Professional Documents
Culture Documents
Ions and Salts
Ions and Salts
Uploaded by
mansi bavliyaCopyright:
Available Formats
You might also like
- JBL Flip 6Document30 pagesJBL Flip 6maibac3300No ratings yet
- Weld SurveillanceDocument33 pagesWeld SurveillancecanakyuzNo ratings yet
- An Investigation of The Causes That Lead To Burn-InBurn-On in Heavy PDFDocument13 pagesAn Investigation of The Causes That Lead To Burn-InBurn-On in Heavy PDFTraian TiberiuNo ratings yet
- Chemistry 1.10Document2 pagesChemistry 1.10terran2020No ratings yet
- Chemistry 1.10Document2 pagesChemistry 1.10terran2020No ratings yet
- Chemistry 1.10Document2 pagesChemistry 1.10terran2020No ratings yet
- Ions and Saltsaazzqqww: Main ArticleDocument2 pagesIons and Saltsaazzqqww: Main Articleterran2020No ratings yet
- Ions and Saltsaazzqqwwaa: Main ArticleDocument2 pagesIons and Saltsaazzqqwwaa: Main Articleterran2020No ratings yet
- Chemistry 5Document3 pagesChemistry 5EINSTEINNo ratings yet
- 7 Many Reaction IntermediatesDocument2 pages7 Many Reaction IntermediatesYouTubeATPNo ratings yet
- Important Topics of ChemistryDocument7 pagesImportant Topics of ChemistrystrangeankitNo ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Oxidation and Reduction in Organic Chemistry AssignmentDocument9 pagesOxidation and Reduction in Organic Chemistry AssignmentAmanuel TarekegnNo ratings yet
- Chapter 2 Resonance and AcidDocument4 pagesChapter 2 Resonance and AcidChakalo HapalonNo ratings yet
- What Is The Relation Between Normality and MolarityDocument7 pagesWhat Is The Relation Between Normality and MolarityDrAmit VermaNo ratings yet
- REVIEWERDocument3 pagesREVIEWEREmelly PadillaNo ratings yet
- Chemical Bonds: Forming IonsDocument10 pagesChemical Bonds: Forming IonsShiela LumbagaNo ratings yet
- The Chemistry of LifeDocument8 pagesThe Chemistry of LifeAda Gay Olandia Serencio0% (1)
- Chapter 2 The Chemical Basis of LifeDocument11 pagesChapter 2 The Chemical Basis of LifeEuniece AnicocheNo ratings yet
- The Influence of PH On Zeta PotentialDocument24 pagesThe Influence of PH On Zeta PotentialJai MurugeshNo ratings yet
- OxidacionDocument1 pageOxidacionPaulaNo ratings yet
- English For Chemical EngineeringDocument27 pagesEnglish For Chemical EngineeringPham Van Tin B1909842No ratings yet
- Reaction Types: From Wikipedia, The Free EncyclopediaDocument6 pagesReaction Types: From Wikipedia, The Free Encyclopediasharih100% (5)
- 11 ChemistryDocument20 pages11 ChemistrykabhiNo ratings yet
- Redox Titration-Study MaterialDocument21 pagesRedox Titration-Study MaterialUma NathNo ratings yet
- Che ProjectDocument8 pagesChe Projectrudra samadhiyaNo ratings yet
- Lecture 2 - Lifes - Chemical - BasisDocument25 pagesLecture 2 - Lifes - Chemical - BasisPooja ChaudharyNo ratings yet
- Chemistry IntrodutionDocument13 pagesChemistry IntrodutionlalithaNo ratings yet
- Chemistry Terms and DefinitionsDocument6 pagesChemistry Terms and DefinitionsbesamundoNo ratings yet
- Organic Synthesis Note 2Document11 pagesOrganic Synthesis Note 2Otitoloju UsmanNo ratings yet
- Unit 4 Electrochemical EnergyDocument49 pagesUnit 4 Electrochemical EnergyRitchel Conde BoholNo ratings yet
- Class XI Redox Reactions NotesDocument5 pagesClass XI Redox Reactions NoteseasaNo ratings yet
