Professional Documents
Culture Documents
LP F060 CH 6 X 10 ML LP F125 CH 5 X 25 ML: in Vitro Diagnostic Medical Device
LP F060 CH 6 X 10 ML LP F125 CH 5 X 25 ML: in Vitro Diagnostic Medical Device
Uploaded by
Meditech visionbdCopyright:
Available Formats
You might also like
- BARKOVICH Pediatric Neuroimaging 5Document1,145 pagesBARKOVICH Pediatric Neuroimaging 5Pepe Ars100% (1)
- General Zoology SyllabusDocument4 pagesGeneral Zoology SyllabusNL R Q DO100% (4)
- A Amylase DirectDocument1 pageA Amylase DirectRisqon Anjahiranda Adiputra0% (1)
- Α Α Α Α-Amylase-Eps: Biosystems S.ADocument1 pageΑ Α Α Α-Amylase-Eps: Biosystems S.ARisqon Anjahiranda AdiputraNo ratings yet
- Drug Discovery and DevelopmentDocument47 pagesDrug Discovery and DevelopmentSalahBensaber100% (3)
- Latihan Soal Recount TextDocument14 pagesLatihan Soal Recount TextMuhammad Anshory100% (2)
- Manuali PDF 414Document1 pageManuali PDF 414Rakib Hossain 3A-159No ratings yet
- Lipase Spinreact 1x24 ML, 1X48 MLDocument2 pagesLipase Spinreact 1x24 ML, 1X48 MLN. K. MandilNo ratings yet
- Manuali PDF 413Document1 pageManuali PDF 413Rakib Hossain 3A-159No ratings yet
- Clonatest Amylase MRDocument4 pagesClonatest Amylase MRSuprovet LabotatorioNo ratings yet
- 1.alpha AmylaseDocument2 pages1.alpha AmylaseHiếu Chí PhanNo ratings yet
- 1105000I Rev. 02Document2 pages1105000I Rev. 02Riadh BenyoucefNo ratings yet
- Pi e LDH 21 Ifcc 3Document2 pagesPi e LDH 21 Ifcc 3Osama Ben DawNo ratings yet
- Alp Mono VialsDocument3 pagesAlp Mono VialsN. K. MandilNo ratings yet
- Lipase (S.L) : ADL/V.02/060114Document1 pageLipase (S.L) : ADL/V.02/060114Aniket dubeyNo ratings yet
- Budi Altgpt - Doc NewDocument3 pagesBudi Altgpt - Doc NewIrvanda ENVIOUSNo ratings yet
- CK MB enDocument1 pageCK MB enShakilur IslamNo ratings yet
- Applications Mindray BS-300 en 06-07-2010Document43 pagesApplications Mindray BS-300 en 06-07-2010dediNo ratings yet
- Lip 110 - en - Ru - CZ - DDocument5 pagesLip 110 - en - Ru - CZ - DMatibar RahmanNo ratings yet
- Ferritin Ing Rev. 03Document2 pagesFerritin Ing Rev. 03Yousra NanoNo ratings yet
- Ga4910 00 - Got-AstDocument2 pagesGa4910 00 - Got-AstTrần Thanh ViệnNo ratings yet
- Uric Acid: (Uricase / Pod Method)Document1 pageUric Acid: (Uricase / Pod Method)Dharmesh Patel100% (3)
- KR10150 Rev02Document2 pagesKR10150 Rev02KOUAME EDYMAIN FRANCISNo ratings yet
- Lyphase 11760I-Reagent A 510 Reagent B 300 Sample 9 Standard 113Document1 pageLyphase 11760I-Reagent A 510 Reagent B 300 Sample 9 Standard 113mahinNo ratings yet
- Crea 500 - en - Ru - CZ - SK - CDocument5 pagesCrea 500 - en - Ru - CZ - SK - CLouis MiuNo ratings yet
- PDF Acid Phosphatase Acp - CompressDocument1 pagePDF Acid Phosphatase Acp - CompressRizki YuliaNo ratings yet
- Bedtt19 Lipase - LQDocument2 pagesBedtt19 Lipase - LQrankedmlbbNo ratings yet
- PHP UYjhb 7Document2 pagesPHP UYjhb 7BerhaneNo ratings yet
- Aspartate Aminotransferase (AST-GOT) - ColorimetricDocument2 pagesAspartate Aminotransferase (AST-GOT) - ColorimetricGuneyden Guneyden0% (1)
- Measure AmyDocument2 pagesMeasure Amytuan vănNo ratings yet
- Pi e AP Ifcc 2Document2 pagesPi e AP Ifcc 2muhammadsufyanfp12No ratings yet
- Acid Phosphatase (Acp)Document1 pageAcid Phosphatase (Acp)Risqon Anjahiranda AdiputraNo ratings yet
- BIOLABO - Applications Humastar 180Document32 pagesBIOLABO - Applications Humastar 180Nguyễn Bảo Huân100% (1)
- Colorimetric-Kinetic: Clinical Significance Materials Required But Not ProvidedDocument2 pagesColorimetric-Kinetic: Clinical Significance Materials Required But Not ProvidedNavin kumarNo ratings yet
