Professional Documents
Culture Documents
0 1 2 3 4 5 6 7 8 9 CM Ruler Wooden Block Beaker Bench
0 1 2 3 4 5 6 7 8 9 CM Ruler Wooden Block Beaker Bench
Uploaded by
lemonCopyright:
Available Formats
You might also like
- 6 Using Moles: Extended CurriculumDocument2 pages6 Using Moles: Extended CurriculumkamalNo ratings yet
- QP Csci Test FebDocument13 pagesQP Csci Test FeblemonNo ratings yet
- 2010CIE GCE 5129 s10 QP 1Document16 pages2010CIE GCE 5129 s10 QP 1葡萄蘿蔔No ratings yet
- Physics Ch2ForcesAndMotionp2p4p6worksheetwithanswersDocument14 pagesPhysics Ch2ForcesAndMotionp2p4p6worksheetwithanswerssupratik30No ratings yet
- Physics: PAPER 1 Multiple ChoiceDocument21 pagesPhysics: PAPER 1 Multiple ChoiceZubairHassanNo ratings yet
- 0625 m20 QP 12 PDFDocument20 pages0625 m20 QP 12 PDFDesire KandawasvikaNo ratings yet
- Science Physics Chemistry October 2005 Paper 1Document20 pagesScience Physics Chemistry October 2005 Paper 1Phiri AgnesNo ratings yet
- Peperiksaan Pertengahan Tahun 2011Document17 pagesPeperiksaan Pertengahan Tahun 2011Sue Suraya NazaNo ratings yet
- Physics Paper 1 C 1 August PDFDocument16 pagesPhysics Paper 1 C 1 August PDFMohammad IrfanNo ratings yet
- CGS 09 5116 S4 Prelim P1 With Ans.Document15 pagesCGS 09 5116 S4 Prelim P1 With Ans.topcatNo ratings yet
- Paper 1 Nov 2000 PhysicsDocument16 pagesPaper 1 Nov 2000 Physicssolarixe100% (1)
- Physics: PAPER 1 Multiple ChoiceDocument20 pagesPhysics: PAPER 1 Multiple ChoiceIslamabad ALMA SchoolNo ratings yet
- O-Levels Physics ExemplarDocument56 pagesO-Levels Physics Exemplarbrendoored2No ratings yet
- Maths IGCSE Quick RevisionDocument16 pagesMaths IGCSE Quick Revisionlindy musekaNo ratings yet
- Cambridge IGCSE: Physics 0625/22Document16 pagesCambridge IGCSE: Physics 0625/22...No ratings yet
- Yr 2 IP Physical Science EOY 2020 SingaporeDocument19 pagesYr 2 IP Physical Science EOY 2020 Singaporecatsarepro17No ratings yet
- 2023 Specimen Paper 1Document16 pages2023 Specimen Paper 1Jąhąnząib Khąn KąkąrNo ratings yet
- Yr1 Physics Induction WorkDocument15 pagesYr1 Physics Induction Workalex.yori123456789No ratings yet
- June 2023 Question Paper 13Document16 pagesJune 2023 Question Paper 13Asmaa JoudehNo ratings yet
- امتحان رامز 25-1Document17 pagesامتحان رامز 25-1Ahmed AliNo ratings yet
- 2019 Sec 4 Pure Physics SA2 CHIJ ST Nicholas GirlsDocument42 pages2019 Sec 4 Pure Physics SA2 CHIJ ST Nicholas GirlsFrancis Ho HoNo ratings yet
- Sci Physics Prelim 09 4E5NA P1Document7 pagesSci Physics Prelim 09 4E5NA P1topcatNo ratings yet
- Work-Sheet For Surveying - IDocument6 pagesWork-Sheet For Surveying - IMaruf Jemal100% (1)
- Cambridge IGCSE: Physics 0625/22Document16 pagesCambridge IGCSE: Physics 0625/22Nisha zehraNo ratings yet
- Cambridge O Level: Physics 5054/12Document20 pagesCambridge O Level: Physics 5054/12iman jamilNo ratings yet
- Phy o 22 20Document16 pagesPhy o 22 20Sadman E AlamNo ratings yet
- OL Physics Book 1 (MCQ Theory) 2008 Till 2021Document409 pagesOL Physics Book 1 (MCQ Theory) 2008 Till 2021ABDULLAH'S SWEET VIDEOSNo ratings yet
- 0625 Paper 2Document527 pages0625 Paper 2Niki SNo ratings yet
- Cambridge International General Certificate of Secondary EducationDocument16 pagesCambridge International General Certificate of Secondary EducationfitriNo ratings yet
- P 1Document16 pagesP 1Chaos GamingNo ratings yet
