Professional Documents
Culture Documents
26 Apr 2019 Ccid 198081 Delayed Hypersensitivity Reaction To Hyaluronic Acid Dermal
26 Apr 2019 Ccid 198081 Delayed Hypersensitivity Reaction To Hyaluronic Acid Dermal
Uploaded by
dominiqueOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
26 Apr 2019 Ccid 198081 Delayed Hypersensitivity Reaction To Hyaluronic Acid Dermal
26 Apr 2019 Ccid 198081 Delayed Hypersensitivity Reaction To Hyaluronic Acid Dermal
Uploaded by
dominiqueCopyright:
Available Formats
Clinical, Cosmetic and Investigational Dermatology Dovepress
open access to scientific and medical research
Open Access Full Text Article
ORIGINAL RESEARCH
Delayed hypersensitivity reaction to hyaluronic
Clinical, Cosmetic and Investigational Dermatology downloaded from https://www.dovepress.com/ by 92.186.191.200 on 19-Jul-2021
acid dermal filler following influenza-like illness
This article was published in the following Dove Press journal:
Clinical, Cosmetic and Investigational Dermatology
Mohammed G Turkmani 1 Background: Delayed reactions after facial hyaluronic acid injection are relatively rare
Koenraad De Boulle 2 complications. Their cause may be infectious or immune-mediated in origin, and their
Wolfgang G Philipp- outbreak can be triggered, for example, by an influenza-like illness.
Dormston 3 Objective: To describe potential adverse event of influenza like illness following dermal
1
filler injection.
Derma Clinic, Riyadh, Saudi Arabia;
2
Aalst Dermatology Clinic, Aalst,
Methods: We report fourteen unusual cases of delayed hypersensitivity reaction to several
brands of hyaluronic acid dermal filler following influenza like illness.
For personal use only.
Belgium; 3Cologne Dermatology,
Cologne, Germany Results: Increasing evidence implicates influenza infection in the pathogenesis of late onset
filler reaction.
Conclusion: Although there is a low risk of late onset adverse reaction with hyaluronic acid
fillers, injecting physicians must be aware of the possible filler reaction following the
influenza infection.
Keywords: fillers, hyaluronic acid, reaction, hypersensitivity, complications, influenza,
infection
Introduction
Fillers have become an important treatment for patients who seek noninvasive
rejuvenation. The American Society of Plastic Surgeons (ASPS) reported that
approximately 2.7 million dermal filler procedures were performed by plastic
surgeons and other board-certified physicians in 2017. Moreover, the
American Society for Dermatologic Surgery ASDS reported that dermatologic
surgeons performed 1.64 million dermal filler injections in the same year
(over 10% more than 2016 and doubled since 2012).1,2 As the field of soft
tissue augmentation has become increasingly popular, reports of adverse
events have also risen.
Post filler injection complications vary and can be categorized based on their
timing in relation to the filler injection as early events (occurring up to several
days post treatment) or delayed events (occurring weeks to years post
treatment).3
Delayed hypersensitivity reactions are characterized by induration, erythema,
and edema and are mediated by T lymphocytes rather than antibodies.4 They
typically occur 48–72 hrs after injection but may be seen as late as several weeks
Correspondence: Mohammed post injection and can persist for months.5
G Turkmani
Derma Clinic, P.O Box 20632, Riyadh Herein we describe fourteen cases of delayed hypersensitivity reaction to
11465, Saudi Arabia hyaluronic acid (HA) dermal filler following influenza-like illness from two der-
Tel +96 650 416 1171
Email mgtderma@yahoo.com matology centers in Saudi Arabia and Belgium.
submit your manuscript | www.dovepress.com Clinical, Cosmetic and Investigational Dermatology 2019:12 277–283 277
DovePress © 2019 Turkmani et al. This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/
terms.php and incorporate the Creative Commons Attribution – Non Commercial (unported, v3.0) License (http://creativecommons.org/licenses/by-nc/3.0/). By accessing
http://doi.org/10.2147/CCID.S198081
the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed.
For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms (https://www.dovepress.com/terms.php).
Powered by TCPDF (www.tcpdf.org)
Turkmani et al Dovepress
Methods experienced influenza-like illness (fever, headache, sore
A total of fourteen females, 22–65 years old, were throat, cough, and fatigue).
presented to our clinics between September 2016 and All patients described their history of multiple injec-
September 2018 complaining of localized redness and tions (between 2–6) over the last four years prior to onset
firm, painful swelling on their faces at the sites of of their symptoms in different sites of their face. Duration
Clinical, Cosmetic and Investigational Dermatology downloaded from https://www.dovepress.com/ by 92.186.191.200 on 19-Jul-2021
previously injected fillers (Figure 1). In all cases, the of the last filler injection varied from 2–10 months before
reactions had started 3–5 days after patients had experiencing the reaction (Table 1).
For personal use only.