- 1116005I Rev. 02Document2 pages1116005I Rev. 02kirubel demelashNo ratings yet
- 1133505I Rev. 04Document2 pages1133505I Rev. 04Nguyễn HuynhNo ratings yet
- AMYLASE CNPG Liquiform: InsertDocument6 pagesAMYLASE CNPG Liquiform: Insertsarajuddin sarajNo ratings yet
- Crea 500 S - en - Ru - CZ - SK - B1Document5 pagesCrea 500 S - en - Ru - CZ - SK - B1Louis MiuNo ratings yet
- Budi Creatinine - Doc NewDocument3 pagesBudi Creatinine - Doc NewIrvanda ENVIOUSNo ratings yet
- SGOT LyphoDocument2 pagesSGOT LyphoNonameNo ratings yet
- SGOT Lypho DikonversiDocument3 pagesSGOT Lypho DikonversiNonameNo ratings yet
- Ck-Nac FsDocument2 pagesCk-Nac Fsadamalay wardiwiraNo ratings yet
- ALP Single ReagentDocument2 pagesALP Single ReagentJames 'jps' SimanjuntakNo ratings yet
- IFU - BX e CK 2Document6 pagesIFU - BX e CK 2betsabevegaaNo ratings yet
- Creatine Kinase (CK)Document2 pagesCreatine Kinase (CK)Mira SafwatNo ratings yet
- Lipase: Calib. Conc. Check Vial LabelDocument2 pagesLipase: Calib. Conc. Check Vial LabelDinesh SreedharanNo ratings yet
- CT10152 Creatinine EDocument2 pagesCT10152 Creatinine ELaboratorio BianucciNo ratings yet
- 11592i PDFDocument1 page11592i PDFKristoforusNo ratings yet
- GGT InsertDocument2 pagesGGT InsertsharmashyamsinghNo ratings yet
- Gpt-11-Alt (GPT) Uv 2V-02,12,2014 - 5Document2 pagesGpt-11-Alt (GPT) Uv 2V-02,12,2014 - 5dung nguyen danhNo ratings yet
- Amylase enDocument1 pageAmylase enRakib Hossain 3A-159No ratings yet
- PotasiumDocument2 pagesPotasiumlabextreme29No ratings yet
- AlkPhos FL 51 SFBC TV-005-CE-003Document1 pageAlkPhos FL 51 SFBC TV-005-CE-003Blas Cuba SanchezNo ratings yet
- ACCENT-200 ALAT ADocument2 pagesACCENT-200 ALAT Aemc.medicalserviceNo ratings yet
- CALCIUM CPC+STD ANG 2015 02 17Document2 pagesCALCIUM CPC+STD ANG 2015 02 17yehiwal756No ratings yet
- AlkPhos FL 51 DGKC TV-004-CE-003Document1 pageAlkPhos FL 51 DGKC TV-004-CE-003Blas Cuba SanchezNo ratings yet
- Alpha-Amylase: For In-Vitro Diagnostic and Professional Use OnlyDocument2 pagesAlpha-Amylase: For In-Vitro Diagnostic and Professional Use OnlyPark YuriNo ratings yet
- 1141010I Rev. 02Document2 pages1141010I Rev. 02Nguyễn HuynhNo ratings yet
- Alp RandoxDocument2 pagesAlp Randoxabdulrahmanaminu0199No ratings yet
- Budi-ASTGOT docNEWDocument3 pagesBudi-ASTGOT docNEWIrvanda ENVIOUSNo ratings yet
- SGPTDocument2 pagesSGPTNeña Lozada TabinasNo ratings yet
- Alkaline Phosphatase FS : Order Information SpecimenDocument3 pagesAlkaline Phosphatase FS : Order Information SpecimenmnemonicsNo ratings yet
- Metodo Cuantificar LipasaDocument1 pageMetodo Cuantificar Lipasa1718h9798s100% (1)
- Practical Handbook of Pharmaceutical Chemistry for M.PharmFrom EverandPractical Handbook of Pharmaceutical Chemistry for M.PharmNo ratings yet
- Infinosis HCG - IN027702 - enDocument2 pagesInfinosis HCG - IN027702 - enMeditech visionbdNo ratings yet
- Infinosis Total IgE IN067705 enDocument2 pagesInfinosis Total IgE IN067705 enMeditech visionbdNo ratings yet
- Infinosis HbA1c IN067701 enDocument2 pagesInfinosis HbA1c IN067701 enMeditech visionbdNo ratings yet
- Infinosis T3-In017702 enDocument2 pagesInfinosis T3-In017702 enMeditech visionbdNo ratings yet
- Infinosis Troponin I IN047701 enDocument2 pagesInfinosis Troponin I IN047701 enMeditech visionbdNo ratings yet
- Infinosis NT-proBNP IN047705 enDocument2 pagesInfinosis NT-proBNP IN047705 enMeditech visionbdNo ratings yet