- Cambridge IGCSE: PHYSICS 0625/01Document18 pagesCambridge IGCSE: PHYSICS 0625/01Aditya RaoNo ratings yet
- Cambridge IGCSE: Physics 0625/21Document16 pagesCambridge IGCSE: Physics 0625/21fayyaz haqueNo ratings yet
- Additional Combined Science: Cambridge International Examinations General Certificate of Education Ordinary LevelDocument20 pagesAdditional Combined Science: Cambridge International Examinations General Certificate of Education Ordinary Levelrodel.verzosaNo ratings yet
- March 2020 (v2) QP - Paper 1 CIE Physics IGCSEDocument20 pagesMarch 2020 (v2) QP - Paper 1 CIE Physics IGCSEjanggodanxdNo ratings yet
- Sec 4 Physics SA2 2018 Ahmad IbrahimDocument52 pagesSec 4 Physics SA2 2018 Ahmad Ibrahim또몽No ratings yet
- © Ucles 2022 0625/21/o/n/22Document5 pages© Ucles 2022 0625/21/o/n/22Md. Zubaer Mahmud RonyNo ratings yet
- Sda 15Document26 pagesSda 15Nothando PhologotswanaNo ratings yet
- Gce O-Level Preliminary Examination 2018: Ahmad Ibrahim Secondary SchoolDocument107 pagesGce O-Level Preliminary Examination 2018: Ahmad Ibrahim Secondary SchoolNamarr SinghNo ratings yet
- Revision Sec3Document14 pagesRevision Sec3โหน่ง บัวรดน้ำNo ratings yet
- Physics OneDocument8 pagesPhysics OneMUSOKE GERALDNo ratings yet
- Forces P2Document76 pagesForces P2arjunboruto1234No ratings yet
- Cambridge IGCSE: PHYSICS 0625/02Document14 pagesCambridge IGCSE: PHYSICS 0625/02Adedamola AdenugaNo ratings yet
- UntitledDocument20 pagesUntitledmorongwa malepengNo ratings yet
- QSS 5116 ScPhy P1 Prelim 09Document8 pagesQSS 5116 ScPhy P1 Prelim 09topcatNo ratings yet
- Physics Y13 Final Exam May 2021Document19 pagesPhysics Y13 Final Exam May 2021John samNo ratings yet
- Intensive CST-03 - (Code-A) - 13-06-2022Document23 pagesIntensive CST-03 - (Code-A) - 13-06-2022The BoNg FOOD recipesNo ratings yet
- 5054 w06 QP 1Document16 pages5054 w06 QP 1darkcastle100% (1)
- 1.1 Physical Quantities & Measurement TechniquesDocument39 pages1.1 Physical Quantities & Measurement TechniquesFatima Sohail 6aNo ratings yet
- Paper 1 May 2006 PhysicsDocument20 pagesPaper 1 May 2006 PhysicssolarixeNo ratings yet
- Physics P-12 IgcseDocument20 pagesPhysics P-12 IgcseareebNo ratings yet
- Physics 6091/01Document16 pagesPhysics 6091/01YogiNo ratings yet
- JEE Mains Sample Paper 3Document14 pagesJEE Mains Sample Paper 3random id100% (1)
- MCQ20BDocument16 pagesMCQ20BMELODY CHENNo ratings yet
- Ccss Phy MCQDocument8 pagesCcss Phy MCQoAeonNo ratings yet
- Unit 1 Physical Quantities, Units and MeasurementsDocument3 pagesUnit 1 Physical Quantities, Units and MeasurementsrafeyNo ratings yet
- Cambridge IGCSE: Physics 0625/11Document16 pagesCambridge IGCSE: Physics 0625/11tccrgvyvuubNo ratings yet
- 0625 m18 QP 22 PDFDocument16 pages0625 m18 QP 22 PDFamalNo ratings yet
- O level Physics Questions And Answer Practice Papers 3From EverandO level Physics Questions And Answer Practice Papers 3Rating: 3 out of 5 stars3/5 (1)
- Che Day1Document22 pagesChe Day1Janrey DohinogNo ratings yet
- Periodic Classification of Elements: Multiple Choice QuestionsDocument10 pagesPeriodic Classification of Elements: Multiple Choice QuestionsAryanNo ratings yet
- Astm A659-18Document3 pagesAstm A659-18Ryan ZhangNo ratings yet
- ReferenceDocument4 pagesReferenceMaries San PedroNo ratings yet
- Itec 2018-Akc-IrDocument63 pagesItec 2018-Akc-IrJohn F ServilleNo ratings yet
