Professional Documents
Culture Documents
A Radiologist's Guide To The 2021 WHO Central Nervous System Tumor Classification
A Radiologist's Guide To The 2021 WHO Central Nervous System Tumor Classification
Uploaded by
MANGCopyright:
Available Formats
You might also like
- 2019 July Ethiopia EHealth Architecture Document 1.5Document31 pages2019 July Ethiopia EHealth Architecture Document 1.5Tinsae Getachew100% (1)
- Brio UK Lit Dec 2007Document8 pagesBrio UK Lit Dec 2007Neil HoggNo ratings yet
- 9 - Urban & Rural Development in BangladeshDocument21 pages9 - Urban & Rural Development in BangladeshMd Omar Kaiser MahinNo ratings yet
- Retail Permit To Work System - Job Hazard AnalysisDocument7 pagesRetail Permit To Work System - Job Hazard AnalysisSanthosh Kumar100% (1)
- French CuisineDocument21 pagesFrench CuisineLea Rachelle VillarmenteNo ratings yet
- TNM 2017Document11 pagesTNM 2017David MaynéNo ratings yet
- EdCaN Specialty Module 4Document23 pagesEdCaN Specialty Module 4cindy8127No ratings yet
- 2022 - Horbinsky - Clin ImpliDocument15 pages2022 - Horbinsky - Clin ImpliRimpa Basu AchariNo ratings yet
- Integrating Pathology and Radiology Disciplines: An Emerging Opportunity?Document7 pagesIntegrating Pathology and Radiology Disciplines: An Emerging Opportunity?Peko PekoNo ratings yet
- Diagnostic Value of Circulating Tumor DNA in Molecular Characterization of GliomaDocument8 pagesDiagnostic Value of Circulating Tumor DNA in Molecular Characterization of GliomaAnyaNo ratings yet
- The 2021 WHO Classification of Tumors of The Central Nervous System, A SumaryDocument21 pagesThe 2021 WHO Classification of Tumors of The Central Nervous System, A SumaryDTNo ratings yet
- Principles Cancer StagingDocument28 pagesPrinciples Cancer Stagingcheatingw995No ratings yet
- Neuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryDocument21 pagesNeuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryJoao FabricioNo ratings yet
- Principles of Cancer StagingDocument28 pagesPrinciples of Cancer StagingAnonymous PKE8zOXNo ratings yet
- Neuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryDocument21 pagesNeuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryArif Susilo RahadiNo ratings yet
- Nejm - SobrevivienteDocument13 pagesNejm - SobrevivienteKathyMarambioNo ratings yet
- Colorectal Cancer StagingDocument14 pagesColorectal Cancer StagingRonnie Alexander Reyes MartínezNo ratings yet
- Underwood Et Al 2019 Liquid Biopsy For Cancer Review and Implications For The RadiologistDocument13 pagesUnderwood Et Al 2019 Liquid Biopsy For Cancer Review and Implications For The RadiologistpakhadkikarNo ratings yet
- 01 Purpose and Principles of Cancer Staging PDFDocument12 pages01 Purpose and Principles of Cancer Staging PDFalyson100% (1)
- Paper 2 Cancer DicoveryDocument18 pagesPaper 2 Cancer DicoverySuraj ShahNo ratings yet
- AllisonDocument11 pagesAllisonNabila NatasyaNo ratings yet
- Clinical and Research Applications of A Brain Tumor Tissue Bank in The Age of Precision MedicineDocument12 pagesClinical and Research Applications of A Brain Tumor Tissue Bank in The Age of Precision MedicineSandra Milena Sanabria BarreraNo ratings yet
- Adressing Suicidal Ideation in Patients With CancerDocument3 pagesAdressing Suicidal Ideation in Patients With CancerJosé Carlos Sánchez-RamirezNo ratings yet
- CNS SummaryDocument21 pagesCNS SummaryshokoNo ratings yet
- Imaging Diagnosis and Treatment Selection For Brain Tumors in The Era of Molecular TherapeuticsDocument14 pagesImaging Diagnosis and Treatment Selection For Brain Tumors in The Era of Molecular Therapeuticsmarwa humairaNo ratings yet
- Neuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryDocument21 pagesNeuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryAndrei PatrichiNo ratings yet
- Artcle 2 PDFDocument11 pagesArtcle 2 PDFAnirban PaulNo ratings yet
- 79 6690033a PDFDocument13 pages79 6690033a PDFEdward Arthur IskandarNo ratings yet
- Koch 2009Document9 pagesKoch 2009Intan Kartika NursyahbaniNo ratings yet
- 02-1.WHO 2022-PitNETDocument21 pages02-1.WHO 2022-PitNETJihuhn YuNo ratings yet
- How To Read A Next-Generation Sequencing Report-What Oncologists Need To KnowDocument9 pagesHow To Read A Next-Generation Sequencing Report-What Oncologists Need To KnowsamuelNo ratings yet
- Cancer Biomarkers: Easier Said Than Done: Kenneth P.H. PritzkerDocument4 pagesCancer Biomarkers: Easier Said Than Done: Kenneth P.H. Pritzkerdumboo21No ratings yet
- Radiation Therapy-Guidelines For PhysiotherapistsDocument9 pagesRadiation Therapy-Guidelines For PhysiotherapistsSofia Adão da FonsecaNo ratings yet
- 2020 Terri Glioma PRODocument7 pages2020 Terri Glioma PRORimpa Basu AchariNo ratings yet
- Radiotherapy and OncologyDocument14 pagesRadiotherapy and OncologyJavier RodríguezNo ratings yet
- Melanoma Pathology Reporting and StagingDocument10 pagesMelanoma Pathology Reporting and StagingLizbeth QuinteroNo ratings yet
- Hematolrep 03 E2 v2Document3 pagesHematolrep 03 E2 v2772450336No ratings yet
- 2013 ASTRO e ACR - Practice Guideline For The Performance TBIDocument5 pages2013 ASTRO e ACR - Practice Guideline For The Performance TBIAndre BanhateNo ratings yet
- Systemic Therapy For Advanced Appendiceal Adenocarcinoma: An Analysis From The NCCN Oncology Outcomes Database For Colorectal CancerDocument8 pagesSystemic Therapy For Advanced Appendiceal Adenocarcinoma: An Analysis From The NCCN Oncology Outcomes Database For Colorectal Canceralberto cabelloNo ratings yet
- Prognostic Biomarkers For Survival in Nasopharyngeal Carcinoma - A Systematic Review of The LiteratureDocument14 pagesPrognostic Biomarkers For Survival in Nasopharyngeal Carcinoma - A Systematic Review of The LiteratureWening Dewi HapsariNo ratings yet
- Precision Oncology in Low and Middle Income Countries A Word of CautionDocument5 pagesPrecision Oncology in Low and Middle Income Countries A Word of Cautionymourjany87No ratings yet
- Histological Molecular and Functional Subtypes ofDocument7 pagesHistological Molecular and Functional Subtypes ofjumed21No ratings yet
- Mizuno Et Al 2021Document20 pagesMizuno Et Al 2021Abner PortilhoNo ratings yet
- Jco 2009 27 0660Document9 pagesJco 2009 27 0660KAREN JAZMIN RUIZ MONROYNo ratings yet
- Diagnosisofrenalcell Carcinoma: A Clinician'S PerspectiveDocument6 pagesDiagnosisofrenalcell Carcinoma: A Clinician'S PerspectiveGangsar DamaiNo ratings yet
- s00261 023 03893 2Document15 pagess00261 023 03893 2sulbey878No ratings yet
- Meningiomas: Knowledge Base, Treatment Outcomes, and Uncertainties. A RANO ReviewDocument20 pagesMeningiomas: Knowledge Base, Treatment Outcomes, and Uncertainties. A RANO ReviewLuwiNo ratings yet
- Cancer of Unknown PrimaryDocument12 pagesCancer of Unknown Primaryraul gutierrezNo ratings yet
- Tearing Down The Walls: FDA Approves Next Generation Sequencing (NGS) Assays For Actionable Cancer Genomic AberrationsDocument3 pagesTearing Down The Walls: FDA Approves Next Generation Sequencing (NGS) Assays For Actionable Cancer Genomic AberrationsAdrián CLNo ratings yet
- Explore Rare Cancer Medicine MutationsDocument6 pagesExplore Rare Cancer Medicine MutationsMikel AngelNo ratings yet
- Cancer Awareness - 25 February 2021Document4 pagesCancer Awareness - 25 February 2021Times MediaNo ratings yet
- GMM. WHO CNS5 Classification (English Version)Document8 pagesGMM. WHO CNS5 Classification (English Version)Melissa Garcia LezamaNo ratings yet
- Bussines EngineringDocument9 pagesBussines EngineringWhulan Rhana TonapaNo ratings yet
- A Systematic Review On Barriers To Palliative Care in OncologyDocument17 pagesA Systematic Review On Barriers To Palliative Care in Oncologyw89qyccn6sNo ratings yet
- Dec 1 Family - Tree Chung 2002Document8 pagesDec 1 Family - Tree Chung 2002onlydarkheartNo ratings yet
- Document 1Document8 pagesDocument 1JOSE PORTILLONo ratings yet
- Colorectal Carcinoma: Diagnostic, Prognostic, and Molecular FeaturesDocument13 pagesColorectal Carcinoma: Diagnostic, Prognostic, and Molecular FeaturesEdward Arthur IskandarNo ratings yet
- Pain - Assessment JE DWDocument8 pagesPain - Assessment JE DWAshesh DahalNo ratings yet
- Head and Neck Cancers, Version 2.2020: NCCN Clinical Practice Guidelines in OncologyDocument26 pagesHead and Neck Cancers, Version 2.2020: NCCN Clinical Practice Guidelines in OncologympNo ratings yet
- Cell Specific Frequency As New HallmarkDocument8 pagesCell Specific Frequency As New Hallmarkjongsu kimNo ratings yet
- Functional and Mollecular ImagingDocument15 pagesFunctional and Mollecular Imagingstepanus massoraNo ratings yet
- Medical Oncologists' Perspectives of The Veterans Affairs National Precision Oncology ProgramDocument15 pagesMedical Oncologists' Perspectives of The Veterans Affairs National Precision Oncology ProgramFredrick WallinNo ratings yet
- Xi, 2021 Tumor Associated Collagen SignaturesDocument15 pagesXi, 2021 Tumor Associated Collagen Signaturesevahendrickx03No ratings yet
- A Patient's Guide to Cancer: Understanding the Causes and Treatments of a Complex DiseaseFrom EverandA Patient's Guide to Cancer: Understanding the Causes and Treatments of a Complex DiseaseNo ratings yet
- Glioma Imaging: Physiologic, Metabolic, and Molecular ApproachesFrom EverandGlioma Imaging: Physiologic, Metabolic, and Molecular ApproachesWhitney B. PopeNo ratings yet
- Interventional Radiology Management of High Flow PriapismDocument11 pagesInterventional Radiology Management of High Flow PriapismMANGNo ratings yet
- Complications of Central Venous Port Systems: A Pictorial ReviewDocument12 pagesComplications of Central Venous Port Systems: A Pictorial ReviewMANGNo ratings yet
- Complementary Roles of Interventional Radiology and Therapeutic Endoscopy in GastroenterologyDocument16 pagesComplementary Roles of Interventional Radiology and Therapeutic Endoscopy in GastroenterologyMANGNo ratings yet
- Basic Principles of Hematology: Section 13Document6 pagesBasic Principles of Hematology: Section 13MANGNo ratings yet
- CT Findings of Pulmonary NocardiosisDocument7 pagesCT Findings of Pulmonary NocardiosisMANGNo ratings yet
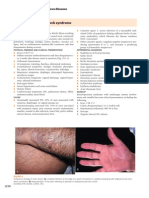
- 14.drug Eruption 2005Document6 pages14.drug Eruption 2005MANGNo ratings yet
- Paroxysmal Nocturnal Hemoglobinuria: Section 13Document2 pagesParoxysmal Nocturnal Hemoglobinuria: Section 13MANGNo ratings yet
- Diagnosis and Classification in MRI of Brucellar SpondylitisDocument6 pagesDiagnosis and Classification in MRI of Brucellar SpondylitisMANGNo ratings yet
- Apr-44 Sep-46 Sep-47 Jun-48 Dec-49 Mar-50 Oct-51 Jan-54 Mar-54 Apr-54 May-54 Sep-54 Jan-55Document4 pagesApr-44 Sep-46 Sep-47 Jun-48 Dec-49 Mar-50 Oct-51 Jan-54 Mar-54 Apr-54 May-54 Sep-54 Jan-55MANGNo ratings yet
- Dual-Energy CT: General PrinciplesDocument6 pagesDual-Energy CT: General PrinciplesMANGNo ratings yet
- PostDocument3 pagesPostMANGNo ratings yet
- Dual-Energy Spectral Computed Tomography To Thoracic Oncology Imaging KJRDocument13 pagesDual-Energy Spectral Computed Tomography To Thoracic Oncology Imaging KJRMANGNo ratings yet
- UCSF ED US Protocol LE Venous - FinalDocument8 pagesUCSF ED US Protocol LE Venous - FinalMANGNo ratings yet
- Toxic Shock Syndrome: Section 14Document2 pagesToxic Shock Syndrome: Section 14MANGNo ratings yet
- B9781416049197X50014 B978141604919750178X MainDocument4 pagesB9781416049197X50014 B978141604919750178X MainMANGNo ratings yet
- Colorectal Cancer: Section 7Document3 pagesColorectal Cancer: Section 7MANGNo ratings yet