- Chemical" Redirects Here. For Other Uses, SeeDocument7 pagesChemical" Redirects Here. For Other Uses, SeeZumilda ZainolNo ratings yet
- Lewis Concept of Acids and BasesDocument6 pagesLewis Concept of Acids and Basescayla mae carlosNo ratings yet
- CHEMISTRY TerminologiesDocument5 pagesCHEMISTRY TerminologiesSharreah LimNo ratings yet
- Ionic EquilibriumDocument13 pagesIonic EquilibriumTrupti ChavanNo ratings yet
- Redox Reactions: Reduction and Oxidation of The ReactantsDocument43 pagesRedox Reactions: Reduction and Oxidation of The ReactantsNur Atiqah Azmi100% (1)
- Group 7 Gen BioDocument27 pagesGroup 7 Gen BiojohndominicmmalubagoNo ratings yet
- Acidity and BasicityDocument89 pagesAcidity and Basicityjzllfrncsc100% (4)
- OxidationDocument5 pagesOxidationKudzanai MuchatoraNo ratings yet
- KBTU - 2023 - Fall - C&C - Lectures 19-21 - Acid-Base+redox CatalDocument38 pagesKBTU - 2023 - Fall - C&C - Lectures 19-21 - Acid-Base+redox CatalAkerke RamazanovaNo ratings yet
- Redox Matkul AnorganikDocument10 pagesRedox Matkul AnorganikAflah Lana Qistina SalsabilaNo ratings yet
- Chemistry Project FileDocument9 pagesChemistry Project FileRadhikaNo ratings yet
- 1-Basic of CorrosionDocument41 pages1-Basic of CorrosionMohamed Gawad ARayaNo ratings yet
- Chemical Bond Valence: Df8f7rf6f56d5dDocument91 pagesChemical Bond Valence: Df8f7rf6f56d5dfitradikojerdsswNo ratings yet
- English 2Document49 pagesEnglish 2mujieNo ratings yet
- 3 Chemical Basis of LifeDocument42 pages3 Chemical Basis of Lifemikel owen castilloNo ratings yet
- List of Definitions Chemistry MDocument8 pagesList of Definitions Chemistry Mlucky Dino54No ratings yet
- ChemicalsDocument6 pagesChemicalsArghadeep GhoshNo ratings yet
- 19.1. Definition of Oxidation State For Carbon - Organic Chemistry IIDocument9 pages19.1. Definition of Oxidation State For Carbon - Organic Chemistry IIMirza Bilal MughalNo ratings yet
- Hsslive Xi Chem Notes Anil CH 8. REDOX REACTIONSDocument6 pagesHsslive Xi Chem Notes Anil CH 8. REDOX REACTIONSKrishnendu NairNo ratings yet
- Chapter 4HSAB TheoryDocument15 pagesChapter 4HSAB TheoryAnurag NihalNo ratings yet
- Basics of Water ChemistryDocument51 pagesBasics of Water ChemistryRavi ShankarNo ratings yet
- Schneider - Chillers RangeDocument10 pagesSchneider - Chillers RangeerreagaaNo ratings yet
- Chemistry Bond NotesDocument5 pagesChemistry Bond NotesfffffNo ratings yet
- Reaction Mechanism.2Document68 pagesReaction Mechanism.2pasatiemporoseanneNo ratings yet
- 3-Classical Analytical MethodsDocument35 pages3-Classical Analytical MethodsAlice MakeNo ratings yet
- Lewis and Bronsted Concept of Acids and BasesDocument5 pagesLewis and Bronsted Concept of Acids and BasesPratiwi Indah SariNo ratings yet
- GCSE Chemistry Revision: Cheeky Revision ShortcutsFrom EverandGCSE Chemistry Revision: Cheeky Revision ShortcutsRating: 4.5 out of 5 stars4.5/5 (3)
- Combining Chemicals - Fun Chemistry Book for 4th Graders | Children's Chemistry BooksFrom EverandCombining Chemicals - Fun Chemistry Book for 4th Graders | Children's Chemistry BooksNo ratings yet
- ProcessesDocument2 pagesProcessesmansi bavliyaNo ratings yet
- The UserDocument1 pageThe Usermansi bavliyaNo ratings yet