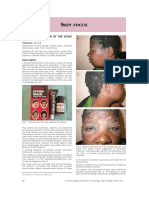
Patient 1. Cheeks and tear trough Patient 2. Cheeks and tear trough Patient 3. Tear trough
Patient 5. Left side of the face (tear trough
Patient 4. Lips Patient 6. Tear trough
cheeks and upper lip)
Patient 7. Cheeks Patient 8. Tear trough
Figure 1 Sites of reaction.
submit your manuscript | www.dovepress.com Clinical, Cosmetic and Investigational Dermatology 2019:12
278
DovePress
Powered by TCPDF (www.tcpdf.org)
Dovepress Turkmani et al
Table 1 The injected fillers, number of injections, and time to onset
Patient Patient Injected product(s) Number of treat- Time to onset of Treatment(s)
number age ments in the 4 years symptoms received
prior to onset of (months after
symptoms final treatment)
Clinical, Cosmetic and Investigational Dermatology downloaded from https://www.dovepress.com/ by 92.186.191.200 on 19-Jul-2021
1 32 Teosyal Deep Lines 4 4 Prednisolone
Belotero Intense Hyaluronidase
Belotero Volume
2 31 Teosyal Deep Lines 3 2 Prednisolone
Juvederm Volbella
Belotero Balance
3 29 Teosyal Puresense 2 10 Prednisolone
Juvederm Volift
4 22 Juvederm Volbella 2 4 Prednisolone
Teosyal Kiss
5 40 Juvederm Volbella 6 6 Prednisolone
Juvederm Voluma Hyaluronidase
Juvederm Volift
For personal use only.
6 65 Teosyal 2 4 Methyl Prednisolone
7 57 Hylaform 8 7 Methyl Prednisolone
Surgiderm
8 63 Juvederm Volbella 5 9 Prednisolone
Belotero Balance Hyaluronidase
9 41 Juvederm Volbella 3 3 Prednisolone
Juvederm Voluma Hyaluronidase
Juvederm Volift
10 25 Restylane Volyme Restylane Defyne 4 5 Prednisolone
Belotero Intense
11 34 Juvederm Voluma 6 3 Methyl Prednisolone
12 45 Belotero Balance 4 5 Methyl Prednisolone
13 23 Belotero Balance 2 6 Methyl Prednisolone
14 61 Restylane Refyne 6 4 Methyl Prednisolone
Restylane Defyne
The injections were performed by dermatologists or plastic Inamed (Nasdaq:IMDC) (Table 2). The most common sites of
surgeons using needles or cannula in different clinics. Patients’ the reactions were the cheeks (n=9), tear trough (n=8), and
files depicted no history of allergies or autoimmune disease and lips (n=3).
none of patients were injected with permanent fillers. All of the Patients were treated with oral prednisolone 20–30 mg
patients were unaware of the amounts of the injected fillers but or Methyl prednisolone 16–24 mg daily for 5 days, fol-
fully aware of the number of injections on their face, and the lowed by tempering of the dose for another 5 days. After
filler brands which were Juvederm (Allergan Inc. Pringy, a 2 week follow up, there was a complete resolution of the
France), Teosyal (Teoxane S.A., Geneva, Switzerland), symptoms in 10 patients following the oral steroid therapy.
Belotero (Anteis S.A, Geneva, Switzerland), Surgiderm The remaining 4 patients still presented minimal swelling
(Allergan Inc.) Restylane (Q-med AB Uppsala, Sweden) and that was treated with hyaluronidase one month after the
submit your manuscript | www.dovepress.com
Clinical, Cosmetic and Investigational Dermatology 2019:12 279
DovePress
Powered by TCPDF (www.tcpdf.org)
Turkmani et al Dovepress
Table 2 Number of injections in each anatomical site of each mediated by CD4+ cells. The memory of macrophages and
kind of filler a trigger, such as an infection or a drug-drug interaction,
Site of Brand of filler Number of could explain the unpredictability of foreign body granu-
injection injections in loma, which may occur even many years after the biode-
each site gradation of the implant.11 Macrophages are known to be
Clinical, Cosmetic and Investigational Dermatology downloaded from https://www.dovepress.com/ by 92.186.191.200 on 19-Jul-2021
Tear trough Juvederm Volbella 6 memory cells even when they move away from their site
Belotero Balance 4 of action once degradation of their target is complete.12
Teosyal Puresense 2 Still, the exact etiology of delayed hypersensitivity in
Lip Teosyal Kiss 1 relation to HA fillers and the influenza virus infection
Belotero intense 2 remains not completely understood.6,13
Juvederm Volbella 2 HA molecules in all fillers are the same polysaccharide
Cheeks Juvederm Voluma 6 molecules that compose a major part of our skin.
Restylane Refyne 3 Therefore, HA molecule itself is not usually considered
Restylane Defyne 1 an immunogen. As suggested by Bitterman-Deutsch et al,
Belotero Volume 1 however, in some conditions, glycosaminoglycans, such as
Nasolabial folds Restylane Refyne 2 HA, could directly trigger a specific immune response
Belotero Balance 2 without the primary phase of inflammation, like a -
Chin Juvederm Voluma 3 “superantigen”.14 Other components that are added to sta-
For personal use only.
Teosyal Deep Lines 3 bilize HA molecules in soft tissue fillers (eg, crosslinkers,
conserving) may also be immunogenic.