- Infinosis PCT IN057701 enDocument2 pagesInfinosis PCT IN057701 enMeditech visionbdNo ratings yet
- Infinosis LH IN027703 enDocument2 pagesInfinosis LH IN027703 enMeditech visionbdNo ratings yet
- Welq T KV Ri Avb ': Qvî/Qvîxi BVG T Gvzvi BVG T Wczvi BVG T Ivj Bs TDocument19 pagesWelq T KV Ri Avb ': Qvî/Qvîxi BVG T Gvzvi BVG T Wczvi BVG T Ivj Bs TMeditech visionbdNo ratings yet
- Qvî/Qvîxi BVG T Ivj Bs Köbx WelqDocument13 pagesQvî/Qvîxi BVG T Ivj Bs Köbx WelqMeditech visionbdNo ratings yet
- Biochemistry Chema, Italy: (IFCC) (IFCC) (IFCC)Document2 pagesBiochemistry Chema, Italy: (IFCC) (IFCC) (IFCC)Meditech visionbdNo ratings yet
- Price List Wholesale and Retail (June-2021)Document2 pagesPrice List Wholesale and Retail (June-2021)Meditech visionbdNo ratings yet
- M T Jute Trading: Count & Quality: 10/1 LBS 1 PLY HessianDocument4 pagesM T Jute Trading: Count & Quality: 10/1 LBS 1 PLY HessianMeditech visionbdNo ratings yet
- SUBJECT:-APPOINTMENT TO THE POST OF Executive (Sales & Marketing)Document2 pagesSUBJECT:-APPOINTMENT TO THE POST OF Executive (Sales & Marketing)Meditech visionbdNo ratings yet
- Febrile AntigenbnkggDocument4 pagesFebrile AntigenbnkggMeditech visionbdNo ratings yet
- Price Quotation For Pathological Reagents. Sir,: (IFCC) (IFCC) (IFCC)Document1 pagePrice Quotation For Pathological Reagents. Sir,: (IFCC) (IFCC) (IFCC)Meditech visionbdNo ratings yet
- 7.1 Respiratory Process in Energy ProductionDocument13 pages7.1 Respiratory Process in Energy ProductionNor Rafidah Che YusofNo ratings yet
- Sohail Reports PDFDocument2 pagesSohail Reports PDFMunazza SohailNo ratings yet
- Full Download Solution Manual For Human Anatomy Physiology Main Version 4th Edition Terry Martin Cynthia Prentice Crave PDF Full ChapterDocument35 pagesFull Download Solution Manual For Human Anatomy Physiology Main Version 4th Edition Terry Martin Cynthia Prentice Crave PDF Full Chaptergabertconsomme.gygy100% (24)
- 1.3 Main Branches of AnthropologyDocument9 pages1.3 Main Branches of AnthropologyRohanNo ratings yet
- r127092130 Gabriel Martinez CUR127092130Document2 pagesr127092130 Gabriel Martinez CUR127092130GabrielNo ratings yet
- Lessonia-Fucoreverse BrochureDocument5 pagesLessonia-Fucoreverse BrochureElaineSilvestriniNo ratings yet
- Spiritual Psychology PDFDocument5 pagesSpiritual Psychology PDFKedar Kelkar100% (2)
- Goering AAO Convocation2012LectureDocument110 pagesGoering AAO Convocation2012Lectureperrolobo73No ratings yet
- Bulleted InfoDocument6 pagesBulleted InfoNiño James AceronNo ratings yet
- Co-Enzymes Role in Metabolic PathwaysDocument23 pagesCo-Enzymes Role in Metabolic PathwaysHarini BalasubramanianNo ratings yet
- Bi-Bi RXNDocument12 pagesBi-Bi RXNbelabelawNo ratings yet
- PIIS2211124721001704Document21 pagesPIIS22111247210017041813975862yvyvNo ratings yet
- SIM TestDocument5 pagesSIM TestBundi Gonzalez MartinNo ratings yet
- Types of SoilDocument2 pagesTypes of SoilCharlon RamiscalNo ratings yet
- ANALYSIS AND DENATURATION OF PROTEINS AnswersDocument5 pagesANALYSIS AND DENATURATION OF PROTEINS AnswersdgfdgsdfgsdsdgNo ratings yet
- Histo SupDocument8 pagesHisto SupAbdoulhaleem MoNo ratings yet
- MicrobiologyDocument3 pagesMicrobiologyRavi SharmaNo ratings yet
- Performance Task QuestionsDocument4 pagesPerformance Task Questionsapi-537184012No ratings yet
- Mendel's Law of SegregationDocument6 pagesMendel's Law of Segregationhanfa zamanNo ratings yet
- Test I. Puzzle Protein: Activity No. 4.1Document2 pagesTest I. Puzzle Protein: Activity No. 4.1Osas OsasNo ratings yet
