Professional Documents
Culture Documents
3-Electrochemical Energy
3-Electrochemical Energy
Uploaded by
Jennifer Tamayo Guzman - EmperadorOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
3-Electrochemical Energy
3-Electrochemical Energy
Uploaded by
Jennifer Tamayo Guzman - EmperadorCopyright:
Available Formats
CHEMISTRY FOR ENGINEERS
MODULE 3
ELECTROCHEMICAL ENERGY
WHAT IS ELECTROCHEMICAL ENERGY?
The Electrochemical energy is defined as “the energy which converts electrical energy to chemical energy
and vice versa.” The electrochemical energy is related to fuel cells, photo electrochemical, and energy storage
systems such as batteries, super capacitors or ultra-capacitors.
RECALL:
REDOX (REDUCTION-OXIDATION) REACTION
The reaction that happens during the production of electrochemical energy is called REDOX (REDUCTION
OXIDATION) reactions which includes the transfer of electrons.
EXAMPLE:
CHEMICAL EQUATION: 𝐹𝑒(𝑠) + 𝐶𝑢𝑆𝑂4(𝑎𝑞) → 𝐹𝑒𝑆𝑂4(𝑎𝑞) + 𝐶𝑢(𝑠)
IONIC CHEMICAL EQUATION: 𝐹𝑒(𝑠) + 𝐶𝑢(𝑎𝑞)
2+ 2−
+ 𝑆𝑂4(𝑎𝑞) 2+
→ 𝐹𝑒(𝑎𝑞) 2−
+ 𝑆𝑂4(𝑎𝑞) + 𝐶𝑢(𝑠)
2+ 2+
𝐹𝑒(𝑠) + 𝐶𝑢(𝑎𝑞) → 𝐹𝑒(𝑎𝑞) + 𝐶𝑢(𝑠)
THE TWO HALF REACTIONS:
2+
𝐹𝑒(𝑠) → 𝐹𝑒(𝑎𝑞) + 2𝑒 −(OXIDATON/RA)
2+
𝐶𝑢(𝑎𝑞) + 2𝑒 − → 𝐶𝑢(𝑠) (REDUCTION/OA)
REDUCTION – occurs when a specie gains electrons and decreases its oxidation number.
OXIDATION – occurs when a specie loses electrons and increases its oxidation number.
REDUCING AGENT (RA) – ion/molecule that donates electrons
OXIDIZING AGENT (OA) – ion/molecule that accepts electrons
REMEMBER!!!
GEROA- Gain of Electrons Reduction Oxidizing Agent
LEORA- Loss of Electrons Oxidation Reducing Agent
ENGR. ARVEY DWAYNE P. BUEN Page 1
CHEMISTRY FOR ENGINEERS
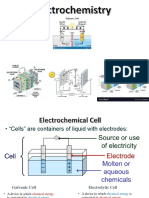
TYPES OF CELLS
1. VOLTAIC/GALVANIC CELL – the reaction generates current
Anode reaction is OXIDATION Cathode reaction is REDUCTION
Zn atoms are oxidized to Zn2+ Cu2+ ions are Reduced to Cu atoms
𝒁𝒏(𝒔) → 𝒁𝒏𝟐+ (𝒂𝒒) + 𝟐𝒆
−
𝑪𝒖𝟐+ −
(𝒂𝒒) + 𝟐𝒆 → 𝑪𝒖(𝒔)
Balancing HALF CELL REACTION:
Cathode: 𝒁𝒏(𝒔) → 𝒁𝒏𝟐+ (𝒂𝒒) + 𝟐𝒆
−
Anode: 𝑪𝒖𝟐+ −
(𝒂𝒒) + 𝟐𝒆 → 𝑪𝒖(𝒔)
REACTION: 𝒁𝒏(𝒔) + 𝑪𝒖𝟐+ 𝟐+
(𝒂𝒒) → 𝒁𝒏(𝒂𝒒) + 𝑪𝒖(𝒔)
Simplifying it into this notation: |- Phase Boundary
Zn|Zn 2+
|| Cu |Cu
2+
||- Salt Bridge
HOW IT WORKS?