- Drilling Practical Manual - Drilling PDFDocument51 pagesDrilling Practical Manual - Drilling PDFAarzoo JobanputraNo ratings yet
- RPT Active OperatorsDocument120 pagesRPT Active Operatorsgijanie93No ratings yet
- Salting and CuringDocument13 pagesSalting and CuringSherylleneAguilera100% (1)
- Preventing Polymer Degradation During Mixing.Document8 pagesPreventing Polymer Degradation During Mixing.विनय शर्माNo ratings yet
- CHE 123 HWK Back and Redox TitrationsDocument3 pagesCHE 123 HWK Back and Redox TitrationsJuiloNo ratings yet
- 5th Grade - Mixing Substances - Lab ActivityDocument6 pages5th Grade - Mixing Substances - Lab Activityapi-254428474No ratings yet
- Scenario 1 S: Lamelar Tearing Occur in Set In? orDocument6 pagesScenario 1 S: Lamelar Tearing Occur in Set In? orBeny ZulNo ratings yet
- 12-19 Limit Test of ArsenicDocument1 page12-19 Limit Test of ArsenicBhupendra TomarNo ratings yet
- Al - Metal MatrixDocument38 pagesAl - Metal Matrixpramo_dassNo ratings yet
- Organic ExamDocument16 pagesOrganic ExamMnawaru KhalfaniNo ratings yet
- TDS - Total - Lubrilam Cleaner H1 - 1XQ - 201412 - enDocument1 pageTDS - Total - Lubrilam Cleaner H1 - 1XQ - 201412 - enVelibor KaranovicNo ratings yet
- Free Energies of Proton-Coupled Electron Transfer Reagents and Their ApplicationsDocument49 pagesFree Energies of Proton-Coupled Electron Transfer Reagents and Their ApplicationsZongxin JinNo ratings yet
- Standards Testing Plastic Tanks and VesselsDocument3 pagesStandards Testing Plastic Tanks and VesselsFaraj MohamedNo ratings yet
- Catalog Uk DrillingDocument226 pagesCatalog Uk DrillingJuanNo ratings yet
- Color Beta Carotene 2009 TRDocument20 pagesColor Beta Carotene 2009 TRUlfah Nurul MutmainnahNo ratings yet
- Nitration of BromobenzeneDocument3 pagesNitration of BromobenzeneLedina BanushllariNo ratings yet
- Basalt FiberDocument19 pagesBasalt FiberHitesh Shah100% (1)
- SDMC Heavy Metal SequestrationDocument1 pageSDMC Heavy Metal Sequestrationwacsii ccasullaNo ratings yet
- Biofuel Production From Mesua Ferrea L Seed OilDocument6 pagesBiofuel Production From Mesua Ferrea L Seed OilerpublicationNo ratings yet
- Eggs by Tonophore: Activation of Sea-Urchin CalciumDocument5 pagesEggs by Tonophore: Activation of Sea-Urchin CalciumCoCo GaldosNo ratings yet
- Liquid SulfurDocument20 pagesLiquid SulfurMarcin MaruchaNo ratings yet
- Incropera Appendix - 2011Document32 pagesIncropera Appendix - 2011Gracylla RoseNo ratings yet
- Zytel LCPA Product Reference GuideDocument2 pagesZytel LCPA Product Reference GuideProvocateur SamaraNo ratings yet
- VisbreakingDocument16 pagesVisbreakingAbdulAbdul100% (1)
0 1 2 3 4 5 6 7 8 9 CM Ruler Wooden Block Beaker Bench
0 1 2 3 4 5 6 7 8 9 CM Ruler Wooden Block Beaker Bench
Uploaded by
lemonOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
0 1 2 3 4 5 6 7 8 9 CM Ruler Wooden Block Beaker Bench
0 1 2 3 4 5 6 7 8 9 CM Ruler Wooden Block Beaker Bench
Uploaded by
lemonCopyright:
Available Formats
2
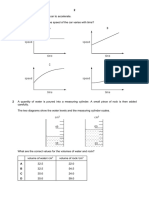
1 The diagram shows a method of measuring the diameter of a beaker.
ruler
0 1 2 3 4 5 6 7 8 9
cm
wooden block
beaker
bench
What is the diameter of the beaker?
A 4.5 cm B 5.0 cm C 5.5 cm D 8.0 cm
2 A block of mass 1 kg is pushed across a frictionless surface with a force of 2 N.
What is the acceleration of the block?