- Interaction StyleDocument2 pagesInteraction Stylemansi bavliyaNo ratings yet
- Draft Version TimelineDocument2 pagesDraft Version Timelinemansi bavliyaNo ratings yet
- XHTML VersionsDocument2 pagesXHTML Versionsmansi bavliyaNo ratings yet
- SourcesDocument2 pagesSourcesmansi bavliyaNo ratings yet
- Always Design For Mobile FirstDocument5 pagesAlways Design For Mobile Firstmansi bavliyaNo ratings yet
- Growth and StandardizationDocument1 pageGrowth and Standardizationmansi bavliyaNo ratings yet
- Java ScriptDocument4 pagesJava Scriptmansi bavliyaNo ratings yet
- Modern Culinary UseDocument4 pagesModern Culinary Usemansi bavliyaNo ratings yet
- Field PeasDocument2 pagesField Peasmansi bavliyaNo ratings yet
- HTML Draft Version TimelineDocument2 pagesHTML Draft Version Timelinemansi bavliyaNo ratings yet
- In ScienceDocument2 pagesIn Sciencemansi bavliyaNo ratings yet
- HistoryDocument1 pageHistorymansi bavliyaNo ratings yet
- Semantic HTMLDocument1 pageSemantic HTMLmansi bavliyaNo ratings yet
- GradingDocument2 pagesGradingmansi bavliyaNo ratings yet
- Languages Where Green and Blue Are One ColorDocument2 pagesLanguages Where Green and Blue Are One Colormansi bavliyaNo ratings yet
- Jump To Navigation Jump To Search: For Other Uses, SeeDocument2 pagesJump To Navigation Jump To Search: For Other Uses, Seemansi bavliyaNo ratings yet
- HistoryDocument2 pagesHistorymansi bavliyaNo ratings yet
- GreenDocument3 pagesGreenmansi bavliyaNo ratings yet
- TrunkDocument2 pagesTrunkmansi bavliyaNo ratings yet
- HTMLDocument3 pagesHTMLmansi bavliyaNo ratings yet
- Experimental Test and Numerical Simulation of The Initial Crack Reinforced Concrete Beam in BendingDocument6 pagesExperimental Test and Numerical Simulation of The Initial Crack Reinforced Concrete Beam in BendingErik Wahyu PradanaNo ratings yet
- Brunnels@uccs - Edu Solids - Uccs.edu: B R, P DDocument3 pagesBrunnels@uccs - Edu Solids - Uccs.edu: B R, P DSheikh NasiruddinNo ratings yet
- How To Turn Plastic Waste Into Diesel Fuel CheaplyDocument25 pagesHow To Turn Plastic Waste Into Diesel Fuel CheaplyJeno Zsolt DudasNo ratings yet
- SFBM BTechDocument59 pagesSFBM BTechSaurav PaulNo ratings yet
- New Generation Calcium CarbonateDocument17 pagesNew Generation Calcium CarbonateVaibhav D RavalNo ratings yet
- Element - 111 - 128-Petrol Station BOQDocument6 pagesElement - 111 - 128-Petrol Station BOQMrweNo ratings yet
- Research and Project Work On The Rate of Evaporation of Different LiquidsDocument24 pagesResearch and Project Work On The Rate of Evaporation of Different LiquidsSamuelNo ratings yet
- Properties of WaterDocument6 pagesProperties of WaterNicoleNo ratings yet
- Corrosion: 2/engr Leonardo C Cuerdo, MSCDocument43 pagesCorrosion: 2/engr Leonardo C Cuerdo, MSCJayvee MauricioNo ratings yet
- How To Build A Volcano Science ProjectDocument5 pagesHow To Build A Volcano Science ProjectAnonymous hb3Dvgc7No ratings yet
- Subject: Method Statement For Shuttering WorksDocument5 pagesSubject: Method Statement For Shuttering WorksRounak MaheshwariNo ratings yet
- Chapter - 1: Shot BlastingDocument23 pagesChapter - 1: Shot BlastingsrinivasNo ratings yet
