Professional Documents
Culture Documents
Using Density To Determine The Sugar Content in Commercial Beverages
Using Density To Determine The Sugar Content in Commercial Beverages
Uploaded by
Max DornelesOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Using Density To Determine The Sugar Content in Commercial Beverages
Using Density To Determine The Sugar Content in Commercial Beverages
Uploaded by
Max DornelesCopyright:
Available Formats
PROP
CER · Modular · Laboratory · Program · in · Chemistry
editor: Henry D. Schreiber
5155
Using Density to Determine the Sugar Content
in Commercial Beverages
prepared by S. K. Henderson, C. A. Fenn, and J. D. Domijan, Quinnipiac College
Purpose of the Experiment Measure densities of solutions with known sugar content. Prepare a graph
relating density to sugar content. Determine densities and corresponding
sugar contents of various commercial beverages.
Background Required You should be familiar with techniques for measuring mass and volume,
and for calculating density. You should also be familiar with graphing tech-
niques, either manual or computerized.
Background Information We define density as the ratio of a substance’s mass to its volume, as shown
in Equation 1.
mass, g
density, g/mL = (Eq. 1)
volume, mL
We can use Equation 1 to calculate a solution’s density, once we have mea-
sured the mass of a known volume of the solution.
Suppose we prepare a solution by dissolving sugar in water. The solu-
tion’s density depends on how much sugar we dissolve in the water. As we
increase the solution’s sugar content, the solution’s density also increases.
In order to determine the exact relationship between a solution’s den-
sity and sugar content, we first prepare solutions with known sugar con-
tent. We express the sugar content in terms of mass percent, as defined by
Equation 2.
mass of sugar
mass percent sugar, % = (100%) (Eq. 2)
mass of solution
We then determine the densities of these solutions. On a graph, we plot each
solution’s density on the y-axis and its mass percent sugar on the x-axis. The
resulting graph is a straight line with positive slope.
If we then determine the density of a commercial beverage, we can use
this graph to determine the mass percent sugar in the beverage. Water and
sugar are the major components of beverages such as fruit juices, iced tea,
lemonade, and sodas. Other ingredients, such as flavorings, do not contrib-
ute measurably to these beverages’ densities.
Copyright 2002 Wadsworth Group. Brooks/Cole is an imprint of the Wadsworth Group, a division of Thomson Learning, Inc.
TM
Thomson Learning is a trademark used herein under license. For more information about this or any other Brooks/Cole products,
contact: BROOKS/COLE, 511 Forest Lodge Road, Pacific Grove, CA 93950 USA; www.brookscole.com; 1-800-423-0563 (Thomson
Learning Academic Resource Center). ALL RIGHTS RESERVED. No part of this work may be reproduced, transcribed, or used
in any form or by any means—graphic, electronic, or mechanical, including photocopying, recording, taping, Web distribution, or information storage and/or re-
trieval systems—without the prior written permission of the publisher. For permission to use material from this work, contact us by www.thomsonrights.com; fax:
1-800-730-2215; phone: 1-800-730-2214. Printed in United States of America.
2 PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages
Example Problem 1 Suppose a student prepares a solution of sodium chloride
(NaCl) in water. The student dissolves 1.05 g of NaCl in about 50 mL of wa-
ter. The mass of the final solution is 51.98 g. Determine the mass percent
NaCl in the solution.
Solution We adapt Equation 2 to show NaCl in water.
mass percent NaCl, %
mass of NaCl 1.05 g
= (100%) = (100%) = 2.02%
mass of solution 51.98 g
Problem 2 The student uses a pipet to transfer 10.00 mL of the NaCl solu-
tion to a beaker. The student determines that the mass of this volume of so-
lution is 10.08 g. Calculate the density of the solution.
Solution We use Equation 1.
mass, g 10.08 g
density, g/mL = = = 1.008 g/mL
volume, mL 10.00 mL
Problem 3 The student prepares two other NaCl–H2O solutions. The stu-
dent determines the densities of these solutions, as well as that of distilled
water. The student’s results are compiled in the following table.
solution
water 1 2 3
mass percent NaCl, % 0 2.02 4.07 6.16
density, g/mL 0.993 1.008 1.021 1.034
Prepare a graph of density versus NaCl content for these solutions,
and use it to determine the relationship between density and NaCl
concentration.