- B9781416049197X50014 B9781416049197502152 MainDocument2 pagesB9781416049197X50014 B9781416049197502152 MainMANGNo ratings yet
- B9781416049197X50014 B9781416049197500396 MainDocument1 pageB9781416049197X50014 B9781416049197500396 MainMANGNo ratings yet
- Diabetes Mellitus: Disorders of Carbohydrate and Lipid MetabolismDocument5 pagesDiabetes Mellitus: Disorders of Carbohydrate and Lipid MetabolismMANGNo ratings yet
- B9781416049197X50014 B9781416049197501791 MainDocument2 pagesB9781416049197X50014 B9781416049197501791 MainMANGNo ratings yet
- Diagnostic Tests and Procedures: Section 2Document7 pagesDiagnostic Tests and Procedures: Section 2MANGNo ratings yet
- B9781416049197X50014 B9781416049197501985 MainDocument2 pagesB9781416049197X50014 B9781416049197501985 MainMANGNo ratings yet
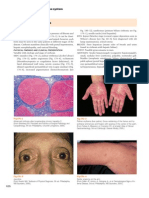
- The Structure and Function of Skin Normal Anatomy: Section 2Document6 pagesThe Structure and Function of Skin Normal Anatomy: Section 2MANGNo ratings yet
- B9781416049197X50014 B9781416049197502772 MainDocument2 pagesB9781416049197X50014 B9781416049197502772 MainMANGNo ratings yet
- B9781416049197X50014 B9781416049197503534 MainDocument2 pagesB9781416049197X50014 B9781416049197503534 MainMANGNo ratings yet
- The Structure and Function of Skin: Section 1Document2 pagesThe Structure and Function of Skin: Section 1MANGNo ratings yet
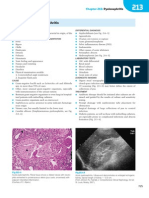
- Familial Polyposis Syndrome: Section 7Document1 pageFamilial Polyposis Syndrome: Section 7MANGNo ratings yet
- B9781416049197X50014 B9781416049197502097 MainDocument2 pagesB9781416049197X50014 B9781416049197502097 MainMANGNo ratings yet
- Reaction Paper KyleDocument4 pagesReaction Paper KyleRonaliza Durano SanchezNo ratings yet
- CHN RleDocument5 pagesCHN RleAntoinette Peleña100% (1)
- KS3 Human Reproduction Exam HigherDocument4 pagesKS3 Human Reproduction Exam HigherSarah KKCNo ratings yet
- TariffDocument254 pagesTariffrehmannazirNo ratings yet
- Paragraphs: A. B. C. D. E. FDocument4 pagesParagraphs: A. B. C. D. E. FAnh Cường ĐỗNo ratings yet
- Band III Core Word ListDocument40 pagesBand III Core Word ListKatya RipsNo ratings yet
- Icsh Hemostasis Critical ValuesDocument12 pagesIcsh Hemostasis Critical Valuescitometria prolabNo ratings yet
- ASSESSOR ScriptDocument3 pagesASSESSOR ScriptJonalyn Lajera BasayaNo ratings yet
- Meeting 6 Understanding The MenuDocument3 pagesMeeting 6 Understanding The Menujurnal mumtazNo ratings yet
- Polyhydraminos and OligohydraminosDocument11 pagesPolyhydraminos and OligohydraminosMelissa Catherine ChinNo ratings yet
- 152 Experiment 3Document2 pages152 Experiment 3Earl CañoneroNo ratings yet
- Final Hospital Pharmacy Module 1Document7 pagesFinal Hospital Pharmacy Module 1Meryl Ann Ibarra-InganNo ratings yet
- 61-12 (PART 0) /issue 1 6 November 1992 Wires Cords and Cables Electrical - Metric Units Part 0:general Requirements and Test Methods For Qualification ApprovalDocument82 pages61-12 (PART 0) /issue 1 6 November 1992 Wires Cords and Cables Electrical - Metric Units Part 0:general Requirements and Test Methods For Qualification Approvaluğur SevgenNo ratings yet
- Feb28.2015 B.docmandatory Employment of Lifeguards in Public Pools SoughtDocument2 pagesFeb28.2015 B.docmandatory Employment of Lifeguards in Public Pools Soughtpribhor2No ratings yet
- Aptitude TestDocument8 pagesAptitude TestKhalidNo ratings yet
- Disease Detectives C ExamDocument34 pagesDisease Detectives C ExamGustavo Pacheco0% (1)
- RoR March Tongan-5Document80 pagesRoR March Tongan-5netanevete020No ratings yet
- University of Caloocan CityDocument9 pagesUniversity of Caloocan CityFudge FajardoNo ratings yet
- Engineering Manual: Variable Refrigerant Flow Outdoor Units 6.0 To 42.0 TonsDocument156 pagesEngineering Manual: Variable Refrigerant Flow Outdoor Units 6.0 To 42.0 TonsNey Raul Benji JoaquinNo ratings yet
- Carbon Offsets: Farming Carbon in The SoilDocument9 pagesCarbon Offsets: Farming Carbon in The Soildanmorgan1968No ratings yet
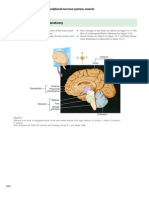
- Lab 2: Anatomy of The Heart: General ConsiderationsDocument19 pagesLab 2: Anatomy of The Heart: General ConsiderationsDisshiNo ratings yet
- GWYB 14-15 KarnatakaDocument226 pagesGWYB 14-15 Karnatakahram_phdNo ratings yet
- No Faster Prop On The Water: Step 1. Engine/DRIVE Step 2. Installation Kit Step 3. PropellerDocument2 pagesNo Faster Prop On The Water: Step 1. Engine/DRIVE Step 2. Installation Kit Step 3. PropellerjitmarineNo ratings yet
- Bartending 101Document15 pagesBartending 101Samuel SanguinoNo ratings yet
- Summary of AFSCME Tentative MOUDocument5 pagesSummary of AFSCME Tentative MOUjon_ortizNo ratings yet
A Radiologist's Guide To The 2021 WHO Central Nervous System Tumor Classification
A Radiologist's Guide To The 2021 WHO Central Nervous System Tumor Classification
Uploaded by
MANGOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
A Radiologist's Guide To The 2021 WHO Central Nervous System Tumor Classification
A Radiologist's Guide To The 2021 WHO Central Nervous System Tumor Classification
Uploaded by
MANGCopyright:
Available Formats
REVIEWS AND COMMENTARY • REVIEW
A Radiologist’s Guide to the 2021 WHO Central Nervous
System Tumor Classification: Part I—Key Concepts and the
Spectrum of Diffuse Gliomas
Derek R. Johnson, MD • Caterina Giannini, MD, PhD • Rachael A. Vaubel, MD, PhD • Jonathan M. Morris, MD •
Laurence J. Eckel, MD • Timothy J. Kaufmann, MD • Julie B. Guerin, MD
From the Departments of Radiology (D.R.J., J.M.M., L.J.E., T.J.K., J.B.G.), Neurology (D.R.J.), and Laboratory Medicine and Pathology (C.G., R.A.V.), Mayo Clinic,
200 First St SW, Rochester, MN 55905; and Department of Biomedical and Neuromotor Sciences, University of Bologna, Bologna, Italy (C.G.). Received December 13,
2021; revision requested January 13, 2022; revision received May 9; accepted May 18. Address correspondence to D.R.J. (email: Johnson.derek1@mayo.edu).
Conflicts of interest are listed at the end of this article.
Radiology 2022; 304:494–508 • https://doi.org/10.1148/radiol.213063 • Content code:
The fifth edition of the World Health Organization (WHO) classification of tumors of the central nervous system, published in
2021, contains substantial updates in the classification of tumor types. Many of these changes are relevant to radiologists, including
“big picture” changes to tumor diagnosis methods, nomenclature, and grading, which apply broadly to many or all central nervous
system tumor types, as well as the addition, elimination, and renaming of multiple specific tumor types. Radiologists are integral
in interpreting brain tumor imaging studies and have a considerable impact on patient care. Thus, radiologists must be aware of
pertinent changes in the field. Staying updated with the most current guidelines allows radiologists to be informed and effective at
multidisciplinary tumor boards and in interactions with colleagues in neuro-oncology, neurosurgery, radiation oncology, and neu-
ropathology. This review represents the first of a two-installment review series on the most recent changes to the WHO brain tumor
classification system. This first installment focuses on the changes to the classification of adult and pediatric gliomas of greatest
relevance for radiologists.
© RSNA, 2022
Online SA-CME • See www.rsna.org/learning-center-ry
Learning Objectives:
After reading the article and taking the test, the reader will be able to:
n Describe the rationale for periodic updates to the World Health Organization (WHO) tumor classification systems
n List key changes to the classification of diffuse gliomas in the fifth edition of the WHO classification of central nervous system tumors, including new tumor types and now-obsolete tumor types
n Explain the increasing importance of molecular markers in the specific diagnosis of diffuse gliomas
Accreditation and Designation Statement
The RSNA is accredited by the Accreditation Council for Continuing Medical Education (ACCME) to provide continuing medical education for physicians. The RSNA designates this journal-based SA-CME activity for a maximum of 1.0 AMA
PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Disclosure Statement
The ACCME requires that the RSNA, as an accredited provider of CME, obtain signed disclosure statements from the authors, editors, and reviewers for this activity. For this journal-based CME activity, author disclosures are listed at the end
of this article.
T he fifth edition of the World Health Organization
(WHO) classification of tumors of the central nervous
system (CNS), published in November 2021, contains
the types and subtypes reflect definitional truths, akin to a
“periodic table” of tumors, but reality is more complicated.
Tumor classification systems provide a framework to ratio-
many changes relevant to radiologists (1). Because imag- nally subdivide the broad spectrum of tumors into specific
ing and radiologists are integral in distinguishing brain types with similar pathologic characteristics, natural his-
tumor mimics, guiding brain tumor management, and tory, and patient demographics. This subdivision is useful
optimizing treatment planning to improve patient out- in both practice and research, allowing clinicians to offer
comes, awareness of these changes is essential. Knowledge accurate prognostic predictions and tumor-specific treat-
of current WHO classification and awareness of imaging ments, clinical researchers to design trials enriched with the
features in tumor-specific therapeutic responses is crucial patient populations most likely to benefit, and basic science
for differentiating tumor recurrence from treatment-related researchers to focus their investigations on specific tumor
changes (2). Understanding and speaking the common types. The WHO has published a classification system for
language of the 2021 fifth edition is essential for effective CNS tumors for more than 40 years, and this system has
communication with neurologists, oncologists, neurosur- become the international reference standard. The WHO
geons, radiation oncologists, and pathologists and also for also publishes tumor classification systems for many other
optimal patient care. organ systems, and new editions of the classification sys-
tems are generally released in a fixed order rather than on
Tumor Classification Principles an as-needed basis. As a result, increasing knowledge re-
To understand the rationale for tumor classification changes garding CNS tumors has outpaced the WHO classification
in the 2021 fifth edition relative to previous editions, it is publication schedule in recent years. In recognition of this
useful to consider why tumor classification systems exist. situation, the Consortium to Inform Molecular and Practi-
Some people conceive of tumor classifications as though cal Approaches to CNS Tumor Taxonomy—Not Official
This copy is for personal use only. To order printed copies, contact reprints@rsna.org
Johnson et al
insufficient tissue for testing or assay failure, it is characterized
Abbreviations by means of histopathologic findings with the designation “not
CNS = central nervous system, FLAIR = fluid-attenuated inversion otherwise specified,” or NOS. If a tumor can be characterized
recovery, IDH = isocitrate dehydrogenase, MAPK = mitogen-activated
protein kinase, WHO = World Health Organization genetically and results do not fit into any WHO-recognized tu-
mor type, it is again characterized by histopathology with the
Summary designation “not elsewhere classified,” or NEC.