Nose Juvedrem Volift 2
Restylane Refyne 1
Beleznay et al have suggested a mechanism involving
the release of pro-inflammatory low molecular weight HA
fragments during an accelerated breakdown of HA gels
onset of symptoms (Figure 2). Patients were followed up triggered by a systemic inflammatory response to an
two months following the complete resolution of their unknown antigen.7 The definition of low molecular weight
symptoms and experienced another influenza-like infection HA, however, is not well defined in clinical research.
or a recurrence of symptoms at the sites of filler injections. Furthermore, whether low molecular weight HA fragments
Each individual patient from all the clinics provided cause a pro-inflammatory response is open to debate in
written informed consent for the case details and accom- scientific literature. Therefore, due to lack of a clear under-
panying images to be published. standing of the exact etiology of delayed hypersensitivity
Institutional approval from all clinics was not required reactions in some filler patients, it is difficult to conclude
to publish the case details. that specific filler technologies are more prone to induce
these reactions in comparison with others.15
Discussion The majority of patients here developed reactions at all
Delayed hypersensitivity may manifest from weeks to the previously injected sites at the same time (except
months after an uneventful HA filler injection. Hence, it is patient 7 who developed the reaction only in one side of
impossible to predict the probability of occurrence. Several the face) regardless of filler type, number of injections, or
published reports, however, have attempted to understand the injected volumes. Interestingly, two patients (patient
the etiology in relation to HA soft tissue fillers.6–9 It has #1 and #3) did not react at the previously injected sites
been suggested that biological/patient factors (eg, previous (nasolabial fold and nose, respectively). This is likely due
skin or systemic conditions such as infections and to the fact that the injections were done more than two
trauma),10 injection technique (eg, filler volume, repeat years from the time of the hypersensitivity reaction, and so
treatments, intramuscular implantation), and the different most of the injected fillers can be expected to have been
properties of HA fillers may explain the etiology. degraded and removed by then.
Type IV hypersensitivity following HA implantation An immunologic interaction between a concomitant amino
and influenza infection seems to be the most probable penicillin treatment and an Ebstein–Barr viral infection (ie,
explanation for our observed late-onset events. This rare sore throat) is known to induce maculopapular exanthems.16
systemic response may be initiated by T-lymphocytes and The exact mechanism behind the interaction is unclear and it is
submit your manuscript | www.dovepress.com Clinical, Cosmetic and Investigational Dermatology 2019:12
280
DovePress
Powered by TCPDF (www.tcpdf.org)
Dovepress Turkmani et al
Clinical, Cosmetic and Investigational Dermatology downloaded from https://www.dovepress.com/ by 92.186.191.200 on 19-Jul-2021
Patient 9. Cheeks and tear trough
For personal use only.
Patient 10. Cheeks, lips, and chin
Figure 2 Patient 1 and patient 2: sites of reaction before (left) and two weeks after (right) treatment.
not yet well explained whether a true allergic drug reaction, asymptomatic granulomas that may even subside on
infectious-dependent eruption or transient loss of drug toler- their own, especially in the case of hyaluronic acid.19
ance due to the infection, is responsible for the symptoms.17 Delayed complications of the soft tissue fillers are
The use of other medications such as antibiotics, antipyretic particularly difficult to diagnose and treat due to the
and non-steroidal anti-inflammatory drugs are commonly seen time lapse from the procedure.20 Type IV hypersensitiv-
in daily practice for treatment of acute respiratory tract infec- ity reactions are unresponsive to antihistamines.21
tions. HA, like viral infection, is known to activate in vitro Steroids are required to alleviate the inflammatory
T-lymphocytes via CD44.18 Therefore, HA could be consid- signs. Hyaluronidase may be injected to remove the
ered as a risk factor in the development of hypersensitivity allergen in some cases.22,23
reactions when any medication is introduced. Delayed hypersensitivity reaction to hyaluronic acid
The differential diagnosis of late onset nodules following infection is relatively rare. Homsy et al24
include infection (including biofilm), foreign body gran- reported multiple inflammatory collections on the face
ulomatous reaction, and immune-mediated delayed following sore throat in two patients. One case of cold
hypersensitivity reactions. Exact diagnosis may be diffi- sore and flu-like illness has been reported by Dr. Bhojani-
cult since delayed filler reactions can present in many Lynch,25 and one more case described a patient who
ways. Biofilms can present as erythematous, tender developed a granulomatous lip 8 months after silicone
nodules and papules.19 They can also present as inert, injections and 1 week after a flu-like syndrome.24 Here,
submit your manuscript | www.dovepress.com
Clinical, Cosmetic and Investigational Dermatology 2019:12 281
DovePress
Powered by TCPDF (www.tcpdf.org)
Turkmani et al Dovepress
we present 10 rare cases of late onset hypersensitivity to
hyaluronic acid injection following influenza-like illness. References
The main limitation of this article is the absence of 1. The American Society of Plastic Surgeons. Plastic Surgery
StatisticsReport. Available from: https://www.plasticsurgery.org/
a histological analysis. Biopsies were not performed due to
news/press-releases/new-statistics-reveal-theshape-of-plastic-surgery.
the patients’ desire to have minimally invasive resolutions Accessed March 1, 2017.
Clinical, Cosmetic and Investigational Dermatology downloaded from https://www.dovepress.com/ by 92.186.191.200 on 19-Jul-2021
for their symptoms as quickly as possible. 2. The American Society for Dermatologic Surgery. ASDS Survey.