Solution We plot density on the y-axis and mass percent NaCl on the
x-axis, as shown in Figure 1. We then draw the best straight line through
these points.
1.040
1.035
1.030
1.025
density, g/mL
1.020
1.015
1.010
1.005
COPYRIGHT 2002 Wadsworth Group
1.000
0.995
0.990
0 1 2 3 4 5 6 7
mass percent NaCl, %
Figure 1 Plot of student-prepared NaCl solution concentrations and correspond-
ing densities
PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages 3
If we determine the trend line for these data via regression analysis, us-
ing a software spreadsheet, we find that:
density, g/mL = (0.0066 g/mL•%)(mass percent NaCl) + 0.994 g/mL
Problem 4 The student obtains an unknown NaCl–H2O sample and deter-
mines that its density is 1.015 g/mL. Determine the mass percent NaCl in
this sample.
Solution We locate the density of 1.015 g/mL on the y-axis of the graph
in Problem 3. We position a ruler horizontally at that point on the y-axis, and
note where it intersects the straight line graph. We then drop the ruler verti-
cally from the intersection point to the x-axis. We read the mass percent
NaCl on the x-axis, which appears to be 3.2%.
Alternatively, we substitute the solution’s density into the straight-line
equation relating density to mass percent NaCl, as determined in Problem 3.
1.015 g/mL = (0.0066 g/mL•%)(mass percent NaCl) + 0.994 g/mL
(1.015 g/mL – 0.994 g/mL )
mass percent NaCl, % = = 3.2%
0.0066 g/mL • %
Procedure Caution: Wear departmentally approved safety goggles while doing
this experiment.
Avoid ingesting all solids and solutions used in the laboratory. Never
drink from laboratory glassware, because of possible contamination.
Note: Record all measurements on your Data Sheet. Record all masses to ei-
ther the nearest milligram (0.001 g) or the nearest centigram (0.01 g), as
indicated by your laboratory instructor.
I. Preparing Solutions of Known Sugar Content
1. Obtain a small bottle, and label it “A”. Tare the bottle on a balance.
2. To prepare solution A, place about 2.5 g of sugar in the bottle. Record the
actual mass of sugar added.
3. Using a graduated cylinder, transfer about 48 mL of distilled or
deionized water to the bottle. Record the total mass of the solution.
4. Cap the bottle, and shake thoroughly to dissolve the sugar.
5. To prepare solution B, repeat Steps 1–4, using about 5.0 g of sugar and
NOTE 1: Record the actual 45 mL of distilled water [NOTE 1]. Label bottle “B”.
mass of sugar added and the
6. To prepare solution C, repeat Steps 1–4, using about 7.5 g of sugar and
total mass of solution for solu-
tions B and C. 42 mL of distilled water [NOTE 1]. Label bottle “C”.
COPYRIGHT 2002 Wadsworth Group
7. Solution D is pure water. Pour 50 mL of distilled water into a small bot-
tle. Label bottle “D”. This solution is your 0.00% sugar solution.
II. Determining the Densities of Your Sugar Solutions
8. Tare a clean 50-mL beaker.
9. Draw up enough solution D to half fill a clean, dry 10-mL pipet. Use this
solution to rinse the inside of the pipet. Allow the rinse solution to drain
into a 150-mL beaker, labeled “Discard Solutions”.
4 PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages
10. Use the rinsed 10-mL pipet to transfer 10.00 mL of solution D to the
tared 50-mL beaker. Measure and record the mass. Pour the contents of the
beaker into the drain, diluting with a large amount of running water. Wash
the beaker, then rinse with distilled water for reuse.
11. Repeat Steps 8–10 at least two more times. Perform additional determi-
nations, if necessary, until you have obtained three masses that agree within
the limits specified by your laboratory instructor.
12. Repeat Steps 8–11, using solutions A, B, and C.
III. Determining the Densities of Commercial Beverage Samples
13. Obtain three commercial beverage samples from your laboratory in-
structor. Record the identification codes or names of your three samples.