1. At the Zinc anode (- pole), electrons are produced by the oxidation half reaction:
2+
𝑍𝑛(𝑠) → 𝑍𝑛(𝑎𝑞) + 2𝑒 −
2. Electrons generated at the anode move to the Copper cathode (+ pole) thru a wire to voltmeter giving a
reduction reaction:
2+
𝐶𝑢(𝑎𝑞) + 2𝑒 − → 𝐶𝑢(𝑠)
3. To maintain balance or electrical neutrality, Cations (+charged ions) move toward move to Copper Cathode.
Anions (- charged ions) move toward Zinc Anode.
2. ELECTROLYTIC CELL – current drives the reaction
ENGR. ARVEY DWAYNE P. BUEN Page 2
CHEMISTRY FOR ENGINEERS
CELL POTENTIAL, Ecell
Electrons will always flow from a high electric potential to a low electric potential and the difference
between the two is called POTENTIAL DIFFERENCE.
𝑬𝒄𝒆𝒍𝒍 = 𝑬𝒐𝒙 + 𝑬𝒓𝒆𝒅
Where the Ered may be obtained from the of STANDARD REDUCTION POTENTIALS TABLE.
The larger the Ecell value, the greater the current it can generate.
GIBB’S FREE ENERGY, ΔG°
The change in Gibb’s Free Energy pertains to the maximum work a voltaic cell is capable of.
∆𝑮° = −𝒏𝑭𝑬°
Where:
n – moles of electrons exchanged in the reaction
𝐽
F – Faraday’s constant, 96,485 𝑉.𝑚𝑜𝑙 𝑒 −
E° – standard cell potential
NERST EQUATION:
The relationship between the cell potential and the standard cell potential
𝑹𝑻 𝐥𝐧(𝑸)
𝑬 = 𝑬° −
𝒏𝑭
Where:
E – Cell Potential
E° - Standard Cell Potential (at standard atmospheric pressure and temperature)
𝐽
R – Universal Gas Constant (8.314 𝑚𝑜𝑙.𝐾)
T – Absolute Temperature (Kelvin)
Q – Reaction Quotient
n – moles of electrons exchanged in the reaction
𝐽
F – Faraday’s constant, 96,485
𝑉.𝑚𝑜𝑙 𝑒 −
PROBLEMS:
1. Consider the following voltaic cell:
3+ 2+
𝐴𝑙(𝑠) | 𝐴𝑙(𝑎𝑞) || 𝐹𝑒(𝑎𝑞) | 𝐹𝑒(𝑠)
a. Which electrode is the anode and which is the cathode?
b. Write the overall reaction.
c. Using the STANDARD REDUCTION POTENTIAL TABLE, calculate the cell potential.
ENGR. ARVEY DWAYNE P. BUEN Page 3
CHEMISTRY FOR ENGINEERS
d. Calculate the Gibb’s Free Energy and the Reaction Quotient (Q).