Available from: https://www.asds.net/_Media.aspx?id=9449, https://
An immunologic interaction between dermal fillers www.asds.net/. Accessed May 17, 2018.
and influenza infection may be the cause of the delayed 3. Lowe NJ, Maxwell CA, Patnaik R. Adverse reactions to dermal
fillers: review. Dermatol Surg. 2005;31(11 Pt 2):1616–1625.
hypersensitivity. The exact mechanism is unclear and
4. De Boulle K. Management of complications after implantations of
not yet well understood; it is unknown whether the fillers. J Cosmet Dermatol. 2004;3(1):2–15. doi:10.1111/j.1473-
hypersensitivity is due to a true allergic filler reaction 2130.2004.00058.x
5. Arron ST, Neuhaus IM. Persistent delayed-type hypersensitivity reac-
or to an influenza infection. To verify this hypothesis, an tion to injectable non-animal-stabilized hyaluronic acid. J Cosmet
allergic work-up can be performed with the same HA Dermatol. 2007;6:167–171. doi:10.1111/j.1473-2165.2007.00331.x
6. Rongioletti F. Complications granulomateuses des techniques de
that was used in each patient. It is, however, difficult to
comblement. Ann Dermatol Venereol. 2008;135:59–65. doi:10.1016/
make definite conclusions from this test, as even if the S0151-9638(08)70213-8
results are negative, a hypersensitivity reaction cannot 7. Beleznay K, Carruthers JD, Carruthers A, Mummert ME,
Humphrey S. Delayed-onset nodules secondary to a smooth cohesive
be excluded due to the limited sensitivity of these 20 mg/mL hyaluronic acid filler: cause and management. Dermatol
tests.26–28 Commercially available HA – based fillers Surg. 2015;41:929–939. doi:10.1097/DSS.0000000000000418
For personal use only.
8. André P. Evaluation of the safety of a non-animal stabilized hyaluro-
have a wide variety of properties including the degree
nic acid (NASHA – Q-Medical, Sweden) in European countries:
of cross-linking and gel concentration that have an a retrospective study from 1997 to 2001. J Eur Acad Dermatol
extensive effect on their expected clinical outcome. Venereol. 2004;18:422–425.
9. Lee JM, Kim YJ. Foreign body granulomas after the use of dermal fillers:
Understanding these characteristics may be important pathophysiology, clinical appearance, histologic features, and treatment.
in deciphering the etiology of delayed hypersensitivity Arch Plast Surg. 2015;42:232–239. doi:10.5999/aps.2015.42.5.596
10. De Boulle K, Heydenrych I. Patient factors influencing dermal filler
reactions. It should be note that the actual causes are
complications: prevention, assessment, and treatment. Clin Cosmet
dependent not only on the characteristics of the HA Investig Dermatol. 2015;15(8):205–214. doi:10.2147/CCID.S80446
fillers, but also on the response of the biological host. 11. Moscona RR, Bergman R, Friedman-Birnbaum R. An unusual late
reaction to Zyderm I injections: A challenge for treatment. Plast
Other possible causes of delayed hypersensitivity in our Reconstr Surg. 1993;92:331. doi:10.1097/00006534-199308000-00021
patients may be injections of large volumes of dermal 12. Williams CT, Williams WJ. Granulomatous inflammation: a review.
J Clin Pathol. 1983;36:723.
filler in many facial sites and repeated treatments. Many
13. Alijotas-Reig J, Fernández-Figueras MT, Puig L. Late-onset inflamma-
questions still remain; for example: Why all the patients tory adverse reactions related to soft tissue filler injections. Clin Rev
injected with HA fillers do not display a hypersensitivity Allergy Immunol. 2013;45:97–108. doi:10.1007/s12016-012-8348-5
14. Bitterman-Deutsch O, Kogan L, Nasser F Delayed immune mediated
response when having influenza or influenza- like ill- adverse effects to hyaluronic acid fillers: report of five cases and review
ness? Why patient number 7 was presented with the of the literature. Dermatol Report. 2015;7:5851. doi:10.4081/
dr.2015.5851
symptoms only on the left side of the face although
15. Cyphert JM, Trempus CS, Garantziotis S. Size matters: molecular
she was injected with the same filler equally in both weight specificity of Hyaluronan effects in cell biology. Int J Cell
sides? What will happen if these patients get another flu Biol. 2015;2015:563818. doi:10.1155/2015/563818
16. Gonzalez-Delgado P, Blanes M, Soriano V, Montoro D, Loeda C,
attack? Are these patients proper candidates for receiv- Niveiro E. Erythema multiforme to amoxicillin with concurrent infec-
ing HA-based fillers again? We believe that more stu- tion by Epstein-Barr virus. Allergol Immunopathol. 2006;34:76–78.
doi:10.1157/13086752
dies should be conducted to explain the mechanism of
17. Onodi-Nagy K, Kinyo A, Meszes A, et al. Amoxicillin rash in patients
this reaction. with infectious mononucleosis: evidence of true drug sensitization. Allergy
Asthma Clin Immunol. 2015;11:1. doi:10.1186/s13223-015-0099-4
18. Jordan AR, Racine RR, Hennig MJ, Lokeshwar VB. The role of
Disclosure CD44 in disease pathophysiology and targeted treatment. Front
Wolfgang G Philipp-Dormston reports being a clinical Immunol. 2015;6:182. doi:10.3389/fimmu.2015.00182
19. Wagner RD, Fakhro A, Cox JA, Izaddoost SA. Etiology, prevention,
trial investigator, scientific advisor, and/or speaker for and management of infectious complications of dermal fillers. Semin
Allergan, Galderma, and Merz, and no other conflicts of Plast Surg. 2016;30:83–86. doi:10.1055/s-0036-1580734
20. Tseng CHW, Chen AM, Chang JYF. Hyaluronic acid injectionin-
interest or payments for this publication. The other duced delayed-onset foreign body granuloma. J Dental Sci.
