Professional Documents
Culture Documents
Covid
Covid
Uploaded by
Bryanna Feliciano0 ratings0% found this document useful (0 votes)
13 views1 pageThe SARS-CoV-2 (COVID-19) RT-PCR test results for Isaiah Mendoza, a 5-year-old male, were negative. The test used was the iAMP COVID-19 Detection assay which utilizes Powergene PCR to detect SARS-CoV-2 nucleic acid in nasopharyngeal or oropharyngeal samples. Negative results do not rule out infection and should not be the sole basis for patient management decisions. Factors like improper specimen collection and storage or use of certain drugs could impact test accuracy.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe SARS-CoV-2 (COVID-19) RT-PCR test results for Isaiah Mendoza, a 5-year-old male, were negative. The test used was the iAMP COVID-19 Detection assay which utilizes Powergene PCR to detect SARS-CoV-2 nucleic acid in nasopharyngeal or oropharyngeal samples. Negative results do not rule out infection and should not be the sole basis for patient management decisions. Factors like improper specimen collection and storage or use of certain drugs could impact test accuracy.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
13 views1 pageCovid
Covid
Uploaded by
Bryanna FelicianoThe SARS-CoV-2 (COVID-19) RT-PCR test results for Isaiah Mendoza, a 5-year-old male, were negative. The test used was the iAMP COVID-19 Detection assay which utilizes Powergene PCR to detect SARS-CoV-2 nucleic acid in nasopharyngeal or oropharyngeal samples. Negative results do not rule out infection and should not be the sole basis for patient management decisions. Factors like improper specimen collection and storage or use of certain drugs could impact test accuracy.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
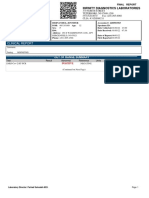
FINAL REPORT
22 Meridian Road, Suite 7 | Edison, NJ 08820 | (732)474-1120
AR EX PHARMACY MENDOZA, ISAIAH Accession #: 2111160482
370 New Brunswick Ave DOB: 05/11/2016 Age: 5 Specimen ID:
FORDS, NJ 08863- Sex: Male Date Collected: 11/16/21
Acct: 406 ID#: Date Received: 11/16/21 23:00
Phone: (732) 738-1085 Address: 859 KREIL ST Printed Date: 11/17/21
PERTH AMBOY, NJ 08861 Date of Report: 01/11/22
Phys: Phone: (347) 744-1099 Report Date: 11/17/21
CLINICAL REPORT
Comments:
Fasting: NONFASTING
SARS-CoV-2 (COVID-19) RT-PCR Tech:AMIT Date:11/17/21 12:18
Test Result Abnormal Reference Units Previous Result Date
SARS-CoV-2, RT-PCR Negative Negative
iAMP COVID-19 Detection assay from Atilla Biosystems using Powergene
PCR is utilized in detection of nucleic acid from the SARS-Cov-2 in
nasopharyngeal swab or oropharyngeal dry swab specimens collected from
individuals suspected of COVID-19 by their healthcare provider. This test
is a qualitative invitro diagnostic test that has been authorized by FDA
under an EUA for use by authorized laboratories.
-Negative results do not preclude infection with SARS-COV-2 virus and
should not be the sole basis of a patient management decision.
-Improper collection, transport, or storage of specimens may impact the
ability of the assay to perform as indicated.
-The impacts of vaccines, antiviral theraputics, antibiotics,
chemotheraputic, or immunosuppressant drugs have not been evaluated.
- The iAMP COVID-19 Detection cannot rule out respiratory diseases caused
by other bacterial or viral pathogens.
