Professional Documents
Culture Documents
PRE FINAL Pathogenesis
PRE FINAL Pathogenesis
Uploaded by
pvsvr0 ratings0% found this document useful (0 votes)
29 views9 pagesMain stay of pathogesis of HCM is due to mutations in sarcomeric proteins causing changes in sarcomere structure and increased muscle cell size. These mutations lead to disarray of cardiomyocytes and fibrosis. Histopathological examination shows irregular arrangement of cardiomyocytes and accumulation of extracellular tissue. These changes cause obstruction during systole and increased ventricular volumes and wall thickness, leading to diastolic dysfunction, ischemia, and mitral regurgitation. Increased thickness also causes abnormalities in electrical conduction promoting atrial fibrillation.
Original Description:
Original Title
PRE FINAL pathogenesis
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentMain stay of pathogesis of HCM is due to mutations in sarcomeric proteins causing changes in sarcomere structure and increased muscle cell size. These mutations lead to disarray of cardiomyocytes and fibrosis. Histopathological examination shows irregular arrangement of cardiomyocytes and accumulation of extracellular tissue. These changes cause obstruction during systole and increased ventricular volumes and wall thickness, leading to diastolic dysfunction, ischemia, and mitral regurgitation. Increased thickness also causes abnormalities in electrical conduction promoting atrial fibrillation.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
29 views9 pagesPRE FINAL Pathogenesis
PRE FINAL Pathogenesis
Uploaded by
pvsvrMain stay of pathogesis of HCM is due to mutations in sarcomeric proteins causing changes in sarcomere structure and increased muscle cell size. These mutations lead to disarray of cardiomyocytes and fibrosis. Histopathological examination shows irregular arrangement of cardiomyocytes and accumulation of extracellular tissue. These changes cause obstruction during systole and increased ventricular volumes and wall thickness, leading to diastolic dysfunction, ischemia, and mitral regurgitation. Increased thickness also causes abnormalities in electrical conduction promoting atrial fibrillation.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 9
Main stay of pathogesis of HCM is due to its
mutation in the sarcomer protein structure causes
the change in structural protein sarcomere which
leads to increase in the muscle cell size [13].Due to
these mutations cadiac muscle muscles undergoes
very huge diarrangment of large muscle cells and
leading to cause fibrosis in structural components of
cardiac muscle cell causing left ventriculat
hypertrophy and increase in interventricular
septum thickness [14]. On histopathological
examination arrangement of cardiomyocytes are in
irregular arrangemt and there is accumulation of
extracellular connective tissue leading to fibrosis and
there nuclei is lost its normal structure [15].
These changes manifests obstruction to outflow
tract during systolic stage (contraction stage) of
cardiac cycle which increases the Left ventricular
end systolic volume [figure 1] ,increase in thickness
of the left ventricular wall progress to dysfunction
in diastole(relaxing stage),and increase in thickness
may also cause the increase in need of blood supply
if not accomapained may cause ischemia of the
tissue,and valves are moved anteriorly leading to
mitral regurgitation [16].
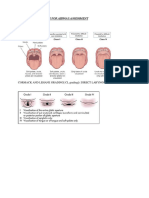
FIGURE 2
Increase in thickness of left ventricular wall and
interventricular septum causes the obstruction of
Aorta,causes increase in left ventricular diastolic
volume and pressure,because of movement of mitral
valve anteriorly cause the mitral regurgitation.
The incidence of atrail fibrillation is increases in
patients with HOCM ,the main reason is changes in
left ventricular thickness leading to increase in left
atrium promotes the abnormalities in
electricalcondiction in left atrial contraction [17].
MECHANISMS CASUING FOR ECTOPIC ATRAIL
FIBRILLATION
There are few machanims that leads to change in normal
automcity of cardiac muscle cell and causing Atrial
fibrillation.
Action potential of atrial muscle cells are maintained by
inward rectifier K+ current and pacemaker current
(If),through these channels atomicity is attained[18].