authors report no conflicts of interest in this work. 2015;10:341–343. doi:10.1016/j.jds.2015.03.008
submit your manuscript | www.dovepress.com Clinical, Cosmetic and Investigational Dermatology 2019:12
282
DovePress
Powered by TCPDF (www.tcpdf.org)
Dovepress Turkmani et al
21. Funt D, Pavicic T. Dermal fillers in aesthetics: an overview of 25. Bhojani-Lynch T. Late-onset inflammatory response to hyal-
adverse events and treatment approaches. Plast Surg Nurs. uronic acid fillers. Dermal Plast Reconstruc Surg. 2017;5(12):
2015;35:13–32. doi:10.1097/PSN.0000000000000087 e1532.
22. Signorini M, Liew S, Sundaram H, et al.; Global Aesthetics Consensus 26. Wolkenstein P, Chosidow O, Flechet ML, et al. Patch testing in severe
Group. Global aesthetics consensus: avoidance and management of cutaneous adverse drug reactions, including Stevens-Johnson syndrome
complications from hyaluronic acid fillers-evidence and opinion-based and toxic epidermal necrolysis. Contact Derm. 1996;35:234–236.
Clinical, Cosmetic and Investigational Dermatology downloaded from https://www.dovepress.com/ by 92.186.191.200 on 19-Jul-2021
review and consensus recommendations. Plast Reconstr Surg. doi:10.1111/j.1600-0536.1996.tb02364.x
2016;137:961e–971e. doi:10.1097/PRS.0000000000002184 27. Britschgi M, Steiner UC, Schmid S, et al. T-cell involvement in
23. Alijotas-Reig J, Fernández-Figueras MT, Puig L. Pseudocystic encap- drug-induced acute generalized exanthematous pustulosis. J Clin
sulation: a late noninflammatory complication of hyaluronic acid filler Invest. 2001;107:1433–1441. doi:10.1172/JCI10501
injections. Dermatol Surg. 2013;39:1726–1728. doi:10.1111/dsu.12316 28. Padial MA, Alvarez-Ferreira J, Tapia B, et al. Acute generalized
24. Homsy A, Rüegg EM, Jandus P, Pittet-Cuénod B, Modarressi exanthematous pustulosis associated with pseudoephedrine. Br
A. Immunoligical reaction after facial hyaluronic acid injection. Case J Dermatol. 2004;150:139. doi:10.1111/j.1365-2133.2004.05717.x
Reports Plast Surg Hand Surg. 2017;4(1):68–72.
For personal use only.
Clinical, Cosmetic and Investigational Dermatology Dovepress
Publish your work in this journal
Clinical, Cosmetic and Investigational Dermatology is an interna- The manuscript management system is completely online and
tional, peer-reviewed, open access, online journal that focuses on includes a very quick and fair peer-review system, which is all easy
the latest clinical and experimental research in all aspects of skin to use. Visit http://www.dovepress.com/testimonials.php to read real
disease and cosmetic interventions. This journal is indexed on CAS. quotes from published authors.
Submit your manuscript here: https://www.dovepress.com/clinical-cosmetic-and-investigational-dermatology-journal
submit your manuscript | www.dovepress.com
Clinical, Cosmetic and Investigational Dermatology 2019:12 283
DovePress
Powered by TCPDF (www.tcpdf.org)
You might also like
- Case Analysis Team CollapseDocument6 pagesCase Analysis Team CollapseRchard Gomez100% (2)
- Breville BCI600 Ice Cream Maker User Manual PDFDocument40 pagesBreville BCI600 Ice Cream Maker User Manual PDFTon TonNo ratings yet
- GD Portfolio Self Critique Report SampleDocument5 pagesGD Portfolio Self Critique Report Sampleirawan50% (2)
- Delayed Hypersensitivity Reaction To Hyaluronic Acid DermalDocument7 pagesDelayed Hypersensitivity Reaction To Hyaluronic Acid DermalJosefa VenegasNo ratings yet
- Vitiligo - New Treatment ApproachDocument7 pagesVitiligo - New Treatment Approachsawaira khanNo ratings yet
- A 10-Point Plan For Avoiding Hyaluronic Acid Dermal Filler-Related Complications During Facial Aesthetic Procedures and Algorithms For ManagementDocument9 pagesA 10-Point Plan For Avoiding Hyaluronic Acid Dermal Filler-Related Complications During Facial Aesthetic Procedures and Algorithms For Managementleenatalia93100% (1)
- Jurnal 9Document8 pagesJurnal 9rizkaNo ratings yet
- Ccid 179430 A Series of in Vitro and Human Studies of A Novel Lip Cream 032119Document16 pagesCcid 179430 A Series of in Vitro and Human Studies of A Novel Lip Cream 032119Lina WinartiNo ratings yet
- Treatment-Resistant Prurigo Nodularis: Challenges and SolutionsDocument10 pagesTreatment-Resistant Prurigo Nodularis: Challenges and SolutionsIBHNo ratings yet
