Professional Documents
Culture Documents
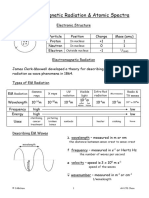
Calculate The Wave Number, Wavelength and Frequency of Second Member (H - Line) of Balmer Series of Hydrogen Atom. Given: R 1.097x10 M
Calculate The Wave Number, Wavelength and Frequency of Second Member (H - Line) of Balmer Series of Hydrogen Atom. Given: R 1.097x10 M
Uploaded by
Kumkum KumbarahalliCopyright:
Available Formats
You might also like
- PS2Document2 pagesPS2Truong CaiNo ratings yet
- MATE1000 Physics and Engineering of Materials: Semester 2 2007 Tutorial 2 (Atomic Structure 2)Document1 pageMATE1000 Physics and Engineering of Materials: Semester 2 2007 Tutorial 2 (Atomic Structure 2)clearcastingNo ratings yet
- 2.1 Atomic StructureDocument60 pages2.1 Atomic StructureKamariah IsmailNo ratings yet
- ATOMS TestDocument2 pagesATOMS TestChitesh MarmatNo ratings yet
- AtomDocument6 pagesAtomShivani Ekant YadavNo ratings yet
- Atomic SpectraDocument12 pagesAtomic SpectraHindi ScientoNo ratings yet
- Written By: MR Chew Sensei Estimate Time Complete: 10 PeriodsDocument28 pagesWritten By: MR Chew Sensei Estimate Time Complete: 10 PeriodsHappy KingNo ratings yet
- Atomic StructureDocument49 pagesAtomic StructureFatimaNo ratings yet
- Serie TD 3 AnglaisDocument3 pagesSerie TD 3 AnglaisjosbenachenhouNo ratings yet
- NCERT Solutions For Class 12 Physics Chapter 12 AtomsDocument14 pagesNCERT Solutions For Class 12 Physics Chapter 12 Atoms943 Shivu 10cNo ratings yet
- Warrior: Physical ChemistryDocument7 pagesWarrior: Physical ChemistryGowri ShankarNo ratings yet
- Atomic PhysicsDocument128 pagesAtomic PhysicsApoorva PrakashNo ratings yet
- Chapter-5 - Periodicity & Electronic Structure of AtomsDocument14 pagesChapter-5 - Periodicity & Electronic Structure of AtomsV KumarNo ratings yet
- Bohr's Theory of The Hydrogen Atom - Physics IIDocument22 pagesBohr's Theory of The Hydrogen Atom - Physics IISayyad aliNo ratings yet
- Models 2017 Complete Model AnswerDocument6 pagesModels 2017 Complete Model AnswersamirNo ratings yet
- Atomic StructureDocument85 pagesAtomic StructureMUHAMMAD FAHMI MOHD ZAMZANINo ratings yet
- Tutorial 1: Subatomicphysics: Nuclear PhysicsDocument8 pagesTutorial 1: Subatomicphysics: Nuclear PhysicsGregory HillhouseNo ratings yet
- Physics Notes Chapter - 18 The Atomic Spectra PDFDocument10 pagesPhysics Notes Chapter - 18 The Atomic Spectra PDFranaateeqNo ratings yet
- 3.3 BohrÆs Atomic ModelDocument35 pages3.3 BohrÆs Atomic ModelAnisha Syazwana Binti RoslyNo ratings yet
- Atomic Structure, X-Ray & Radio Activity: P F F Iit-JDocument3 pagesAtomic Structure, X-Ray & Radio Activity: P F F Iit-JsayondeepNo ratings yet
- Quantum ChemistryDocument71 pagesQuantum Chemistrybinseung skzNo ratings yet
- Class 11 Chemistry Chapter 2 Structure of AtomDocument15 pagesClass 11 Chemistry Chapter 2 Structure of Atomgokul100% (1)
- Lecture Note On Atomic Structure (Part 2)Document23 pagesLecture Note On Atomic Structure (Part 2)matthewamechinaNo ratings yet
- Chemistry Pre-U Chemistry Sem 1 Chap 2Document48 pagesChemistry Pre-U Chemistry Sem 1 Chap 2Jia HuiNo ratings yet
- Atomic PhysicsDocument58 pagesAtomic PhysicsBasitNo ratings yet