- Aluminum Glass and Windows For QuotationDocument2 pagesAluminum Glass and Windows For Quotationjarellano08No ratings yet
- HBI TestingDocument15 pagesHBI Testingreva cmNo ratings yet
- Metallurgical Defects of Cast Steel: SeminarDocument26 pagesMetallurgical Defects of Cast Steel: SeminarFacebookNo ratings yet
- Chapter 5Document22 pagesChapter 5Aftab AhmedNo ratings yet
- Titanium IIDocument1 pageTitanium IIsje productionNo ratings yet
- Presentation DelcorteDocument15 pagesPresentation Delcortecarlos2685No ratings yet
- Waterproofing SystemsDocument4 pagesWaterproofing SystemsfairmatechemicalNo ratings yet
- SLK G7 Q3wk8 Mongcopa RevisedDocument11 pagesSLK G7 Q3wk8 Mongcopa RevisedTabada NickyNo ratings yet
- Pressure VesselDocument114 pagesPressure Vesseldanemsal100% (3)
- Experiment No 8 Case Study On The Construction of Burj KhalifaDocument4 pagesExperiment No 8 Case Study On The Construction of Burj KhalifaBrijNo ratings yet
- Double 3 2011 PDFDocument19 pagesDouble 3 2011 PDFmalebogo makalareNo ratings yet
- 41xx SteelDocument2 pages41xx Steelkumar rajaNo ratings yet
- Edexcel Igcse ElectrolysisDocument86 pagesEdexcel Igcse ElectrolysisshoshoNo ratings yet
- Pipeline Design For Pigging: Pipeline Dimention Pipeline Materials DivertersDocument13 pagesPipeline Design For Pigging: Pipeline Dimention Pipeline Materials DivertersSazzadNo ratings yet
- Bankole Et Al. (2018)Document18 pagesBankole Et Al. (2018)alhassanNo ratings yet
Ions and Salts
Ions and Salts
Uploaded by
mansi bavliyaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Ions and Salts
Ions and Salts
Uploaded by
mansi bavliyaCopyright:
Available Formats
Ions and salts
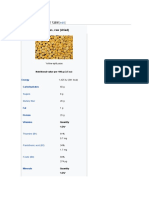
The crystal lattice structure of potassium chloride (KCl), a salt which is formed due to the attraction of
K+ cations and Cl− anions. Note how the overall charge of the ionic compound is zero.
Main article: Ion
An ion is a charged species, an atom or a molecule, that has lost or gained one or more
electrons. When an atom loses an electron and thus has more protons than electrons, the atom
is a positively charged ion or cation. When an atom gains an electron and thus has more
electrons than protons, the atom is a negatively charged ion or anion. Cations and anions can
form a crystalline lattice of neutral salts, such as the Na+ and Cl− ions forming sodium chloride, or
NaCl. Examples of polyatomic ions that do not split up during acid–base
reactions are hydroxide (OH−) and phosphate (PO43−).
Plasma is composed of gaseous matter that has been completely ionized, usually through high
temperature.
Acidity and basicity
Hydrogen bromide exists in the gas phase as a diatomic molecule
Main article: Acid–base reaction
A substance can often be classified as an acid or a base. There are several different theories
which explain acid–base behavior. The simplest is Arrhenius theory, which states that acid is a
substance that produces hydronium ions when it is dissolved in water, and a base is one that
produces hydroxide ions when dissolved in water. According to Brønsted–Lowry acid–base
theory, acids are substances that donate a positive hydrogen ion to another substance in a
chemical reaction; by extension, a base is the substance which receives that hydrogen ion.
