 100
0.4930 6.670
0.400 50
0.3330 40
0.2890 3.330
0.2270 2.50
a. Is0the0inhibitor0competitive0or0noncompetitive?0
b. Find0KI.0
6. Protease0 (an0 enzyme0 that0 breaks0 down0 proteins)0 is0 being0 inactivated0 by0 UV0 light.00
The0 deactivation0 is0 assumed0 to0 be0 a0 first0 order0 reaction0 with0 deactivation0 rate0
constant0 =0 0.050 minJ1.0 0 A0 protein0 degradation0 reaction0 was0 started0 with0 10mM0 of0
enzyme0 and0 1000 mM0 of0 substrate.0 0 The0 catalytic0 constant0 for0 the0 reaction0 is0 0.030
minJ10 and0 the0 Michaelis0 constant0 is0 50mM.0 0 0 After0 10 hour0 of0 UV0 irradiation,0 how0
much0undegraded0protein0(substrate)0is0left?000
7. An0enzyme0ATPase0has0a0molecular0weight0of050x01040daltons,0a0KM0value0of010J40M,0
and0 a0 k20 value0 of0 1040 molecules0 of0 ATP/(min*molecule0 of0 enzyme)0 at0 37ºC.0 0 The0
reaction0catalyzed0is0the0following:0
ATP0(with0ATPase)0!0ADP0+0Pi0
0 which0can0also0be0represented0as:0
E0+0S0⇔0ES0!0E0+0P0
where0 S0 is0 ATP.0 0 The0 enzyme0 at0 this0 temperature0 is0 unstable.0 0 The0 enzyme0
inactivation0kinetcs0are0first0order:0
[Ea]0=0[Eo]exp(Jkdt)0
where0[Eo]0is0the0initial0enzyme0concentration0and0kd0=00.10minJ1.00In0an0experiment0
with0 a0 partially0 pure0 enzyme0 preparation,0 100 µg0 of0 total0 crude0 protein0 (containing0
enzyme)0is0added0to0a010ml0reaction0mixture0containing00.020M0ATP0and0incubated0at0
37ºC.00After0120hours0the0reaction0ends0(i.e.,0t0!0∞)0and0the0inorganic0phosphate0(Pi)0
concentration0is0found0to0be00.0020M,0which0was0initially0zero.00What0fraction0of0the0
crude0protein0preparation0was0the0enzyme?00Hint:00Since0[S]>>Km,0the0reaction0rate0
can0be0represented0by:0
0 0 d[P]/dt0=0k2[Ea]0
0
CHEN382_SP17 Problem set 4 KAO
8. Show0why0d[P]/dt0=0k2[Ea]0when0[S]>>Km.00
0
9. Lipase0is0being0investigated0as0an0additive0to0laundry0detergent0for0removal0of0stains0
from0fabric.00The0general0reaction0is:0
0
Fats0!0fatty0acids0+0glycerol0
0
The0 MichaelisJMenten0 constant0 for0 pancreatic0 lipase0 is0 50 mM.0 0 At0 60ºC,0 lipase0 is0
subject0 to0 deactivation0 with0 a0 halfJlife0 of0 80 min.0 0 Fat0 hydrolysis0 is0 carried0 out0 in0 a0
wellJmixed0batch0reactor0that0simulates0a0topJloading0washing0machine.00The0initial0
fat0 concentration0 is0 450 gmol0 mJ3.0 0 At0 the0 beginning0 of0 the0 reaction,0 the0 rate0 of0
hydrolysis0is00.070mmol0LJ10sJ1.00How0long0does0it0take0for0the0enzyme0to0hydrolyze0
80%0of0the0fat0present?0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
0
You might also like
- Toshiba Case 3Document4 pagesToshiba Case 3Deta Detade100% (1)
- BS 57 (1951)Document23 pagesBS 57 (1951)dapsiduNo ratings yet
- Part 4 Grade 6 Edumate Introduction On MelodyDocument3 pagesPart 4 Grade 6 Edumate Introduction On MelodyAndrewOribianaNo ratings yet
- BP Intern InterviewDocument5 pagesBP Intern InterviewSergey ParsegovNo ratings yet
- Ic 01 Last Day Test 3Document30 pagesIc 01 Last Day Test 3Sohini0% (1)