The 2021 fifth edition of the World Health Organization Classification
of Tumors of the Central Nervous System includes many “big picture” Tumor Grading
changes relevant to radiologists relating to tumor diagnosis and grad-
ing, as well as the addition, elimination, and renaming of multiple The grading of CNS tumors has changed substantially in the
tumor types. 2021 fifth edition. A highly visible change is the replacement
of Roman numerals (ie, I, II, III, and IV) by Arabic numerals
Essentials (ie, 1, 2, 3, and 4). This shift both partially harmonizes CNS
N Central nervous system (CNS) tumors are increasingly defined by tumor grading nomenclature with that used for other tumor
molecular markers.
types and theoretically reduces the risk of error when reading
N Glioblastoma, isocitrate dehydrogenase (IDH)-wildtype is now the
only recognized adult-type diffuse glioma without IDH mutation. or transcribing tumor grades. Because grading of CNS tumors
N The term glioblastoma is no longer applied to IDH-mutant continues to differ from that of other organ systems in some
tumors; prior IDH-mutant glioblastomas are now designated as regards, for clarity, the WHO endorses the use of the phrase
astrocytoma, IDH-mutant, CNS World Health Organization “CNS WHO grade” rather than just “WHO grade” or “grade.”
grade 4. A more fundamental change in the 2021 fifth edition is
N New “families” of pediatric-type high-grade and low-grade diffuse the move to assigning grade within rather than across tu-
gliomas contain multiple new tumor types, some with characteristic
imaging features. mor types, a concept first introduced to CNS tumor grading
in the 2016 edition with respect to solitary fibrous tumor/
hemangiopericytoma and now broadly applied in the 2021
WHO, or cIMPACT-NOW, was organized to provide practical update (11). For example, in previous versions of the WHO
recommendations and updates to the neuro-oncology commu- classification system, WHO grade I, II, and III meningiomas
nity in the interval between WHO updates (3–10). were separate tumor types, with WHO grade II meningioma
representing “atypical” meningioma and WHO grade III
“Big Picture” Changes representing “anaplastic” or “malignant” meningioma. In this
system, the word atypical was a formal part of the tumor
Increased Emphasis on Molecular Diagnostics name, not simply a redundant way of stating that a menin-
In the fourth edition update of the WHO classification of tu- gioma was WHO grade II. In the fifth edition, meningioma
mors of the CNS, published in 2016, several brain tumors were is a single tumor type, with possible CNS WHO grades of 1,
defined by using molecular features for the first time (11). For 2, or 3. The same concept is applicable to the diffuse gliomas,
example, oligodendroglioma was redefined as diffuse glioma making terms such as “anaplastic astrocytoma” and “anaplas-
containing both isocitrate dehydrogenase (IDH) mutation and tic oligodendroglioma” obsolete. This change does not imply
1p/19q codeletion, even if classic histologic features were not that multiple grade options exist for all tumor types; some
present. The 2021 fifth edition continues to expand the role tumor types with predictably favorable or aggressive natural
of molecular features in tumor diagnosis, with a much larger histories may only be assigned CNS WHO grades of 1 or
number of tumor types defined by molecular features. In other 4, respectively, and other tumors may be assigned a limited
tumors, molecular features impact grading, even if not part of range of grades.
the definition of the tumor. A partial list of diffuse gliomas An additional change to CNS tumor grading in the 2021
recognized in the fifth edition and associated molecular fea- fifth edition relevant to radiologists is the use of both histologic
tures is shown in Table 1. Several common molecular features and molecular features to assign grades to several tumor types.
seen in glioma and their mechanisms of action are displayed For example, before 2021, the diagnosis of glioblastoma, IDH-
in Table 2. wildtype, required the histologic features of necrosis and/or
Although molecular methods are used more frequently now microvascular proliferation. In the fifth edition, IDH-wildtype
than in the past, they have not displaced traditional techniques diffuse astrocytic tumors harboring certain molecular features
such as histologic analysis and immunohistochemistry, and are diagnosed as glioblastoma, IDH-wildtype even without
many tumor types are adequately classified with these tools, with either of these histologic findings.
molecular testing supportive although not required for tumor As originally conceived, tumor grades were intended to
diagnosis. Histologic and molecular features are integrated into correspond with the natural history of a tumor. However,
a framework of a layered diagnosis. This concept, introduced in advances in therapy combined with changes in tumor clas-
the 2016 edition and carried forward in the 2021 fifth edition, sification have weakened this relationship. Although some
is where tumors are characterized based on both conventional tumor grades were changed for 2021, such as the assignment
histopathologic characteristics and molecular markers, with of myxopapillary ependymoma as CNS WHO grade 2 rather
the final diagnosis representing a synthesis of data. If a tumor than the previous WHO grade of I, the overall system was not
cannot be fully characterized due to technical reasons such as fundamentally re-evaluated.
Radiology: Volume 304: Number 3—September 2022 n radiology.rsna.org 495
Radiologist’s Guide to the 2021 WHO Central Nervous System Tumor Classification
Table 1: Diffuse Gliomas and Their Associated Molecular Alterations
Diagnostic Criteria
Tumor Family and
Tumor Type Required Desirable
Adult-type
diffuse gliomas
Astrocytoma, Diffusely infiltrating glioma AND TP53 mutation or strong p53 nuclear expression
IDH-mutant IDH mutation* AND (.10%)
Loss of nuclear ATRX expression or ATRX mutation DNA methylation profile of astrocytoma, IDH-
OR mutant
NO 1p/19q codeletion Astrocytic differentiation by morphology
CDKN2A/B homozygous deletion†
Oligodendroglioma, IDH- Diffusely infiltrating glioma AND TERT promoter mutation
mutant and IDH mutation* AND Retained nuclear ATRX expression
1p/19q-codeleted 1p/19q codeletion Methylation profile of oligodendroglioma IDH-
CDKN2A/B homozygous deletion‡ mutant and 1p/19q codeleted
Glioblastoma, Diffuse astrocytic glioma IDH-wildtype, H3-wildtype AND one or DNA methylation profile of glioblastoma, IDH-
IDH-wildtype more among the following: wildtype
Microvascular proliferation
Necrosis
TERT promoter mutation†
EGFR amplification†
17/210 chromosomal copy number changes†
Pediatric-type diffuse
low-grade gliomas
Diffuse astrocytoma, Diffuse astrocytoma without histologic features of anaplasia AND Absence of OLIG2 and MAP2 expression
MYB- or MYBL1- No mutations in IDH or H3 gene AND
altered Structural variant of MYB or MYBL OR
Diagnostic methylation profile
Angiocentric glioma Glioma with diffuse architecture and a focal angiocentric pattern AND Lack of anaplastic features
Monomorphic spindle cells with immunophenotype and/or ultrastructure of MYB rearrangement (MYB::QKI most common)
astrocytic and ependymal differentiation DNA, ethylation profile aligned with diffuse glioma,
MYB- or MYBL1-altered
Polymorphous low-grade Diffuse pattern of growth (at least regionally) AND Conspicuous calcifications
neuroepithelial Oligodendroglioma-like component (even minor) AND Absence of 1p/19q codeletion
tumor of the young Few (if any) mitoses AND
CD34 expression by tumor cells and ramified neural cells AND
IDH-wildtype status AND
Unequivocal BRAF p.V600E expression OR
BRAF p.V600E, FGFR2, FGFR3, or other MAPK alteration
Diffuse low-grade Diffuse glioma with absent or minimal mitoses, no microvascular proliferation Onset in children, adolescent or young adults
glioma, MAPK or necrosis AND Absence of morphologic features or DNA
pathway–altered MAPK pathway alteration AND methylation profile suggestive of an alternative
NO IDH or H3 mutation AND tumor in which FGFR or BRAF abnormalities
NO CDKN2A homozygous deletion occur
Pediatric-type diffuse
high-grade gliomas
Diffuse midline Diffuse glioma AND Results from molecular analyses that enable
glioma, H3 Loss of H3 p.K28me3 (K27me3) expression AND discrimination of the H3.1 or H3.2 p.K28 (K27)-
K27-altered Midline location AND mutant subtype
H3 p.K28M (K27M) or p.K28I (K27I) mutation, OR
EGFR alteration, OR
EZHIP overexpression, OR
Diagnostic methylation profile
Diffuse hemispheric Cellular infiltrative glioma with mitotic activity AND OLIG2 immunonegative
glioma, H3 H3.3 p.G34R or p.G34 V mutation AND Loss of ATRX expression
G34-mutant Hemispheric location AND (for unresolved lesions) Diffuse p53 immunopositive
Methylation profile of diffuse hemispheric glioma, H3 G34-mutant
Table 1 (continues)
496 radiology.rsna.org n Radiology: Volume 304: Number 3—September 2022
Johnson et al
Table 1 (continued): Diffuse Gliomas and Their Associated Molecular Alterations
Diagnostic Criteria
Tumor Family and
Tumor Type Required Desirable
Diffuse pediatric- Diffuse glioma with mitoses occurring in a child or young adult Microvascular proliferation
type high-grade AND Necrosis, typically palisading H3 p.K28me3
glioma, H3-wildtype NO IDH or Histone H3 mutation AND (K27me3) retained
and IDH-wildtype Methylation profile aligned with pHGG RTK1, pHGG RTK2, or pHGG MYC
OR
Key molecular features PDGFRA alteration, EGFR alteration or MYCN
amplification
Infant-type Cellular astrocytoma AND …
hemispheric glioma Presentation in early childhood AND
Cerebral hemisphere location AND
Receptor tyrosine kinase abnormality (NTRK family, ROS1, MET1, or ALK)
OR
Methylation profile aligned with infant-type hemispheric glioma
Note.—IDH = isocitrate dehydrogenase, MAPK = mitogen-activated protein kinase.
* Canonical (IDH1 p.R132H) or noncanonical (other IDH1 p.R1372).
†
Sufficient for a central nervous system (CNS) World Health Organization (WHO) grade 4 designation in the absence of grade 4 histologic
finding (microvascular proliferation and necrosis).
‡
CDKN2A/B homozygous deletion indicates a CNS WHO grade 3 oligodendroglioma, IDH-mutant and 1p/19q codeleted.
Table 2: Mechanisms of Molecular Alterations in Diffuse Gliomas
Molecular Alteration Function
IDH1 p.R132 or IDH2 IDH mutations generate the oncometabolite 2-hydroxyglutarate, resulting in DNA hypermethylation
p.R172 mutation (G-CIMP phenotype)
ATRX mutation Activates alternative lengthening of telomeres (ALT) pathway to maintain telomere length
TERT promoter mutation Activates telomerase to maintain telomere length
CDKN2A/B homozygous deletion Results in loss of tumor suppressor and cell cycle regulator p16
EGFR amplification Activates receptor tyrosine kinase pathway
H3 K27M mutation Histone H3 mutations result in widespread loss of H3 K27-trimethylation and aberrant gene transcription
H3 G34R or G34 V mutation Histone H3 mutation alters H3 K36 methylation and results in aberrant gene transcription
Note.—IDH = isocitrate dehydrogenase, MAPK = mitogen-activated protein kinase.
Table 3: WHO Classification of Tumors of the CNS, Fifth Edition
CNS Tumor Chapter Tumor Families within Chapter
Gliomas, glioneuronal tumors, Adult-type diffuse gliomas, pediatric-type diffuse low-grade gliomas, pediatric-type diffuse
and neuronal tumors high-grade gliomas, circumscribed astrocytic gliomas, glioneuronal and neuronal tumors,
ependymal tumors
Choroid plexus tumors …
Embryonal tumors Medulloblastoma, molecularly and histologically defined; other CNS embryonal tumors
Pineal tumors …
Cranial and paraspinal nerve tumors …
Meningiomas …
Mesenchymal, nonmeningothelial tumors Soft-tissue tumors, chondro-osseous tumors, notochordal tumors
Melanocytic tumors …
Hematolymphoid tumors Lymphomas, histiocytic tumors
Germ cell tumors …
Tumors of the sellar region …
Metastases to the CNS …
Note.—The spectrum of nervous system tumors is subdivided by chapter and family where applicable. CNS = central nervous system,
WHO = World Health Organization.
Radiology: Volume 304: Number 3—September 2022 n radiology.rsna.org 497
Radiologist’s Guide to the 2021 WHO Central Nervous System Tumor Classification
Figure 1: Images in a 63-year-old man with a left frontal glioblastoma, IDH-wildtype, with necrosis and microvascular proliferation documented
at histologic examination. (A) T1-weighted gadolinium-enhanced axial MRI scan demonstrates classic ring enhancement (arrows). (B) T2-weighted
fluid-attenuated inversion recovery axial MRI scan shows surrounding vasogenic edema (arrows). (C) Diffusion-weighted axial MRI scan displays
restriction in the peripheral region of enhancement (arrowheads).
Molecular Diagnostic Methods ependymoma, and atypical teratoid rhabdoid tumor. However,
Multiple methods are available to detect molecular alterations methylation profiling is currently available at only a limited
in CNS tumors, and the WHO does not endorse any specific number of institutions and, while helpful in some clinical sce-
techniques or technology. Several common molecular altera- narios, such as those described earlier, it may provide mislead-
tions are assessed directly by means of immunohistochemistry ing or noncontributory profiles in others and a subset of tumors
using mutation-specific antibodies, including IDH1 R132H, cannot be definitively classified by using methylation profiling
BRAF V600E, and H3 K27M, and additional alterations are (ranging from 12% to 44% across studies) (14–16). As with all
inferred by loss of normal immunohistochemistry expression the molecular techniques, methylation profiling is most power-
patterns, including ATRX, BAP1, and INI-1 (SMARCB1). In ful when integrated with histologic and other molecular infor-
patients older than 54 years with a histologic diagnosis of glio- mation; it should not be viewed as a replacement for existing
blastoma, a negative IDH1 R132H immunostain is sufficient for classification schemes.
an IDH-wildtype designation (12), while in younger patients or
lower-grade tumors, assessment for noncanonical IDH1/IDH2 Tumor “Families”
(ie, non-IDH1 R132H) mutations with further molecular anal- The 12 chapters of the 2021 fifth edition cover the full spec-
ysis is mandatory. trum of CNS neoplasms, including metastatic disease. Each
Multiple techniques are available to assess copy number chapter encompasses multiple tumor types, some grouped
alterations, such as EGFR amplification, CDKN2A/B homo- into tumor “families.” Chapters and main tumor families are
zygous deletion, and 1p/19q codeletion. Fluorescence in situ shown in Table 3. The chapter on gliomas, glioneuronal tu-
hybridization remains a commonly used technique, with the mors, and neuronal tumors collectively accounts for a major-
advantage of being relatively inexpensive with faster turn- ity of primary intra-axial CNS tumors and is divided into
around times, although false-positive and false-negative find- six different tumor families, recognizing major differences in
ings may occur (13). Genome-wide copy number arrays are biology, growth patterns, and typical patient demographics.
