Professional Documents
Culture Documents
Installation Qualification 2022.11.30 15.10.42
Installation Qualification 2022.11.30 15.10.42
Uploaded by
Labovida RoraimaOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Installation Qualification 2022.11.30 15.10.42
Installation Qualification 2022.11.30 15.10.42
Uploaded by
Labovida RoraimaCopyright:
Available Formats
830194 - LABOVIDA - BOA VISTA RR - BRASIL 30/11/22 15:10:42
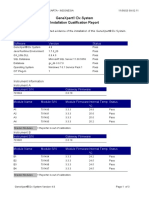
GeneXpert® Dx System
Installation Qualification Report
This report provides documented evidence of the installation of this GeneXpert® Dx System.
System Information
Software Version Status
GeneXpert® Dx System 4.8 Pass
Java Runtime Environment 1.7.0_60 Pass
GX_Utils.DLL 0.8.4.0 Pass
SQL Database Microsoft SQL Server 11.00.5058 Pass
Database gx_db 3.0.7.0 Pass
Operating System Windows 7 6.1 Service Pack 1 Pass
CIT Plug-In 1 Pass
Instrument Information
Instrument A
Instrument S/N Gateway Firmware
830194 2.0.18
Module Name Module S/N Module Firmware Internal Temp Status
°C
A1 618778 3.3.3 25.9 Pass
A2 744476 3.3.3 20.5 Pass
Shaded Modules = Reporter is out of calibration.
Available Assays
Assay Name Version Assay Type
Xpert CT_NG 3 In Vitro Diagnostic
Xpert FII & FV Combo 1 In Vitro Diagnostic
Xpert Flu CE-IVD 7 In Vitro Diagnostic
Xpert GBS G3 4 In Vitro Diagnostic
Xpert HPV HR_16_18-45 1 In Vitro Diagnostic
Xpert Xpress Flu-RSV 5 In Vitro Diagnostic
Xpert Xpress SARS-CoV-2 2 In Vitro Diagnostic
Xpert Xpress_Flu 5 In Vitro Diagnostic
Xpert Xpress_RSV 5 In Vitro Diagnostic
GxReliabilityTestFATA NA Research
GeneXpert® Dx System Version 4.8 Page 1 of 2
830194 - LABOVIDA - BOA VISTA RR - BRASIL 30/11/22 15:10:42
GeneXpert® Dx System
Installation Qualification Report
Installation of networked instruments complies with the setup requirements specified in the GeneXpert® Dx
System Operator Manual, 'Installation' section.
Verified by Date
This IQ is acceptable if all System Information and Instrument Information are listed as 'Pass'. All instrument
modules that are listed as 'Pass' are available for use.
Acceptance: [ ] Acceptable [ ] Not Acceptable
Performed by Date
Reviewed and approved by Date
GeneXpert® Dx System Version 4.8 Page 2 of 2
You might also like
- GUIDE Cancer by - Click MeDocument9 pagesGUIDE Cancer by - Click MeFlavio Galib100% (2)
- Ge Lunar Prodigy Dxse0012Document10 pagesGe Lunar Prodigy Dxse0012Jairo ManzanedaNo ratings yet
- RTLS Installation Manual: Date Modified: June 4, 2014Document52 pagesRTLS Installation Manual: Date Modified: June 4, 2014Asif Belal100% (1)
- Synopsis (Customer Billing System)Document3 pagesSynopsis (Customer Billing System)Akash Vashisht50% (2)
- Installation Qualification 2017.12.13 16.37.32Document2 pagesInstallation Qualification 2017.12.13 16.37.32Saad EL Mousadeq FilaliNo ratings yet
- Genexpert® DX System Installation Qualification ReportDocument2 pagesGenexpert® DX System Installation Qualification ReportBryan GonzalesNo ratings yet
- Installation Qualification 2023.09.04 09.36.41Document2 pagesInstallation Qualification 2023.09.04 09.36.41Satriawan SyahNo ratings yet
- Installation Qualification 2022.12.05 08.47.29Document2 pagesInstallation Qualification 2022.12.05 08.47.29Samuel BaidooNo ratings yet
- Genexpert® DX System Installation Qualification ReportDocument2 pagesGenexpert® DX System Installation Qualification Reportrsud abadi0% (1)
- Installation Qualification 2023.01.19 10.34.31Document2 pagesInstallation Qualification 2023.01.19 10.34.31Eep Prihatin KoserisNo ratings yet
- Installation - Qualification - 2023.02.02 - 08.14.18 Modul 1Document2 pagesInstallation - Qualification - 2023.02.02 - 08.14.18 Modul 1RoslinaNo ratings yet
- Genexpert® DX System Installation Qualification ReportDocument3 pagesGenexpert® DX System Installation Qualification Reportdar zipNo ratings yet
- Pdpro enDocument8 pagesPdpro ennikolalukovicNo ratings yet
- BD Veritor Plus System Overview BrochureDocument8 pagesBD Veritor Plus System Overview BrochureacroleewpaNo ratings yet
- 3Document6 pages3Jose F. Ramirez MendozaNo ratings yet
- DRCRnet Optovue RTVue Procedures 4222010Document27 pagesDRCRnet Optovue RTVue Procedures 4222010Yoselin Herrera Guzmán100% (2)
- Hasil Running Amplirun Di Alat PCRDocument8 pagesHasil Running Amplirun Di Alat PCRandi takwaNo ratings yet
- Rsi Sa 12-10-2023Document6 pagesRsi Sa 12-10-2023Laboratorium RSI Sultan Agung BanjarbaruNo ratings yet