- Eshmuno S ResinDocument4 pagesEshmuno S ResinTuyền KimNo ratings yet
- Piaget's Theory of Cognitive DevelopmentDocument19 pagesPiaget's Theory of Cognitive DevelopmentGuru Khb PblNo ratings yet
- Chapter 11Document11 pagesChapter 11christian anchetaNo ratings yet
- Mep 2 Assesemnt CC NewDocument12 pagesMep 2 Assesemnt CC NewPrecious Bardon-MempinNo ratings yet
- Chapter 1 Textile IndustryDocument9 pagesChapter 1 Textile IndustryAlice ChepkuruiNo ratings yet
- Clostridium, Called "TU-103", That Can Convert Nearly Any Form ofDocument1 pageClostridium, Called "TU-103", That Can Convert Nearly Any Form ofRiaNo ratings yet
LP F060 CH 6 X 10 ML LP F125 CH 5 X 25 ML: in Vitro Diagnostic Medical Device
LP F060 CH 6 X 10 ML LP F125 CH 5 X 25 ML: in Vitro Diagnostic Medical Device
Uploaded by
Meditech visionbdOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
LP F060 CH 6 X 10 ML LP F125 CH 5 X 25 ML: in Vitro Diagnostic Medical Device
LP F060 CH 6 X 10 ML LP F125 CH 5 X 25 ML: in Vitro Diagnostic Medical Device
Uploaded by
Meditech visionbdCopyright:
Available Formats
TEST PROCEDURE
LIPASE FL REFERENCES
Tietz Textbook of Clinical Chemistry and Molecular Dia-
Wavelength: 580 nm gnostics, Sixth Edition, Rifai-Horvath-Wittwer (2017) 421-
LP F060 CH 6 x 10 ml Lightpath: 1 cm 424
LP F125 CH 5 x 25 ml Temperature: 37°C Tietz N. and Shuey DF. - Clin. Chem. 1993, 39, 746-756
dispense: blank calibrator sample MANUFACTURER
INTENDED USE reagent R1 1 ml 1 ml 1 ml Chema Diagnostica
Reagent for quantitative in vitro determination of lipase in water 20 µl - - Via Campania 2/4
biological fluids. 60030 Monsano (AN) - ITALY - EU
calibrator - 20 µl -
phone +39 0731 605064
SUMMARY OF TEST sample - - 20 µl fax +39 0731 605672
Human lipase is a glycoprotein with a molecular weight e-mail: mail@chema.com
of 48000 and an isoelectric point of about 5.8. For full Mix carefully (do not shake), incubate at 37°C for 5 minutes.
website: http://www.chema.com
catalytic activity and greatest specificity, the presence
of bile salts and a cofactor, called colipase, is required. dispense: blank calibrator sample SYMBOLS
Lipase measurement on serum, plasma, and ascitic and reagent R2 250 µl 250 µl 250 µl
pleural fluid is used to investigate pancreatic disorders,
Mix, execute a first reading of absorbance after 2 minute, in vitro diagnostic medical device
usually pancreatitis.
incubating at 37°C. Perform other 2 readings at 60
batch code
PRINCIPLE OF THE METHOD seconds intervals. Calculate the ∆A/min.
The colorimetric substrate 1,2-O-Dilauryl-rac-glycero-3- catalogue number
glutaric acid-(6’methyl-resorufin)-ester is cleaved by pan- RESULTS CALCULATION
temperature limit
creatic lipase and the resulting dicarboxilic acid ester is ∆A/min = ∆A/min(calibrator or sample) - ∆A/min(blank)
hydrolysed under the alkaline test conditions to yield the use by date
chromophore methylresorufin. The kinetic of colour forma- serum/plasma sample: caution
tion at 580 nm is monitored and it is proportional to lipase
∆A/min(sample) consult instructions for use
activity in sample. U/l (methylresorufin 37°C) = x Calibrator value
KIT COMPONENTS ∆A/min(calibrator)
For in vitro diagnostic use only.
The components of the kit are stable until expiration date EXPECTED VALUES
on the label. Keep away from direct light sources. normal subjects: ≤ 60 U/l (methylresorufin 37°C)
LIP R1 F060: 6 x 8 ml (liquid) blue cap Each laboratory should establish appropriate reference
F125: 4 x 25 ml (liquid) blue cap intervals related to its population.