- CHM131 - CH 3 - The Electronic Structure of Atoms and Periodic Table PDFDocument102 pagesCHM131 - CH 3 - The Electronic Structure of Atoms and Periodic Table PDFRabiatul AdawiyyahNo ratings yet
- 2B Atomic StructureDocument5 pages2B Atomic StructureSoham Thinks DrdzNo ratings yet
- Unit 2 The Electronic Structure of Atoms and The Periodic TableDocument22 pagesUnit 2 The Electronic Structure of Atoms and The Periodic TableAragorn ChanNo ratings yet
- Topic 10.quantumDocument34 pagesTopic 10.quantumNOR AZAM BIN ENDOT / FSNo ratings yet
- Classnote 523c02d03b526 PDFDocument30 pagesClassnote 523c02d03b526 PDFmirsNo ratings yet
- Tomic Tructure: Etoos Academy Pvt. LTDDocument30 pagesTomic Tructure: Etoos Academy Pvt. LTDDivyanshu RajNo ratings yet
- Structure of AtomDocument20 pagesStructure of AtomSachin KumarNo ratings yet
- JotterPad - JotterPad - FinalDocument26 pagesJotterPad - JotterPad - FinalOlajide HeritageNo ratings yet
- ATOMIC SPECTRA 2012 W AnswerDocument29 pagesATOMIC SPECTRA 2012 W AnswerAng chong bengNo ratings yet
- 1 Basics of Atomic Structure-3-19Document17 pages1 Basics of Atomic Structure-3-19Raj KishoreNo ratings yet
- Hydrogen Spectrum Chemistry Questions With Solutions - Docx 2Document7 pagesHydrogen Spectrum Chemistry Questions With Solutions - Docx 2nethmiayodyamrNo ratings yet
- Atomic Structure: Chapter - 1Document14 pagesAtomic Structure: Chapter - 1Cube WorldNo ratings yet
- Worksheet 10 PDFDocument4 pagesWorksheet 10 PDFJosh FlorentinoNo ratings yet
- Edited Assignment Atomic Structure JH Sir-2611Document30 pagesEdited Assignment Atomic Structure JH Sir-2611d anjilappaNo ratings yet
- Cbse World: NCERT Solution Part - 1Document17 pagesCbse World: NCERT Solution Part - 1Bibha KumariNo ratings yet
- Mona NotesDocument2 pagesMona NotessnyvalkNo ratings yet
- Chemisty Chapter 5sept8cDocument13 pagesChemisty Chapter 5sept8cVictoria MooreNo ratings yet
- Kisi Kisi FT TERM 4Document13 pagesKisi Kisi FT TERM 4Owain Cato DaniwanNo ratings yet
- Dhselect 5Document9 pagesDhselect 5Biswanath Gouda (Biswanath)No ratings yet
- VT Sir (Atomic Structure) + PYQDocument70 pagesVT Sir (Atomic Structure) + PYQkrarchna115No ratings yet
- 1 IntroductionDocument42 pages1 IntroductionJustine CabuayNo ratings yet
- Modern Physics and Quantum Mechanics Mod-2 PDFDocument28 pagesModern Physics and Quantum Mechanics Mod-2 PDFShreyas SeshadriNo ratings yet
- NCERT Solutions For Class 12 Physics Chapter 12 AtomsDocument14 pagesNCERT Solutions For Class 12 Physics Chapter 12 AtomsKritika MishraNo ratings yet
- EFFICIENCY - OF - SOLITARY-WAVES - RADIATED - BY - THE - DISCH - Explications Effet PrioreDocument10 pagesEFFICIENCY - OF - SOLITARY-WAVES - RADIATED - BY - THE - DISCH - Explications Effet Priorem18999843No ratings yet
- Section:BOYS' &GIRLS' Date:7-04-20 Class & Div.:XI (All Divisions) Subject:CHEMISTRY Lesson / Topic: ATOMIC STRUCTURE - 1Document8 pagesSection:BOYS' &GIRLS' Date:7-04-20 Class & Div.:XI (All Divisions) Subject:CHEMISTRY Lesson / Topic: ATOMIC STRUCTURE - 1Mohammed IliasNo ratings yet
- Bohr's TheoryDocument26 pagesBohr's TheorySukhwinder Singh GillNo ratings yet
- Chemistry Chapter 5Document11 pagesChemistry Chapter 5Victoria MooreNo ratings yet