also widely used to detect copy number changes and are more Importantly, although some tumor family names contain ref-
specific in the detection of whole chromosome and/or chromo- erences to patient demographics, exceptions do occur, with
somal arm gains or losses. Many next-generation sequencing children developing adult-type diffuse gliomas and adults de-
panels are validated to detect copy number alterations in addi- veloping pediatric-type tumors. As such, it is important for
tion to sequence alterations. Copy number can also be derived radiologists to be familiar with all the diffuse gliomas, regard-
from methylation array, an emerging technology newly incor- less of whether they primarily interpret images from adults
porated into the 2021 fifth edition. or children. Clinically, a distinction into three age groups
DNA methylation profiling has emerged as a powerful new instead of two groups including children (,15 years old),
technique in the identification of new entities and molecular adolescent and young adults (15–39 years old), and adults
subgroups and the classification of CNS tumors (14). A meth- (40 years and older) better reflects tumor epidemiology.
ylation profile is generated from tumor DNA, and a machine
learning algorithm matches the profile to a reference set to gen- Adult-type Diffuse Gliomas
erate a calibrated score, with a score of 0.84 or higher or 0.9
or higher commonly used as a cutoff to define a “match” to a Glioblastoma, IDH-wildtype
particular entity. Methylation profiling is particularly useful for Historically, glioblastomas have been defined as diffuse astro-
pediatric and embryonal tumors, including medulloblastoma, cytic tumors demonstrating mitotic activity as well as micro-
498 radiology.rsna.org n Radiology: Volume 304: Number 3—September 2022
Johnson et al
edition, glioblastoma was formally subdivided on
the presence or absence of IDH gene mutations
(either IDH1 or IDH2), given the recognition
that IDH-mutant and IDH-wildtype glioblasto-
mas were fundamentally different with distinct
tumorigenesis, natural history, and treatment re-
sponse. In the 2021 fifth edition, the definition
of glioblastoma is further refined to include only
IDH-wildtype tumors. Tumors previously diag-
nosed as glioblastoma, IDH-mutant, under the
2016 edition are now reclassified as astrocytoma,
IDH-mutant, CNS WHO grade 4. In addition,
the designation of glioblastoma, IDH-wildtype,
is applied to IDH-wildtype astrocytic tumors
harboring any of the following alterations: TERT
promoter mutation, EGFR amplification, and
17/210 chromosomal copy number changes. Of
note, as in prior versions of the WHO tumor clas-
sification system, gliosarcoma is considered a vari-
ant of glioblastoma, IDH-wildtype, rather than an
independent tumor type.
Radiologists are impacted by these changes in
several ways. First, there will be a period of transi-
tion where follow-up imaging reports on many pa-
tients require revision to reflect changes in tumor
type nomenclature. The 2021 fifth edition desig-
nation of some tumors will be readily apparent.
For example, a tumor previously diagnosed as glio-
blastoma, IDH-mutant, WHO grade IV, is reason-
ably assumed to represent an astrocytoma, IDH-
mutant, CNS WHO grade 4. However, it may be
unclear whether a previously diagnosed anaplastic
astrocytoma, IDH-wildtype, WHO grade III,
meets molecular criteria for glioblastoma, IDH-
wildtype, CNS WHO grade 4, without review-
ing the case with pathologic examination or ad-
ditional testing. When interpreting imaging of an
unknown tumor type, it is important to remember
that while the absence of gadolinium enhancement
on MRI scans has never excluded the possibility
of high-grade glioma, molecular glioblastomas are
commonly nonenhancing. Figures 1 and 2 demon-
Figure 2: Images in three different patients with of glioblastoma, IDH-wildtype, with diagnosis strate the differing typical imaging appearances of
confirmed using molecular criteria. A, C, and E are T2-weighted fluid-attenuated inversion re-
covery (FLAIR) MRI scans, and B, D, and F are T1-weighted gadolinium-enhanced MRI scans.
glioblastoma, IDH-wildtype, based on histologic
(A, B) Images in a 55-year-old man with an astrocytic glioma, IDH-wildtype, histopathologically and molecular criteria, respectively.
consistent with anaplastic astrocytoma, harboring TERT c.-124C . T mutation. (C, D) Images in a
68-year-old man with an IDH-wild-type glioma also histopathologically consistent with anaplastic Astrocytoma, IDH-mutant
astrocytoma, harboring TERT c.-146C . T. (E, F) Images in a 39-year-old man with an astrocytic In the 2021 fifth edition, astrocytoma, IDH-mu-
glioma, IDH-wildtype, histopathologically “low grade,” that, in addition to TERT c.-124C . T (also
known as C228T) mutation, shows presence of EGFR amplification. The FLAIR scans demonstrate
tant, is defined as an adult-type diffuse glioma har-
varying degrees of circumscription (arrows). The T1-weighted gadolinium-enhanced scans show boring IDH1 or IDH2 mutation without 1p/19q
lack of contrast enhancement. codeletion, with possible CNS WHO grades of
2–4. Most astrocytoma, IDH-mutant, also har-
bor ATRX and TP53 gene mutations. As in pre-
vascular proliferation and/or necrosis. Tumors were clinically vious editions, grading is primarily based on histologic find-
subdivided into primary and secondary glioblastoma based on ings; however, the fifth edition also incorporates CDKN2A/B
whether they were thought to have arisen de novo as glioblas- homozygous deletion as a marker of poor prognosis sufficient
toma or progressed from lower-grade glial tumors. In the 2016 to indicate CNS WHO grade 4 even without microvascular
Radiology: Volume 304: Number 3—September 2022 n radiology.rsna.org 499
Radiologist’s Guide to the 2021 WHO Central Nervous System Tumor Classification
Figure 3: Images in a 32-year-old man with astrocytoma, IDH-mutant, central nervous system World Health Organization grade 2, harboring a
canonical IDH1 (p.R132H) mutation as well as TP53 and ATRX mutations. No CDKN2A/B homozygous deletion was detected. (A) T2-weighted
MRI and (B) T2-weighted fluid-attenuated inversion recovery (FLAIR) axial MRI scans demonstrate the presence of the T2-FLAIR mismatch sign (ar-
rows), which is highly specific for astrocytoma, IDH-mutant. (C) T1-weighted gadolinium-enhanced axial MRI scan shows signal hypointensity (ar-
rows) and lack of enhancement, which is typical in tumors that demonstrate true T2-FLAIR mismatch.
Figure 4: Images in two different patients with oligodendroglioma, IDH-mutant and 1p/19q codeleted; both had canonical IDH1 mutation
at immunohistochemistry and sequencing as well as 1p/19q codeletion at fluorescence in situ hybridization analysis. (A) Unenhanced axial
head CT scan in a 41-year-old man with a central nervous system (CNS) World Health Organization (WHO) grade 2 tumor shows prominent
calcification (arrow). (B) T2-weighted fluid-attenuated inversion recovery (FLAIR) axial MRI scan demonstrates poorly circumscribed margins (ar-
rows), and (C) T1-weighted gadolinium-enhanced axial MRI scan shows a small region of enhancement (arrowhead). (D) Unenhanced axial
head CT scan in a 42-year-old man with a bifrontal CNS WHO grade 3 tumor shows prominent calcification (arrow). (E) T2-weighted FLAIR
axial MRI scan demonstrates a hyperintense internal cystic portion (arrow), and (F) T1-weighted gadolinium-enhanced axial MRI scan shows
avid enhancement (arrowheads).
500 radiology.rsna.org n Radiology: Volume 304: Number 3—September 2022
Johnson et al
proliferation or necrosis. Consequently, IDH-mu-
tant astrocytomas previously diagnosed as WHO
grade IV should now all represent CNS WHO
grade 4 tumors, and those previously assigned
WHO grades II or III should not be assumed to
correspond to CNS WHO grade 2 and 3 tumors
unless CDKN2A/B deletion status is known.
CDKN2A/B homozygous deletion results in disrup-
tion in the production of multiple tumor suppres-
sor proteins and is seen in a wide variety of tumors.
Acquired CDKN2A deletions have been reported
to occur in IDH-mutant gliomas following radia-
tion and are associated with poor outcomes (17).
Of note, alternations in the CDK pathway may be Figure 5: Images in a 2-year-old girl with diffuse midline glioma, H3 K27-altered. (A) T2-
therapeutically targetable, and preclinical work in weighted fluid-attenuated inversion recovery (FLAIR) axial MRI scan displays an expansile, homo-
this area is ongoing (18). geneous, nonenhancing lesion centered in the pons engulfing the basilar artery flow void (arrow),
a classic appearance of diffuse intrinsic pontine glioma. (B) T1-weighted gadolinium-enhanced
The imaging characteristics of astrocytoma,
sagittal MRI scan shows lack of enhancement and effacement of the lower fourth ventricle (arrow)
IDH-mutant, are variable and influenced by tu- resulting in obstructive hydrocephalus and cerebellar tonsillar herniation.
mor grade. A recent systematic review and meta-
analysis on the radiologic differences between
subtypes of grade II and III gliomas, as defined
in the 2016 edition, suggested that astrocytoma,
IDH-mutant, is more often well-circumscribed
than either astrocytoma, IDH-wildtype, or oligo-
dendroglioma and displays less frequent contrast
enhancement when considering grade II and III
tumors under the 2016 WHO classification sys-
tem (19). The “T2-FLAIR” mismatch sign occurs
in well-circumscribed tumors that show homoge-
neous hyperintensity on T2-weighted images with
central signal drop-out and a thin rim of residual
hyperintensity on T2-weighted fluid-attenuated in-
version recovery (FLAIR) images. The appearance
of mismatch has been suggested to be caused by the
extremely long MRI T1 and T2 relaxation times Figure 6: Images in a 38-year-old man with diffuse midline glioma, H3 K27-altered. (A) T2-
of IDH-mutant gliomas relative to IDH-wildtype weighted coronal MRI scan shows a markedly heterogeneous mass involving the left thalamus and
tumors (20). This sign is a highly specific and mod- temporal lobe (arrows). (B) T1-weighted gadolinium-enhanced coronal MRI scan demonstrates
erately sensitive imaging marker of astrocytoma, peripheral enhancement (arrows).
IDH-mutant, although false-positive findings can
occur, particularly in children (21–23). An ex-
ample of the T2-FLAIR mismatch sign is shown in Figure 3. necessary or sufficient for diagnosis. These tumors may be as-
Tumor enhancement is frequently seen in WHO grade 3 and signed CNS WHO grades of 2 or 3, and in the latter case the
4 tumors. However, the imaging features of tumors designated previous term “anaplastic oligodendroglioma” is no longer for-
as astrocytoma, IDH-mutant, CNS WHO grade 4 based on mally applied. Grading remains primarily based on histologic
CDKN2A/B homozygous deletion rather than microvascular criteria: mitotic activity, microvascular proliferation, and necro-
proliferation or necrosis may be visibly indistinguishable from sis. As oligodendrogliomas have relatively favorable biologic be-
lower-grade tumors, analogous to the situation in glioblastoma, havior and response to treatment, they cannot be CNS WHO
IDH-wildtype diagnosed with molecular criteria. grade 4. Similar to IDH-mutant astrocytomas, CDKN2A/B
homozygous deletion may be used as a molecular marker of
Oligodendroglioma, IDH-mutant and 1p/19q-codeleted CNS WHO grade 3, particularly in those IDH-mutant and
After substantial redefinition in the 2016 edition, the diagnosis 1p/19q-codeleted oligodendrogliomas that are difficult to assign
of oligodendroglioma, IDH-mutant and 1p/19q-codeleted, is to either CNS WHO grade 2 or CNS WHO grade 3 purely
not markedly changed in the 2021 fifth edition. As the name based on histologic findings. Among CNS WHO grade 3 oligo-
states, IDH mutation and 1p/19q codeletion are required for dendrogliomas, homozygous deletion involving the CDKN2A
the diagnosis of oligodendrogliomas. Historical pathologic fea- and/or CDKN2B locus is present in a small subset of tumors
tures such as “perinuclear halos” creating “fried egg cells” and (,10%) and is associated with decreased survival, independent
“chicken wire vasculature” remain characteristic although not of microvascular proliferation with or without necrosis (24).
Radiology: Volume 304: Number 3—September 2022 n radiology.rsna.org 501
Radiologist’s Guide to the 2021 WHO Central Nervous System Tumor Classification
Figure 7: Images in a 20-year-old woman with diffuse hemispheric glioma, H3 G34-mutant harboring H3–3A p.G35 (G34) gene mutation in
the context of ATRX mutation and homozygous and/or biallelic deletion of the TP53 gene. (A) T2-weighted fast spin-echo coronal MRI scan shows
a well-delineated expansile mass situated peripherally in the posterosuperior left cerebral hemisphere with broad-based leptomeningeal contact
(arrows). (B) T1-weighted gadolinium-enhanced coronal MRI scan demonstrates a focal area of contrast enhancement (arrow), while (C) diffusion-
weighted axial MRI scan shows tumor hyperintensity due to diffusion restriction (arrowhead).
Characteristic imaging findings of oligodendro-
glioma, IDH-mutant and 1p/19q-codeleted, include
internal heterogeneity, poorly circumscribed margins
on T1- and T2-weighted images, internal cysts, and
calcification. In the previously mentioned systematic
review and meta-analysis of grade II and III gliomas,
as defined under the previous 2016 WHO classifica-
tion system, it was reported that heterogeneity was
96% sensitive for oligodendroglioma, while calcifica-
tion was 88% specific in patients with glioma (19).