2. Consider the voltaic cell at 25°C and 1 atm:
+ −
𝑂2(𝑔,0.98 𝑎𝑡𝑚) + 4𝐻(𝑎𝑞,𝑝𝐻=1.24) + 4𝐵𝑟(𝑎𝑞,0.15 𝑀) → 2𝐻2 𝑂 + 2𝐵𝑟2(𝑙)
Solve:
a. Ecell
a. ΔGo
b. Q (search the internet how to calculate for the Reaction Quotient)
REFERENCES:
https://energyfaculty.com/electrochemical-energy/
https://www.youtube.com/watch?v=2VT5rl8P84M
ENGR. ARVEY DWAYNE P. BUEN Page 4
You might also like
- Electrochemistry RevisedDocument79 pagesElectrochemistry RevisedTuna ShinNo ratings yet
- 3 ElectrochemDocument4 pages3 ElectrochemFelven Leo AbayaNo ratings yet
- Electrochemistry 153strippedDocument65 pagesElectrochemistry 153strippedJasonTenebrosoNo ratings yet
- SK0014 - Lecture 8 - ElectrochemDocument63 pagesSK0014 - Lecture 8 - ElectrochemEvaNo ratings yet
- 2 Electrochemistry (Voltaic Cells)Document46 pages2 Electrochemistry (Voltaic Cells)Gerald Paul SumagpaoNo ratings yet
- ELECTROCHEMISTRY Module 2 Lecture NoteDocument7 pagesELECTROCHEMISTRY Module 2 Lecture Notex seyiNo ratings yet
- Electrolysis in Aqueous SolutionDocument15 pagesElectrolysis in Aqueous SolutionEdon BediNo ratings yet
- CHAPTER 2 2023 ElectrochemistryDocument46 pagesCHAPTER 2 2023 Electrochemistrym.yassinmansor19No ratings yet
- Electrochemistry: Applications of RedoxDocument40 pagesElectrochemistry: Applications of Redoxcatsathish1No ratings yet
- Chapter6-Electrochemistry (Part 3)Document33 pagesChapter6-Electrochemistry (Part 3)BagusprPrasetyoNo ratings yet
- Chapter7 Electrochemistry (Part3)Document13 pagesChapter7 Electrochemistry (Part3)Christoval PandilalaNo ratings yet
- Chapter6-Electrochemistry (Part 2)Document27 pagesChapter6-Electrochemistry (Part 2)BagusprPrasetyoNo ratings yet
- Lecture 09. 28122020 E&MEDocument41 pagesLecture 09. 28122020 E&MEMuhammad TalhaNo ratings yet
- Electrochemistry 2Document42 pagesElectrochemistry 2Sara FatimaNo ratings yet
- CHM 101 ELECTROCHEMISTRY Lecture 2Document18 pagesCHM 101 ELECTROCHEMISTRY Lecture 2Kehinde AladetoyinboNo ratings yet
- Electrochemical MethodsDocument70 pagesElectrochemical MethodsBüşra DOĞRUNo ratings yet
- Cell PotentialDocument39 pagesCell Potentialellamae sanbuenaventuraNo ratings yet
- Chemistry Long Quiz 5Document11 pagesChemistry Long Quiz 5Ervin MogarNo ratings yet
- Electrochemistry StudentDocument88 pagesElectrochemistry StudentCtNabihahAmilaMarminNo ratings yet
- CH 11Document12 pagesCH 11MrsriyansyahNo ratings yet
- Electrochemistry: Syllabus Theme 5Document63 pagesElectrochemistry: Syllabus Theme 5Jack WilliamsNo ratings yet
- Electrochemistry, PPT 3Document33 pagesElectrochemistry, PPT 3Ernest Nana Yaw AggreyNo ratings yet
- Physical Chemistry Chapter 4 - ElectrochemistryDocument36 pagesPhysical Chemistry Chapter 4 - Electrochemistryjatropos6810No ratings yet
- Chem 114 Electrochemical Energy 2Document32 pagesChem 114 Electrochemical Energy 2KaizNo ratings yet
- ElectroChemistry Slides PDFDocument44 pagesElectroChemistry Slides PDFHenry OkoyeNo ratings yet
- Electrochemistry and NanomaterialsDocument76 pagesElectrochemistry and NanomaterialsCharles Arthel ReyNo ratings yet
- Chapter6-Electrochemistry (Part 2)Document27 pagesChapter6-Electrochemistry (Part 2)Uswatun KhasanahNo ratings yet
- ElectrochemistryDocument39 pagesElectrochemistryHaider AliNo ratings yet
- The Electrochemical Cell Module 2 AralinksDocument38 pagesThe Electrochemical Cell Module 2 AralinksJohn OliquianoNo ratings yet