- 1.1 AH CfE Chemistry NotesDocument7 pages1.1 AH CfE Chemistry Noteskira.zavyalova24No ratings yet
- Temu 1Document36 pagesTemu 1Farida UtamiNo ratings yet
- L - 19-21 - Modern - Physics - Atomic - Structure AMPDocument25 pagesL - 19-21 - Modern - Physics - Atomic - Structure AMPAnindya BiswasNo ratings yet
- Atoms and NucleiDocument18 pagesAtoms and NucleihumanruhulNo ratings yet
- Physics II (4103) Part10-1Document9 pagesPhysics II (4103) Part10-1wafa nagiNo ratings yet
- HC Verma Solutions Class 12 Physics Chapter 21 Bohr's Theory and Physics of AtomDocument12 pagesHC Verma Solutions Class 12 Physics Chapter 21 Bohr's Theory and Physics of Atomtarun.8287yashNo ratings yet
- Feynman Lectures Simplified 2C: Electromagnetism: in Relativity & in Dense MatterFrom EverandFeynman Lectures Simplified 2C: Electromagnetism: in Relativity & in Dense MatterNo ratings yet
- LAB 1 To LAB 10 - Jupyter NotebookDocument20 pagesLAB 1 To LAB 10 - Jupyter NotebookKumkum KumbarahalliNo ratings yet
- ChemDocument7 pagesChemKumkum KumbarahalliNo ratings yet
- BPWSK106Document2 pagesBPWSK106Kumkum KumbarahalliNo ratings yet
- SF of HealthDocument2 pagesSF of HealthKumkum KumbarahalliNo ratings yet
- M5 Ece Imp ADocument10 pagesM5 Ece Imp AKumkum KumbarahalliNo ratings yet
- Chem Viva - OrganizedDocument8 pagesChem Viva - OrganizedKumkum KumbarahalliNo ratings yet
- Sri Chaitanya IIT Academy., India.: JEE-MAIN (WTM-1)Document1 pageSri Chaitanya IIT Academy., India.: JEE-MAIN (WTM-1)Kumkum KumbarahalliNo ratings yet
- II PUC - Mathematics (TARGET CENTUM) - Question BankDocument68 pagesII PUC - Mathematics (TARGET CENTUM) - Question BankKumkum Kumbarahalli100% (1)
- Kcet - Paper Analysis: - Total Number of Questions 60Document9 pagesKcet - Paper Analysis: - Total Number of Questions 60Kumkum KumbarahalliNo ratings yet
- Misce-Exe-robbillh: SolnDocument12 pagesMisce-Exe-robbillh: SolnKumkum KumbarahalliNo ratings yet
- New Doc 12-Oct-2021 10.33 AmDocument3 pagesNew Doc 12-Oct-2021 10.33 AmKumkum KumbarahalliNo ratings yet
- Yds ProblemsDocument3 pagesYds ProblemsKumkum KumbarahalliNo ratings yet
- Q No 44 and 45 Formulae: Department of Physics Pradeep PadmanabhanDocument5 pagesQ No 44 and 45 Formulae: Department of Physics Pradeep PadmanabhanKumkum KumbarahalliNo ratings yet
- NUCLEUS ProblemsDocument3 pagesNUCLEUS ProblemsKumkum KumbarahalliNo ratings yet
Calculate The Wave Number, Wavelength and Frequency of Second Member (H - Line) of Balmer Series of Hydrogen Atom. Given: R 1.097x10 M
Calculate The Wave Number, Wavelength and Frequency of Second Member (H - Line) of Balmer Series of Hydrogen Atom. Given: R 1.097x10 M
Uploaded by
Kumkum KumbarahalliOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Calculate The Wave Number, Wavelength and Frequency of Second Member (H - Line) of Balmer Series of Hydrogen Atom. Given: R 1.097x10 M
Calculate The Wave Number, Wavelength and Frequency of Second Member (H - Line) of Balmer Series of Hydrogen Atom. Given: R 1.097x10 M
Uploaded by
Kumkum KumbarahalliCopyright:
Available Formats
1
ATOMS
1. Calculate the wave number, wavelength and frequency of second member
[H𝛽—line] of Balmer series of hydrogen atom. Given: R=1.097x107 m- 1 .