A 0.5 m / s2 B 1.0 m / s2 C 2.0 m / s2 D 3.0 m / s2
3 The diagram shows three pieces of apparatus.
1 2 3
0.0g
balance measuring cylinder vernier calipers
Which instruments are required to measure the density of an irregular piece of rock?
A 1 and 2 only B 1 and 3 only C 2 and 3 only D 1, 2 and 3
© UCLES 2010 5129/01/M/J/10
3
4 A diver, weighing 720 N, stands at the end of a springboard measuring 3 m long.
3m
What is the moment about the support?
A 720 Nm B 720 × 3 Nm C 3 Nm D 720 Nm
720 3
5 Which energy source is used in a nuclear power station?
A coal
B hydrogen
C natural gas
D uranium
6 Equal volumes of four substances are heated at atmospheric pressure.
The temperature rise is the same for each substance.
Which substance expands the most?
A air
B mercury
C steel
D water
© UCLES 2010 5129/01/M/J/10 [Turn over
4
7 The diagram shows a plane mirror and the position of an image.
Where must the object be placed to form this image?
A B
D image
mirror
8 A battery moves a charge of 60 C around a circuit in a time of 20 s.
What is the current in the circuit?
A 0.3 A B 3.0 A C 40 A D 1200 A
9 A current of 6 A flows in the circuit shown. The current splits up when it enters parallel branches
of resistors.
3Ω
A
6A
3Ω
What is the reading on the ammeter?
A 2A B 3A C 6A D 12 A
10 A small heater operates at 12 V, 2 A.
How much energy will it use when it is used for 5 minutes?
A 30 J B 120 J C 1800 J D 7200 J
© UCLES 2010 5129/01/M/J/10
2
1 The diagram shows part of a vernier scale.
mm
30
0
35
40
10
45
What is the correct reading?
A 30.5 mm B 33.5 mm C 38.0 mm D 42.5 mm
2 The gradient of the line on a graph gives the acceleration of a moving object.
What are the quantities on the horizontal and vertical axes of this graph?
quantity on quantity on
horizontal axis vertical axis
A speed distance
B speed time
C time distance
D time speed
© UCLES 2010 5129/01/O/N/10
3
3 The gravitational field strength is 2 N / kg on the Moon and 10 N / kg on the Earth.
An astronaut returns from the Moon to the Earth.
What effect does this have on the astronaut’s mass and weight?
mass weight
A less on Earth same on Earth and Moon
B more on Earth same on Earth and Moon
C same on Earth and Moon less on Earth
D same on Earth and Moon more on Earth
4 The diagrams show how a spring extends when a weight of 6.0 N is hung on it.
10 cm
12 cm
6.0 N
Which weight hanging from the spring causes the length to become 15 cm?
A 7.5 N B 15 N C 30 N D 45 N
5 An electric motor lifts a weight of 8 N through a height of 5 m in 4 s.
What is the power developed?
A 2.5 W B 6.4 W C 10 W D 40 W
© UCLES 2010 5129/01/O/N/10 [Turn over
4
6 To protect a polished table, a cork mat may be put on the table underneath a mug containing hot
liquid.
mug of hot liquid
cork mat
polished table
Why is this effective?
A Cork is a good conductor.
B Cork is a good radiator.
C Cork is a poor conductor.
D Cork is a poor radiator.
7 What is the angle of refraction for this ray of light moving from glass to air?
glass block
A
normal B
C normal
D
8 Electric current is defined as rate of flow of charge and is measured in amperes, A.
How can the unit of current also be written?
A Cm B C/m C Cs D C/s
9 Two resistors are connected in series with a 9 V supply.
9V
1.5 Ω 3Ω
What is the current flowing in the circuit?
A 2.0 A B 3.0 A C 4.5 A D 6.0 A
© UCLES 2010 5129/01/O/N/10
5
10 A circuit consists of a battery and four resistors.
2.0 A 3.5 A
3.0 A X
The current in three of the resistors is shown.
What is the current in X?
A 1.5 A B 2.0 A C 3.0 A D 5.0 A
11 A 2 kW appliance is to be connected to the 240 V mains supply.
Which fuse should be fitted in the plug?
A 1A B 3A C 5A D 10 A
12 What is the nucleon number of a nuclide?
A the number of neutrons
B the number of protons
C the total number of neutrons and protons
D the total number of protons and electrons
13 A radioactive material gives a count rate of 8000 counts per minute.
After 20 days, it gives a count rate of 500 counts per minute.
What is the half-life of the material?
A 4 days B 5 days C 20 days D 80 days
© UCLES 2010 5129/01/O/N/10 [Turn over
2
1 A plumber needs to measure the internal diameter of a water tap as accurately as possible.
Which instrument should be used?