Composition: Good’s Buffer pH 8.0, colipase ≥ 1 mg/l, QUALITY CONTROL AND CALIBRATION
desoxycholate ≥ 1.0 mM, taurodesoxycholate ≥ 1.0 mM, It is suggested to perform an internal quality control. For
calcium ions ≥ 1 mM, detergent and preservative. this purpose the following human based control sera are
available:
LIP R2 F060: 1 x 12 ml (liquid) red cap QUANTINORM CHEMA
F125: 1 x 25 ml (liquid) redcap with normal or close to normal control values
QUANTIPATH CHEMA
Composition: Tartrate buffer pH 4.0, lipase substrate ≥ 0.1 with pathological control values.
mM, stabilizer and preservative. If required, a multiparametric, human based calibrator is
available:
Calibrator: lyophilized (value on label) - 3 ml AUTOCAL H
Store all components at 2-8°C and do not freeze. Please contact Customer Care for further information.
MATERIALS REQUIRED BUT NOT SUPPLIED TEST PERFORMANCE
Current laboratory instrumentation. Spectrophotometer
Linearity
UV/VIS with thermostatic cuvette holder. Automatic micro-
The method is linear up to 300 U/l.
pipettes. Glass or high quality polystyrene cuvettes. Saline
If the limit value is exceeded, it is suggested to dilute
solution.
sample 1+1 with saline solution (9 g/l) and to repeat the
REAGENT PREPARATION test, multiplying the result by 2.
Use separate reagents ready to use.
Stability: up to expiration date on labels at 2-8°C; Sensitivity/limit of detection (LOD)
Stability since first opening of vials: use preferably within The limit of detection is 1 U/l.
60 days at 2-8°C.
Caution: reagent R2 is a microemulsion. Therefore, a Interferences
slight apparent precipitation could occur, showing a light No interference was observed by the presence of:
red deposit on the bottom of vial. It is a normal behaviour ascorbic acid ≤ 50 mg/dl
and it is recommended to resuspend solution before analy- hemoglobin ≤ 400 mg/dl
sis, with a mild shaking. bilirubin ≤ 50 mg/dl
lipids ≤ 1000 mg/dl
PRECAUTIONS
LIP R1: It is not classified as hazardous. Precision
intra-assay (n=10) mean (U/l) SD (U/l) CV%
LIP R2: Danger. Causes serious eye damage (H318). sample 1 49.9 0.65 1.30
Wear protective gloves. Eye protection (P280). sample 2 110.5 1.69 1.53
IF IN EYES: Rinse cautiously with water for
several minutes. Remove contact lenses, if pre- inter-assay (n=20) mean (U/l) SD (U/l) CV%
sent and easy to do. Continue rinsing (P305+P351+P338). sample 1 50.0 1.43 2.87
Immediately call a doctor (P310). sample 2 110.9 3.91 3.53
Calibrator: It is not classified as hazardous. Methods comparison
A comparison between Chema and a commercially availa-
Some commercial reagents for triglycerides, HDL and ble product gave the following results:
LDL determination could contain microbial lipases, whose
could stick on surface of instrument plastic cuvettes. It Lipase Chema = y
is recommended to program a “wash” procedure before Lipase competitor = x
lipase determination, if a contamination is suspected. n = 94
Perform the test according to the general “Good Labora-
tory Practice” (GLP) guidelines. y = 0.93 x + 5.50 U/l r2 = 0.99
SPECIMEN WASTE DISPOSAL
Serum, plasma heparinate. This product is made to be used in professional labora-
Samples are stable 7 days at 2-8°C. tories.
P501: Dispose of contents according to national/interna-
tional regulations.