________________________________ END OF REPORT __________________________
Laboratory Director: Edward S. Liu MD
Avantic Medical Lab ; CLIA: 31D2077017 Page 1
22 Meridian Road, Suite 7 | Edison, NJ 08820 | (732)474-1120 Printed: 01/11/22 14:10
You might also like
- SITXFSA008 - Unit Assessment Tool (UAT) V3Document71 pagesSITXFSA008 - Unit Assessment Tool (UAT) V3Stacy ParkerNo ratings yet
- Mathematical Methods in Chemical EngineeringDocument576 pagesMathematical Methods in Chemical EngineeringAnonymous TwY8spHZJT86% (14)
- Unit Plan SainsDocument10 pagesUnit Plan Sainsajuy88No ratings yet
- Negative: Patient Information Specimen InformationDocument1 pageNegative: Patient Information Specimen InformationDanuardi WijayaNo ratings yet
- Philippine Red Cross Molecular Laboratory: Covid-19 RT-PCR Test Report & CertificationDocument1 pagePhilippine Red Cross Molecular Laboratory: Covid-19 RT-PCR Test Report & CertificationFrancis SevillenoNo ratings yet
- Department of Genetics: Covid-19 RT PCRDocument1 pageDepartment of Genetics: Covid-19 RT PCRAswathy LNo ratings yet
- Medical Officer KPJ Pasir Gudang Specialist Hospital Jalan Persiaran Dahlia 2, 81700 Pasir GudangDocument1 pageMedical Officer KPJ Pasir Gudang Specialist Hospital Jalan Persiaran Dahlia 2, 81700 Pasir GudangNooh DinNo ratings yet
- Covid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedDocument1 pageCovid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedRodel OrtegaNo ratings yet
- I DCD 0067262239Document1 pageI DCD 0067262239abhinavNo ratings yet
- Covid-19 Test Report: Patient Name: Akash GargDocument1 pageCovid-19 Test Report: Patient Name: Akash GargGenestrings Diagnostic CenterNo ratings yet
- Department of Genetics: Covid-19 RT PCRDocument1 pageDepartment of Genetics: Covid-19 RT PCRAjo Jose100% (1)
- KM Instructor Course Phase 1aDocument22 pagesKM Instructor Course Phase 1anozza_1No ratings yet
- 26 - LCCI L3 AC - Sep 2019 - ASE 20104 - MSDocument15 pages26 - LCCI L3 AC - Sep 2019 - ASE 20104 - MSKhin Zaw Htwe100% (6)
- TM355: Communications Technology: Take Home Exam For Final Assignment 2020-2021/ FallDocument7 pagesTM355: Communications Technology: Take Home Exam For Final Assignment 2020-2021/ FallHusseinJdeedNo ratings yet
- Drilling Fluids ManualDocument486 pagesDrilling Fluids ManualParaZzzit100% (12)
- Screenshot 2022-02-08 at 12.19.07 AMDocument1 pageScreenshot 2022-02-08 at 12.19.07 AMManal MazoziNo ratings yet
- FadiDocument2 pagesFadisafi alsafiNo ratings yet
- KPJ Perdana Specialist Hospital Lot PT.37 & PT.600, Seksyen 14, Jalan Bayam, Kota Bahru, KelantanDocument2 pagesKPJ Perdana Specialist Hospital Lot PT.37 & PT.600, Seksyen 14, Jalan Bayam, Kota Bahru, Kelantananuarhussaini.abdullatifNo ratings yet
- Department of Molecular Biology: Mr. Rajendra Ashokrao Chavan 52 Y(s) /male OB312901 / P00000122169 06-Sep-202011:03 AmDocument1 pageDepartment of Molecular Biology: Mr. Rajendra Ashokrao Chavan 52 Y(s) /male OB312901 / P00000122169 06-Sep-202011:03 AmRajendra ChavanNo ratings yet