A third common theory is Lewis acid–base theory, which is based on the formation of new
chemical bonds. Lewis theory explains that an acid is a substance which is capable of accepting
a pair of electrons from another substance during the process of bond formation, while a base is
a substance which can provide a pair of electrons to form a new bond. According to this theory,
the crucial things being exchanged are charges.[32][unreliable source?] There are several other ways in
which a substance may be classified as an acid or a base, as is evident in the history of this
concept.[33]
Acid strength is commonly measured by two methods. One measurement, based on the
Arrhenius definition of acidity, is pH, which is a measurement of the hydronium ion concentration
in a solution, as expressed on a negative logarithmic scale. Thus, solutions that have a low pH
have a high hydronium ion concentration and can be said to be more acidic. The other
measurement, based on the Brønsted–Lowry definition, is the acid dissociation constant (Ka),
which measures the relative ability of a substance to act as an acid under the Brønsted–Lowry
definition of an acid. That is, substances with a higher Ka are more likely to donate hydrogen ions
in chemical reactions than those with lower Ka values.
Redox
Main article: Redox
Redox (reduction-oxidation) reactions include all chemical reactions in which atoms have
their oxidation state changed by either gaining electrons (reduction) or losing electrons
(oxidation). Substances that have the ability to oxidize other substances are said to be oxidative
and are known as oxidizing agents, oxidants or oxidizers. An oxidant removes electrons from
another substance. Similarly, substances that have the ability to reduce other substances are
said to be reductive and are known as reducing agents, reductants, or reducers.
A reductant transfers electrons to another substance and is thus oxidized itself. And because it
"donates" electrons it is also called an electron donor. Oxidation and reduction properly refer to a
change in oxidation number—the actual transfer of electrons may never occur. Thus, oxidation is
better defined as an increase in oxidation number, and reduction as a decrease in oxidation
number.
Equilibrium
Main article: Chemical equilibrium
Although the concept of equilibrium is widely used across sciences, in the context of chemistry, it
arises whenever a number of different states of the chemical composition are possible, as for
example, in a mixture of several chemical compounds that can react with one another, or when a
substance can be present in more than one kind of phase.
A system of chemical substances at equilibrium, even though having an unchanging
composition, is most often not static; molecules of the substances continue to react with one
another thus giving rise to a dynamic equilibrium. Thus the concept describes the state in which
the parameters such as chemical composition remain unchanged over time.
You might also like
- JBL Flip 6Document30 pagesJBL Flip 6maibac3300No ratings yet
- Weld SurveillanceDocument33 pagesWeld SurveillancecanakyuzNo ratings yet
- An Investigation of The Causes That Lead To Burn-InBurn-On in Heavy PDFDocument13 pagesAn Investigation of The Causes That Lead To Burn-InBurn-On in Heavy PDFTraian TiberiuNo ratings yet
- Chemistry 1.10Document2 pagesChemistry 1.10terran2020No ratings yet
- Chemistry 1.10Document2 pagesChemistry 1.10terran2020No ratings yet