Of note, enhancement occurs in roughly half of oli-
Figure 8: Images in a 3-year-old girl with diffuse pediatric-type high-grade glioma, H3-wildtype
godendrogliomas and is frequently partial; ring-en-
and IDH-wildtype, in the posterior left temporal lobe. The tumor showed presence of MYCN amplifi-
hancing or predominantly enhancing oligodendro- cation and could be further subclassified as diffuse pediatric-type high-grade glioma MYCN subtype.
gliomas are not typical (19,25). The MRI features of (A) T2-weighted axial MRI scan shows a well-circumscribed solid and cystic mass (arrows). (B) T1-
oligodendroglioma are shown in Figure 4. weighted gadolinium-enhanced axial MRI scan demonstrates thin peripheral enhancement (arrows).
Astrocytoma, IDH-wildtype
In the 2021 fifth edition, astrocytoma, IDH-wildtype, is not a this family, it is used to describe the nature of tumor growth at
recognized tumor family. The only IDH-wildtype adult-type dif- pathologic examination. These tumors may sometimes appear
fuse glioma is glioblastoma, IDH-wildtype. Any tumor previously well-circumscribed at imaging.
diagnosed as diffuse astrocytoma, IDH-wildtype, WHO grade II,
or anaplastic astrocytoma, IDH-wildtype, WHO grade III, will Diffuse Midline Glioma, H3 K27-altered
require reclassification. Many of these tumors will meet the crite- In the 2016 edition of the WHO classification, diffuse midline
ria for glioblastoma, IDH-wildtype, based on molecular features glioma, H3 K27M-mutant, was introduced as an infiltrative mid-
(pTERT mutation, 17/−10, or EGFR amplification). Others will line tumor harboring a K27M mutation in either H3.1 or H3.2.
have molecular features that will allow for an alternative diagnosis Since that time, additional tumor-defining molecular features have
such as a tumor in the “pediatric-type diffuse low-grade glioma” been described and the name of the tumor slightly modified (26).
or “pediatric-type diffuse high-grade glioma” families. Still, other EGFR-altered tumors arising in children in the midline structures
tumors will be given descriptive histologic diagnoses and qualified may also fall within this group. A uniform histopathologic feature
as “not otherwise specified” or “not elsewhere classified” based on regardless of the underlying driver alteration is the loss of expres-
the molecular evaluation. sion of H3 K27me3 at immunohistochemistry. The presence of
this mutation confers a poor prognosis (27) and designates this as
Pediatric-type Diffuse High-Grade Gliomas a CNS WHO grade 4 tumor. Although typically arising from the
The tumor family of pediatric type diffuse high-grade gliomas is brainstem, thalamus, or spinal cord, rare off-midline tumors are
new for the 2021 fifth edition. This group of tumors comprises reported. The imaging appearance of diffuse midline glioma, H3
the vast majority of high-grade gliomas in children. Of note, al- K27-altered, is widely variable, ranging from nonenhancing and
though the term “diffuse” appears both in the name of the tumor infiltrative to enhancing and necrotic tumors (28,29), as shown in
family and in the name of several specific tumor types within Figures 5 and 6. In a large registry study on diffuse intrinsic pon-
502 radiology.rsna.org n Radiology: Volume 304: Number 3—September 2022
Johnson et al
Figure 9: Images in a 5-month-old boy with infant-type hemispheric glioma. This tumor shows ALK overexpression and harbors a fusion between the
DYNC1I2 gene and ALK gene, consistent with infant-type hemispheric glioma, ALK-rearranged subtype. (A) Unenhanced axial head CT scan demon-
strates a heterogeneous right frontal tumor with a hyperattenuating solid component, multiple cystic spaces, and a small region of dense calcification (ar-
row). (B) T2-weighted fluid-attenuated inversion recovery (FLAIR) axial MRI scan shows hyperintense periphery of tumor cysts (arrows) which have pro-
teinaceous contents. Marked enlargement of the ventricular system (arrowheads) without periventricular edema is greater than expected for tumor loca-
tion, possibly reflecting longstanding communicating hydrocephalus from tumor seeding and/or underlying parenchymal volume loss. (C) T1-weighted
gadolinium-enhanced sagittal MRI scan demonstrates further the extent of complex heterogenous tumor. Arrowhead indicates ventricular enlargement.
Figure 10: Images in a 6-year-old boy with angiocentric glioma. (A) T2-weighted axial MRI scan shows a predominately hyperintense lesion
(arrows) involving cortex and subcortical white matter in the right middle and superior temporal gyri. (B) T1-weighted axial MRI scan obtained
before administration of a gadolinium-based contrast agent demonstrates intrinsic T1 hyperintensity (arrows), while (C) gadolinium-enhanced
T1-weighted axial MRI scan shows no superimposed enhancement.
tine gliomas, the only imaging feature associated with histone sta- and/or microcalcification. However, some tumors were diffusely
tus was an ill-defined infiltrating signal in the pontine fibers (29). infiltrative or showed both a well-defined mass and prominent ad-
jacent infiltrative signal. Leptomeningeal contact was noted in all
Diffuse Hemispheric Glioma, H3 G34-mutant cases. Enhancement of these tumors was variable, ranging from
Diffuse hemispheric glioma, H3 G34-mutant, is a new tumor intense to absent. The study by Kurokawa et al (33) evaluated
type described in the 2021 fifth edition. This aggressive infiltrat- 59 pediatric and adult cases; they described contact of leptomen-
ing tumor generally occurs in the supratentorial brain of older inges and/or ependymal margins in a large majority. Picart and
children and young adults and is always designated as CNS colleagues (30) limited their study to adult patients and reported
WHO grade 4, although the prognosis is variable. The pathog- most tumors were infiltrative in nature with faint or no contrast
nomonic molecular feature is a missense mutation at position 34 enhancement, although most harbored regions of diffusion re-
of the histone H3.3 protein, and IDH mutation is never present. striction. Figure 7 shows an example of this tumor entity.
Several studies have examined the radiologic features of diffuse
hemispheric glioma, H3 G34-mutant (30–32). As stated in the Diffuse Pediatric-type High-Grade Glioma, H3-wildtype and
name, tumors are hemispheric (supratentorial nonmidline) in ori- IDH-wildtype
gin, although midline extension is sometimes seen. The study by Diffuse pediatric-type high-grade glioma, H3-wildtype and
Puntonet et al (31) evaluated both pediatric and adult patients, re- IDH-wildtype, is a newly described entity in the 2021 fifth edi-
porting marked variability in imaging features. Most tumors were tion, representing an aggressive pediatric brain tumor lacking
well-delineated and expansile, with associated regions of necrosis mutations in either IDH or histone 3. Most of these tumors
Radiology: Volume 304: Number 3—September 2022 n radiology.rsna.org 503
Radiologist’s Guide to the 2021 WHO Central Nervous System Tumor Classification
have high-grade histologic features, with either a
glial or at times a primitive, poorly differentiated
appearance in association with microvascular prolif-
eration and/or necrosis. Three molecular subgroups
are identified that differ in important ways, and it
is possible that future editions of the WHO CNS
classification system will formally subdivide this
tumor type further (34). Of note, most pediatric
radiation-induced high-grade gliomas are H3-wild-
type and IDH-wildtype (35). Radiographically,
diffuse pediatric-type high-grade glioma, H3-
wildtype and IDH-wildtype, often resembles adult
glioblastoma, as displayed in Figure 8 (36).
Infant-type Hemispheric Glioma
Infant-type hemispheric glioma is a malignant
glioma, often presenting in the 1st year of life as a
large supratentorial mass. A definitive CNS WHO
grade has not yet been assigned. Characteristic re-
ceptor tyrosine kinase fusions involve the NTRK
family, ALK, ROS, or MET (37). Relatively little
has been published concerning imaging findings;
however, histopathologically the tumors can dem-
onstrate cystic and solid portions, necrosis, and
hemorrhage, and the images should reflect this, as Figure 11: Images in a boy with diffuse astrocytoma, MYB-altered. (A) T2-weighted axial MRI
demonstrated in Figure 9. Leptomeningeal dissemi- scan and (B) T1-weighted gadolinium-enhanced sagittal MRI scan obtained when the patient was
nation has been reported, and as such spinal imag- 2 years old demonstrate an expansile, T2-hyperintense nonenhancing mass centered in the superior
ing should be considered (38). right parietal lobe with well-circumscribed margins (arrows). Initial pathologic diagnosis following
biopsy was anaplastic astrocytoma, fibrillary type (World Health Organization [WHO] grade III) in
accordance with the 2007 WHO classification of central nervous system tumors based exclusively
Pediatric-type Diffuse Low-Grade on histopathologic diagnostic criteria. The patient received antiepileptic medication for associated
Gliomas seizure, although no chemotherapies or radiation therapy was given, and he remained clinically well.
The family of pediatric-type diffuse low-grade glio- (C, D) Follow-up T2-weighted axial MRI scan (C) and T1-weighted gadolinium-enhanced sagittal
mas is a newly recognized group of tumors in the MRI scan (D) in the same patient at 9 years of age demonstrate decreased size and extent of the
tumor (arrows), with a more conspicuous central cystic focus (arrowhead in C). Re-evaluation of the
2021 fifth edition, as are three of the four tumor en- original biopsy in conjunction with molecular analysis, including next-generation sequencing and
tities that comprise this group. While these tumors whole genome methylation profiling, disclosed the tumor to harbor a MYB-PCDHGA1 fusion and a
vary in molecular characteristics and imaging, they genome whole methylation profile matching “low-grade glioma, MYB/MYBL1” class.
share the common features of being CNS WHO
grade 1 tumors and frequently present clinically as
epileptogenic lesions. and may be surgically curable (43). Imaging typically demon-
strates a supratentorial T2-hyperintense, T1-hypointense mass
Angiocentric Glioma without enhancement. Margins are often well-circumscribed,
Angiocentric glioma is a rare epilepsy-associated neoplasm oc- and cysts can be seen (44). Figure 11 shows an example of typi-
curring primarily in children and young adults and has been cal imaging findings.
recognized by the WHO since 2007. Molecularly, angiocentric
glioma is characterized by MYB gene alterations. This CNS Polymorphous Low-Grade Neuroepithelial Tumor of the Young
WHO grade 1 tumor is typically peripherally located in the Polymorphous low-grade neuroepithelial tumor of the young,
supratentorial brain with T2 prolongation, facilitated diffu- or PLNTY, is a new tumor type in the 2021 fifth edition, origi-
sion, and lack of enhancement on MRI scans (Fig 10) (39,40). nally described in 2017 (45). This CNS WHO grade 1 neo-
Additional imaging features include ribbon-like intrinsic T1 plasm is molecularly characterized by aberrant CD34 expression
hyperintensity and T2-hyperintense stalk-like extension to the and a distinct DNA methylation signature. Frequent mitogen-
underlying ventricle (41). Gross total resection typically results activated protein kinase (MAPK) pathway activation includes
in freedom from seizures (42). FGFR genes with a unique FGFR2-CTNNA3 fusion (46). These
tumors most frequently occur in the superficial cerebral hemi-
Diffuse Astrocytoma, MYB- or MYBL1-altered spheres, with a predilection for the temporal lobes. They tend
Diffuse astrocytoma, MYB- or MYBL1-altered, is a rare CNS to be well-circumscribed with heterogeneous signal, peripheral
WHO grade 1 tumor newly recognized in the WHO fifth edi- cysts, and frequent central coarse calcification, which is the dom-
tion. This highly differentiated glioma often manifests as seizures inant imaging feature of this tumor (47), as seen in Figure 12.
504 radiology.rsna.org n Radiology: Volume 304: Number 3—September 2022
Johnson et al
Figure 12: Images in a 20-year-old woman with polymorphous low-grade neuroepithelial tumor of the young, or PLNTY, harboring BRAF
p.V600E mutation identified with immunohistochemistry. (A) T2-weighted axial MRI scan shows a well-circumscribed right temporal mass with hyper-
intense cystic components (arrow), while (B) susceptibility-weighted axial MRI scan demonstrates a focal central T2-hypointensity corresponding to
dense calcification (arrow). (C) T1-weighted gadolinium-enhanced axial MRI scan displays a small amount of enhancement along the tumor margin
(arrowhead). Essential diagnostic criteria of this tumor include histopathologic features such as diffuse growth pattern (at least in part), oligodendrog-
lial-like component, low (if any) mitotic activity, and CD34 expression in tumor cells, in conjunction with immunohistochemical or molecular evidence
of BRAF p.V600E mutations, FGFR2 or FGFR3 fusions, or potentially other mitogen-activated protein kinase pathway–driving genetic abnormalities.
Figure 13: Images in a 14-month-old girl with diffuse low-grade glioma, mitogen-activated protein kinase pathway-altered. This diffuse glioma
harbors BRAFV600E mutation (evaluated with immunohistochemistry) and lacks CDKN2A/B homozygous deletion. The precise subtype is diffuse low-
grade glioma, BRAF p.V600E–mutant. A central nervous system (CNS) World Health Organization (WHO) grade is not assigned to this tumor at pres-
ent, but this tumor type seems to have a more favorable prognosis than the adult-type CNS WHO grade 2 IDH-mutant diffuse gliomas. (A) T2-weighted
coronal MRI scan shows a predominantly hyperintense mass in the medial right temporal lobe with poorly defined margins and a cystic component (ar-
row). (B) T1-weighted gadolinium-enhanced coronal MRI scan demonstrates a focal region of solid enhancement within the mass (arrow).