IUS-7.5 EN rev. 26/04/2021 ©
You might also like
- BARKOVICH Pediatric Neuroimaging 5Document1,145 pagesBARKOVICH Pediatric Neuroimaging 5Pepe Ars100% (1)
- General Zoology SyllabusDocument4 pagesGeneral Zoology SyllabusNL R Q DO100% (4)
- A Amylase DirectDocument1 pageA Amylase DirectRisqon Anjahiranda Adiputra0% (1)
- Α Α Α Α-Amylase-Eps: Biosystems S.ADocument1 pageΑ Α Α Α-Amylase-Eps: Biosystems S.ARisqon Anjahiranda AdiputraNo ratings yet
- Drug Discovery and DevelopmentDocument47 pagesDrug Discovery and DevelopmentSalahBensaber100% (3)
- Latihan Soal Recount TextDocument14 pagesLatihan Soal Recount TextMuhammad Anshory100% (2)
- Manuali PDF 414Document1 pageManuali PDF 414Rakib Hossain 3A-159No ratings yet
- Lipase Spinreact 1x24 ML, 1X48 MLDocument2 pagesLipase Spinreact 1x24 ML, 1X48 MLN. K. MandilNo ratings yet
- Manuali PDF 413Document1 pageManuali PDF 413Rakib Hossain 3A-159No ratings yet
- Clonatest Amylase MRDocument4 pagesClonatest Amylase MRSuprovet LabotatorioNo ratings yet
- 1.alpha AmylaseDocument2 pages1.alpha AmylaseHiếu Chí PhanNo ratings yet
- 1105000I Rev. 02Document2 pages1105000I Rev. 02Riadh BenyoucefNo ratings yet
- Pi e LDH 21 Ifcc 3Document2 pagesPi e LDH 21 Ifcc 3Osama Ben DawNo ratings yet
- Alp Mono VialsDocument3 pagesAlp Mono VialsN. K. MandilNo ratings yet
- Lipase (S.L) : ADL/V.02/060114Document1 pageLipase (S.L) : ADL/V.02/060114Aniket dubeyNo ratings yet
- Budi Altgpt - Doc NewDocument3 pagesBudi Altgpt - Doc NewIrvanda ENVIOUSNo ratings yet
- CK MB enDocument1 pageCK MB enShakilur IslamNo ratings yet
- Applications Mindray BS-300 en 06-07-2010Document43 pagesApplications Mindray BS-300 en 06-07-2010dediNo ratings yet
- Lip 110 - en - Ru - CZ - DDocument5 pagesLip 110 - en - Ru - CZ - DMatibar RahmanNo ratings yet
- Ferritin Ing Rev. 03Document2 pagesFerritin Ing Rev. 03Yousra NanoNo ratings yet
- Ga4910 00 - Got-AstDocument2 pagesGa4910 00 - Got-AstTrần Thanh ViệnNo ratings yet
- Uric Acid: (Uricase / Pod Method)Document1 pageUric Acid: (Uricase / Pod Method)Dharmesh Patel100% (3)
- KR10150 Rev02Document2 pagesKR10150 Rev02KOUAME EDYMAIN FRANCISNo ratings yet
- Lyphase 11760I-Reagent A 510 Reagent B 300 Sample 9 Standard 113Document1 pageLyphase 11760I-Reagent A 510 Reagent B 300 Sample 9 Standard 113mahinNo ratings yet
- Crea 500 - en - Ru - CZ - SK - CDocument5 pagesCrea 500 - en - Ru - CZ - SK - CLouis MiuNo ratings yet
- PDF Acid Phosphatase Acp - CompressDocument1 pagePDF Acid Phosphatase Acp - CompressRizki YuliaNo ratings yet
- Bedtt19 Lipase - LQDocument2 pagesBedtt19 Lipase - LQrankedmlbbNo ratings yet
- PHP UYjhb 7Document2 pagesPHP UYjhb 7BerhaneNo ratings yet
- Aspartate Aminotransferase (AST-GOT) - ColorimetricDocument2 pagesAspartate Aminotransferase (AST-GOT) - ColorimetricGuneyden Guneyden0% (1)
- Measure AmyDocument2 pagesMeasure Amytuan vănNo ratings yet
- Pi e AP Ifcc 2Document2 pagesPi e AP Ifcc 2muhammadsufyanfp12No ratings yet
- Acid Phosphatase (Acp)Document1 pageAcid Phosphatase (Acp)Risqon Anjahiranda AdiputraNo ratings yet
- BIOLABO - Applications Humastar 180Document32 pagesBIOLABO - Applications Humastar 180Nguyễn Bảo Huân100% (1)
- Colorimetric-Kinetic: Clinical Significance Materials Required But Not ProvidedDocument2 pagesColorimetric-Kinetic: Clinical Significance Materials Required But Not ProvidedNavin kumarNo ratings yet
- 1116005I Rev. 02Document2 pages1116005I Rev. 02kirubel demelashNo ratings yet
- 1133505I Rev. 04Document2 pages1133505I Rev. 04Nguyễn HuynhNo ratings yet
- AMYLASE CNPG Liquiform: InsertDocument6 pagesAMYLASE CNPG Liquiform: Insertsarajuddin sarajNo ratings yet