- Glass Railing - Structural CalculationDocument36 pagesGlass Railing - Structural CalculationHSY79No ratings yet
- Espectro de AceleracionDocument1 pageEspectro de AceleracionYanely FalcònNo ratings yet
- Sistema: 1-Propanol (A) - Agua (B) : Práctica #9. Equilibrio Líquido - VaporDocument22 pagesSistema: 1-Propanol (A) - Agua (B) : Práctica #9. Equilibrio Líquido - Vaporwaldir marcelinoNo ratings yet
- Simulación Reacción EDocument67 pagesSimulación Reacción EMilagros Morales VergarayNo ratings yet
- Calibration LinearDocument15 pagesCalibration LinearThusith WijayawardenaNo ratings yet
- CostsDocument6 pagesCostsChalaNo ratings yet
- Thermometer Indikator : Penunjukan Alat Standard Koreksi Ketidakpastian (°C) (°C) (°C) (°C)Document20 pagesThermometer Indikator : Penunjukan Alat Standard Koreksi Ketidakpastian (°C) (°C) (°C) (°C)lailiNo ratings yet
- 2023-08-24 Ukur Debit Current Meter DI KarangtalunDocument15 pages2023-08-24 Ukur Debit Current Meter DI KarangtalunrahmawatiNo ratings yet
- Spectru ProiectareDocument27 pagesSpectru ProiectareHoria Ionut MihneaNo ratings yet
- Spectru ProiectareDocument27 pagesSpectru ProiectareHoria Ionut MihneaNo ratings yet
- Loading: Displacement / Micrometer Force / Micro NewtonDocument9 pagesLoading: Displacement / Micrometer Force / Micro NewtonasamNo ratings yet
- Blade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsDocument3 pagesBlade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsBülent KabadayiNo ratings yet
- Evaluacion de La Integral de Duhamel para Cargas SismicasDocument6 pagesEvaluacion de La Integral de Duhamel para Cargas SismicasAngela Odría RubioNo ratings yet
- VLE Using UNIFAC in ExcelDocument55 pagesVLE Using UNIFAC in Exceljdgh1986No ratings yet
- LebensmittelDocument582 pagesLebensmittelmaria budiNo ratings yet
- Blade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsDocument3 pagesBlade Design Spreadsheet: 7 2.40 2 4 0.8 6 Blue CellsbadmaNo ratings yet
- Group 7 - Data SheetDocument9 pagesGroup 7 - Data SheetJeremy Kyle Edson AustriaNo ratings yet
- Tablas de TermodinamicaDocument55 pagesTablas de TermodinamicadeymarNo ratings yet
- P' (KN/m2)Document1 pageP' (KN/m2)Andriie DassieNo ratings yet
- Physical Organic 1 Post-LabDocument7 pagesPhysical Organic 1 Post-LabsamNo ratings yet
- Blade Design Spreadsheet Shape: 7 2.40 2 4 0.8 6 Blue CellsDocument2 pagesBlade Design Spreadsheet Shape: 7 2.40 2 4 0.8 6 Blue CellsOptimuz TsNo ratings yet
- Feeding Program FixDocument9 pagesFeeding Program FixLo Christian DjunaidiNo ratings yet
- Calculating-ISO-7886-TOGCDocument9 pagesCalculating-ISO-7886-TOGCﺵﻓﻴﺮﺍ ﺭﺷﻲ ﺍﻧﺎNo ratings yet
- 001 AggregateDocument4 pages001 Aggregatevanessa ImlerNo ratings yet
- Laporan Modul 5Document4 pagesLaporan Modul 5Adryan Fathur RahmanNo ratings yet
- Cut Off PointDocument8 pagesCut Off Pointfiruzz1No ratings yet
- Factor XP 02Document2 pagesFactor XP 02Javier FcNo ratings yet