- The 10-Point Plan 2021 Updated Concepts For ImprovDocument36 pagesThe 10-Point Plan 2021 Updated Concepts For ImprovdnaielNo ratings yet
- Wollenberg Et Al 2000 AllergyDocument9 pagesWollenberg Et Al 2000 AllergyArturo VeraNo ratings yet
- Ccid 12 911Document7 pagesCcid 12 911Imanuel CristiantoNo ratings yet
- CCID 37327 Use of Tazarotene Foam For The Treatment of Acne Vulgaris 052714Document6 pagesCCID 37327 Use of Tazarotene Foam For The Treatment of Acne Vulgaris 052714Camila torresNo ratings yet
- CCID 84903 The Role of Vitamin C in Pushing Back The Boundaries of Skin - 090215Document8 pagesCCID 84903 The Role of Vitamin C in Pushing Back The Boundaries of Skin - 090215sebastianNo ratings yet
- Distinguishing Truly Recalcitrant Prurigo Nodularis From Poor Treatment Adherence - A Response To Treatment-Resistant Prurigo Nodularis - Response To LetterDocument3 pagesDistinguishing Truly Recalcitrant Prurigo Nodularis From Poor Treatment Adherence - A Response To Treatment-Resistant Prurigo Nodularis - Response To Letterfatchurhamzah62No ratings yet
- Dupilumab Side Effect in A Patient With Atopic Dermatitis: A Case Report StudyDocument4 pagesDupilumab Side Effect in A Patient With Atopic Dermatitis: A Case Report StudyKihyun YooNo ratings yet
- Ccid 281901 The One21 Technique An Individualized Treatment For GlabellDocument9 pagesCcid 281901 The One21 Technique An Individualized Treatment For GlabellAtena KhrdmndNo ratings yet
- Microbiome in Atopic Dermatitis: Clinical, Cosmetic and Investigational Dermatology DoveDocument6 pagesMicrobiome in Atopic Dermatitis: Clinical, Cosmetic and Investigational Dermatology DoveTrustia RizqandaruNo ratings yet
- Surgical Mask Contact Dermatitis and Epidemiology of Contact Dermatitis in Healthcare WorkersDocument6 pagesSurgical Mask Contact Dermatitis and Epidemiology of Contact Dermatitis in Healthcare WorkersJuan Stuart Xaverius JohanesNo ratings yet
- Baru 1Document6 pagesBaru 1Nazihan Safitri AlkatiriNo ratings yet
- InJPharPract 13 4 355Document4 pagesInJPharPract 13 4 355Yeol LoeyNo ratings yet
- A Series of in Vitro and Human Studies of A Novel Lip Cream FormulationDocument16 pagesA Series of in Vitro and Human Studies of A Novel Lip Cream FormulationDummy CipawNo ratings yet
- The Efficacy of Colloidal Oatmeal Cream 1 Percent As Addon Therapy in The Management of Chronic Irritant Hand Eczema - A Double-Blind StudyDocument11 pagesThe Efficacy of Colloidal Oatmeal Cream 1 Percent As Addon Therapy in The Management of Chronic Irritant Hand Eczema - A Double-Blind StudyQualityMattersNo ratings yet
- Treatment-Resistant Prurigo Nodularis: Challenges and SolutionsDocument10 pagesTreatment-Resistant Prurigo Nodularis: Challenges and Solutionsranz ibonkNo ratings yet
- Ad 26 462Document7 pagesAd 26 462AmelyalesmanaNo ratings yet
- Chemical Peels in The Treatment of Acne PDFDocument8 pagesChemical Peels in The Treatment of Acne PDFSpica AdharaNo ratings yet
- "前路"化学磨削治疗痤疮疤痕Document9 pages"前路"化学磨削治疗痤疮疤痕kkNo ratings yet
- Ccid 8 257 PDFDocument9 pagesCcid 8 257 PDFFahmil AgungNo ratings yet
- Allergic Contact Dermatitis - TranslateDocument9 pagesAllergic Contact Dermatitis - Translatewahyuni taslimNo ratings yet
- Ccid 11 175Document11 pagesCcid 11 175Remo Alnovryanda PutraNo ratings yet
- CCID 111019 Hidradenitis Suppurativa From Pathogenesis To Diagnosis and 041917Document11 pagesCCID 111019 Hidradenitis Suppurativa From Pathogenesis To Diagnosis and 041917Popi ZeniusaNo ratings yet
- 2010 - Octenidine Dihydrochloride, A Modern Antiseptic For Skin, Mucous Membranes and WoundsDocument15 pages2010 - Octenidine Dihydrochloride, A Modern Antiseptic For Skin, Mucous Membranes and Woundsnovia100% (2)
- Gau Glitz 2013Document12 pagesGau Glitz 2013Stella SunurNo ratings yet
- Efficacy and Safety of Cream Containing Climbazole/Piroctone Olamine For Facial Seborrheic Dermatitis: A Single-Center, Open-Label Split-Face Clinical StudyDocument7 pagesEfficacy and Safety of Cream Containing Climbazole/Piroctone Olamine For Facial Seborrheic Dermatitis: A Single-Center, Open-Label Split-Face Clinical StudynurulNo ratings yet
- Treatmen-Resistant Bacterial Keratitis, Challenges and SolutuionsDocument11 pagesTreatmen-Resistant Bacterial Keratitis, Challenges and SolutuionsGraitaaNo ratings yet