- Department of Serology Covid-19 Antigen Test: Covid 19 Ag Test, Test Name Result Unit Bio. Ref. Range MethodDocument1 pageDepartment of Serology Covid-19 Antigen Test: Covid 19 Ag Test, Test Name Result Unit Bio. Ref. Range MethodPradeep VunnamNo ratings yet
- Patients Profile: Not Detected NegativeDocument2 pagesPatients Profile: Not Detected NegativeELLIE JAMES PLACIONo ratings yet
- Covid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedDocument1 pageCovid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedGEr JrvillaruElNo ratings yet
- Molecular Laboratory Test Result: de Loreto, San Isidro, City of Parañaque, NCR, Fourth District (Not A Province)Document2 pagesMolecular Laboratory Test Result: de Loreto, San Isidro, City of Parañaque, NCR, Fourth District (Not A Province)JJS INTERNATIONAL PLACEMENT AGENCY COMPANYNo ratings yet
- Clinical Report: Infinity Diagnostics LaboratoriesDocument2 pagesClinical Report: Infinity Diagnostics LaboratorieswponiofniwNo ratings yet
- Chandan Speciality Lab: Department of Molecular TestingDocument1 pageChandan Speciality Lab: Department of Molecular TestingZeeshan AliNo ratings yet
- Covidl 9 Test - Google Drive: St. Luke'sDocument2 pagesCovidl 9 Test - Google Drive: St. Luke'sAya BeeNo ratings yet
- This Is A Computer Generated Form and If Issued Without Any Alteration, This Does Not Require A SignatureDocument1 pageThis Is A Computer Generated Form and If Issued Without Any Alteration, This Does Not Require A SignatureIssa LlamasNo ratings yet
- Webrep - Cgi 4Document1 pageWebrep - Cgi 4viridiana bonillaNo ratings yet
- Patient Name: Mohit Raj Address: H-155, Longowal, Sangrur, PunjabDocument1 pagePatient Name: Mohit Raj Address: H-155, Longowal, Sangrur, PunjabMohitrajranikashyapNo ratings yet
- National Institute of Health: Sars-Cov-2 PCRDocument1 pageNational Institute of Health: Sars-Cov-2 PCRUmar BadshahNo ratings yet
- Patients Profile: Not Detected NegativeDocument2 pagesPatients Profile: Not Detected NegativeELLIE JAMES PLACIONo ratings yet
- Not Infected (Negative) : ResultDocument1 pageNot Infected (Negative) : ResultAllyssa Francheska OlitaNo ratings yet
- MR - AJINKYA KASAR LabReportNew-4Document2 pagesMR - AJINKYA KASAR LabReportNew-4Ajinkya kasarNo ratings yet
- RESULTDocument1 pageRESULTjenifer bongaoNo ratings yet
- LabReport 3557734Document1 pageLabReport 3557734Mahmoud Abed ElbestawyNo ratings yet
- Bugtong, Ergin May, DungcaDocument1 pageBugtong, Ergin May, DungcaGedion DelosreyesNo ratings yet
- Molecular Biology Report: Test Result MethodologyDocument1 pageMolecular Biology Report: Test Result MethodologySuhail AhamedNo ratings yet
- Take Care Sa GensanDocument1 pageTake Care Sa GensanAya BeeNo ratings yet
- Fusion Diagnostics Lingad, Joselito Bohots: Sars-Cov-2 PCRDocument2 pagesFusion Diagnostics Lingad, Joselito Bohots: Sars-Cov-2 PCRjb lingadNo ratings yet
- Department of Molecular Biology. Covid 19 RTPCR With Home Collection Test Name Result Unit Bio. Ref. Range MethodDocument2 pagesDepartment of Molecular Biology. Covid 19 RTPCR With Home Collection Test Name Result Unit Bio. Ref. Range MethodPritam JanaNo ratings yet
- WebrepDocument1 pageWebrepMayur ThebossNo ratings yet