2. Calculate longest and shortest wavelength of Balmer series. Given:
R=1.097x 107 m-1.
ATOMS DEPARTMENT OF PHYSICS Prescribed by PRADEEP P
2
3. When certain energy is supplied to hydrogen atom, electron jumps from n=1
to n=2 state. Find
a) Energy absorbed by the electron and b) The wavelength of the radiation
emitted when the electron comes to ground state.
Given: Ground state energy of hydrogen atom, E1 = -13.6 eV, Planck’s
constant, h = 6. 625 x 10-34 Js and Speed of light,
c = 3 x 10B m/s
ATOMS DEPARTMENT OF PHYSICS Prescribed by PRADEEP P
You might also like
- PS2Document2 pagesPS2Truong CaiNo ratings yet
- MATE1000 Physics and Engineering of Materials: Semester 2 2007 Tutorial 2 (Atomic Structure 2)Document1 pageMATE1000 Physics and Engineering of Materials: Semester 2 2007 Tutorial 2 (Atomic Structure 2)clearcastingNo ratings yet
- 2.1 Atomic StructureDocument60 pages2.1 Atomic StructureKamariah IsmailNo ratings yet
- ATOMS TestDocument2 pagesATOMS TestChitesh MarmatNo ratings yet
- AtomDocument6 pagesAtomShivani Ekant YadavNo ratings yet
- Atomic SpectraDocument12 pagesAtomic SpectraHindi ScientoNo ratings yet
- Written By: MR Chew Sensei Estimate Time Complete: 10 PeriodsDocument28 pagesWritten By: MR Chew Sensei Estimate Time Complete: 10 PeriodsHappy KingNo ratings yet
- Atomic StructureDocument49 pagesAtomic StructureFatimaNo ratings yet
- Serie TD 3 AnglaisDocument3 pagesSerie TD 3 AnglaisjosbenachenhouNo ratings yet
- NCERT Solutions For Class 12 Physics Chapter 12 AtomsDocument14 pagesNCERT Solutions For Class 12 Physics Chapter 12 Atoms943 Shivu 10cNo ratings yet
- Warrior: Physical ChemistryDocument7 pagesWarrior: Physical ChemistryGowri ShankarNo ratings yet
- Atomic PhysicsDocument128 pagesAtomic PhysicsApoorva PrakashNo ratings yet
- Chapter-5 - Periodicity & Electronic Structure of AtomsDocument14 pagesChapter-5 - Periodicity & Electronic Structure of AtomsV KumarNo ratings yet
- Bohr's Theory of The Hydrogen Atom - Physics IIDocument22 pagesBohr's Theory of The Hydrogen Atom - Physics IISayyad aliNo ratings yet
- Models 2017 Complete Model AnswerDocument6 pagesModels 2017 Complete Model AnswersamirNo ratings yet
- Atomic StructureDocument85 pagesAtomic StructureMUHAMMAD FAHMI MOHD ZAMZANINo ratings yet
- Tutorial 1: Subatomicphysics: Nuclear PhysicsDocument8 pagesTutorial 1: Subatomicphysics: Nuclear PhysicsGregory HillhouseNo ratings yet
- Physics Notes Chapter - 18 The Atomic Spectra PDFDocument10 pagesPhysics Notes Chapter - 18 The Atomic Spectra PDFranaateeqNo ratings yet
- 3.3 BohrÆs Atomic ModelDocument35 pages3.3 BohrÆs Atomic ModelAnisha Syazwana Binti RoslyNo ratings yet
- Atomic Structure, X-Ray & Radio Activity: P F F Iit-JDocument3 pagesAtomic Structure, X-Ray & Radio Activity: P F F Iit-JsayondeepNo ratings yet
- Quantum ChemistryDocument71 pagesQuantum Chemistrybinseung skzNo ratings yet
- Class 11 Chemistry Chapter 2 Structure of AtomDocument15 pagesClass 11 Chemistry Chapter 2 Structure of Atomgokul100% (1)