- Chart TitleDocument11 pagesChart Titlemuhammad alfatihNo ratings yet
- Member Name Member # Steel N Total Bar (#) HA Doux Bar L (CM) Total L (CM) Poids/mLDocument269 pagesMember Name Member # Steel N Total Bar (#) HA Doux Bar L (CM) Total L (CM) Poids/mLYoumna ShatilaNo ratings yet
- Ejercicio 1 Metodos NumericosDocument17 pagesEjercicio 1 Metodos Numericosjader florezNo ratings yet
- Assignment 2 Final Version1Document78 pagesAssignment 2 Final Version1Christopher Bayo DaramolaNo ratings yet
- Armaduras en 2D PerfectDocument48 pagesArmaduras en 2D Perfectjosluis fNo ratings yet
- N FourierDocument6 pagesN FourierZokiNo ratings yet
- Επιφάνεια Βαφής Και Βάρος Δοκών Μεταλλικής ΚατασκευήςDocument27 pagesΕπιφάνεια Βαφής Και Βάρος Δοκών Μεταλλικής ΚατασκευήςTheodoros AtheridisNo ratings yet
- EstadisticaDocument2 pagesEstadisticaJuan BonifazNo ratings yet
- Spun 500 Load Settlement CurveDocument2 pagesSpun 500 Load Settlement Curveyin hoe ongNo ratings yet
- Dow Engineering InformationDocument29 pagesDow Engineering InformationbenakiaNo ratings yet
- Loading: Displacement / Micrometer Force / Micro NewtonDocument11 pagesLoading: Displacement / Micrometer Force / Micro NewtonasamNo ratings yet
- Loading: Displacement / Micrometer Force / Micro NewtonDocument7 pagesLoading: Displacement / Micrometer Force / Micro NewtonasamNo ratings yet
- Vrednosti Odgovora U Desetinama Pojedinih Delova SpektraDocument4 pagesVrednosti Odgovora U Desetinama Pojedinih Delova SpektraNiko NištićNo ratings yet
- Plan Is Acceptable: Sample Size 200 Acceptable Quality Level 1 % Alpha 5 % Unacceptable Quality Level 5 % Beta 5 %Document2 pagesPlan Is Acceptable: Sample Size 200 Acceptable Quality Level 1 % Alpha 5 % Unacceptable Quality Level 5 % Beta 5 %Cellulose NitroNo ratings yet
- Curva Fe FabianDocument11 pagesCurva Fe FabianFabian RodriguezNo ratings yet
- Hasil Regresi Soal No.2 Fix Tinggal (P)Document3 pagesHasil Regresi Soal No.2 Fix Tinggal (P)Neiscya Dhitya AmritaNo ratings yet
- Dow Water Solutions DOWEX™ Ion Exchange Resins and FILMTEC™ Membranes Engineering InformationDocument29 pagesDow Water Solutions DOWEX™ Ion Exchange Resins and FILMTEC™ Membranes Engineering Informationirshad sondeNo ratings yet
- Specifikacija paraDocument5 pagesSpecifikacija paraLjiljana Radović BukaNo ratings yet
- Reserve Estimation Unit: Ooip Recov Oil (MMSTB) (MMSTB)Document8 pagesReserve Estimation Unit: Ooip Recov Oil (MMSTB) (MMSTB)FreddyCorredorNo ratings yet
- Variograma Regular: DistanciasDocument3 pagesVariograma Regular: DistanciasyulianNo ratings yet
- Experiment 1: Physical Properties of Water ObjectivesDocument12 pagesExperiment 1: Physical Properties of Water ObjectivesWan XïnNo ratings yet
- Datos: T/TP Q/QP TDocument9 pagesDatos: T/TP Q/QP TManuel CerveraNo ratings yet
- 5.1 Holding Period ReturnDocument48 pages5.1 Holding Period ReturnSrilekha BasavojuNo ratings yet