The resting action potential is stabilized due to high
flow of K+ ions through respective channel, although
there is high pacemaker current but inward rectifier
K+ current is much larger than this and maintaining
the stable automacity moreover unbalance in this
mainly cause of increased automcity by decrease in
K+ current and increase in pacemaker current [18].
Early afterdepolarizations is caused mainly by
prolongation of actinpotential duraration due to
allowing of L-type Ca2+ current (ICaL) to recover from
inactivation causing depolarizing inward movement
of Ca2+ ions.These changes that take place in atrila
muscle cell is main reason for the increase in
prevalence of atrail fibrillation in congenital long-QT
syndrome patients[19].
Delayed afterdepolarizations due to abnoarmal
release of calcium Ca2+ from sarcoplasmic reticulum
stores during diastole, Specialized sarcoplasmic
reticulum Ca2+ channels are called ryanodine
receptors [RyRs].This abnormal release is due to
response to Ca2+ in the transmembrane. RyRs are
closed during diastole but opened due to channel
abnormality or overlead of calcium in the
sarcoplasmic reticulum.so there is an Na+-
Ca2+ exchanger where one Ca2+ released is
exchanged with 3 extracellular Na+ leading to net
depolarizing inward positive-ion movement,These
changes leads to delayed afterdepolarizations[20].
13 Marian AJ, Braunwald E. Hypertrophic
Cardiomyopathy: Genetics, Pathogenesis, Clinical
Manifestations, Diagnosis, and Therapy. Circ Res.
2017 Sep 15;121(7):749-770. doi:
10.1161/CIRCRESAHA.117.311059. PMID:
28912181; PMCID: PMC5654557.
14 Brigden W. Hypertrophic cardiomyopathy. Br
Heart J. 1987 Oct;58(4):299-302. doi:
10.1136/hrt.58.4.299. PMID: 3314948; PMCID:
PMC1277258.
15 Varnava AM, Elliott PM, Mahon N, Davies MJ,
McKenna WJ. Relation between myocyte disarray
and outcome in hypertrophic cardiomyopathy. Am J
Cardiol. 2001 Aug 1;88(3):275-9. doi: 10.1016/s0002-
9149(01)01640-x. PMID: 11472707.
16 Medical Masterclass contributors, Firth J.
Cardiology: hypertrophic cardiomyopathy. Clin Med
(Lond). 2019 Jan;19(1):61-63. doi:
10.7861/clinmedicine.19-1-61. PMID: 30651247;
PMCID: PMC6399630.
17 Paur H, Wright PT, Sikkel MB, Tranter MH,
Mansfield C, O'gara P, Stuckey DJ, Nikolaev
VO, Diakonov I, Pannell L, Gong H. High levels
of circulating epinephrine trigger apical
cardiodepression in a β2-adrenergic
receptor/Gi–dependent manner: A new model
of Takotsubo cardiomyopathy. Circulation. 2012
Aug 7;126(6):697-706.
18 Stillitano F, Lonardo G, Zicha S, Varro A, Cerbai
E, Mugelli A, Nattel S. Molecular basis of funny
current (If) in normal and failing human heart.
Journal of molecular and cellular cardiology. 2008
Aug 1;45(2):289-99.
19 Johnson JN, Tester DJ, Perry J, Salisbury BA,
Reed CR, Ackerman MJ. Prevalence of early-onset
atrial fibrillation in congenital long QT syndrome.
Heart Rhythm. 2008 May 1;5(5):704-9.
20 Yeh YH, Wakili R, Qi XY, Chartier D, Boknik P,
Kääb S, Ravens U, Coutu P, Dobrev D, Nattel S.
Calcium-handling abnormalities underlying atrial
arrhythmogenesis and contractile dysfunction in
dogs with congestive heart failure. Circulation:
Arrhythmia and Electrophysiology. 2008 Jun
1;1(2):93-102.