Diffuse Low-Grade Glioma, MAPK Pathway–altered Tumor Classification in the Reading Room
Diffuse low-grade glioma, MAPK pathway–altered, is a newly rec- As discussed, many CNS tumors demonstrate characteristic
ognized tumor in the 2021 fifth edition, characterized by MAPK imaging findings, and some findings specifically reflect molecu-
pathway alteration. These rare tumors may exhibit astrocytic lar tumor diagnoses, such as the T2-FLAIR mismatch sign in
or oligodendroglial morphologic characteristics and can occur astrocytoma, IDH-mutant (21). Table 4 summarizes the com-
throughout the craniospinal axis, although associations between mon imaging features of several adult-type and pediatric-type
morphologic characteristics, location, and molecular features are diffuse gliomas. Despite the frequent ability of imaging to help
still being defined and it is possible that this tumor type will be predict tumor pathologic characteristics, the growing primacy of
subdivided further in the future (48). This heterogeneity is re- molecular characterization in tumor classification makes it clear
flected in the imaging characteristics, described as similar to those that pathologic analysis will remain the standard of care in brain
of pilocytic astrocytoma, although with a more diffuse pattern (1). tumor diagnosis for the foreseeable future and will not soon be
Although heterogeneous enhancement and cystic elements are of- displaced by noninvasive imaging diagnosis or radiogenomic
ten seen, these are not always present, as shown in Figure 13. analysis. This is not to say that the role of radiology in the pre-
Radiology: Volume 304: Number 3—September 2022 n radiology.rsna.org 505
Radiologist’s Guide to the 2021 WHO Central Nervous System Tumor Classification
Table 4: Imaging Features of Common Diffuse Gliomas
Key Imaging
Tumor Type Location Typical Imaging Features References
Adult-type diffuse
glioma family
Astrocytoma, IDH-mutant Cerebral hemispheres CNS WHO grade 2: 19, 21, 57
Frontoinsular predominance (51) Relatively circumscribed and heterogeneous
Enhancement very rare
T2-FLAIR mismatch sign (sensitive, not specific)
CNS WHO grade 3 and 4:
Increased heterogeneity
Enhancement common; various patterns
Local vasogenic edema
May be indistinguishable from GBM, IDH-wt
Oligodendroglioma, Cerebral hemispheres CNS WHO grade 2: 19, 57, 58
IDH-mutant and Frontal lobe predominance (51) Poorly circumscribed
1p/19q-codeleted Often peripheral and/or cortically Heterogeneous appearance
based Calcification common
Cystic component common
Enhancement can occur CNS
WHO grade 3:
Enhancement more common than CNS WHO
grade 2
Otherwise similar features
Glioblastoma, Cerebral hemispheres Diagnosis with histologic examination: 52–54
IDH-wildtype Enhancement common, “ring” pattern classic
Diffusion restriction
Increased perfusion
Local vasogenic edema
Associated hemorrhage can occur
Diagnosis with molecular features:
Enhancement variable
Similar to low-grade gliomas
Pediatric-type diffuse
low-grade glioma family
Diffuse astrocytoma, Cerebral hemispheres Well circumscribed 43, 44
MYB- or MYBL1-altered Enhancement uncommon
T2 hyperintense
Angiocentric glioma Cerebral hemispheres Well circumscribed 39, 40
Cortical and/or subcortical location T2 hyperintense
Stalk-like extension to ventricle
Facilitated diffusion
Rare enhancement
Polymorphous low-grade Cerebral hemispheres Calcification common 47
neuroepithelial tumor Temporal lobe predominance (47) Cystic component common
of the young (PLNTY) Enhancement uncommon
Diffuse low-grade glioma, Throughout craniospinal axis Similar to pilocytic astrocytoma Limited literature
MAPK pathway-altered Cerebral hemispheres most common Heterogeneous enhancement
Cystic component common
Pediatric-type diffuse
high-grade glioma family
Diffuse midline glioma, Midline Highly variable (see Fig 4) 28, 56
H3 K27-altered Brainstem, thalamus, Classic appearance is expansile nonenhancing
and spinal cord mass in the pons
Table 4 (continues)
506 radiology.rsna.org n Radiology: Volume 304: Number 3—September 2022
Johnson et al
Table 4 (continued): Imaging Features of Common Diffuse Gliomas
Key Imaging
Tumor Type Location Typical Imaging Features References
Diffuse hemispheric Cerebral hemispheres Often well-defined margins 30, 31, 33
glioma, H3 G34-mutant Leptomeningeal contact common Occasionally diffusely infiltrative
Variable enhancement
Expansile
Necrosis or microcalcification
Diffuse pediatric-type Cerebral hemispheres, brainstem, Heterogeneous 36
high-grade glioma, and cerebellum Similar to other high-grade gliomas
H3-wildtype and
IDH-wildtype
Infant-type hemispheric Cerebral hemispheres Often large at presentation Limited literature
glioma Leptomeningeal contact common
Note.—CNS = central nervous system, FLAIR = fluid-attenuated inversion recovery, GBM = glioblastoma, IDH = isocitrate
dehydrogenase, MAPK = mitogen-activated protein kinase, WHO = World Health Organization, wt = wildtype.
operative setting is not vital. Radiologists add significant value Acknowledgment: The authors acknowledge the assistance of Sonia Watson,
PhD (Department of Radiology, Mayo Clinic, Rochester, Minn), in preparation
by helping differentiate tumors from tumor mimics, detailing of the manuscript.
anatomic considerations important for surgical planning, pro-
viding preliminary information about suspected tumor abnor- Disclosures of conflicts of interest: D.R.J. No relevant relationships. C.G. No
mality, and informing the risk-reward analysis of various treat- relevant relationships. R.A.V. No relevant relationships. J.M.M. Participation on a
data safety monitoring board or advisory board at Mayo Clinic. L.J.E. No relevant
ment strategies. relationships. T.J.K. Co-chair, NeuroOncology Imaging Subcommittee, Alliance
Knowledge of tumor classification is also important when for Clinical Trials in Oncology; stock or stock options in SpineThera. J.B.G. No
interpreting follow-up images because it helps predict and un- relevant relationships.
derstand tumor behavior. For example, oligodendroglioma,
IDH-mutant and 1p/19q-codeleted, CNS WHO grade 2, and References
1. WHO Classification of Tumours Editorial Board. WHO classification of
glioblastoma, IDH-wildtype, CNS WHO grade 4, diagnosed tumours series: central nervous system tumours, 5th ed, volume 6. Lyon,
with molecular criteria can both manifest as nonenhancing France: International Agency for Research on Cancer; 2021. https://pub-
poorly circumscribed tumors, but median overall survival for lications.iarc.fr/601
2. Weller M, van den Bent M, Preusser M, et al. EANO guidelines on the
the former is more than a decade with appropriate treatment, diagnosis and treatment of diffuse gliomas of adulthood. Nat Rev Clin
whereas the median overall survival for the latter is less than 2 Oncol 2021;18(3):170–186 [Published correction appears in Nat Rev
years (49,50). Knowing this information, a radiologist may be Clin Oncol 2022;19(5):357–358.].
3. Louis DN, Aldape K, Brat DJ, et al. Announcing cIMPACT-NOW: the
more likely to raise the possibility of treatment effect and recom- Consortium to Inform Molecular and Practical Approaches to CNS Tu-
mend follow-up imaging in the setting of equivocal progression mor Taxonomy. Acta Neuropathol (Berl) 2017;133(1):1–3.
of oligodendroglioma, while true and clinically important pro- 4. Louis DN, Aldape K, Brat DJ, et al. cIMPACT-NOW (the consortium
to inform molecular and practical approaches to CNS tumor taxonomy):
gression may be suspected in glioblastoma. a new initiative in advancing nervous system tumor classification. Brain
Pathol 2017;27(6):851–852.
Conclusion 5. Louis DN, Giannini C, Capper D, et al. cIMPACT-NOW update 2: di-
agnostic clarifications for diffuse midline glioma, H3 K27M-mutant and
Radiologists play a crucial role in the care of patients with brain diffuse astrocytoma/anaplastic astrocytoma, IDH-mutant. Acta Neuro-
tumors, making it vital to stay current with advances in the field. pathol (Berl) 2018;135(4):639–642.
6. Louis DN, Wesseling P, Paulus W, et al. cIMPACT-NOW update 1: Not
Awareness of changes in brain tumor classification, such as those Otherwise Specified (NOS) and Not Elsewhere Classified (NEC). Acta
contained within the fifth edition of the World Health Orga- Neuropathol (Berl) 2018;135(3):481–484.
nization classification of tumors of the central nervous system, 7. Brat DJ, Aldape K, Colman H, et al. cIMPACT-NOW update 5: recom-
mended grading criteria and terminologies for IDH-mutant astrocytomas.
represents an important baseline level of knowledge. However, Acta Neuropathol (Berl) 2020;139(3):603–608.
knowledge acquisition must be an ongoing iterative process. 8. Ellison DW, Aldape KD, Capper D, et al. cIMPACT-NOW update 7:
Molecular techniques are enhancing our understanding of brain advancing the molecular classification of ependymal tumors. Brain Pathol
2020;30(5):863–866.
tumor pathology more quickly than ever before. These fast ad- 9. Ellison DW, Hawkins C, Jones DTW, et al. cIMPACT-NOW update 4:
vancements require radiologists to stay abreast of neuro-oncology diffuse gliomas characterized by MYB, MYBL1, or FGFR1 alterations or
developments. This includes monitoring the results of clinical BRAFV600E mutation. Acta Neuropathol (Berl) 2019;137(4):683–687.
10. Louis DN, Wesseling P, Aldape K, et al. cIMPACT-NOW update 6:
trials and publications by the Consortium to Inform Molecular new entity and diagnostic principle recommendations of the cIMPACT-
and Practical Approaches to CNS Tumor Taxonomy—Not Of- Utrecht meeting on future CNS tumor classification and grading. Brain
ficial WHO, or cIMPACT-NOW, group. Although this com- Pathol 2020;30(4):844–856.
11. Louis DN, Perry A, Reifenberger G, et al. The 2016 World Health Orga-
mitment requires additional time and effort, it is imperative for nization Classification of Tumors of the Central Nervous System: a sum-
providing the best patient clinical care possible. mary. Acta Neuropathol (Berl) 2016;131(6):803–820.
Radiology: Volume 304: Number 3—September 2022 n radiology.rsna.org 507
Radiologist’s Guide to the 2021 WHO Central Nervous System Tumor Classification
12. Barresi V, Eccher A, Simbolo M, et al. Diffuse gliomas in patients aged 36. Tauziède-Espariat A, Debily MA, Castel D, et al. The pediatric supra-
55 years or over: A suggestion for IDH mutation testing. Neuropathology tentorial MYCN-amplified high-grade gliomas methylation class presents
2020;40(1):68–74. the same radiological, histopathological and molecular features as their
13. Ball MK, Kollmeyer TM, Praska CE, et al. Frequency of false-positive pontine counterparts. Acta Neuropathol Commun 2020;8(1):104.
FISH 1p/19q codeletion in adult diffuse astrocytic gliomas. Neurooncol 37. Guerreiro Stucklin AS, Ryall S, Fukuoka K, et al. Alterations in ALK/
Adv 2020;2(1):vdaa109. ROS1/NTRK/MET drive a group of infantile hemispheric gliomas. Nat
14. Capper D, Jones DTW, Sill M, et al. DNA methylation-based classification of Commun 2019;10(1):4343.
central nervous system tumours. Nature 2018;555(7697):469–474. 38. Olsen TK, Panagopoulos I, Meling TR, et al. Fusion genes with ALK as
15. JaunmuktaneZ,CapperD,JonesDTW,et al.Methylationarrayprofilingofadult recurrent partner in ependymoma-like gliomas: a new brain tumor entity?
brain tumours: diagnostic outcomes in a large, single centre. Acta Neuropathol Neuro Oncol 2015;17(10):1365–1373.
Commun 2019;7(1):24. 39. Han G, Zhang J, Ma Y, Gui Q, Yin S. Clinical characteristics, treatment and
16. Wu Z, Abdullaev Z, Pratt D, et al. Impact of the methylation clas- prognosis of angiocentric glioma. Oncol Lett 2020;20(2):1641–1648.
sifier and ancillary methods on CNS tumor diagnostics. Neuro Oncol 40. Koral K, Koral KM, Sklar F. Angiocentric glioma in a 4-year-old boy:
2022;24(4):571–581. imaging characteristics and review of the literature. Clin Imaging
17. Kocakavuk E, Anderson KJ, Varn FS, et al. Radiotherapy is associated 2012;36(1):61–64.
with a deletion signature that contributes to poor outcomes in patients 41. Shakur SF, McGirt MJ, Johnson MW, et al. Angiocentric glioma: a case
with cancer. Nat Genet 2021;53(7):1088–1096. series. J Neurosurg Pediatr 2009;3(3):197–202.
18. Cao Y, Li X, Kong S, Shang S, Qi Y. CDK4/6 inhibition suppresses tu- 42. Preusser M, Hoischen A, Novak K, et al. Angiocentric glioma: report of clini-
mour growth and enhances the effect of temozolomide in glioma cells. J co-pathologic and genetic findings in 8 cases. Am J Surg Pathol 2007;31(11):
Cell Mol Med 2020;24(9):5135–5145. 1709–1718.