- High Frequency Equipment Promotes Antibacterial Effects Dependent On Intensity and Exposure TimeDocument5 pagesHigh Frequency Equipment Promotes Antibacterial Effects Dependent On Intensity and Exposure TimeJéssica CarvalhalNo ratings yet
- Letters To The Editor: Hand Eczema Due To Hygiene and Antisepsis Products: Not Only An Irritative EtiologyDocument2 pagesLetters To The Editor: Hand Eczema Due To Hygiene and Antisepsis Products: Not Only An Irritative Etiologysaka wibawaNo ratings yet
- 2023 Article 1070Document12 pages2023 Article 1070Noema AmorochoNo ratings yet
- Contact Dermatitis: Allergic and IrritantDocument9 pagesContact Dermatitis: Allergic and IrritantatikaNo ratings yet
- 2020 Cutaneous Protothecosis As An Unusual Complication Following Dermal Filler Injection A Case Report.Document4 pages2020 Cutaneous Protothecosis As An Unusual Complication Following Dermal Filler Injection A Case Report.Dimitris RodriguezNo ratings yet
- A Hands On Approach To Patch TestingDocument11 pagesA Hands On Approach To Patch TestingSundar RamanathanNo ratings yet
- Ccid 237519 One21 A Novel Customizable Injection Protocol For TreatmenDocument10 pagesCcid 237519 One21 A Novel Customizable Injection Protocol For TreatmenRafael SanchezNo ratings yet
- Gloves Handling Chemotherapeutic: AgentsDocument4 pagesGloves Handling Chemotherapeutic: AgentsDarren TanNo ratings yet
- Contact Dermatitis in The Patient With Atopic DermatitisDocument9 pagesContact Dermatitis in The Patient With Atopic Dermatitislina7soteroNo ratings yet
- Reading AsthmaDocument7 pagesReading AsthmaLovely Grace PoreNo ratings yet
- Jurnal Dermatitis KontakDocument3 pagesJurnal Dermatitis KontakretrianisutrisnoNo ratings yet
- Epidemiological Characterization of Dermatomycosis in EthiopiaDocument7 pagesEpidemiological Characterization of Dermatomycosis in EthiopiaLuthfiyah SalsabillaNo ratings yet
- Consensus Statement On Prevention and Management of Adverse Effects Following Rejuvenation Procedures With Hyaluronic Acid Based FillersDocument26 pagesConsensus Statement On Prevention and Management of Adverse Effects Following Rejuvenation Procedures With Hyaluronic Acid Based FillersArmin ArceoNo ratings yet
- OPTH 80503 Effect of Alcaftadine 0 25 On Ocular Itch Associated With S - 050215Document8 pagesOPTH 80503 Effect of Alcaftadine 0 25 On Ocular Itch Associated With S - 050215Ravi RanjanNo ratings yet
- A 10% Glycolic Acid Containing Oil in Water Emulsion Improves Mild Acne A Randomized Double Blind Placebo Controlled TriaDocument8 pagesA 10% Glycolic Acid Containing Oil in Water Emulsion Improves Mild Acne A Randomized Double Blind Placebo Controlled TriaGabriela ArthusoNo ratings yet
- Ceramid DADocument4 pagesCeramid DAAli HadjinNo ratings yet
- Newly Developed Anti-Dandruff RegimenDocument9 pagesNewly Developed Anti-Dandruff RegimenHilda FitriaNo ratings yet
- System Disorder: Dermatitis and Acne: Atopic Dermatitis 57Document1 pageSystem Disorder: Dermatitis and Acne: Atopic Dermatitis 57Kassandra MerrillNo ratings yet
- Niculet2020 Glucocorticoid Induced Skin AtrophyDocument10 pagesNiculet2020 Glucocorticoid Induced Skin AtrophyYuri YogyaNo ratings yet
- Ccid 12 759Document11 pagesCcid 12 759dimiz77No ratings yet
- Choudhury 2015Document7 pagesChoudhury 2015KreAch3RNo ratings yet
- Dermatology Module 3Document7 pagesDermatology Module 3joseph joshua ngushualNo ratings yet
- Kumpulan Resep Ilmu Penyakit Kulit Dan KelaminDocument9 pagesKumpulan Resep Ilmu Penyakit Kulit Dan KelaminLevaszkaNo ratings yet
- CCID 75908 Management of Pemphigus Vulgaris Challenges and Solutions 102115Document7 pagesCCID 75908 Management of Pemphigus Vulgaris Challenges and Solutions 102115M Marliando Satria PangestuNo ratings yet
- Contact Dermatitis - 2016 - Verhulst - Cosmetic Components Causing Contact Urticaria A Review and UpdateDocument13 pagesContact Dermatitis - 2016 - Verhulst - Cosmetic Components Causing Contact Urticaria A Review and UpdateВалентина ПлукчиNo ratings yet
- Pathophysiology of Allergic and Irritant Contact Dermatitis: January 2012Document9 pagesPathophysiology of Allergic and Irritant Contact Dermatitis: January 2012cemm_11No ratings yet
- Clinical and Instrumental Assessment of The Effects of A New ProductDocument5 pagesClinical and Instrumental Assessment of The Effects of A New ProductMyroslava KvitchukNo ratings yet