- Lecture Note On Atomic Structure (Part 2)Document23 pagesLecture Note On Atomic Structure (Part 2)matthewamechinaNo ratings yet
- Chemistry Pre-U Chemistry Sem 1 Chap 2Document48 pagesChemistry Pre-U Chemistry Sem 1 Chap 2Jia HuiNo ratings yet
- Atomic PhysicsDocument58 pagesAtomic PhysicsBasitNo ratings yet
- CHM131 - CH 3 - The Electronic Structure of Atoms and Periodic Table PDFDocument102 pagesCHM131 - CH 3 - The Electronic Structure of Atoms and Periodic Table PDFRabiatul AdawiyyahNo ratings yet
- 2B Atomic StructureDocument5 pages2B Atomic StructureSoham Thinks DrdzNo ratings yet
- Unit 2 The Electronic Structure of Atoms and The Periodic TableDocument22 pagesUnit 2 The Electronic Structure of Atoms and The Periodic TableAragorn ChanNo ratings yet
- Topic 10.quantumDocument34 pagesTopic 10.quantumNOR AZAM BIN ENDOT / FSNo ratings yet
- Classnote 523c02d03b526 PDFDocument30 pagesClassnote 523c02d03b526 PDFmirsNo ratings yet
- Tomic Tructure: Etoos Academy Pvt. LTDDocument30 pagesTomic Tructure: Etoos Academy Pvt. LTDDivyanshu RajNo ratings yet
- Structure of AtomDocument20 pagesStructure of AtomSachin KumarNo ratings yet
- JotterPad - JotterPad - FinalDocument26 pagesJotterPad - JotterPad - FinalOlajide HeritageNo ratings yet
- ATOMIC SPECTRA 2012 W AnswerDocument29 pagesATOMIC SPECTRA 2012 W AnswerAng chong bengNo ratings yet
- 1 Basics of Atomic Structure-3-19Document17 pages1 Basics of Atomic Structure-3-19Raj KishoreNo ratings yet
- Hydrogen Spectrum Chemistry Questions With Solutions - Docx 2Document7 pagesHydrogen Spectrum Chemistry Questions With Solutions - Docx 2nethmiayodyamrNo ratings yet
- Atomic Structure: Chapter - 1Document14 pagesAtomic Structure: Chapter - 1Cube WorldNo ratings yet
- Worksheet 10 PDFDocument4 pagesWorksheet 10 PDFJosh FlorentinoNo ratings yet
- Edited Assignment Atomic Structure JH Sir-2611Document30 pagesEdited Assignment Atomic Structure JH Sir-2611d anjilappaNo ratings yet
- Cbse World: NCERT Solution Part - 1Document17 pagesCbse World: NCERT Solution Part - 1Bibha KumariNo ratings yet
- Mona NotesDocument2 pagesMona NotessnyvalkNo ratings yet
- Chemisty Chapter 5sept8cDocument13 pagesChemisty Chapter 5sept8cVictoria MooreNo ratings yet
- Kisi Kisi FT TERM 4Document13 pagesKisi Kisi FT TERM 4Owain Cato DaniwanNo ratings yet
- Dhselect 5Document9 pagesDhselect 5Biswanath Gouda (Biswanath)No ratings yet
- VT Sir (Atomic Structure) + PYQDocument70 pagesVT Sir (Atomic Structure) + PYQkrarchna115No ratings yet
- 1 IntroductionDocument42 pages1 IntroductionJustine CabuayNo ratings yet
- Modern Physics and Quantum Mechanics Mod-2 PDFDocument28 pagesModern Physics and Quantum Mechanics Mod-2 PDFShreyas SeshadriNo ratings yet
- NCERT Solutions For Class 12 Physics Chapter 12 AtomsDocument14 pagesNCERT Solutions For Class 12 Physics Chapter 12 AtomsKritika MishraNo ratings yet