- Velasco, Crestita VelosoDocument1 pageVelasco, Crestita VelosoAdan NunungNo ratings yet
- This Is A Computer Generated Form and If Issued Without Any Alteration, This Does Not Require A SignatureDocument1 pageThis Is A Computer Generated Form and If Issued Without Any Alteration, This Does Not Require A SignatureRyan FernandezNo ratings yet
- BooksDocument1 pageBooksCollin LongNo ratings yet
- LabResultTempPDF CJ0304865Document2 pagesLabResultTempPDF CJ0304865Jahred EstebanNo ratings yet
- Covid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedDocument1 pageCovid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not Detectedpogito ramosNo ratings yet
- National Institute of Health: Sars-Cov-2 PCRDocument1 pageNational Institute of Health: Sars-Cov-2 PCRDRSM QAUNo ratings yet
- April Jane: Cellular Immunology and ImmunogeneticsDocument2 pagesApril Jane: Cellular Immunology and ImmunogeneticsAya BeeNo ratings yet
- Moleculardiagnostics: Assayname Result Sarscov-2 (Realtimert-Pcr)Document2 pagesMoleculardiagnostics: Assayname Result Sarscov-2 (Realtimert-Pcr)Clash ClansNo ratings yet
- EH21718010 Report 1Document1 pageEH21718010 Report 1Aravind AjaiNo ratings yet
- Test Report: Ms - Ankita Ghosh (29/F)Document2 pagesTest Report: Ms - Ankita Ghosh (29/F)Aeio SavaNo ratings yet
- Covid 9.171021Document1 pageCovid 9.171021abid gulNo ratings yet
- PCR COVID-19: Negative: If You Have Any Questions Regarding This Report Please Contact Your ProviderDocument1 pagePCR COVID-19: Negative: If You Have Any Questions Regarding This Report Please Contact Your ProviderLuis ReyesNo ratings yet
- ResultsDocument1 pageResultsAlejandro OsunaNo ratings yet
- Patients Profile: Not Detected NegativeDocument2 pagesPatients Profile: Not Detected NegativeELLIE JAMES PLACIONo ratings yet
- 20-CVD-294738 - Cayanan PDFDocument1 page20-CVD-294738 - Cayanan PDFLyan CayananNo ratings yet
- The Nairobi West Hospital LTD: P.O.BOX: 43375-00100, NAIROBI, KENYA ISO 15189-2012 ACCREDITEDDocument1 pageThe Nairobi West Hospital LTD: P.O.BOX: 43375-00100, NAIROBI, KENYA ISO 15189-2012 ACCREDITEDAbba MustaphaNo ratings yet
- Philippine Red Cross Molecular Laboratory: Covid-19 RT-PCR Test Report & CertificationDocument1 pagePhilippine Red Cross Molecular Laboratory: Covid-19 RT-PCR Test Report & CertificationAldrin TevesNo ratings yet
- Sars-Cov-2 (Covid 19) Detection (Qualitative) by Real Time RT PCRDocument2 pagesSars-Cov-2 (Covid 19) Detection (Qualitative) by Real Time RT PCRRajat RanjanNo ratings yet
- Vargas, Juana Dr. Thornton, Karen 17675278Document1 pageVargas, Juana Dr. Thornton, Karen 17675278ahmedNo ratings yet
- Qualitative Detection of COVID-19Document1 pageQualitative Detection of COVID-19asdoiNo ratings yet
- SMSHLD tPEkEzDocument1 pageSMSHLD tPEkEzPranjal JindalNo ratings yet
- Negative: Patient Information Specimen InformationDocument1 pageNegative: Patient Information Specimen InformationojanNo ratings yet
- !58256 ! - !2439 !SelfPay !784-1988-0826532-0 !TRENCY FELIX FERNANDES ANTONIO ROSARIO FERNANDES !201222 !NULL ! - MOLECULAR BIOLOGY - New - Test !0Document1 page!58256 ! - !2439 !SelfPay !784-1988-0826532-0 !TRENCY FELIX FERNANDES ANTONIO ROSARIO FERNANDES !201222 !NULL ! - MOLECULAR BIOLOGY - New - Test !0Trency FernandesNo ratings yet