- 1 s2.0 S002239132100634X MainDocument12 pages1 s2.0 S002239132100634X MaindominiqueNo ratings yet
- Botox Masetero 2Document9 pagesBotox Masetero 2dominiqueNo ratings yet
- Hand Injury Clinical Case by SlidesgoDocument57 pagesHand Injury Clinical Case by SlidesgodominiqueNo ratings yet
- Lee 2020Document10 pagesLee 2020dominiqueNo ratings yet
- Paper Lips1Document11 pagesPaper Lips1dominiqueNo ratings yet
- Complicaciones Hilos PdoDocument3 pagesComplicaciones Hilos PdodominiqueNo ratings yet
- Research Article Safety Assessment of High-And Low-Molecular-Weight Hyaluronans (Profhilo®) As Derived From Worldwide Postmarketing DataDocument9 pagesResearch Article Safety Assessment of High-And Low-Molecular-Weight Hyaluronans (Profhilo®) As Derived From Worldwide Postmarketing DatadominiqueNo ratings yet
- Three Winding or Tertiary Winding TransformerDocument7 pagesThree Winding or Tertiary Winding TransformergopalakrishnanNo ratings yet
- Pharmacology of AntidepressantsDocument28 pagesPharmacology of Antidepressantsحيدر كريم سعيد حمزهNo ratings yet
- FMCG Industry Review of Bangladesh PDFDocument33 pagesFMCG Industry Review of Bangladesh PDFShoaib HussainNo ratings yet
- Kadar Protein Terlarut Dalam Albumin Ikan Gabus (Channa Striata Dan Channa Micropeltes) Asal BogorDocument9 pagesKadar Protein Terlarut Dalam Albumin Ikan Gabus (Channa Striata Dan Channa Micropeltes) Asal BogorAnkaranaRenvaNo ratings yet
- Dry Well PSDocument7 pagesDry Well PSprajmenNo ratings yet
- 15 Minutes Test I. Circle The Word Whose Underlined Part Is Pronounced Differently From The OthersDocument2 pages15 Minutes Test I. Circle The Word Whose Underlined Part Is Pronounced Differently From The OthersLann Anhh NguyễnNo ratings yet
- Cube Test Letter - SignedDocument2 pagesCube Test Letter - Signedsamarsandip1No ratings yet
- Ponedjeljak: Mišićna Skupina Upper BodyDocument12 pagesPonedjeljak: Mišićna Skupina Upper BodyVedran Pisarek100% (1)
- Hypertension FamcoDocument42 pagesHypertension FamcoMusleh Al MusalhiNo ratings yet
- Primary Frca Help Plain UpdatedDocument5 pagesPrimary Frca Help Plain UpdatedDavid PappinNo ratings yet
- Compatibility Testing - BloodDocument5 pagesCompatibility Testing - BloodMunish DograNo ratings yet
- Sample QuestionnaireDocument5 pagesSample QuestionnaireAudi Kyle SaydovenNo ratings yet
- 0628-334-01 - D LLHS Operator ManualDocument48 pages0628-334-01 - D LLHS Operator Manualtecvision.assistenciaNo ratings yet
- Program Kreativitas MahasiswaDocument28 pagesProgram Kreativitas Mahasiswawendi gunawanNo ratings yet
- Know All Men by These PresentsDocument7 pagesKnow All Men by These PresentsKyle GuerreroNo ratings yet
- State Wise Members of JAC For Organizing Senior Citizens National Protest Day - 16 Aufgust 2010Document2 pagesState Wise Members of JAC For Organizing Senior Citizens National Protest Day - 16 Aufgust 2010api-27224201No ratings yet
- 01 Health Safety & Environmental MGMTDocument134 pages01 Health Safety & Environmental MGMTLau Siong Kwong67% (3)
- NCP Risk For InfectionDocument1 pageNCP Risk For InfectionBianca MaeNo ratings yet
- Sourse File Foe Ekta 3Document64 pagesSourse File Foe Ekta 3SUBHI AWASTHINo ratings yet
- 150 de Thi Hoc Sinh Gioi Tieng Anh Lop 6 Kem Dap AnDocument58 pages150 de Thi Hoc Sinh Gioi Tieng Anh Lop 6 Kem Dap AnChung Thuỷ NguyễnNo ratings yet
- api/resources/file/CH604MM/ATA 100/CH604/AMM/49/AMM49 10 00 04.fullDocument40 pagesapi/resources/file/CH604MM/ATA 100/CH604/AMM/49/AMM49 10 00 04.fullRahul VaishnavNo ratings yet
- TOPIC: Exceptionalities and Interventions of Special EducationDocument7 pagesTOPIC: Exceptionalities and Interventions of Special EducationRaymart EstavilloNo ratings yet
- 2&3&4&5 Burner Induction+Infrared Cooker Quotations-Alan Liu (LONGSTAR)Document9 pages2&3&4&5 Burner Induction+Infrared Cooker Quotations-Alan Liu (LONGSTAR)Dương Nguyễn ĐắcNo ratings yet
- Bill of Lading / Guía de Carga: Corpus Christi, TX 78466 Tel. (361) 814-7867 TAX ID:81-3031542 TMF Ticket#Document1 pageBill of Lading / Guía de Carga: Corpus Christi, TX 78466 Tel. (361) 814-7867 TAX ID:81-3031542 TMF Ticket#Victor Ingmar abisaiNo ratings yet
- UK DynaSand C0209Document2 pagesUK DynaSand C0209Mee DepoNo ratings yet
- Eti Final MPDocument19 pagesEti Final MPAstel DmelloNo ratings yet
- Secondary RefrigerantsDocument15 pagesSecondary Refrigerantse4erkNo ratings yet