- Chapter 08 EditedDocument39 pagesChapter 08 EditedqamaralmahseriNo ratings yet
- Redox Reaction and Electrochemistry 2018Document66 pagesRedox Reaction and Electrochemistry 2018Jonathan AndikaNo ratings yet
- 3.A. Electrochemistry: Topic 3: EnergyDocument67 pages3.A. Electrochemistry: Topic 3: EnergyJeam Russell AlfaroNo ratings yet
- Electrochemistry and Redox Titration: Dr. Cherrie Pascual Institute of Chemistry UP DilimanDocument57 pagesElectrochemistry and Redox Titration: Dr. Cherrie Pascual Institute of Chemistry UP DilimanAcadGucciManeNo ratings yet
- Ion Mobility: Ue S Zee F Rs FDocument5 pagesIon Mobility: Ue S Zee F Rs FJohnS.GallianoNo ratings yet
- ElectrochemistryDocument53 pagesElectrochemistryRosemarie DalupangNo ratings yet
- Module 1 Electrochemistry PPT Slides Part 2V2Document36 pagesModule 1 Electrochemistry PPT Slides Part 2V2May TampusNo ratings yet
- UntitledDocument90 pagesUntitledS KamalNo ratings yet
- CH 20 Electrochemistry:: Review Redox ReactionsDocument27 pagesCH 20 Electrochemistry:: Review Redox ReactionsAtlas shabuNo ratings yet
- Electrochemistry LECTURE NOTESDocument45 pagesElectrochemistry LECTURE NOTESDalitso SimonNo ratings yet
- Chapter 18: Electrochemistry: Key TopicsDocument25 pagesChapter 18: Electrochemistry: Key TopicsMoises Tapia BaezNo ratings yet
- Electrochemical Cells: Dr. B. FarrellDocument0 pagesElectrochemical Cells: Dr. B. FarrellborgiamatriceNo ratings yet
- KimiaDocument46 pagesKimiaErvina RetnaningtyasNo ratings yet
- Test - ElectrochemistryDocument3 pagesTest - ElectrochemistryAdrian AlvaNo ratings yet
- Chapter 18 ElectrochemistryDocument49 pagesChapter 18 ElectrochemistryDwivelia AftikaNo ratings yet
- Chapter 2electrochemical Cells EditedDocument50 pagesChapter 2electrochemical Cells EditedAdugnaw BiksNo ratings yet
- Electrochemistry 12Document19 pagesElectrochemistry 12Manas ChhabraNo ratings yet
- Electrochemistry: Compiled By: E.S. EspirituDocument123 pagesElectrochemistry: Compiled By: E.S. EspirituCyrus Vizon100% (1)
- MM 222 - Lecture 3Document11 pagesMM 222 - Lecture 312 34No ratings yet
- Chapter 17 ElectrochemistryDocument68 pagesChapter 17 ElectrochemistryBeastUnleashed28No ratings yet
- LU 5.1 ElectrochemistryDocument32 pagesLU 5.1 ElectrochemistryNurAkila Mohd YasirNo ratings yet
- Chapter 18 ElectrochemistryDocument53 pagesChapter 18 ElectrochemistryKatarina WuriyaniNo ratings yet
- Electrochemical CellDocument30 pagesElectrochemical CellSubhu100% (1)
- Chapter 3 Electrochemistry (Notes)Document14 pagesChapter 3 Electrochemistry (Notes)DivyanshuNo ratings yet
- (2091) Lecture Notes Electrochemistry E.pdf - TMPDocument43 pages(2091) Lecture Notes Electrochemistry E.pdf - TMPRamJiPandeyNo ratings yet
- ElectrochemistryDocument49 pagesElectrochemistrySafril JuntakNo ratings yet
- ElectrochemistryDocument9 pagesElectrochemistryavrildominique.abcejo.engNo ratings yet
- NOTES ElectrochemistryDocument30 pagesNOTES ElectrochemistryAlexander LeeNo ratings yet
- Electromotiveforcefinal 200727 135128Document15 pagesElectromotiveforcefinal 200727 135128Mohit AgarwalNo ratings yet
- Practice Makes Perfect in Chemistry: Oxidation-ReductionFrom EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionRating: 5 out of 5 stars5/5 (1)
- Practice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersFrom EverandPractice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersNo ratings yet