- EFFICIENCY - OF - SOLITARY-WAVES - RADIATED - BY - THE - DISCH - Explications Effet PrioreDocument10 pagesEFFICIENCY - OF - SOLITARY-WAVES - RADIATED - BY - THE - DISCH - Explications Effet Priorem18999843No ratings yet
- Section:BOYS' &GIRLS' Date:7-04-20 Class & Div.:XI (All Divisions) Subject:CHEMISTRY Lesson / Topic: ATOMIC STRUCTURE - 1Document8 pagesSection:BOYS' &GIRLS' Date:7-04-20 Class & Div.:XI (All Divisions) Subject:CHEMISTRY Lesson / Topic: ATOMIC STRUCTURE - 1Mohammed IliasNo ratings yet
- Bohr's TheoryDocument26 pagesBohr's TheorySukhwinder Singh GillNo ratings yet
- Chemistry Chapter 5Document11 pagesChemistry Chapter 5Victoria MooreNo ratings yet
- 1.1 AH CfE Chemistry NotesDocument7 pages1.1 AH CfE Chemistry Noteskira.zavyalova24No ratings yet
- Temu 1Document36 pagesTemu 1Farida UtamiNo ratings yet
- L - 19-21 - Modern - Physics - Atomic - Structure AMPDocument25 pagesL - 19-21 - Modern - Physics - Atomic - Structure AMPAnindya BiswasNo ratings yet
- Atoms and NucleiDocument18 pagesAtoms and NucleihumanruhulNo ratings yet
- Physics II (4103) Part10-1Document9 pagesPhysics II (4103) Part10-1wafa nagiNo ratings yet
- HC Verma Solutions Class 12 Physics Chapter 21 Bohr's Theory and Physics of AtomDocument12 pagesHC Verma Solutions Class 12 Physics Chapter 21 Bohr's Theory and Physics of Atomtarun.8287yashNo ratings yet
- Feynman Lectures Simplified 2C: Electromagnetism: in Relativity & in Dense MatterFrom EverandFeynman Lectures Simplified 2C: Electromagnetism: in Relativity & in Dense MatterNo ratings yet
- LAB 1 To LAB 10 - Jupyter NotebookDocument20 pagesLAB 1 To LAB 10 - Jupyter NotebookKumkum KumbarahalliNo ratings yet
- ChemDocument7 pagesChemKumkum KumbarahalliNo ratings yet
- BPWSK106Document2 pagesBPWSK106Kumkum KumbarahalliNo ratings yet
- SF of HealthDocument2 pagesSF of HealthKumkum KumbarahalliNo ratings yet
- M5 Ece Imp ADocument10 pagesM5 Ece Imp AKumkum KumbarahalliNo ratings yet
- Chem Viva - OrganizedDocument8 pagesChem Viva - OrganizedKumkum KumbarahalliNo ratings yet
- Sri Chaitanya IIT Academy., India.: JEE-MAIN (WTM-1)Document1 pageSri Chaitanya IIT Academy., India.: JEE-MAIN (WTM-1)Kumkum KumbarahalliNo ratings yet
- II PUC - Mathematics (TARGET CENTUM) - Question BankDocument68 pagesII PUC - Mathematics (TARGET CENTUM) - Question BankKumkum Kumbarahalli100% (1)
- Kcet - Paper Analysis: - Total Number of Questions 60Document9 pagesKcet - Paper Analysis: - Total Number of Questions 60Kumkum KumbarahalliNo ratings yet
- Misce-Exe-robbillh: SolnDocument12 pagesMisce-Exe-robbillh: SolnKumkum KumbarahalliNo ratings yet
- New Doc 12-Oct-2021 10.33 AmDocument3 pagesNew Doc 12-Oct-2021 10.33 AmKumkum KumbarahalliNo ratings yet
- Yds ProblemsDocument3 pagesYds ProblemsKumkum KumbarahalliNo ratings yet
- Q No 44 and 45 Formulae: Department of Physics Pradeep PadmanabhanDocument5 pagesQ No 44 and 45 Formulae: Department of Physics Pradeep PadmanabhanKumkum KumbarahalliNo ratings yet
- NUCLEUS ProblemsDocument3 pagesNUCLEUS ProblemsKumkum KumbarahalliNo ratings yet