19. van Lent DI, van Baarsen KM, Snijders TJ, Robe P. Radiological differ- 43. Wefers AK, Stichel D, Schrimpf D, et al. Isomorphic diffuse glioma is a
ences between subtypes of WHO 2016 grade II-III gliomas: a systematic morphologically and molecularly distinct tumour entity with recurrent gene
review and meta-analysis. Neurooncol Adv 2020;2(1):vdaa044. fusions of MYBL1 or MYB and a benign disease course. Acta Neuropathol
20. Kinoshita M, Uchikoshi M, Sakai M, Kanemura Y, Kishima H, Nakanishi K. (Berl) 2020;139(1):193–209.
T2-FLAIR Mismatch Sign Is Caused by Long T1 and T2 of IDH- 44. Chiang J, Harreld JH, Tinkle CL, et al. A single-center study of the clini-
mutant, 1p19q Non-codeleted Astrocytoma. Magn Reson Med Sci copathologic correlates of gliomas with a MYB or MYBL1 alteration. Acta
2021;20(1):119–123. Neuropathol (Berl) 2019;138(6):1091–1092.
21. Jain R, Johnson DR, Patel SH, et al. “Real world” use of a highly reliable 45. Huse JT, Snuderl M, Jones DT, et al. Polymorphous low-grade neuro-
imaging sign: “T2-FLAIR mismatch” for identification of IDH mutant epithelial tumor of the young (PLNTY): an epileptogenic neoplasm with
astrocytomas. Neuro Oncol 2020;22(7):936–943. oligodendroglioma-like components, aberrant CD34 expression, and ge-
22. Johnson DR, Kaufmann TJ, Patel SH, Chi AS, Snuderl M, Jain R. There netic alterations involving the MAP kinase pathway. Acta Neuropathol
is an exception to every rule-T2-FLAIR mismatch sign in gliomas. Neuro- (Berl) 2017;133(3):417–429.
radiology 2019;61(2):225–227. 46. Ida CM, Johnson DR, Nair AA, et al. Polymorphous Low-Grade Neu-
23. Patel SH, Poisson LM, Brat DJ, et al. T2-FLAIR Mismatch, an Imag- roepithelial Tumor of the Young (PLNTY): Molecular Profiling Con-
ing Biomarker for IDH and 1p/19q Status in Lower-grade Gliomas: A firms Frequent MAPK Pathway Activation. J Neuropathol Exp Neurol
TCGA/TCIA Project. Clin Cancer Res 2017;23(20):6078–6085. 2021;80(9):821–829.
24. Appay R, Dehais C, Maurage CA, et al. CDKN2A homozygous deletion 47. Johnson DR, Giannini C, Jenkins RB, Kim DK, Kaufmann TJ. Plenty of
is a strong adverse prognosis factor in diffuse malignant IDH-mutant glio- calcification: imaging characterization of polymorphous low-grade neuro-
mas. Neuro Oncol 2019;21(12):1519–1528. epithelial tumor of the young. Neuroradiology 2019;61(11):1327–1332.
25. Nam YK, Park JE, Park SY, et al. Reproducible imaging-based predic- 48. Ryall S, Zapotocky M, Fukuoka K, et al. Integrated Molecular and
tion of molecular subtype and risk stratification of gliomas across dif- Clinical Analysis of 1,000 Pediatric Low-Grade Gliomas. Cancer Cell
ferent experience levels using a structured reporting system. Eur Radiol 2020;37(4):569–583.e5.
2021;31(10):7374–7385. 49. Buckner JC, Shaw EG, Pugh SL, et al. Radiation plus Procarba-
26. Findlay IJ, De Iuliis GN, Duchatel RJ, et al. Pharmaco-proteogenomic zine, CCNU, and Vincristine in Low-Grade Glioma. N Engl J Med
profiling of pediatric diffuse midline glioma to inform future treatment 2016;374(14):1344–1355.
strategies. Oncogene 2022;41(4):461–475. 50. Johnson DR, Leeper HE, Uhm JH. Glioblastoma survival in the United States
27. Lu VM, Alvi MA, McDonald KL, Daniels DJ. Impact of the H3K27M improved after Food and Drug Administration approval of bevacizumab:
mutation on survival in pediatric high-grade glioma: a systematic review a population-based analysis. Cancer 2013;119(19):3489–3495.
and meta-analysis. J Neurosurg Pediatr 2018;23(3):308–316. 51. Wijnenga MMJ, van der Voort SR, French PJ, et al. Differences in spatial
28. Aboian MS, Solomon DA, Felton E, et al. Imaging Characteristics of distribution between WHO 2016 low-grade glioma molecular subgroups.
Pediatric Diffuse Midline Gliomas with Histone H3 K27M Mutation. Neurooncol Adv 2019;1(1):vdz001.
AJNR Am J Neuroradiol 2017;38(4):795–800. 52. Lundy P, Domino J, Ryken T, et al. The role of imaging for the man-
29. Leach JL, Roebker J, Schafer A, et al. MR imaging features of diffuse agement of newly diagnosed glioblastoma in adults: a systematic review
intrinsic pontine glioma and relationship to overall survival: report from and evidence-based clinical practice guideline update. J Neurooncol
the International DIPG Registry. Neuro Oncol 2020;22(11):1647–1657. 2020;150(2):95–120.
30. Picart T, Barritault M, Poncet D, et al. Characteristics of diffuse 53. Nelson SJ, Cha S. Imaging glioblastoma multiforme. Cancer J
hemispheric gliomas, H3 G34-mutant in adults. Neurooncol Adv 2003;9(2):134–145.
2021;3(1):vdab061. 54. Park CJ, Han K, Kim H, et al. MRI Features May Predict Molecular Fea-
31. Puntonet J, Dangouloff-Ros V, Saffroy R, et al. Historadiological correla- tures of Glioblastoma in Isocitrate Dehydrogenase Wild-Type Lower-Grade
tions in high-grade glioma with the histone 3.3 G34R mutation. J Neuro- Gliomas. AJNR Am J Neuroradiol 2021;42(3):448–456.
radiol 2018;45(5):316–322. 55. Kurokawa M, Kurokawa R, Capizzano AA, et al. Neuroradiological fea-
32. Yoshimoto K, Hatae R, Sangatsuda Y, et al. Prevalence and clinicopathologi- tures of the polymorphous low-grade neuroepithelial tumor of the young:
cal features of H3.3 G34-mutant high-grade gliomas: a retrospective study five new cases with a systematic review of the literature. Neuroradiology
of 411 consecutive glioma cases in a single institution. Brain Tumor Pathol 2022;64(6):1255–1264.
2017;34(3):103–112. 56. Meyronet D, Esteban-Mader M, Bonnet C, et al. Characteristics of H3
33. Kurokawa R, Baba A, Kurokawa M, et al. Neuroimaging features of diffuse K27M-mutant gliomas in adults. Neuro Oncol 2017;19(8):1127–1134.
hemispheric glioma, H3 G34-mutant: A case series and systematic review. 57. Lasocki A, Anjari M, rs Kokurcan S, et al. Conventional MRI features of
J Neuroimaging 2022;32(1):17–27. adult diffuse glioma molecular subtypes: a systematic review. Neuroradiol-
34. Korshunov A, Schrimpf D, Ryzhova M, et al. H3-/IDH-wild type pediatric ogy 2021;63(3):353–362.
glioblastoma is comprised of molecularly and prognostically distinct 58. Lasocki A, Gaillard F, Gorelik A, et al. MRI Features Can Predict
subtypes with associated oncogenic drivers. Acta Neuropathol (Berl) 1p/19q Status in Intracranial Gliomas. AJNR Am J Neuroradiol
2017;134(3):507–516. 2018;39(4):687–692.
35. DeSisto J, Lucas JT Jr, Xu K, et al. Comprehensive molecular character-
ization of pediatric radiation-induced high-grade glioma. Nat Commun
2021;12(1):5531.
508 radiology.rsna.org n Radiology: Volume 304: Number 3—September 2022
You might also like
- 2019 July Ethiopia EHealth Architecture Document 1.5Document31 pages2019 July Ethiopia EHealth Architecture Document 1.5Tinsae Getachew100% (1)
- Brio UK Lit Dec 2007Document8 pagesBrio UK Lit Dec 2007Neil HoggNo ratings yet
- 9 - Urban & Rural Development in BangladeshDocument21 pages9 - Urban & Rural Development in BangladeshMd Omar Kaiser MahinNo ratings yet
- Retail Permit To Work System - Job Hazard AnalysisDocument7 pagesRetail Permit To Work System - Job Hazard AnalysisSanthosh Kumar100% (1)
- French CuisineDocument21 pagesFrench CuisineLea Rachelle VillarmenteNo ratings yet
- TNM 2017Document11 pagesTNM 2017David MaynéNo ratings yet
- EdCaN Specialty Module 4Document23 pagesEdCaN Specialty Module 4cindy8127No ratings yet
- 2022 - Horbinsky - Clin ImpliDocument15 pages2022 - Horbinsky - Clin ImpliRimpa Basu AchariNo ratings yet
- Integrating Pathology and Radiology Disciplines: An Emerging Opportunity?Document7 pagesIntegrating Pathology and Radiology Disciplines: An Emerging Opportunity?Peko PekoNo ratings yet
- Diagnostic Value of Circulating Tumor DNA in Molecular Characterization of GliomaDocument8 pagesDiagnostic Value of Circulating Tumor DNA in Molecular Characterization of GliomaAnyaNo ratings yet
- The 2021 WHO Classification of Tumors of The Central Nervous System, A SumaryDocument21 pagesThe 2021 WHO Classification of Tumors of The Central Nervous System, A SumaryDTNo ratings yet
- Principles Cancer StagingDocument28 pagesPrinciples Cancer Stagingcheatingw995No ratings yet
- Neuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryDocument21 pagesNeuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryJoao FabricioNo ratings yet
- Principles of Cancer StagingDocument28 pagesPrinciples of Cancer StagingAnonymous PKE8zOXNo ratings yet
- Neuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryDocument21 pagesNeuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryArif Susilo RahadiNo ratings yet
- Nejm - SobrevivienteDocument13 pagesNejm - SobrevivienteKathyMarambioNo ratings yet
- Colorectal Cancer StagingDocument14 pagesColorectal Cancer StagingRonnie Alexander Reyes MartínezNo ratings yet
- Underwood Et Al 2019 Liquid Biopsy For Cancer Review and Implications For The RadiologistDocument13 pagesUnderwood Et Al 2019 Liquid Biopsy For Cancer Review and Implications For The RadiologistpakhadkikarNo ratings yet
- 01 Purpose and Principles of Cancer Staging PDFDocument12 pages01 Purpose and Principles of Cancer Staging PDFalyson100% (1)
- Paper 2 Cancer DicoveryDocument18 pagesPaper 2 Cancer DicoverySuraj ShahNo ratings yet
- AllisonDocument11 pagesAllisonNabila NatasyaNo ratings yet
- Clinical and Research Applications of A Brain Tumor Tissue Bank in The Age of Precision MedicineDocument12 pagesClinical and Research Applications of A Brain Tumor Tissue Bank in The Age of Precision MedicineSandra Milena Sanabria BarreraNo ratings yet
- Adressing Suicidal Ideation in Patients With CancerDocument3 pagesAdressing Suicidal Ideation in Patients With CancerJosé Carlos Sánchez-RamirezNo ratings yet
- CNS SummaryDocument21 pagesCNS SummaryshokoNo ratings yet
- Imaging Diagnosis and Treatment Selection For Brain Tumors in The Era of Molecular TherapeuticsDocument14 pagesImaging Diagnosis and Treatment Selection For Brain Tumors in The Era of Molecular Therapeuticsmarwa humairaNo ratings yet
- Neuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryDocument21 pagesNeuro-Oncology: The 2021 WHO Classification of Tumors of The Central Nervous System: A SummaryAndrei PatrichiNo ratings yet
- Artcle 2 PDFDocument11 pagesArtcle 2 PDFAnirban PaulNo ratings yet
- 79 6690033a PDFDocument13 pages79 6690033a PDFEdward Arthur IskandarNo ratings yet
- Koch 2009Document9 pagesKoch 2009Intan Kartika NursyahbaniNo ratings yet
- 02-1.WHO 2022-PitNETDocument21 pages02-1.WHO 2022-PitNETJihuhn YuNo ratings yet
- How To Read A Next-Generation Sequencing Report-What Oncologists Need To KnowDocument9 pagesHow To Read A Next-Generation Sequencing Report-What Oncologists Need To KnowsamuelNo ratings yet
- Cancer Biomarkers: Easier Said Than Done: Kenneth P.H. PritzkerDocument4 pagesCancer Biomarkers: Easier Said Than Done: Kenneth P.H. Pritzkerdumboo21No ratings yet
- Radiation Therapy-Guidelines For PhysiotherapistsDocument9 pagesRadiation Therapy-Guidelines For PhysiotherapistsSofia Adão da FonsecaNo ratings yet
- 2020 Terri Glioma PRODocument7 pages2020 Terri Glioma PRORimpa Basu AchariNo ratings yet
- Radiotherapy and OncologyDocument14 pagesRadiotherapy and OncologyJavier RodríguezNo ratings yet