A measuring tape
B metre rule
C micrometer
D vernier calipers
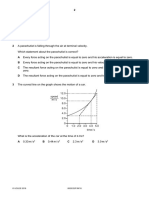
2 A speed-time graph for a car starting from rest is shown.
20
speed
m / s 15
10
0 1 2 3 4 5 6 7 8
time / s
What is the acceleration of the car between 4 s and 8 s?
A 0 m / s2 B 2.5 m / s2 C 5 m / s2 D 10 m / s2
3 A rectangular metal block measures 4 cm × 5 cm × 10 cm. The mass of the block is 800 g.
5 cm
4 cm
10 cm
What is the density of the metal?
A 0.25 g / cm3 B 2.5 g / cm3 C 4.0 g / cm3 D 40 g / cm3
© UCLES 2011 5129/01/M/J/11
3
4 A horseshoe can be made from a piece of metal by first heating and then hammering the metal.
Which property of the metal changes during the hammering action?
A density
B mass
C shape
D volume
5 A box is subjected to a force of 60 N and moves a distance of 15 m in the direction of the force.
What is the work done?
A 0.25 J B 4.0 J C 75 J D 900 J
6 Density changes are responsible for which method of thermal energy transfer?
A conduction only
B convection only
C radiation only
D conduction, convection and radiation
7 The diagram shows the displacement across a wave pattern.
Which value is multiplied by the frequency to give the speed of the wave?
displacement
A
0
0 B C distance
D
© UCLES 2011 5129/01/M/J/11 [Turn over
4
8 The diagram shows the path of a ray of light travelling towards and into a pool of water.
Four angles are labelled.
P
Q
S
R water
Which two angles would be correctly used in the equation sin i = constant?
sin r
A P and R B P and S C Q and R D Q and S
9 Which type of electromagnetic radiation travels at the highest speed through a vacuum?
A gamma rays
B light waves
C radio waves
D none – all have the same speed
10 The diagram shows an electrical circuit.
4Ω
A1 A4
A2 6Ω
A3 3Ω
The reading of ammeter A2 is 1 A and of A4 is 3 A.
What are the readings of ammeters A1 and A3?
A1 / A A3 / A
A 1.5 0.5
B 2 1
C 3 1
D 3 2
© UCLES 2011 5129/01/M/J/11
5
11 A 750 W microwave oven is used in a house where the mains voltage is 240 V.
Which fuse should be used in the plug?
A 3A B 5A C 7A D 10 A
12 Which nuclide has equal numbers of neutrons and protons?
1 4 7 9
A 1H B 2 He C 3 Li D 4 Be
13 How do the ionising abilities of beta-particles and gamma-rays compare with the ionising abilities
of alpha-particles?
beta-particles gamma-rays
A less less
B less more
C more less
D more more
14 Which property shows that a liquid is pure?
A It turns anhydrous copper(II) sulfate blue.
B It is colourless and odourless.
C It has no effect on red or blue litmus paper.
D It boils at a fixed temperature at a given pressure.
15 Which particle has the smallest mass?
A electron
B hydrogen ion
C neutron
D proton
© UCLES 2011 5129/01/M/J/11 [Turn over
DATA SHEET
The Periodic Table of the Elements
Group
I II III IV V VI VII 0
1 4
H He
Hydrogen Helium
1 2
7 9 11 12 14 16 19 20
Li Be B C N O F Ne
Lithium Beryllium Boron Carbon Nitrogen Oxygen Fluorine Neon
3 4 5 6 7 8 9 10
23 24 27 28 31 32 35.5 40
Na Mg Al Si P S Cl Ar
Sodium Magnesium Aluminium Silicon Phosphorus Sulfur Chlorine Argon
11 12 13 14 15 16 17 18
39 40 45 48 51 52 55 56 59 59 64 65 70 73 75 79 80 84
K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr
Potassium Calcium Scandium Titanium Vanadium Chromium Manganese Iron Cobalt Nickel Copper Zinc Gallium Germanium Arsenic Selenium Bromine Krypton
publisher will be pleased to make amends at the earliest possible opportunity.
19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36
16
85 88 89 91 93 96 101 103 106 108 112 115 119 122 128 127 131
Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe
Rubidium Strontium Yttrium Zirconium Niobium Molybdenum Technetium Ruthenium Rhodium Palladium Silver Cadmium Tin Antimony Tellurium Xenon
5129/01/M/J/10
Indium Iodine
37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54
133 137 139 178 181 184 186 190 192 195 197 201 204 207 209
Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn
Caesium Barium Lanthanum Hafnium Tantalum Tungsten Rhenium Osmium Iridium Platinum Gold Mercury Thallium Lead Bismuth Polonium Astatine Radon
55 56 57 * 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86
226 227
Fr Ra Ac
Cambridge Local Examinations Syndicate (UCLES), which is itself a department of the University of Cambridge.