- PCR Test 20-12-22Document2 pagesPCR Test 20-12-22Trency FernandesNo ratings yet
- Fast Facts: Blastic Plasmacytoid Dendritic Cell Neoplasm: Shedding light on a rare diseaseFrom EverandFast Facts: Blastic Plasmacytoid Dendritic Cell Neoplasm: Shedding light on a rare diseaseNo ratings yet
- Sony Kdl32!37!40s5600 Chassis Ex2n-PeppermintDocument57 pagesSony Kdl32!37!40s5600 Chassis Ex2n-Peppermintvideoson100% (1)
- A6 Standard Lift Assembly m3 CAN Plasma System: Instruction ManualDocument79 pagesA6 Standard Lift Assembly m3 CAN Plasma System: Instruction ManualJulio De la RosaNo ratings yet
- Review LeadershipDocument10 pagesReview LeadershipON ChannelIDNo ratings yet
- Harrison Bergeron - With AnnotationsDocument8 pagesHarrison Bergeron - With Annotationserica.hugoNo ratings yet
- 202221JSA (Job Safety Analysis) and HIRARC (Hazard Identification, Risk Assessment and Risk Control)Document7 pages202221JSA (Job Safety Analysis) and HIRARC (Hazard Identification, Risk Assessment and Risk Control)Khairulz AnuarNo ratings yet
- Mekuria Bulcha Genocidal Violence in The Making of NationDocument54 pagesMekuria Bulcha Genocidal Violence in The Making of NationnasiseidrscribdNo ratings yet
- HP ZBOOK 17 QUICKSPECS c04111344Document45 pagesHP ZBOOK 17 QUICKSPECS c04111344hutz5000No ratings yet
- Hempathane HS 55610Document4 pagesHempathane HS 55610Hussein MohamedNo ratings yet
- DLL - Week - 12 - Statistics and ProbabilityDocument3 pagesDLL - Week - 12 - Statistics and ProbabilityRoboCopyNo ratings yet
- Machine Design Vol 12-2Document26 pagesMachine Design Vol 12-2CikamaksaNo ratings yet
- Engineering Management 5 - ControllingDocument46 pagesEngineering Management 5 - ControllingCraig PeriNo ratings yet
- Swot Analysis 123Document15 pagesSwot Analysis 123Roif SamsulNo ratings yet
- Sika Colorflo Yellow: Safety Data SheetDocument7 pagesSika Colorflo Yellow: Safety Data SheetKartika Setia RiniNo ratings yet
- Optical Communication NetworksDocument4 pagesOptical Communication Networksarivurp100% (1)
- Tourism Management: Emily Ma, Huijun Yang, Yao-Chin Wang, Hanqun SongDocument13 pagesTourism Management: Emily Ma, Huijun Yang, Yao-Chin Wang, Hanqun Song韩意颜No ratings yet
- Shravni Ragoor ResDocument1 pageShravni Ragoor Resapi-705833851No ratings yet
- Sap fb08 Amp f80 Tutorial Document ReversalDocument16 pagesSap fb08 Amp f80 Tutorial Document Reversalsaeedawais47No ratings yet
- 45ACP Hollow PointDocument5 pages45ACP Hollow PointTactic Otd ArgNo ratings yet
- Collaboration Agreement Inpulse World BankDocument1 pageCollaboration Agreement Inpulse World Bankapi-243080967No ratings yet
- Life of PiDocument45 pagesLife of Pisandroenglish100% (1)
- Human Cheek Cell ExpDocument3 pagesHuman Cheek Cell ExpArchna Verma100% (2)
- The Myth of The Neronian Persecution. Brent D. ShawDocument28 pagesThe Myth of The Neronian Persecution. Brent D. ShawDolores MonteroNo ratings yet
- TR - Dressmaking NC IIDocument60 pagesTR - Dressmaking NC IIMR. CHRISTIAN DACORONNo ratings yet