You might also like
- Pathophysiology of Atrial FibrillationDocument12 pagesPathophysiology of Atrial FibrillationAndrew Surya Putra SccNo ratings yet
- Changes in Human Body Structure and Function Due To Prolonged ImmobilizationDocument29 pagesChanges in Human Body Structure and Function Due To Prolonged ImmobilizationReinita Arlin Pringgoredjo100% (2)
- D Received Patient Sitting On Bed With Bottle #Document17 pagesD Received Patient Sitting On Bed With Bottle #jaypee100% (2)
- Lauren Herr Nsg-432cc-Care-Plan-ExemplarDocument11 pagesLauren Herr Nsg-432cc-Care-Plan-Exemplarapi-520453750100% (1)
- DISCUDocument23 pagesDISCUpvsvrNo ratings yet
- Pathogenesis FinalDocument23 pagesPathogenesis FinalpvsvrNo ratings yet
- Kardiologi Klinik LV Remodelling SlideDocument19 pagesKardiologi Klinik LV Remodelling Slidebagus anantaNo ratings yet
- Strait 2012Document22 pagesStrait 2012abraham rumayaraNo ratings yet
- Cardiogenic Shock: Historical AspectsDocument24 pagesCardiogenic Shock: Historical AspectsnugessurNo ratings yet
- Bioquimica de La Isquemia y Reperfusion ArticuloDocument35 pagesBioquimica de La Isquemia y Reperfusion ArticulofelipeNo ratings yet
- Cardiogenic Shock - FINALDocument79 pagesCardiogenic Shock - FINALIgor StefanetNo ratings yet
- Project Injectable Hydrogels As Recent Advances in Treatment of Myocardial InfarctionDocument16 pagesProject Injectable Hydrogels As Recent Advances in Treatment of Myocardial Infarctionvarshankpathak2001No ratings yet
- Fisiopatologia Del Shock Cardiogenico Por Iam PDFDocument12 pagesFisiopatologia Del Shock Cardiogenico Por Iam PDFYESSICA CARENALGANo ratings yet
- Oxen Ham 2003Document9 pagesOxen Ham 2003abraham rumayaraNo ratings yet
- Cardiac Resynchronization Therapy (CRT) - Its History, Indication, Usefulness and ProblemsDocument6 pagesCardiac Resynchronization Therapy (CRT) - Its History, Indication, Usefulness and Problemsvikram.vNo ratings yet
- Myocardial Infarction: Practice Essentials, Background, Definitions PDFDocument12 pagesMyocardial Infarction: Practice Essentials, Background, Definitions PDFMukhtar UllahNo ratings yet
- Anderson 2019Document13 pagesAnderson 2019Cut Keumala PutriNo ratings yet
- Cardiogenic Shock Pathogenesis, Classification, and ManagementDocument20 pagesCardiogenic Shock Pathogenesis, Classification, and Managementjosh FaducNo ratings yet
- Korean J Hepatol 16-4-347 352Document6 pagesKorean J Hepatol 16-4-347 352Jorge DiazNo ratings yet
- Blood Flow ECMDocument31 pagesBlood Flow ECMirene :]No ratings yet
- Ischaemic Cardiogenic ShockDocument4 pagesIschaemic Cardiogenic ShocksuckeydluffyNo ratings yet
- Abstract and History: Table 1. Summary of Haemodynamic Effects of IABP TherapyDocument8 pagesAbstract and History: Table 1. Summary of Haemodynamic Effects of IABP TherapymishelNo ratings yet
- Myocardial InfarctionDocument45 pagesMyocardial InfarctionGopal SinghNo ratings yet
- Wiki Ventricular RemodelingDocument3 pagesWiki Ventricular Remodelingdgina8800No ratings yet
- Makki Et Al 2013 Acute Coronary SyndromeDocument15 pagesMakki Et Al 2013 Acute Coronary SyndromeV ANo ratings yet
- DR Dhiman BanikCariogenic Shock Final 2022 DDDocument59 pagesDR Dhiman BanikCariogenic Shock Final 2022 DDCloudySkyNo ratings yet