Francium Radium Actinium
87 88 89
140 141 144 150 152 157 159 162 165 167 169 173 175
*58-71 Lanthanoid series
Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
90-103 Actinoid series Cerium Praseodymium Neodymium Promethium Samarium Europium Gadolinium Terbium Dysprosium Holmium Erbium Thulium Ytterbium Lutetium
58 59 60 61 62 63 64 65 66 67 68 69 70 71
a a = relative atomic mass 232 238
Key X X = atomic symbol Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
Thorium Protactinium Uranium Neptunium Plutonium Americium Curium Berkelium Californium Einsteinium Fermium Mendelevium Nobelium Lawrencium
b b = proton (atomic) number 90 91 92 93 94 95 96 97 98 99 100 101 102 103
The volume of one mole of any gas is 24 dm3 at room temperature and pressure (r.t.p.).
University of Cambridge International Examinations is part of the Cambridge Assessment Group. Cambridge Assessment is the brand name of University of
reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included, the
Permission to reproduce items where third-party owned material protected by copyright is included has been sought and cleared where possible. Every
You might also like
- 6 Using Moles: Extended CurriculumDocument2 pages6 Using Moles: Extended CurriculumkamalNo ratings yet
- QP Csci Test FebDocument13 pagesQP Csci Test FeblemonNo ratings yet
- 2010CIE GCE 5129 s10 QP 1Document16 pages2010CIE GCE 5129 s10 QP 1葡萄蘿蔔No ratings yet
- Physics Ch2ForcesAndMotionp2p4p6worksheetwithanswersDocument14 pagesPhysics Ch2ForcesAndMotionp2p4p6worksheetwithanswerssupratik30No ratings yet
- Physics: PAPER 1 Multiple ChoiceDocument21 pagesPhysics: PAPER 1 Multiple ChoiceZubairHassanNo ratings yet
- 0625 m20 QP 12 PDFDocument20 pages0625 m20 QP 12 PDFDesire KandawasvikaNo ratings yet
- Science Physics Chemistry October 2005 Paper 1Document20 pagesScience Physics Chemistry October 2005 Paper 1Phiri AgnesNo ratings yet
- Peperiksaan Pertengahan Tahun 2011Document17 pagesPeperiksaan Pertengahan Tahun 2011Sue Suraya NazaNo ratings yet
- Physics Paper 1 C 1 August PDFDocument16 pagesPhysics Paper 1 C 1 August PDFMohammad IrfanNo ratings yet
- CGS 09 5116 S4 Prelim P1 With Ans.Document15 pagesCGS 09 5116 S4 Prelim P1 With Ans.topcatNo ratings yet
- Paper 1 Nov 2000 PhysicsDocument16 pagesPaper 1 Nov 2000 Physicssolarixe100% (1)
- Physics: PAPER 1 Multiple ChoiceDocument20 pagesPhysics: PAPER 1 Multiple ChoiceIslamabad ALMA SchoolNo ratings yet
- O-Levels Physics ExemplarDocument56 pagesO-Levels Physics Exemplarbrendoored2No ratings yet
- Maths IGCSE Quick RevisionDocument16 pagesMaths IGCSE Quick Revisionlindy musekaNo ratings yet
- Cambridge IGCSE: Physics 0625/22Document16 pagesCambridge IGCSE: Physics 0625/22...No ratings yet
- Yr 2 IP Physical Science EOY 2020 SingaporeDocument19 pagesYr 2 IP Physical Science EOY 2020 Singaporecatsarepro17No ratings yet
- 2023 Specimen Paper 1Document16 pages2023 Specimen Paper 1Jąhąnząib Khąn KąkąrNo ratings yet
- Yr1 Physics Induction WorkDocument15 pagesYr1 Physics Induction Workalex.yori123456789No ratings yet
- June 2023 Question Paper 13Document16 pagesJune 2023 Question Paper 13Asmaa JoudehNo ratings yet
- امتحان رامز 25-1Document17 pagesامتحان رامز 25-1Ahmed AliNo ratings yet
- 2019 Sec 4 Pure Physics SA2 CHIJ ST Nicholas GirlsDocument42 pages2019 Sec 4 Pure Physics SA2 CHIJ ST Nicholas GirlsFrancis Ho HoNo ratings yet
- Sci Physics Prelim 09 4E5NA P1Document7 pagesSci Physics Prelim 09 4E5NA P1topcatNo ratings yet