- PolyLED PresentationDocument26 pagesPolyLED PresentationDilip MathuriaNo ratings yet
- Electrochemistry E WADocument92 pagesElectrochemistry E WAlol7755330No ratings yet
- Redox Reaction - Chemical Reactions in Which Both Oxidation and Reduction Occur SimultaneouslyDocument17 pagesRedox Reaction - Chemical Reactions in Which Both Oxidation and Reduction Occur SimultaneouslyJoanne SiaNo ratings yet
- Basic of Corrosion 021211Document18 pagesBasic of Corrosion 021211Sunil DeshpandeNo ratings yet
- Questions & ans-WPS OfficeDocument8 pagesQuestions & ans-WPS OfficeATUKUNDA GIFTNo ratings yet
- Class 11 Chapter Atomic Structure Chemistry Notes 2 1Document35 pagesClass 11 Chapter Atomic Structure Chemistry Notes 2 1Muhammad AbdullahNo ratings yet
- Automatic Emergency LightDocument47 pagesAutomatic Emergency LightSandeep Yadav100% (1)
- Nptel WeldingDocument233 pagesNptel WeldingThasarathan Ravichandran100% (2)
- SOP For RD TankDocument2 pagesSOP For RD TankMogan RajNo ratings yet
- 1 s2.0 S2212982023000884 MainDocument17 pages1 s2.0 S2212982023000884 MainDaniel MontalvoNo ratings yet
- Magnetron TechDocument10 pagesMagnetron TechramjoceNo ratings yet
- Lithium IonDocument19 pagesLithium IonVenkata sathwik100% (2)
- Chem ProjectDocument15 pagesChem ProjectAPARNA GANGWARNo ratings yet
- Glossary PpiDocument24 pagesGlossary PpiJose BustosNo ratings yet
- The Cold Fusion ReactorDocument12 pagesThe Cold Fusion ReactorShahid Z Ansari100% (1)
- Fundamental of Metal Corrosion, Corrosion Theory & Corrosion TypesDocument3 pagesFundamental of Metal Corrosion, Corrosion Theory & Corrosion Typessyahril siddiq arelNo ratings yet
- BITSAT Mock Test: This Section Is Taken From The BookDocument30 pagesBITSAT Mock Test: This Section Is Taken From The BookNekhill KumarNo ratings yet
- Hot vs. Cold Cathode Technology OverviewDocument16 pagesHot vs. Cold Cathode Technology OverviewdavidNo ratings yet
- US2574551 (Electronic Tachometer)Document7 pagesUS2574551 (Electronic Tachometer)Aaron Choco De GuzmanNo ratings yet
- Electrohydrodynamics: Home Search Collections Journals About Contact Us My IopscienceDocument25 pagesElectrohydrodynamics: Home Search Collections Journals About Contact Us My IopscienceANSHUL BANSALNo ratings yet
- Batteries PresentationDocument14 pagesBatteries PresentationMukhtiar HussainNo ratings yet
- Methane Steam ReformingDocument118 pagesMethane Steam Reformingrezaroohollahi100% (2)
- A Comparative Study Between The Efficiency of NaturalDocument20 pagesA Comparative Study Between The Efficiency of Natural11-CG10 Elamparo, Jochelle G.No ratings yet
- Electrolysis of Aqeous SolutionDocument6 pagesElectrolysis of Aqeous Solutionanon_383714618No ratings yet
- How CPRS WorksDocument19 pagesHow CPRS WorksAnonymous i3xDz4TNo ratings yet
- Teaching ProjectDocument10 pagesTeaching ProjectLoraine TangalinNo ratings yet
- Chapter 4 KN Sharma Atomic Structure (ClassicalDocument18 pagesChapter 4 KN Sharma Atomic Structure (Classicalajay mauryaNo ratings yet
- p2 Essay Chap 01 To 09Document23 pagesp2 Essay Chap 01 To 09Ina AmalinaNo ratings yet
- November 2005 QP - Paper 2 CIE Physics IGCSEDocument16 pagesNovember 2005 QP - Paper 2 CIE Physics IGCSEύπατίαNo ratings yet
- CHM409 2020Document2 pagesCHM409 2020Ali AllamNo ratings yet