- Melanoma Pathology Reporting and StagingDocument10 pagesMelanoma Pathology Reporting and StagingLizbeth QuinteroNo ratings yet
- Hematolrep 03 E2 v2Document3 pagesHematolrep 03 E2 v2772450336No ratings yet
- 2013 ASTRO e ACR - Practice Guideline For The Performance TBIDocument5 pages2013 ASTRO e ACR - Practice Guideline For The Performance TBIAndre BanhateNo ratings yet
- Systemic Therapy For Advanced Appendiceal Adenocarcinoma: An Analysis From The NCCN Oncology Outcomes Database For Colorectal CancerDocument8 pagesSystemic Therapy For Advanced Appendiceal Adenocarcinoma: An Analysis From The NCCN Oncology Outcomes Database For Colorectal Canceralberto cabelloNo ratings yet
- Prognostic Biomarkers For Survival in Nasopharyngeal Carcinoma - A Systematic Review of The LiteratureDocument14 pagesPrognostic Biomarkers For Survival in Nasopharyngeal Carcinoma - A Systematic Review of The LiteratureWening Dewi HapsariNo ratings yet
- Precision Oncology in Low and Middle Income Countries A Word of CautionDocument5 pagesPrecision Oncology in Low and Middle Income Countries A Word of Cautionymourjany87No ratings yet
- Histological Molecular and Functional Subtypes ofDocument7 pagesHistological Molecular and Functional Subtypes ofjumed21No ratings yet
- Mizuno Et Al 2021Document20 pagesMizuno Et Al 2021Abner PortilhoNo ratings yet
- Jco 2009 27 0660Document9 pagesJco 2009 27 0660KAREN JAZMIN RUIZ MONROYNo ratings yet
- Diagnosisofrenalcell Carcinoma: A Clinician'S PerspectiveDocument6 pagesDiagnosisofrenalcell Carcinoma: A Clinician'S PerspectiveGangsar DamaiNo ratings yet
- s00261 023 03893 2Document15 pagess00261 023 03893 2sulbey878No ratings yet
- Meningiomas: Knowledge Base, Treatment Outcomes, and Uncertainties. A RANO ReviewDocument20 pagesMeningiomas: Knowledge Base, Treatment Outcomes, and Uncertainties. A RANO ReviewLuwiNo ratings yet
- Cancer of Unknown PrimaryDocument12 pagesCancer of Unknown Primaryraul gutierrezNo ratings yet
- Tearing Down The Walls: FDA Approves Next Generation Sequencing (NGS) Assays For Actionable Cancer Genomic AberrationsDocument3 pagesTearing Down The Walls: FDA Approves Next Generation Sequencing (NGS) Assays For Actionable Cancer Genomic AberrationsAdrián CLNo ratings yet
- Explore Rare Cancer Medicine MutationsDocument6 pagesExplore Rare Cancer Medicine MutationsMikel AngelNo ratings yet
- Cancer Awareness - 25 February 2021Document4 pagesCancer Awareness - 25 February 2021Times MediaNo ratings yet
- GMM. WHO CNS5 Classification (English Version)Document8 pagesGMM. WHO CNS5 Classification (English Version)Melissa Garcia LezamaNo ratings yet
- Bussines EngineringDocument9 pagesBussines EngineringWhulan Rhana TonapaNo ratings yet
- A Systematic Review On Barriers To Palliative Care in OncologyDocument17 pagesA Systematic Review On Barriers To Palliative Care in Oncologyw89qyccn6sNo ratings yet
- Dec 1 Family - Tree Chung 2002Document8 pagesDec 1 Family - Tree Chung 2002onlydarkheartNo ratings yet
- Document 1Document8 pagesDocument 1JOSE PORTILLONo ratings yet
- Colorectal Carcinoma: Diagnostic, Prognostic, and Molecular FeaturesDocument13 pagesColorectal Carcinoma: Diagnostic, Prognostic, and Molecular FeaturesEdward Arthur IskandarNo ratings yet
- Pain - Assessment JE DWDocument8 pagesPain - Assessment JE DWAshesh DahalNo ratings yet
- Head and Neck Cancers, Version 2.2020: NCCN Clinical Practice Guidelines in OncologyDocument26 pagesHead and Neck Cancers, Version 2.2020: NCCN Clinical Practice Guidelines in OncologympNo ratings yet
- Cell Specific Frequency As New HallmarkDocument8 pagesCell Specific Frequency As New Hallmarkjongsu kimNo ratings yet
- Functional and Mollecular ImagingDocument15 pagesFunctional and Mollecular Imagingstepanus massoraNo ratings yet
- Medical Oncologists' Perspectives of The Veterans Affairs National Precision Oncology ProgramDocument15 pagesMedical Oncologists' Perspectives of The Veterans Affairs National Precision Oncology ProgramFredrick WallinNo ratings yet
- Xi, 2021 Tumor Associated Collagen SignaturesDocument15 pagesXi, 2021 Tumor Associated Collagen Signaturesevahendrickx03No ratings yet
- A Patient's Guide to Cancer: Understanding the Causes and Treatments of a Complex DiseaseFrom EverandA Patient's Guide to Cancer: Understanding the Causes and Treatments of a Complex DiseaseNo ratings yet
- Glioma Imaging: Physiologic, Metabolic, and Molecular ApproachesFrom EverandGlioma Imaging: Physiologic, Metabolic, and Molecular ApproachesWhitney B. PopeNo ratings yet
- Interventional Radiology Management of High Flow PriapismDocument11 pagesInterventional Radiology Management of High Flow PriapismMANGNo ratings yet
- Complications of Central Venous Port Systems: A Pictorial ReviewDocument12 pagesComplications of Central Venous Port Systems: A Pictorial ReviewMANGNo ratings yet
- Complementary Roles of Interventional Radiology and Therapeutic Endoscopy in GastroenterologyDocument16 pagesComplementary Roles of Interventional Radiology and Therapeutic Endoscopy in GastroenterologyMANGNo ratings yet
- Basic Principles of Hematology: Section 13Document6 pagesBasic Principles of Hematology: Section 13MANGNo ratings yet
- CT Findings of Pulmonary NocardiosisDocument7 pagesCT Findings of Pulmonary NocardiosisMANGNo ratings yet
- 14.drug Eruption 2005Document6 pages14.drug Eruption 2005MANGNo ratings yet
- Paroxysmal Nocturnal Hemoglobinuria: Section 13Document2 pagesParoxysmal Nocturnal Hemoglobinuria: Section 13MANGNo ratings yet
- Diagnosis and Classification in MRI of Brucellar SpondylitisDocument6 pagesDiagnosis and Classification in MRI of Brucellar SpondylitisMANGNo ratings yet
- Apr-44 Sep-46 Sep-47 Jun-48 Dec-49 Mar-50 Oct-51 Jan-54 Mar-54 Apr-54 May-54 Sep-54 Jan-55Document4 pagesApr-44 Sep-46 Sep-47 Jun-48 Dec-49 Mar-50 Oct-51 Jan-54 Mar-54 Apr-54 May-54 Sep-54 Jan-55MANGNo ratings yet
- Dual-Energy CT: General PrinciplesDocument6 pagesDual-Energy CT: General PrinciplesMANGNo ratings yet
- PostDocument3 pagesPostMANGNo ratings yet
- Dual-Energy Spectral Computed Tomography To Thoracic Oncology Imaging KJRDocument13 pagesDual-Energy Spectral Computed Tomography To Thoracic Oncology Imaging KJRMANGNo ratings yet
- UCSF ED US Protocol LE Venous - FinalDocument8 pagesUCSF ED US Protocol LE Venous - FinalMANGNo ratings yet
- Toxic Shock Syndrome: Section 14Document2 pagesToxic Shock Syndrome: Section 14MANGNo ratings yet
- B9781416049197X50014 B978141604919750178X MainDocument4 pagesB9781416049197X50014 B978141604919750178X MainMANGNo ratings yet
- Colorectal Cancer: Section 7Document3 pagesColorectal Cancer: Section 7MANGNo ratings yet
- B9781416049197X50014 B9781416049197502152 MainDocument2 pagesB9781416049197X50014 B9781416049197502152 MainMANGNo ratings yet
- B9781416049197X50014 B9781416049197500396 MainDocument1 pageB9781416049197X50014 B9781416049197500396 MainMANGNo ratings yet
- Diabetes Mellitus: Disorders of Carbohydrate and Lipid MetabolismDocument5 pagesDiabetes Mellitus: Disorders of Carbohydrate and Lipid MetabolismMANGNo ratings yet
- B9781416049197X50014 B9781416049197501791 MainDocument2 pagesB9781416049197X50014 B9781416049197501791 MainMANGNo ratings yet
- Diagnostic Tests and Procedures: Section 2Document7 pagesDiagnostic Tests and Procedures: Section 2MANGNo ratings yet
- B9781416049197X50014 B9781416049197501985 MainDocument2 pagesB9781416049197X50014 B9781416049197501985 MainMANGNo ratings yet
- The Structure and Function of Skin Normal Anatomy: Section 2Document6 pagesThe Structure and Function of Skin Normal Anatomy: Section 2MANGNo ratings yet
- B9781416049197X50014 B9781416049197502772 MainDocument2 pagesB9781416049197X50014 B9781416049197502772 MainMANGNo ratings yet
- B9781416049197X50014 B9781416049197503534 MainDocument2 pagesB9781416049197X50014 B9781416049197503534 MainMANGNo ratings yet
- The Structure and Function of Skin: Section 1Document2 pagesThe Structure and Function of Skin: Section 1MANGNo ratings yet
- Familial Polyposis Syndrome: Section 7Document1 pageFamilial Polyposis Syndrome: Section 7MANGNo ratings yet
- B9781416049197X50014 B9781416049197502097 MainDocument2 pagesB9781416049197X50014 B9781416049197502097 MainMANGNo ratings yet
- Reaction Paper KyleDocument4 pagesReaction Paper KyleRonaliza Durano SanchezNo ratings yet
- CHN RleDocument5 pagesCHN RleAntoinette Peleña100% (1)
- KS3 Human Reproduction Exam HigherDocument4 pagesKS3 Human Reproduction Exam HigherSarah KKCNo ratings yet
- TariffDocument254 pagesTariffrehmannazirNo ratings yet
- Paragraphs: A. B. C. D. E. FDocument4 pagesParagraphs: A. B. C. D. E. FAnh Cường ĐỗNo ratings yet
- Band III Core Word ListDocument40 pagesBand III Core Word ListKatya RipsNo ratings yet
- Icsh Hemostasis Critical ValuesDocument12 pagesIcsh Hemostasis Critical Valuescitometria prolabNo ratings yet
- ASSESSOR ScriptDocument3 pagesASSESSOR ScriptJonalyn Lajera BasayaNo ratings yet
- Meeting 6 Understanding The MenuDocument3 pagesMeeting 6 Understanding The Menujurnal mumtazNo ratings yet
- Polyhydraminos and OligohydraminosDocument11 pagesPolyhydraminos and OligohydraminosMelissa Catherine ChinNo ratings yet
- 152 Experiment 3Document2 pages152 Experiment 3Earl CañoneroNo ratings yet
- Final Hospital Pharmacy Module 1Document7 pagesFinal Hospital Pharmacy Module 1Meryl Ann Ibarra-InganNo ratings yet
- 61-12 (PART 0) /issue 1 6 November 1992 Wires Cords and Cables Electrical - Metric Units Part 0:general Requirements and Test Methods For Qualification ApprovalDocument82 pages61-12 (PART 0) /issue 1 6 November 1992 Wires Cords and Cables Electrical - Metric Units Part 0:general Requirements and Test Methods For Qualification Approvaluğur SevgenNo ratings yet
- Feb28.2015 B.docmandatory Employment of Lifeguards in Public Pools SoughtDocument2 pagesFeb28.2015 B.docmandatory Employment of Lifeguards in Public Pools Soughtpribhor2No ratings yet
- Aptitude TestDocument8 pagesAptitude TestKhalidNo ratings yet
- Disease Detectives C ExamDocument34 pagesDisease Detectives C ExamGustavo Pacheco0% (1)
- RoR March Tongan-5Document80 pagesRoR March Tongan-5netanevete020No ratings yet
- University of Caloocan CityDocument9 pagesUniversity of Caloocan CityFudge FajardoNo ratings yet
- Engineering Manual: Variable Refrigerant Flow Outdoor Units 6.0 To 42.0 TonsDocument156 pagesEngineering Manual: Variable Refrigerant Flow Outdoor Units 6.0 To 42.0 TonsNey Raul Benji JoaquinNo ratings yet
- Carbon Offsets: Farming Carbon in The SoilDocument9 pagesCarbon Offsets: Farming Carbon in The Soildanmorgan1968No ratings yet
- Lab 2: Anatomy of The Heart: General ConsiderationsDocument19 pagesLab 2: Anatomy of The Heart: General ConsiderationsDisshiNo ratings yet
- GWYB 14-15 KarnatakaDocument226 pagesGWYB 14-15 Karnatakahram_phdNo ratings yet
- No Faster Prop On The Water: Step 1. Engine/DRIVE Step 2. Installation Kit Step 3. PropellerDocument2 pagesNo Faster Prop On The Water: Step 1. Engine/DRIVE Step 2. Installation Kit Step 3. PropellerjitmarineNo ratings yet
- Bartending 101Document15 pagesBartending 101Samuel SanguinoNo ratings yet
- Summary of AFSCME Tentative MOUDocument5 pagesSummary of AFSCME Tentative MOUjon_ortizNo ratings yet