- Work-Sheet For Surveying - IDocument6 pagesWork-Sheet For Surveying - IMaruf Jemal100% (1)
- Cambridge IGCSE: Physics 0625/22Document16 pagesCambridge IGCSE: Physics 0625/22Nisha zehraNo ratings yet
- Cambridge O Level: Physics 5054/12Document20 pagesCambridge O Level: Physics 5054/12iman jamilNo ratings yet
- Phy o 22 20Document16 pagesPhy o 22 20Sadman E AlamNo ratings yet
- OL Physics Book 1 (MCQ Theory) 2008 Till 2021Document409 pagesOL Physics Book 1 (MCQ Theory) 2008 Till 2021ABDULLAH'S SWEET VIDEOSNo ratings yet
- 0625 Paper 2Document527 pages0625 Paper 2Niki SNo ratings yet
- Cambridge International General Certificate of Secondary EducationDocument16 pagesCambridge International General Certificate of Secondary EducationfitriNo ratings yet
- P 1Document16 pagesP 1Chaos GamingNo ratings yet
- Cambridge IGCSE: PHYSICS 0625/01Document18 pagesCambridge IGCSE: PHYSICS 0625/01Aditya RaoNo ratings yet
- Cambridge IGCSE: Physics 0625/21Document16 pagesCambridge IGCSE: Physics 0625/21fayyaz haqueNo ratings yet
- Additional Combined Science: Cambridge International Examinations General Certificate of Education Ordinary LevelDocument20 pagesAdditional Combined Science: Cambridge International Examinations General Certificate of Education Ordinary Levelrodel.verzosaNo ratings yet
- March 2020 (v2) QP - Paper 1 CIE Physics IGCSEDocument20 pagesMarch 2020 (v2) QP - Paper 1 CIE Physics IGCSEjanggodanxdNo ratings yet
- Sec 4 Physics SA2 2018 Ahmad IbrahimDocument52 pagesSec 4 Physics SA2 2018 Ahmad Ibrahim또몽No ratings yet
- © Ucles 2022 0625/21/o/n/22Document5 pages© Ucles 2022 0625/21/o/n/22Md. Zubaer Mahmud RonyNo ratings yet
- Sda 15Document26 pagesSda 15Nothando PhologotswanaNo ratings yet
- Gce O-Level Preliminary Examination 2018: Ahmad Ibrahim Secondary SchoolDocument107 pagesGce O-Level Preliminary Examination 2018: Ahmad Ibrahim Secondary SchoolNamarr SinghNo ratings yet
- Revision Sec3Document14 pagesRevision Sec3โหน่ง บัวรดน้ำNo ratings yet
- Physics OneDocument8 pagesPhysics OneMUSOKE GERALDNo ratings yet
- Forces P2Document76 pagesForces P2arjunboruto1234No ratings yet
- Cambridge IGCSE: PHYSICS 0625/02Document14 pagesCambridge IGCSE: PHYSICS 0625/02Adedamola AdenugaNo ratings yet
- UntitledDocument20 pagesUntitledmorongwa malepengNo ratings yet
- QSS 5116 ScPhy P1 Prelim 09Document8 pagesQSS 5116 ScPhy P1 Prelim 09topcatNo ratings yet
- Physics Y13 Final Exam May 2021Document19 pagesPhysics Y13 Final Exam May 2021John samNo ratings yet
- Intensive CST-03 - (Code-A) - 13-06-2022Document23 pagesIntensive CST-03 - (Code-A) - 13-06-2022The BoNg FOOD recipesNo ratings yet
- 5054 w06 QP 1Document16 pages5054 w06 QP 1darkcastle100% (1)
- 1.1 Physical Quantities & Measurement TechniquesDocument39 pages1.1 Physical Quantities & Measurement TechniquesFatima Sohail 6aNo ratings yet
- Paper 1 May 2006 PhysicsDocument20 pagesPaper 1 May 2006 PhysicssolarixeNo ratings yet
- Physics P-12 IgcseDocument20 pagesPhysics P-12 IgcseareebNo ratings yet
- Physics 6091/01Document16 pagesPhysics 6091/01YogiNo ratings yet
- JEE Mains Sample Paper 3Document14 pagesJEE Mains Sample Paper 3random id100% (1)
- MCQ20BDocument16 pagesMCQ20BMELODY CHENNo ratings yet
- Ccss Phy MCQDocument8 pagesCcss Phy MCQoAeonNo ratings yet
- Unit 1 Physical Quantities, Units and MeasurementsDocument3 pagesUnit 1 Physical Quantities, Units and MeasurementsrafeyNo ratings yet