- Other Numerical Integration ExamplesDocument19 pagesOther Numerical Integration Examplessiswanto.micoNo ratings yet
- FractionatorsDocument9 pagesFractionatorsjoker princeNo ratings yet
- 2 Absorvs ConcentrationDocument2 pages2 Absorvs ConcentrationKristin SheafferNo ratings yet
- Movimeinto de Tierras Ramon CastillaDocument19 pagesMovimeinto de Tierras Ramon CastillaGiancarlos AguinagaNo ratings yet
- Consol Grades Per BlankDocument2 pagesConsol Grades Per BlankSunddee S RomagosNo ratings yet
- LillieforsDocument2 pagesLillieforsGebz Ai BianNo ratings yet
- Polar Fineza: Axis Title Axis TitleDocument4 pagesPolar Fineza: Axis Title Axis TitleUriel Gallegos PérezNo ratings yet
- Grupo Hogares Ingreso % Acumulado de Hogares % Acumulado Del IngresoDocument6 pagesGrupo Hogares Ingreso % Acumulado de Hogares % Acumulado Del IngresoLIZETH KATHERIN MONROY MACIASNo ratings yet
- Class Customer Count Unit Consumed Basic Tariff Service Charge Street Light LevyDocument3 pagesClass Customer Count Unit Consumed Basic Tariff Service Charge Street Light LevySankalp TiwariNo ratings yet
- O Tec Reference 4Document5 pagesO Tec Reference 4Eilyza AballaNo ratings yet
- Soap Detergents and Related Compounds 6Document10 pagesSoap Detergents and Related Compounds 6Eilyza AballaNo ratings yet
- Industrial Acids: I. Properties of AcidsDocument4 pagesIndustrial Acids: I. Properties of AcidsEilyza AballaNo ratings yet
- CHE 410 4 Petroleum ProductsDocument23 pagesCHE 410 4 Petroleum ProductsEilyza AballaNo ratings yet
- Chea 432 Paints Pigmwents and Industrial CoatingsDocument63 pagesChea 432 Paints Pigmwents and Industrial CoatingsEilyza AballaNo ratings yet
- Che 410 Glass Industry 7Document11 pagesChe 410 Glass Industry 7Eilyza AballaNo ratings yet
- NFLX 2019 10-KDocument93 pagesNFLX 2019 10-KEilyza AballaNo ratings yet
- CHE 410 Unit Process and Operations Part2Document20 pagesCHE 410 Unit Process and Operations Part2Eilyza AballaNo ratings yet
- CHE 410 Fundamentals in Plants Equipment InstrumentationDocument34 pagesCHE 410 Fundamentals in Plants Equipment InstrumentationEilyza AballaNo ratings yet
- Industrial Acids and Bases NotesDocument4 pagesIndustrial Acids and Bases NotesEilyza AballaNo ratings yet
- Otec 1Document44 pagesOtec 1Eilyza AballaNo ratings yet
- Food Expo Choconana Ice Cream DiscussionDocument3 pagesFood Expo Choconana Ice Cream DiscussionEilyza AballaNo ratings yet
- Sample/practice Exam 2010, Questions and Answers Sample/practice Exam 2010, Questions and AnswersDocument14 pagesSample/practice Exam 2010, Questions and Answers Sample/practice Exam 2010, Questions and AnswersEilyza Aballa100% (1)
- NFLX 4M 2020-6-16Document23 pagesNFLX 4M 2020-6-16Eilyza AballaNo ratings yet
- 363 P.Casal ST., Quiapo, Manila: Technological Institute of The PhilippinesDocument53 pages363 P.Casal ST., Quiapo, Manila: Technological Institute of The PhilippinesEilyza AballaNo ratings yet