14. Repeat Steps 8–11, using each beverage sample.
15. Pour the contents of your Discard Solutions beaker and any leftover so-
lutions or samples into the drain, diluting with a large amount of running
water.
Caution: Wash your hands thoroughly with soap or detergent before leav-
ing the laboratory.
COPYRIGHT 2002 Wadsworth Group
name section date
Data Sheet
I. Preparing Solutions of Known Sugar Content
solution mass of sugar, g mass of solution, g
A _____________ _____________
B _____________ _____________
C _____________ _____________
D 0
II. Determining the Densities of Your Sugar Solutions
mass of 10.00 mL of solution, g
determination average mass of 10.00 mL
solution 1 2 3 of solution, g
A _____________ _____________ _____________ _____________
B _____________ _____________ _____________ _____________
C _____________ _____________ _____________ _____________
D _____________ _____________ _____________ _____________
III. Determining the Densities of Commercial Beverage Samples
mass of 10.00 mL of beverage, g
identification code determination average mass of 10.00 mL
or name of beverage 1 2 3 of beverage, g
______________ _____________ _____________ _____________ _____________
COPYRIGHT 2002 Wadsworth Group
______________ _____________ _____________ _____________ _____________
______________ _____________ _____________ _____________ _____________
PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages 5
name section date
Results Sheet
II. Determining the Densities of Your Sugar Solutions
1. Calculate mass percent sugar and average density for solutions A–D. Record your results in the follow-
ing table.
solution mass percent sugar, % average density of solution, g/mL
A __________________ __________________
B __________________ __________________
C __________________ __________________
D __________________ __________________
2. On the graph paper supplied, graph your data for solutions A–D. Plot average solution density on the
y-axis and mass percent sugar on the x-axis. Do not start your y-axis scale at 0. Instead, select y-axis scale
units so that the lowest value is slightly below the average density of solution D, and the highest value is
slightly above the average density of solution C. Your x-axis scale should run from 0 to 20 mass percent
sugar. Draw the best straight line defined by your data points.
Alternatively, your laboratory instructor may tell you to use a computer spreadsheet program to
graph the data and determine the following equation for the resulting trend line:
density, g/mL = (_________) (mass percent sugar, %) + _________
slope y-intercept
III. Determining the Densities of Commercial Beverage Samples
3. Calculate the average density for each of your commercial beverages. Record your results in the follow-
ing table. Using either your graph or the trend-line equation that you calculated, determine the mass
percent sugar for each of your beverages. Record your results in the following table.
identification code average density of
or name of beverage beverage, g/mL mass percent sugar, %
__________________ __________________ __________________
COPYRIGHT 2002 Wadsworth Group
__________________ __________________ __________________
__________________ __________________ __________________
PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages 7
8 PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages
COPYRIGHT 2002 Wadsworth Group
name section date
Interpretation of Your Results
Use the spaces provided for the answers and additional paper if necessary.
1. Chemical references list the density of water as 0.998 g/mL at room temperature. Based on this accepted
value, calculate the percent error in your average density (dexperimental) for solution D, using Equation 3:
0.998 g/mL – d experimental
percent error, % = (100%) (Eq. 3)
0.998 g/ mL
2. (a) Read the can or bottle label for each of your commercial beverage samples, in order to find the Calo-
rie content per serving and the serving size in milliliters.* Calculate Calories per milliliter for each of
your beverages.
(b) Prepare a table listing your three beverages, their mass percent sugar, and their Calories/mL. Deter-
mine whether or not the sugar content is roughly proportional to the caloric value of the beverages.
(c) Explain any differences (or similarities) in the sugar contents of these beverages.
3. Would cans of each of your beverages float or sink if placed in a tub of water? Briefly explain.
4. In order to use the Procedure in this experiment to analyze freshly squeezed orange juice, you must first
COPYRIGHT 2002 Wadsworth Group
filter the pulp from the liquid. Explain why this must be done in order for the Procedure to yield accurate
results.
*This information may be supplied by your laboratory instructor, keyed using sample identification codes.
PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages 9
name section date
Pre-Laboratory Assignment
1. What precautions should you take when working with the solids and solutions you will use in this
experiment?
2. In Step 1 of the Procedure, what is meant by the word, “tare”?
3. (a) A student prepared a solution of ethylene glycol (automotive antifreeze) in distilled water. He
mixed about 5 mL of ethylene glycol with about 45 mL of water. The mass of the ethylene glycol was
5.580 g. The total mass of the solution was 49.037 g. Calculate the mass percent ethylene glycol in the
solution.
(b) The student determined the average mass of 10.00 mL of this solution to be 10.063 g. Calculate the
average density of the solution.
(c) In a similar fashion, the student prepared other aqueous solutions of ethylene glycol and measured
their densities, obtaining the data shown in the following table. Record your calculated results from (a)
and (b) in this table.
mass percent ethylene glycol, % 0.00 _____________ 22.75 33.22
average density, g/mL 0.993 _____________ 1.021 1.030
(d) Using the graph paper supplied, plot average density versus mass percent ethylene glycol. Draw
the best straight line through your data points.
Alternatively, your laboratory instructor may tell you to use a computer spreadsheet program to
graph the data and determine the equation of the resulting trend line.
(e) An aqueous solution of ethylene glycol has a density of 1.010 g/mL. What is the mass percent ethyl-
ene glycol in this solution?
4. A student performing this experiment for solutions of sugar in water plotted her data as described in the
Procedure. She found that the slope of her trend line was 0.00401 g/mL•% and the y-intercept was
COPYRIGHT 2002 Wadsworth Group
0.995 g/mL. She also found the average density of a lemonade sample to be 1.031 g/mL. Calculate the
mass percent sugar in this lemonade sample.
PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages 11
12 PROP 515/Using Density to Determine the Sugar Content in Commercial Beverages
COPYRIGHT 2002 Wadsworth Group
ISBN 0-53497-752-9
You might also like
- Jamiolkowski Et Al (1988)Document18 pagesJamiolkowski Et Al (1988)Dariusz GuzikNo ratings yet
- Post-Liquefaction Volumetric Strain of Gravel-Sand Mixtures in Constant Volume Simple ShearDocument12 pagesPost-Liquefaction Volumetric Strain of Gravel-Sand Mixtures in Constant Volume Simple ShearJiaer Wu0% (1)
- Astm D4829Document4 pagesAstm D4829Felipe Ignacio Córdoba rebolledoNo ratings yet
- Assessment of Liquefaction Susceptibility of Fine Grained Soils Bray - SancioDocument187 pagesAssessment of Liquefaction Susceptibility of Fine Grained Soils Bray - SancioFernando ChiribogaNo ratings yet
- Triaxial Testing of Granular Soil (Colliat-Dangus, 1988)Document21 pagesTriaxial Testing of Granular Soil (Colliat-Dangus, 1988)Abraham FIgueroa ARevaloNo ratings yet
- Introduction To Triaxial Testing - Part 3Document5 pagesIntroduction To Triaxial Testing - Part 3Habib Musa MohamadNo ratings yet
- Measurement of Hydraulic Conductivity of Saturated Porous Materials Using A Flexible Wall PermeameterDocument24 pagesMeasurement of Hydraulic Conductivity of Saturated Porous Materials Using A Flexible Wall PermeameterManolo AtarihuanaNo ratings yet
- ASTM Code For CU Triaxial Test PDFDocument13 pagesASTM Code For CU Triaxial Test PDFAhmedHossainNo ratings yet