- Crea 500 S - en - Ru - CZ - SK - B1Document5 pagesCrea 500 S - en - Ru - CZ - SK - B1Louis MiuNo ratings yet
- Budi Creatinine - Doc NewDocument3 pagesBudi Creatinine - Doc NewIrvanda ENVIOUSNo ratings yet
- SGOT LyphoDocument2 pagesSGOT LyphoNonameNo ratings yet
- SGOT Lypho DikonversiDocument3 pagesSGOT Lypho DikonversiNonameNo ratings yet
- Ck-Nac FsDocument2 pagesCk-Nac Fsadamalay wardiwiraNo ratings yet
- ALP Single ReagentDocument2 pagesALP Single ReagentJames 'jps' SimanjuntakNo ratings yet
- IFU - BX e CK 2Document6 pagesIFU - BX e CK 2betsabevegaaNo ratings yet
- Creatine Kinase (CK)Document2 pagesCreatine Kinase (CK)Mira SafwatNo ratings yet
- Lipase: Calib. Conc. Check Vial LabelDocument2 pagesLipase: Calib. Conc. Check Vial LabelDinesh SreedharanNo ratings yet
- CT10152 Creatinine EDocument2 pagesCT10152 Creatinine ELaboratorio BianucciNo ratings yet
- 11592i PDFDocument1 page11592i PDFKristoforusNo ratings yet
- GGT InsertDocument2 pagesGGT InsertsharmashyamsinghNo ratings yet
- Gpt-11-Alt (GPT) Uv 2V-02,12,2014 - 5Document2 pagesGpt-11-Alt (GPT) Uv 2V-02,12,2014 - 5dung nguyen danhNo ratings yet
- Amylase enDocument1 pageAmylase enRakib Hossain 3A-159No ratings yet
- PotasiumDocument2 pagesPotasiumlabextreme29No ratings yet
- AlkPhos FL 51 SFBC TV-005-CE-003Document1 pageAlkPhos FL 51 SFBC TV-005-CE-003Blas Cuba SanchezNo ratings yet
- ACCENT-200 ALAT ADocument2 pagesACCENT-200 ALAT Aemc.medicalserviceNo ratings yet
- CALCIUM CPC+STD ANG 2015 02 17Document2 pagesCALCIUM CPC+STD ANG 2015 02 17yehiwal756No ratings yet
- AlkPhos FL 51 DGKC TV-004-CE-003Document1 pageAlkPhos FL 51 DGKC TV-004-CE-003Blas Cuba SanchezNo ratings yet
- Alpha-Amylase: For In-Vitro Diagnostic and Professional Use OnlyDocument2 pagesAlpha-Amylase: For In-Vitro Diagnostic and Professional Use OnlyPark YuriNo ratings yet
- 1141010I Rev. 02Document2 pages1141010I Rev. 02Nguyễn HuynhNo ratings yet
- Alp RandoxDocument2 pagesAlp Randoxabdulrahmanaminu0199No ratings yet
- Budi-ASTGOT docNEWDocument3 pagesBudi-ASTGOT docNEWIrvanda ENVIOUSNo ratings yet
- SGPTDocument2 pagesSGPTNeña Lozada TabinasNo ratings yet
- Alkaline Phosphatase FS : Order Information SpecimenDocument3 pagesAlkaline Phosphatase FS : Order Information SpecimenmnemonicsNo ratings yet
- Metodo Cuantificar LipasaDocument1 pageMetodo Cuantificar Lipasa1718h9798s100% (1)
- Practical Handbook of Pharmaceutical Chemistry for M.PharmFrom EverandPractical Handbook of Pharmaceutical Chemistry for M.PharmNo ratings yet
- Infinosis HCG - IN027702 - enDocument2 pagesInfinosis HCG - IN027702 - enMeditech visionbdNo ratings yet
- Infinosis Total IgE IN067705 enDocument2 pagesInfinosis Total IgE IN067705 enMeditech visionbdNo ratings yet
- Infinosis HbA1c IN067701 enDocument2 pagesInfinosis HbA1c IN067701 enMeditech visionbdNo ratings yet
- Infinosis T3-In017702 enDocument2 pagesInfinosis T3-In017702 enMeditech visionbdNo ratings yet
- Infinosis Troponin I IN047701 enDocument2 pagesInfinosis Troponin I IN047701 enMeditech visionbdNo ratings yet
- Infinosis NT-proBNP IN047705 enDocument2 pagesInfinosis NT-proBNP IN047705 enMeditech visionbdNo ratings yet
- Infinosis PCT IN057701 enDocument2 pagesInfinosis PCT IN057701 enMeditech visionbdNo ratings yet
- Infinosis LH IN027703 enDocument2 pagesInfinosis LH IN027703 enMeditech visionbdNo ratings yet
- Welq T KV Ri Avb ': Qvî/Qvîxi BVG T Gvzvi BVG T Wczvi BVG T Ivj Bs TDocument19 pagesWelq T KV Ri Avb ': Qvî/Qvîxi BVG T Gvzvi BVG T Wczvi BVG T Ivj Bs TMeditech visionbdNo ratings yet
- Qvî/Qvîxi BVG T Ivj Bs Köbx WelqDocument13 pagesQvî/Qvîxi BVG T Ivj Bs Köbx WelqMeditech visionbdNo ratings yet