- Physiological Changes After Brain Stem DeathDocument5 pagesPhysiological Changes After Brain Stem DeathFraggle100% (1)
- Pi Is 0002934312004901Document2 pagesPi Is 0002934312004901MuliaDharmaSaputraHutapeaNo ratings yet
- Cardio Diabetes 8Document17 pagesCardio Diabetes 8miguelcontreraszambranoNo ratings yet
- Patten 2000Document6 pagesPatten 2000diqa aridaniNo ratings yet
- Advances in Understanding Ischemic Acute Kidney Injury: Minireview Open AccessDocument6 pagesAdvances in Understanding Ischemic Acute Kidney Injury: Minireview Open AccessJack AukiónNo ratings yet
- Nutritional Treatments For Acute Myocardial Infarction: Alan R. Gaby, MDDocument11 pagesNutritional Treatments For Acute Myocardial Infarction: Alan R. Gaby, MDAlmaNo ratings yet
- 1 s2.0 S2667100X22001219 Main PDFDocument10 pages1 s2.0 S2667100X22001219 Main PDFjesusNo ratings yet
- Intra Aortic Balloon PumpDocument5 pagesIntra Aortic Balloon PumpZainal 'babeh' Arifin100% (1)
- Cardiac Torsion and Electromagnetic Fields - The Cardiac Bioinformation HypothesisDocument8 pagesCardiac Torsion and Electromagnetic Fields - The Cardiac Bioinformation HypothesisoscarnineNo ratings yet
- Optimalmedicaltherapyfor Stableischemicheart Disease:: Focus On Anti-Anginal TherapyDocument14 pagesOptimalmedicaltherapyfor Stableischemicheart Disease:: Focus On Anti-Anginal TherapyCarlos Hernan Castañeda RuizNo ratings yet
- Atrial Fibrillation and Heart Failure - Cause or Effect?Document8 pagesAtrial Fibrillation and Heart Failure - Cause or Effect?Cosmin GabrielNo ratings yet
- Cardiogenic ShockDocument20 pagesCardiogenic ShockVasilis VazgiourakisNo ratings yet
- Contemporary Reviews in Cardiovascular Medicine: Cardiogenic ShockDocument32 pagesContemporary Reviews in Cardiovascular Medicine: Cardiogenic ShockSindhuNugrohoMuktiNo ratings yet
- Kharel - Implications of Localized ST Depression in A Vascular Territory and Altered Precordial T-Wave Balance in Ischemic Heart DiseaseDocument6 pagesKharel - Implications of Localized ST Depression in A Vascular Territory and Altered Precordial T-Wave Balance in Ischemic Heart DiseaseM. PurnomoNo ratings yet
- Cardiogenic ShockDocument26 pagesCardiogenic ShockAnggun V. M SitumorangNo ratings yet
- European Journal of Pharmacology: Jenna L. Ross, Susan E. HowlettDocument9 pagesEuropean Journal of Pharmacology: Jenna L. Ross, Susan E. HowlettMarcelo Pires de OliveiraNo ratings yet
- Myocardial Infarction: Khaled Dajani, MD, MBA Division of CardiologyDocument45 pagesMyocardial Infarction: Khaled Dajani, MD, MBA Division of CardiologyNaveen Kumar SharmaNo ratings yet
- Bidirectional VTDocument4 pagesBidirectional VTbgusjklNo ratings yet
- Shock Cardiogenico - IamDocument19 pagesShock Cardiogenico - IamYESSICA CARENALGANo ratings yet
- Ahead of Print Issue-Article 201408-19 PDFDocument26 pagesAhead of Print Issue-Article 201408-19 PDFSyilla PriscilliaNo ratings yet
- Peripheral Edema: ReviewDocument7 pagesPeripheral Edema: ReviewVmsdNo ratings yet
- Japplphysiol 00035 2004Document10 pagesJapplphysiol 00035 2004Kritik KumarNo ratings yet
- Cardiac Arrhythmia PrintDocument7 pagesCardiac Arrhythmia PrintJim clarence palmaNo ratings yet
- How To Diagnose STEMI?Document6 pagesHow To Diagnose STEMI?Adhie BadriNo ratings yet