- Chemistry 1.10Document2 pagesChemistry 1.10terran2020No ratings yet
- Ions and Saltsaazzqqww: Main ArticleDocument2 pagesIons and Saltsaazzqqww: Main Articleterran2020No ratings yet
- Ions and Saltsaazzqqwwaa: Main ArticleDocument2 pagesIons and Saltsaazzqqwwaa: Main Articleterran2020No ratings yet
- Chemistry 5Document3 pagesChemistry 5EINSTEINNo ratings yet
- 7 Many Reaction IntermediatesDocument2 pages7 Many Reaction IntermediatesYouTubeATPNo ratings yet
- Important Topics of ChemistryDocument7 pagesImportant Topics of ChemistrystrangeankitNo ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Chemistry 1.11Document1 pageChemistry 1.11terran2020No ratings yet
- Oxidation and Reduction in Organic Chemistry AssignmentDocument9 pagesOxidation and Reduction in Organic Chemistry AssignmentAmanuel TarekegnNo ratings yet
- Chapter 2 Resonance and AcidDocument4 pagesChapter 2 Resonance and AcidChakalo HapalonNo ratings yet
- What Is The Relation Between Normality and MolarityDocument7 pagesWhat Is The Relation Between Normality and MolarityDrAmit VermaNo ratings yet
- REVIEWERDocument3 pagesREVIEWEREmelly PadillaNo ratings yet
- Chemical Bonds: Forming IonsDocument10 pagesChemical Bonds: Forming IonsShiela LumbagaNo ratings yet
- The Chemistry of LifeDocument8 pagesThe Chemistry of LifeAda Gay Olandia Serencio0% (1)
- Chapter 2 The Chemical Basis of LifeDocument11 pagesChapter 2 The Chemical Basis of LifeEuniece AnicocheNo ratings yet
- The Influence of PH On Zeta PotentialDocument24 pagesThe Influence of PH On Zeta PotentialJai MurugeshNo ratings yet
- OxidacionDocument1 pageOxidacionPaulaNo ratings yet
- English For Chemical EngineeringDocument27 pagesEnglish For Chemical EngineeringPham Van Tin B1909842No ratings yet
- Reaction Types: From Wikipedia, The Free EncyclopediaDocument6 pagesReaction Types: From Wikipedia, The Free Encyclopediasharih100% (5)
- 11 ChemistryDocument20 pages11 ChemistrykabhiNo ratings yet
- Redox Titration-Study MaterialDocument21 pagesRedox Titration-Study MaterialUma NathNo ratings yet
- Che ProjectDocument8 pagesChe Projectrudra samadhiyaNo ratings yet
- Lecture 2 - Lifes - Chemical - BasisDocument25 pagesLecture 2 - Lifes - Chemical - BasisPooja ChaudharyNo ratings yet
- Chemistry IntrodutionDocument13 pagesChemistry IntrodutionlalithaNo ratings yet
- Chemistry Terms and DefinitionsDocument6 pagesChemistry Terms and DefinitionsbesamundoNo ratings yet
- Organic Synthesis Note 2Document11 pagesOrganic Synthesis Note 2Otitoloju UsmanNo ratings yet
- Unit 4 Electrochemical EnergyDocument49 pagesUnit 4 Electrochemical EnergyRitchel Conde BoholNo ratings yet
- Class XI Redox Reactions NotesDocument5 pagesClass XI Redox Reactions NoteseasaNo ratings yet
- Chemical" Redirects Here. For Other Uses, SeeDocument7 pagesChemical" Redirects Here. For Other Uses, SeeZumilda ZainolNo ratings yet
- Lewis Concept of Acids and BasesDocument6 pagesLewis Concept of Acids and Basescayla mae carlosNo ratings yet
- CHEMISTRY TerminologiesDocument5 pagesCHEMISTRY TerminologiesSharreah LimNo ratings yet
- Ionic EquilibriumDocument13 pagesIonic EquilibriumTrupti ChavanNo ratings yet
- Redox Reactions: Reduction and Oxidation of The ReactantsDocument43 pagesRedox Reactions: Reduction and Oxidation of The ReactantsNur Atiqah Azmi100% (1)
- Group 7 Gen BioDocument27 pagesGroup 7 Gen BiojohndominicmmalubagoNo ratings yet
- Acidity and BasicityDocument89 pagesAcidity and Basicityjzllfrncsc100% (4)
- OxidationDocument5 pagesOxidationKudzanai MuchatoraNo ratings yet