- The Direct Shear Strength and Dilatancyof Sand-Gravel MixturesDocument27 pagesThe Direct Shear Strength and Dilatancyof Sand-Gravel MixturesPipatpong NookhaoNo ratings yet
- Astm D4083 - 1 (En)Document6 pagesAstm D4083 - 1 (En)Amm MarakataNo ratings yet
- A Comparative Evaluation of Various AdditivesDocument11 pagesA Comparative Evaluation of Various Additivesagus_ip3808No ratings yet
- Astm D 7263 09Document9 pagesAstm D 7263 09Irna PrasticaNo ratings yet
- Recent Developments in The Techniques of Controlling and Measuring Suction in Unsaturated SoilsDocument20 pagesRecent Developments in The Techniques of Controlling and Measuring Suction in Unsaturated SoilsAmal BhlNo ratings yet
- Water Parameters - RocscienceDocument2 pagesWater Parameters - RocscienceAgha Shafi Jawaid KhanNo ratings yet
- Lugeon TestDocument8 pagesLugeon TestAdly Al-saafinNo ratings yet
- D 2845 - 95 Rdi4ndutukve PDFDocument9 pagesD 2845 - 95 Rdi4ndutukve PDFLupita CarelyNo ratings yet
- D427Document4 pagesD427Nouman KhalidNo ratings yet
- E1266-12 Standard Practice For Processing MixturesDocument4 pagesE1266-12 Standard Practice For Processing Mixturescsharpplus100% (1)
- ASTM - D4253 16E01 - en USDocument16 pagesASTM - D4253 16E01 - en USMatias Leiva FloresNo ratings yet
- D 4767 Test Method For Consolidated Undrained Triaxial Compression Test For Cohesive Soils PDFDocument13 pagesD 4767 Test Method For Consolidated Undrained Triaxial Compression Test For Cohesive Soils PDFjairoNo ratings yet
- Astm D5084-10 Permeability TRXDocument24 pagesAstm D5084-10 Permeability TRXquequma7202100% (1)
- D 6391 - 99 R04 - RdyzoteDocument12 pagesD 6391 - 99 R04 - RdyzoteabualamalNo ratings yet
- 614 Christopher Chud Lundgreen 2010 KRDocument121 pages614 Christopher Chud Lundgreen 2010 KRwaltervillanNo ratings yet
- Job-2 Relative DensityDocument5 pagesJob-2 Relative DensityUmair JuttNo ratings yet
- Astm d5873 Esclerometro PDFDocument4 pagesAstm d5873 Esclerometro PDFJulio Oyola Salvador100% (1)
- Compressive Strength of Bituminous Mixtures: Standard Test Method ForDocument5 pagesCompressive Strength of Bituminous Mixtures: Standard Test Method Foraastha mehtaNo ratings yet
- Aggregate Testing ProcechureDocument19 pagesAggregate Testing ProcechureBalaNguyentriNo ratings yet
- ASTM D4829-08a PDFDocument5 pagesASTM D4829-08a PDFOm BunNo ratings yet
- Measurement of Hydraulic Conductivity of Porous Material Using A Rigid-Wall, Compaction-Mold PermeameterDocument9 pagesMeasurement of Hydraulic Conductivity of Porous Material Using A Rigid-Wall, Compaction-Mold PermeameterhugoNo ratings yet
- IS - Free Swell IndexDocument2 pagesIS - Free Swell IndexDr Wong Kim YuenNo ratings yet
- D 4543 - 85 R91 - (Ingles)Document7 pagesD 4543 - 85 R91 - (Ingles)FrancisNo ratings yet
- Particle-Size Distribution (Gradation) of Fine-Grained Soils Using The Sedimentation (Hydrometer) AnalysisDocument27 pagesParticle-Size Distribution (Gradation) of Fine-Grained Soils Using The Sedimentation (Hydrometer) AnalysisCRISTHIAN JESUS NEGRETE TASAYCONo ratings yet
- Astm D427-Contracción MercurioDocument4 pagesAstm D427-Contracción MercurioDaniel Villagomez SilvaNo ratings yet
- D 2974 - 00 PDFDocument4 pagesD 2974 - 00 PDFashrafNo ratings yet
- Field Behaviour of Stiffened Deep Cement Mixing PilesDocument17 pagesField Behaviour of Stiffened Deep Cement Mixing PilesNguyen Quoc KhanhNo ratings yet