- A Standardized and Comprehensive Approach To The Management of Cardiogenic Shock 2020Document13 pagesA Standardized and Comprehensive Approach To The Management of Cardiogenic Shock 2020gloriaNo ratings yet
- 3-Classification of Noreflow PhenomenonDocument4 pages3-Classification of Noreflow PhenomenonMohamed KhattabNo ratings yet
- Jama 2021 326 1840Document11 pagesJama 2021 326 1840silviaNo ratings yet
- Cardiogenic Shock and Acute Kidney InjuryDocument10 pagesCardiogenic Shock and Acute Kidney InjuryMoises Agustin Preciado AlvarezNo ratings yet
- Chira Karn Jana Korn 2017Document55 pagesChira Karn Jana Korn 2017JoecoNo ratings yet
- Cardiogenic Shock in Children: March 2012Document9 pagesCardiogenic Shock in Children: March 2012Quang TinNo ratings yet
- Aging and Arterial StiffnessDocument6 pagesAging and Arterial StiffnessRiaz AkhtarNo ratings yet
- 2021 Article 1975Document6 pages2021 Article 1975melon segerNo ratings yet
- Vasoconstriction in Male Cardiac Allografts Conjugated Estrogens Acutely Abolish Abnormal Cold-Induced CoronaryDocument4 pagesVasoconstriction in Male Cardiac Allografts Conjugated Estrogens Acutely Abolish Abnormal Cold-Induced Coronarykuttlu pandaNo ratings yet
- Cardiac Resynchronisation Therapy For Patients With Heart FailureDocument5 pagesCardiac Resynchronisation Therapy For Patients With Heart FailureIpar DayNo ratings yet
- ACN Unit3Document35 pagesACN Unit3Kamran AltafNo ratings yet
- Microcirculation: From Bench to BedsideFrom EverandMicrocirculation: From Bench to BedsideMaria DorobantuNo ratings yet
- Pathogenesis FinalDocument23 pagesPathogenesis FinalpvsvrNo ratings yet
- AnatolJCardiol - HCM Embolic Risk and PrognoDocument4 pagesAnatolJCardiol - HCM Embolic Risk and PrognopvsvrNo ratings yet
- New Microsoft Office Word DocumentDocument6 pagesNew Microsoft Office Word DocumentpvsvrNo ratings yet
- Medical Progress: T M H CDocument11 pagesMedical Progress: T M H CpvsvrNo ratings yet
- Pre FinalDocument24 pagesPre FinalpvsvrNo ratings yet
- DISCUDocument23 pagesDISCUpvsvrNo ratings yet
- FINAL DRAFT MistakesDocument28 pagesFINAL DRAFT MistakespvsvrNo ratings yet
- Scifi Atrial Fibrillation in HCM Prevalence Clinical Impact and ManagDocument9 pagesScifi Atrial Fibrillation in HCM Prevalence Clinical Impact and ManagpvsvrNo ratings yet
- Clinical Impact StructuredDocument3 pagesClinical Impact StructuredpvsvrNo ratings yet
- Anti ArrthymicDocument7 pagesAnti ArrthymicpvsvrNo ratings yet
- Risk Factors For The Development of AF and Detection in HCM: Surface Area)Document4 pagesRisk Factors For The Development of AF and Detection in HCM: Surface Area)pvsvrNo ratings yet
- 27 Patients With AF Compared With A Control Group Consisted of 53 Patients Without Any Documented AF Attacks andDocument3 pages27 Patients With AF Compared With A Control Group Consisted of 53 Patients Without Any Documented AF Attacks andpvsvrNo ratings yet
- HCM and Af Final DraftDocument30 pagesHCM and Af Final DraftpvsvrNo ratings yet
- Step 2 Clinical Knowledge: Content Description and General InformationDocument13 pagesStep 2 Clinical Knowledge: Content Description and General InformationpvsvrNo ratings yet
- Obstetrics - OBrationsDocument3 pagesObstetrics - OBrationspvsvrNo ratings yet
- Afib in Hcom Article in PressDocument8 pagesAfib in Hcom Article in PresspvsvrNo ratings yet