- KBTU - 2023 - Fall - C&C - Lectures 19-21 - Acid-Base+redox CatalDocument38 pagesKBTU - 2023 - Fall - C&C - Lectures 19-21 - Acid-Base+redox CatalAkerke RamazanovaNo ratings yet
- Redox Matkul AnorganikDocument10 pagesRedox Matkul AnorganikAflah Lana Qistina SalsabilaNo ratings yet
- Chemistry Project FileDocument9 pagesChemistry Project FileRadhikaNo ratings yet
- 1-Basic of CorrosionDocument41 pages1-Basic of CorrosionMohamed Gawad ARayaNo ratings yet
- Chemical Bond Valence: Df8f7rf6f56d5dDocument91 pagesChemical Bond Valence: Df8f7rf6f56d5dfitradikojerdsswNo ratings yet
- English 2Document49 pagesEnglish 2mujieNo ratings yet
- 3 Chemical Basis of LifeDocument42 pages3 Chemical Basis of Lifemikel owen castilloNo ratings yet
- List of Definitions Chemistry MDocument8 pagesList of Definitions Chemistry Mlucky Dino54No ratings yet
- ChemicalsDocument6 pagesChemicalsArghadeep GhoshNo ratings yet
- 19.1. Definition of Oxidation State For Carbon - Organic Chemistry IIDocument9 pages19.1. Definition of Oxidation State For Carbon - Organic Chemistry IIMirza Bilal MughalNo ratings yet
- Hsslive Xi Chem Notes Anil CH 8. REDOX REACTIONSDocument6 pagesHsslive Xi Chem Notes Anil CH 8. REDOX REACTIONSKrishnendu NairNo ratings yet
- Chapter 4HSAB TheoryDocument15 pagesChapter 4HSAB TheoryAnurag NihalNo ratings yet
- Basics of Water ChemistryDocument51 pagesBasics of Water ChemistryRavi ShankarNo ratings yet
- Schneider - Chillers RangeDocument10 pagesSchneider - Chillers RangeerreagaaNo ratings yet
- Chemistry Bond NotesDocument5 pagesChemistry Bond NotesfffffNo ratings yet
- Reaction Mechanism.2Document68 pagesReaction Mechanism.2pasatiemporoseanneNo ratings yet
- 3-Classical Analytical MethodsDocument35 pages3-Classical Analytical MethodsAlice MakeNo ratings yet
- Lewis and Bronsted Concept of Acids and BasesDocument5 pagesLewis and Bronsted Concept of Acids and BasesPratiwi Indah SariNo ratings yet
- GCSE Chemistry Revision: Cheeky Revision ShortcutsFrom EverandGCSE Chemistry Revision: Cheeky Revision ShortcutsRating: 4.5 out of 5 stars4.5/5 (3)
- Combining Chemicals - Fun Chemistry Book for 4th Graders | Children's Chemistry BooksFrom EverandCombining Chemicals - Fun Chemistry Book for 4th Graders | Children's Chemistry BooksNo ratings yet
- ProcessesDocument2 pagesProcessesmansi bavliyaNo ratings yet
- The UserDocument1 pageThe Usermansi bavliyaNo ratings yet
- Interaction StyleDocument2 pagesInteraction Stylemansi bavliyaNo ratings yet
- Draft Version TimelineDocument2 pagesDraft Version Timelinemansi bavliyaNo ratings yet
- XHTML VersionsDocument2 pagesXHTML Versionsmansi bavliyaNo ratings yet
- SourcesDocument2 pagesSourcesmansi bavliyaNo ratings yet
- Always Design For Mobile FirstDocument5 pagesAlways Design For Mobile Firstmansi bavliyaNo ratings yet
- Growth and StandardizationDocument1 pageGrowth and Standardizationmansi bavliyaNo ratings yet
- Java ScriptDocument4 pagesJava Scriptmansi bavliyaNo ratings yet
- Modern Culinary UseDocument4 pagesModern Culinary Usemansi bavliyaNo ratings yet
- Field PeasDocument2 pagesField Peasmansi bavliyaNo ratings yet
- HTML Draft Version TimelineDocument2 pagesHTML Draft Version Timelinemansi bavliyaNo ratings yet
- In ScienceDocument2 pagesIn Sciencemansi bavliyaNo ratings yet
- HistoryDocument1 pageHistorymansi bavliyaNo ratings yet
- Semantic HTMLDocument1 pageSemantic HTMLmansi bavliyaNo ratings yet
- GradingDocument2 pagesGradingmansi bavliyaNo ratings yet