- Pressure Meter Test in PracticeDocument6 pagesPressure Meter Test in PracticeNathan VincentNo ratings yet
- D 6519 - 02-Sampling of Soil Usingthe Hydraulic OperatedStationary Piston SamplerDocument8 pagesD 6519 - 02-Sampling of Soil Usingthe Hydraulic OperatedStationary Piston SamplerJamie MooreNo ratings yet
- Astm D4718D4718M-15Document3 pagesAstm D4718D4718M-15Jha AcostaNo ratings yet
- Astm d5778 12Document20 pagesAstm d5778 12farquinNo ratings yet
- Swelling Characteristics of Compacted, Expansive Soils: (Received 5 NovemberDocument11 pagesSwelling Characteristics of Compacted, Expansive Soils: (Received 5 NovemberYogesh KapaseNo ratings yet
- D 6836 - 02 - Rdy4mzyDocument20 pagesD 6836 - 02 - Rdy4mzyEmiliano AgostiniNo ratings yet
- D 4647 - 93 R98 RDQ2NDCDocument11 pagesD 4647 - 93 R98 RDQ2NDCJonathan Chacón100% (1)
- Laboratory Determination of Resilient Modulus For Flexible Pavement DesignDocument53 pagesLaboratory Determination of Resilient Modulus For Flexible Pavement DesignprmraoNo ratings yet
- Column Supported Embankments - AsceDocument24 pagesColumn Supported Embankments - AsceAnonymous D5s00DdUNo ratings yet
- Peso Unitario SuelosDocument7 pagesPeso Unitario SuelossergioNo ratings yet
- Sampling Bituminous Paving Mixtures: Standard Practice ForDocument3 pagesSampling Bituminous Paving Mixtures: Standard Practice Foraastha mehtaNo ratings yet
- Estimation of Hydraulic Conductivity From Grain Size AnalysesDocument96 pagesEstimation of Hydraulic Conductivity From Grain Size AnalysesmelvindanginNo ratings yet
- Roberson 2012 PDFDocument22 pagesRoberson 2012 PDFAdy NugrahaNo ratings yet
- Specific Gravity of Soil Solids by Water Pycnometer: Standard Test Methods ForDocument8 pagesSpecific Gravity of Soil Solids by Water Pycnometer: Standard Test Methods ForMon QuinolNo ratings yet
- Andrus 2004 Shear Wave Vs For LiquefDocument24 pagesAndrus 2004 Shear Wave Vs For LiquefAlessandra BortoluzziNo ratings yet
- D4043-17 - Selection Aquifer TestsDocument6 pagesD4043-17 - Selection Aquifer TestsJessikaNo ratings yet
- Expansive Soils PaperDocument7 pagesExpansive Soils PaperM Ramzan QadirNo ratings yet
- Calculo Empujes LateralesDocument24 pagesCalculo Empujes LateralesOscar PelaezNo ratings yet
- Unsat2000 DenverDocument15 pagesUnsat2000 DenverSamuel Laura HuancaNo ratings yet
- Consolidated Drained Triaxial Compression Test For SoilsDocument11 pagesConsolidated Drained Triaxial Compression Test For Soilscristinaabate1No ratings yet
- Em - 1110 2 3506Document159 pagesEm - 1110 2 3506dannychacon27No ratings yet
- Calculating in Situ Equivalent Elastic Moduli of Pavement Materials Using Layered Elastic TheoryDocument7 pagesCalculating in Situ Equivalent Elastic Moduli of Pavement Materials Using Layered Elastic TheoryKevin Darly Chavez ChuzonNo ratings yet
- Design of Piles Under Cyclic Loading: SOLCYP RecommendationsFrom EverandDesign of Piles Under Cyclic Loading: SOLCYP RecommendationsAlain PuechNo ratings yet
- Saliva: DR - Deepshikha Soni Mds 1 YearDocument90 pagesSaliva: DR - Deepshikha Soni Mds 1 Yeardisha 146jandialNo ratings yet
- Research Projects JiitDocument14 pagesResearch Projects JiitSAMAIRA SINGHNo ratings yet
- Causes and Prevention of Paint Failure PDFDocument14 pagesCauses and Prevention of Paint Failure PDFsenthilkumarNo ratings yet