- Neurosurgery - Medicine Simplified (DBMCI)Document5 pagesNeurosurgery - Medicine Simplified (DBMCI)pvsvrNo ratings yet
- Anesthesia Images (All in OnDocument100 pagesAnesthesia Images (All in OnpvsvrNo ratings yet
- ENT Toppers Scribble NotesDocument5 pagesENT Toppers Scribble NotespvsvrNo ratings yet
- UHS 3rd Professional MBBS SyllabusDocument39 pagesUHS 3rd Professional MBBS SyllabusRafey HashmiNo ratings yet
- Degenerative Diseases of The Brain 2Document66 pagesDegenerative Diseases of The Brain 2Janie-Vi Villamor Ismael-GorospeNo ratings yet
- Extrapyramidal System and CerebellumDocument52 pagesExtrapyramidal System and CerebellumVictoriiaNo ratings yet
- Prometric Mcqs Plus CaculationsDocument147 pagesPrometric Mcqs Plus CaculationsShanaz KhanNo ratings yet
- Sas 17Document3 pagesSas 17Sistine Rose LabajoNo ratings yet
- Altered Genitourinary/Renal FunctionDocument14 pagesAltered Genitourinary/Renal FunctionAshleyNo ratings yet
- Neuropatologia AlzheimerDocument27 pagesNeuropatologia AlzheimerSarah JatobáNo ratings yet
- Summary of DrugsDocument19 pagesSummary of DrugsAthena BendoNo ratings yet
- NeuropsychiatryDocument5 pagesNeuropsychiatryNicole TangcoNo ratings yet
- IM RheumatologyDocument56 pagesIM RheumatologySujoud AbuserdanehNo ratings yet
- 1.epithelium, Connective Tissues, Cell JunctionsDocument21 pages1.epithelium, Connective Tissues, Cell JunctionsbrihaspathiacademyNo ratings yet
- Parkinsons Disease Frequently Asked QuestionsDocument52 pagesParkinsons Disease Frequently Asked QuestionsWilliam HuertasNo ratings yet
- HypersensitivityDocument12 pagesHypersensitivityDwi TyoNo ratings yet
- Diabetes Homeopathic RemediesDocument12 pagesDiabetes Homeopathic Remediessksmilyin100% (2)
- Chapter 3 Subcellular Fractionation and Isolation of OrganellesDocument8 pagesChapter 3 Subcellular Fractionation and Isolation of OrganellesrajaNo ratings yet
- Neonatal HypotoniaDocument7 pagesNeonatal HypotoniaShirley KoehNo ratings yet
- CNS Related DiseasesDocument4 pagesCNS Related DiseasesYzabelle EncoyNo ratings yet
- Practice Guidelines For Acute Care Nurse Practitioners E Book 2Nd Edition PDF Full Chapter PDFDocument53 pagesPractice Guidelines For Acute Care Nurse Practitioners E Book 2Nd Edition PDF Full Chapter PDFpitsonmoedi100% (5)
- Nbme 4 Answer CKDocument4 pagesNbme 4 Answer CKNajia Choudhury86% (7)
- Primary Review: Psoas Abscess: Case of The LiteratureDocument3 pagesPrimary Review: Psoas Abscess: Case of The LiteratureDung Tran HoangNo ratings yet
- Physical Examination Techniques and ManueversDocument160 pagesPhysical Examination Techniques and ManueversMa-anJaneDiamosNo ratings yet
- TerminDocument5 pagesTerminAnonymous DA8iQzNo ratings yet
- Syncope: Global Cerebral Blood FlowDocument7 pagesSyncope: Global Cerebral Blood FlowChananNo ratings yet
- DR Rajiv Dhawan 2020 T&D ImagesDocument30 pagesDR Rajiv Dhawan 2020 T&D Imagespalohe1277No ratings yet
- The Child With Respiratory DysfunctionDocument91 pagesThe Child With Respiratory DysfunctionSerbanNo ratings yet
- A Review of Herbal Therapy in Multiple SclerosisDocument16 pagesA Review of Herbal Therapy in Multiple Sclerosissorin.morosan64No ratings yet
- Joy N. Saquibal, M.DDocument78 pagesJoy N. Saquibal, M.DJoy AlmondsNo ratings yet
- Fundamentals in NursingDocument15 pagesFundamentals in NursingAngie Bucio MiraNo ratings yet