- Languages Where Green and Blue Are One ColorDocument2 pagesLanguages Where Green and Blue Are One Colormansi bavliyaNo ratings yet
- Jump To Navigation Jump To Search: For Other Uses, SeeDocument2 pagesJump To Navigation Jump To Search: For Other Uses, Seemansi bavliyaNo ratings yet
- HistoryDocument2 pagesHistorymansi bavliyaNo ratings yet
- GreenDocument3 pagesGreenmansi bavliyaNo ratings yet
- TrunkDocument2 pagesTrunkmansi bavliyaNo ratings yet
- HTMLDocument3 pagesHTMLmansi bavliyaNo ratings yet
- Experimental Test and Numerical Simulation of The Initial Crack Reinforced Concrete Beam in BendingDocument6 pagesExperimental Test and Numerical Simulation of The Initial Crack Reinforced Concrete Beam in BendingErik Wahyu PradanaNo ratings yet
- Brunnels@uccs - Edu Solids - Uccs.edu: B R, P DDocument3 pagesBrunnels@uccs - Edu Solids - Uccs.edu: B R, P DSheikh NasiruddinNo ratings yet
- How To Turn Plastic Waste Into Diesel Fuel CheaplyDocument25 pagesHow To Turn Plastic Waste Into Diesel Fuel CheaplyJeno Zsolt DudasNo ratings yet
- SFBM BTechDocument59 pagesSFBM BTechSaurav PaulNo ratings yet
- New Generation Calcium CarbonateDocument17 pagesNew Generation Calcium CarbonateVaibhav D RavalNo ratings yet
- Element - 111 - 128-Petrol Station BOQDocument6 pagesElement - 111 - 128-Petrol Station BOQMrweNo ratings yet
- Research and Project Work On The Rate of Evaporation of Different LiquidsDocument24 pagesResearch and Project Work On The Rate of Evaporation of Different LiquidsSamuelNo ratings yet
- Properties of WaterDocument6 pagesProperties of WaterNicoleNo ratings yet
- Corrosion: 2/engr Leonardo C Cuerdo, MSCDocument43 pagesCorrosion: 2/engr Leonardo C Cuerdo, MSCJayvee MauricioNo ratings yet
- How To Build A Volcano Science ProjectDocument5 pagesHow To Build A Volcano Science ProjectAnonymous hb3Dvgc7No ratings yet
- Subject: Method Statement For Shuttering WorksDocument5 pagesSubject: Method Statement For Shuttering WorksRounak MaheshwariNo ratings yet
- Chapter - 1: Shot BlastingDocument23 pagesChapter - 1: Shot BlastingsrinivasNo ratings yet
- Aluminum Glass and Windows For QuotationDocument2 pagesAluminum Glass and Windows For Quotationjarellano08No ratings yet
- HBI TestingDocument15 pagesHBI Testingreva cmNo ratings yet
- Metallurgical Defects of Cast Steel: SeminarDocument26 pagesMetallurgical Defects of Cast Steel: SeminarFacebookNo ratings yet
- Chapter 5Document22 pagesChapter 5Aftab AhmedNo ratings yet
- Titanium IIDocument1 pageTitanium IIsje productionNo ratings yet
- Presentation DelcorteDocument15 pagesPresentation Delcortecarlos2685No ratings yet
- Waterproofing SystemsDocument4 pagesWaterproofing SystemsfairmatechemicalNo ratings yet
- SLK G7 Q3wk8 Mongcopa RevisedDocument11 pagesSLK G7 Q3wk8 Mongcopa RevisedTabada NickyNo ratings yet
- Pressure VesselDocument114 pagesPressure Vesseldanemsal100% (3)
- Experiment No 8 Case Study On The Construction of Burj KhalifaDocument4 pagesExperiment No 8 Case Study On The Construction of Burj KhalifaBrijNo ratings yet
- Double 3 2011 PDFDocument19 pagesDouble 3 2011 PDFmalebogo makalareNo ratings yet
- 41xx SteelDocument2 pages41xx Steelkumar rajaNo ratings yet
- Edexcel Igcse ElectrolysisDocument86 pagesEdexcel Igcse ElectrolysisshoshoNo ratings yet
- Pipeline Design For Pigging: Pipeline Dimention Pipeline Materials DivertersDocument13 pagesPipeline Design For Pigging: Pipeline Dimention Pipeline Materials DivertersSazzadNo ratings yet
- Bankole Et Al. (2018)Document18 pagesBankole Et Al. (2018)alhassanNo ratings yet