- Perry-TABS - Tabs For Perry Chemical Engineer's Handbook Perry-TABS - Tabs For Perry Chemical Engineer's HandbookDocument9 pagesPerry-TABS - Tabs For Perry Chemical Engineer's Handbook Perry-TABS - Tabs For Perry Chemical Engineer's HandbookEilyza Aballa100% (1)
- Orange3 Text PDFDocument53 pagesOrange3 Text PDFfajrina rinaNo ratings yet
- A Remote Home Security System Based On Wireless Sensor Network Using GSM TechnologyDocument3 pagesA Remote Home Security System Based On Wireless Sensor Network Using GSM TechnologyAjith Kumar GSNo ratings yet
- Ibot vs. TaycoDocument2 pagesIbot vs. TaycoMishal Oisin100% (1)
- HI FOG For BuildingsDocument32 pagesHI FOG For BuildingsKaustubh BidkarNo ratings yet
- DLL in Solar SystemDocument19 pagesDLL in Solar SystemginalynNo ratings yet
- Programa FinalDocument63 pagesPrograma Finalpatmos666No ratings yet
- PoonamDocument54 pagesPoonamCH Rajan GujjarNo ratings yet
- NMKV - WikipediaDocument17 pagesNMKV - WikipediaNUTHI SIVA SANTHANNo ratings yet
- 0RBIAR0B4E1ADocument321 pages0RBIAR0B4E1AAnkur VermaNo ratings yet
- WH2009 WaterHorseCatalogDocument132 pagesWH2009 WaterHorseCatalogAiko FeroNo ratings yet
- Ib Phys DC ESQ ADocument30 pagesIb Phys DC ESQ AkjsaccsNo ratings yet
- PFW - Vol. 23, Issue 08 (August 18, 2008) Escape To New YorkDocument0 pagesPFW - Vol. 23, Issue 08 (August 18, 2008) Escape To New YorkskanzeniNo ratings yet
- Assessing The Feasibility of A New Venture 1.1 Assessment and Evaluation of Entrepreneurial OpportunitiesDocument13 pagesAssessing The Feasibility of A New Venture 1.1 Assessment and Evaluation of Entrepreneurial OpportunitiesBereket Desalegn100% (1)
- Hydrogen EconomyDocument19 pagesHydrogen EconomyEdgar Gabriel Sanchez DominguezNo ratings yet
- Product Risk Management Report For Spine ProductsDocument25 pagesProduct Risk Management Report For Spine ProductsAlejandro Landinez100% (1)
- Basic Bible SeminarDocument45 pagesBasic Bible SeminarjovinerNo ratings yet
- TCL022150e Theory Past Paper 2019 May A - Grade 3Document8 pagesTCL022150e Theory Past Paper 2019 May A - Grade 3Andrea Price100% (1)
- ObjectivesDocument3 pagesObjectivesSameer IrfanNo ratings yet
- VRF-SLB013-EN - 0805115 - Catalogo Ingles 2015 PDFDocument50 pagesVRF-SLB013-EN - 0805115 - Catalogo Ingles 2015 PDFJhon Lewis PinoNo ratings yet
- Mage Wars: Variant With No Dice: Basic RulesDocument8 pagesMage Wars: Variant With No Dice: Basic RulesMáté BoaNo ratings yet
- Lesson Plan PatrickDocument4 pagesLesson Plan PatrickPatrick Jerome SilvanoNo ratings yet
- Biostat IntroductionDocument31 pagesBiostat IntroductionteklayNo ratings yet
- Uponor Control System Radio Installation Manual Controller and Thermostats, UK, IRLDocument34 pagesUponor Control System Radio Installation Manual Controller and Thermostats, UK, IRLjamppajoo2No ratings yet
- Podman Part4Document5 pagesPodman Part4anbuchennai82No ratings yet
- Botech. IDocument37 pagesBotech. IKevin Rose BarnuevoNo ratings yet