- Biochemistry Chema, Italy: (IFCC) (IFCC) (IFCC)Document2 pagesBiochemistry Chema, Italy: (IFCC) (IFCC) (IFCC)Meditech visionbdNo ratings yet
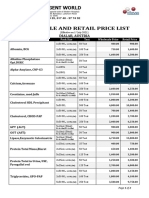
- Price List Wholesale and Retail (June-2021)Document2 pagesPrice List Wholesale and Retail (June-2021)Meditech visionbdNo ratings yet
- M T Jute Trading: Count & Quality: 10/1 LBS 1 PLY HessianDocument4 pagesM T Jute Trading: Count & Quality: 10/1 LBS 1 PLY HessianMeditech visionbdNo ratings yet
- SUBJECT:-APPOINTMENT TO THE POST OF Executive (Sales & Marketing)Document2 pagesSUBJECT:-APPOINTMENT TO THE POST OF Executive (Sales & Marketing)Meditech visionbdNo ratings yet
- Febrile AntigenbnkggDocument4 pagesFebrile AntigenbnkggMeditech visionbdNo ratings yet
- Price Quotation For Pathological Reagents. Sir,: (IFCC) (IFCC) (IFCC)Document1 pagePrice Quotation For Pathological Reagents. Sir,: (IFCC) (IFCC) (IFCC)Meditech visionbdNo ratings yet
- 7.1 Respiratory Process in Energy ProductionDocument13 pages7.1 Respiratory Process in Energy ProductionNor Rafidah Che YusofNo ratings yet
- Sohail Reports PDFDocument2 pagesSohail Reports PDFMunazza SohailNo ratings yet
- Full Download Solution Manual For Human Anatomy Physiology Main Version 4th Edition Terry Martin Cynthia Prentice Crave PDF Full ChapterDocument35 pagesFull Download Solution Manual For Human Anatomy Physiology Main Version 4th Edition Terry Martin Cynthia Prentice Crave PDF Full Chaptergabertconsomme.gygy100% (24)
- 1.3 Main Branches of AnthropologyDocument9 pages1.3 Main Branches of AnthropologyRohanNo ratings yet
- r127092130 Gabriel Martinez CUR127092130Document2 pagesr127092130 Gabriel Martinez CUR127092130GabrielNo ratings yet
- Lessonia-Fucoreverse BrochureDocument5 pagesLessonia-Fucoreverse BrochureElaineSilvestriniNo ratings yet
- Spiritual Psychology PDFDocument5 pagesSpiritual Psychology PDFKedar Kelkar100% (2)
- Goering AAO Convocation2012LectureDocument110 pagesGoering AAO Convocation2012Lectureperrolobo73No ratings yet
- Bulleted InfoDocument6 pagesBulleted InfoNiño James AceronNo ratings yet
- Co-Enzymes Role in Metabolic PathwaysDocument23 pagesCo-Enzymes Role in Metabolic PathwaysHarini BalasubramanianNo ratings yet
- Bi-Bi RXNDocument12 pagesBi-Bi RXNbelabelawNo ratings yet
- PIIS2211124721001704Document21 pagesPIIS22111247210017041813975862yvyvNo ratings yet
- SIM TestDocument5 pagesSIM TestBundi Gonzalez MartinNo ratings yet
- Types of SoilDocument2 pagesTypes of SoilCharlon RamiscalNo ratings yet
- ANALYSIS AND DENATURATION OF PROTEINS AnswersDocument5 pagesANALYSIS AND DENATURATION OF PROTEINS AnswersdgfdgsdfgsdsdgNo ratings yet
- Histo SupDocument8 pagesHisto SupAbdoulhaleem MoNo ratings yet
- MicrobiologyDocument3 pagesMicrobiologyRavi SharmaNo ratings yet
- Performance Task QuestionsDocument4 pagesPerformance Task Questionsapi-537184012No ratings yet
- Mendel's Law of SegregationDocument6 pagesMendel's Law of Segregationhanfa zamanNo ratings yet
- Test I. Puzzle Protein: Activity No. 4.1Document2 pagesTest I. Puzzle Protein: Activity No. 4.1Osas OsasNo ratings yet
- Eshmuno S ResinDocument4 pagesEshmuno S ResinTuyền KimNo ratings yet
- Piaget's Theory of Cognitive DevelopmentDocument19 pagesPiaget's Theory of Cognitive DevelopmentGuru Khb PblNo ratings yet
- Chapter 11Document11 pagesChapter 11christian anchetaNo ratings yet
- Mep 2 Assesemnt CC NewDocument12 pagesMep 2 Assesemnt CC NewPrecious Bardon-MempinNo ratings yet
- Chapter 1 Textile IndustryDocument9 pagesChapter 1 Textile IndustryAlice ChepkuruiNo ratings yet
- Clostridium, Called "TU-103", That Can Convert Nearly Any Form ofDocument1 pageClostridium, Called "TU-103", That Can Convert Nearly Any Form ofRiaNo ratings yet