- Scpa Presentation December 15, 2014: Presented By: Chandra Brown, Internal Audit Monya Wyatt, SCPA Budget ManagerDocument20 pagesScpa Presentation December 15, 2014: Presented By: Chandra Brown, Internal Audit Monya Wyatt, SCPA Budget ManagerCincinnatiEnquirerNo ratings yet
- Medical Marijuana: Social Construction of A Myth Theories, Findings, FutureDocument21 pagesMedical Marijuana: Social Construction of A Myth Theories, Findings, Futurejohn mark demetilloNo ratings yet
- Driving Exam NotesDocument7 pagesDriving Exam Notesreach2ashish5065No ratings yet
- HLS T78Document2 pagesHLS T78Power electronicsNo ratings yet
- Eu 998 PDFDocument2 pagesEu 998 PDFJeanNo ratings yet
- MenstruationDocument4 pagesMenstruationVpln SarmaNo ratings yet
- 6 Types of Cell Culture-2024Document49 pages6 Types of Cell Culture-2024hamza najmNo ratings yet
- Literature Review On Environmental DegradationDocument8 pagesLiterature Review On Environmental Degradationafmzaoahmicfxg100% (1)
- A Systematic Review For Enabling of Develop A Blockchain Technology in Healthcare Application TaxonomyDocument35 pagesA Systematic Review For Enabling of Develop A Blockchain Technology in Healthcare Application TaxonomyAshraf AbdouNo ratings yet
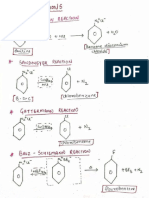
- Named Organic ReactionsDocument8 pagesNamed Organic ReactionscrawlskullNo ratings yet
- Autotrophic Nutrition Heterotrophic NutritionDocument1 pageAutotrophic Nutrition Heterotrophic NutritionAditya KediaNo ratings yet
- Ada363924 S&aDocument30 pagesAda363924 S&asksabinaNo ratings yet
- 8.1 Purpose of The Unit: 8 Design of Distillation ColumnDocument16 pages8.1 Purpose of The Unit: 8 Design of Distillation Columnsami ullahNo ratings yet
- D5462 PDFDocument6 pagesD5462 PDFZamir Danilo Morera ForeroNo ratings yet
- Hitachi Mild Hybrid Press ReleaseDocument4 pagesHitachi Mild Hybrid Press ReleaseRafa SilesNo ratings yet
- Enviroline 199+ds+eng PDFDocument4 pagesEnviroline 199+ds+eng PDFMohamed NouzerNo ratings yet
- Aadhaar Enabled Public Distribution System - Aepds: Food and Consumer Protection Department Government of BiharDocument1 pageAadhaar Enabled Public Distribution System - Aepds: Food and Consumer Protection Department Government of BiharAnil SharmaNo ratings yet
- Michael's On East Lunch MenuDocument2 pagesMichael's On East Lunch Menusupport_local_flavorNo ratings yet
- Marine Refrigeration AND Air ConditioningDocument73 pagesMarine Refrigeration AND Air ConditioningBruno Manestar100% (2)
- Taxes in PhilippinesDocument2 pagesTaxes in PhilippinesJames iKnowNo ratings yet
- Natarajan and Murugavel - 2017 - Thermal Stability and Thermal Degradation KineticsDocument22 pagesNatarajan and Murugavel - 2017 - Thermal Stability and Thermal Degradation Kineticsmartina.cirronis96No ratings yet
- Irrigation Manual FAO Module 7Document169 pagesIrrigation Manual FAO Module 7Zac Mweb86% (7)
- Hazard ManagementDocument18 pagesHazard Managementsthiyagu0% (1)
- Catalogo Nocchi PDFDocument94 pagesCatalogo Nocchi PDFDonovan Newton100% (1)
- Home Care CompetencyDocument54 pagesHome Care Competencyapi-3002188600% (1)
- A Healing Story About GuyabanoDocument4 pagesA Healing Story About GuyabanoZena Bezabih100% (1)
- Problem Set 1 (Review of Fundamental Soil Testing) : Answer Reference Solution File HereDocument3 pagesProblem Set 1 (Review of Fundamental Soil Testing) : Answer Reference Solution File HereJasper Kenneth PeraltaNo ratings yet