- Cambridge IGCSE: Physics 0625/11Document16 pagesCambridge IGCSE: Physics 0625/11tccrgvyvuubNo ratings yet
- 0625 m18 QP 22 PDFDocument16 pages0625 m18 QP 22 PDFamalNo ratings yet
- O level Physics Questions And Answer Practice Papers 3From EverandO level Physics Questions And Answer Practice Papers 3Rating: 3 out of 5 stars3/5 (1)
- Che Day1Document22 pagesChe Day1Janrey DohinogNo ratings yet
- Periodic Classification of Elements: Multiple Choice QuestionsDocument10 pagesPeriodic Classification of Elements: Multiple Choice QuestionsAryanNo ratings yet
- Astm A659-18Document3 pagesAstm A659-18Ryan ZhangNo ratings yet
- ReferenceDocument4 pagesReferenceMaries San PedroNo ratings yet
- Itec 2018-Akc-IrDocument63 pagesItec 2018-Akc-IrJohn F ServilleNo ratings yet
- Drilling Practical Manual - Drilling PDFDocument51 pagesDrilling Practical Manual - Drilling PDFAarzoo JobanputraNo ratings yet
- RPT Active OperatorsDocument120 pagesRPT Active Operatorsgijanie93No ratings yet
- Salting and CuringDocument13 pagesSalting and CuringSherylleneAguilera100% (1)
- Preventing Polymer Degradation During Mixing.Document8 pagesPreventing Polymer Degradation During Mixing.विनय शर्माNo ratings yet
- CHE 123 HWK Back and Redox TitrationsDocument3 pagesCHE 123 HWK Back and Redox TitrationsJuiloNo ratings yet
- 5th Grade - Mixing Substances - Lab ActivityDocument6 pages5th Grade - Mixing Substances - Lab Activityapi-254428474No ratings yet
- Scenario 1 S: Lamelar Tearing Occur in Set In? orDocument6 pagesScenario 1 S: Lamelar Tearing Occur in Set In? orBeny ZulNo ratings yet
- 12-19 Limit Test of ArsenicDocument1 page12-19 Limit Test of ArsenicBhupendra TomarNo ratings yet
- Al - Metal MatrixDocument38 pagesAl - Metal Matrixpramo_dassNo ratings yet
- Organic ExamDocument16 pagesOrganic ExamMnawaru KhalfaniNo ratings yet
- TDS - Total - Lubrilam Cleaner H1 - 1XQ - 201412 - enDocument1 pageTDS - Total - Lubrilam Cleaner H1 - 1XQ - 201412 - enVelibor KaranovicNo ratings yet
- Free Energies of Proton-Coupled Electron Transfer Reagents and Their ApplicationsDocument49 pagesFree Energies of Proton-Coupled Electron Transfer Reagents and Their ApplicationsZongxin JinNo ratings yet
- Standards Testing Plastic Tanks and VesselsDocument3 pagesStandards Testing Plastic Tanks and VesselsFaraj MohamedNo ratings yet
- Catalog Uk DrillingDocument226 pagesCatalog Uk DrillingJuanNo ratings yet
- Color Beta Carotene 2009 TRDocument20 pagesColor Beta Carotene 2009 TRUlfah Nurul MutmainnahNo ratings yet
- Nitration of BromobenzeneDocument3 pagesNitration of BromobenzeneLedina BanushllariNo ratings yet
- Basalt FiberDocument19 pagesBasalt FiberHitesh Shah100% (1)
- SDMC Heavy Metal SequestrationDocument1 pageSDMC Heavy Metal Sequestrationwacsii ccasullaNo ratings yet
- Biofuel Production From Mesua Ferrea L Seed OilDocument6 pagesBiofuel Production From Mesua Ferrea L Seed OilerpublicationNo ratings yet
- Eggs by Tonophore: Activation of Sea-Urchin CalciumDocument5 pagesEggs by Tonophore: Activation of Sea-Urchin CalciumCoCo GaldosNo ratings yet
- Liquid SulfurDocument20 pagesLiquid SulfurMarcin MaruchaNo ratings yet
- Incropera Appendix - 2011Document32 pagesIncropera Appendix - 2011Gracylla RoseNo ratings yet
- Zytel LCPA Product Reference GuideDocument2 pagesZytel LCPA Product Reference GuideProvocateur SamaraNo ratings yet
- VisbreakingDocument16 pagesVisbreakingAbdulAbdul100% (1)