- 06 - PhantomDocument8 pages06 - PhantomOmar Stalin Lucio RonNo ratings yet
- Test Report: Assay Assay Version Assay TypeDocument6 pagesTest Report: Assay Assay Version Assay TypeJose F. Ramirez MendozaNo ratings yet
- Idexx Vetlab QC MaintenancelogDocument21 pagesIdexx Vetlab QC MaintenancelogTesh McGillicuttyNo ratings yet
- STE Practical 11-15Document24 pagesSTE Practical 11-15CO-352 Vedant DeshpandeNo ratings yet
- 2 Streaming To External SystemsDocument1 page2 Streaming To External Systemsanna8cagiNo ratings yet
- DXQC2022Document8 pagesDXQC2022morgan.caldwellNo ratings yet
- Rukovodstvo Po PrimeneniyuDocument12 pagesRukovodstvo Po PrimeneniyuJuanfri CorreaNo ratings yet
- VistA Imaging DICOM Modality Interfaces0721Document38 pagesVistA Imaging DICOM Modality Interfaces0721SOPORTE LINEASNo ratings yet
- Guide c07 744925 - SensorDocument88 pagesGuide c07 744925 - SensorJavierNo ratings yet
- Hasim. S1Document2 pagesHasim. S1pkmsilo1No ratings yet
- Xpert Calibration Package Insert (Ingles)Document48 pagesXpert Calibration Package Insert (Ingles)Luis OvalleNo ratings yet
- VistAImaging DICOM Modality Interfaces1221Document37 pagesVistAImaging DICOM Modality Interfaces1221Bashir MtwaklNo ratings yet
- PROM-12395-002 1114007 BRO QIAstat-Dx 0518 SPREAD WWDocument3 pagesPROM-12395-002 1114007 BRO QIAstat-Dx 0518 SPREAD WWTaty ElejaldeNo ratings yet
- Test Report: Assay Assay Version Assay TypeDocument2 pagesTest Report: Assay Assay Version Assay TypesetyawanankesNo ratings yet
- PKM BetoambariDocument6 pagesPKM Betoambarinur liylaNo ratings yet
- UD2-9301-4 DocUReader2 Leaflet SmallDocument2 pagesUD2-9301-4 DocUReader2 Leaflet SmallKinnari BhattNo ratings yet
- UD2-9301-4 DocUReader2 Leaflet SmallDocument2 pagesUD2-9301-4 DocUReader2 Leaflet SmallKinnari BhattNo ratings yet
- Schneider Electric - EcoStruxure-Security-Expert - SX-SRVRDocument2 pagesSchneider Electric - EcoStruxure-Security-Expert - SX-SRVRmksaudiaidNo ratings yet
- Zkbiosecurity Access Control Devices List: No. Device Type Model Name Firmware Version Software Version RemarkDocument6 pagesZkbiosecurity Access Control Devices List: No. Device Type Model Name Firmware Version Software Version RemarkVanesa Purnama PutriNo ratings yet
- DocUReader 2 PRO Uputstvo enDocument66 pagesDocUReader 2 PRO Uputstvo enDarko MaksimovicNo ratings yet
- Cepheid GeneXpert System Brochure CE IVD 0309 English PDFDocument12 pagesCepheid GeneXpert System Brochure CE IVD 0309 English PDFlaboratoire BEN ARIBIANo ratings yet
- DICOM Conformance Statement MultiDiagnost Eleva FD R6.1Document115 pagesDICOM Conformance Statement MultiDiagnost Eleva FD R6.1azurivrg100% (1)
- DocUReader 2 Operators ManualDocument85 pagesDocUReader 2 Operators ManualKinnari BhattNo ratings yet
- Ananda FebriantiDocument2 pagesAnanda FebriantiInternis RsuyarsiNo ratings yet
- Test Report: Assay Assay Version Assay TypeDocument2 pagesTest Report: Assay Assay Version Assay Typeyossy aprillyaNo ratings yet
- Gen5 Software Features For DetectionDocument7 pagesGen5 Software Features For DetectionmohdNo ratings yet
- 736-144-G1 Nexus DR V3.2 SW Release NotesDocument7 pages736-144-G1 Nexus DR V3.2 SW Release NotesphuonganNo ratings yet
- AN 264 Suprema Biometrics Integration With Protege GXDocument10 pagesAN 264 Suprema Biometrics Integration With Protege GXchars choirNo ratings yet
- 1 - ProTech Product Line Overview Rev - ADocument60 pages1 - ProTech Product Line Overview Rev - ANGUYEN HUU TUANNo ratings yet
- DX-D 100 Wireless V5V6V7V8 - Acceptance TestDocument57 pagesDX-D 100 Wireless V5V6V7V8 - Acceptance TestEng-Eslam HassaanNo ratings yet
- Data Center Intrusion Prevention System Test ReportDocument2 pagesData Center Intrusion Prevention System Test ReportVivek R KoushikNo ratings yet
- Imaging Protocol Loading Guidance - SM - DOC2113485 - 1Document19 pagesImaging Protocol Loading Guidance - SM - DOC2113485 - 1QaiserAnwarNo ratings yet
- Netscout University - Lab - Router Profiled Automatic Threshold ConfigurationDocument6 pagesNetscout University - Lab - Router Profiled Automatic Threshold ConfigurationElaouni AbdessamadNo ratings yet
- Date Author Product: 07/05/2014 14:24:03 CENTREL Solutions 1.21 XIA Configuration Server (6.0.0.25996)Document20 pagesDate Author Product: 07/05/2014 14:24:03 CENTREL Solutions 1.21 XIA Configuration Server (6.0.0.25996)Deepak ChaudhariNo ratings yet
- Installation Instructions SW CRE - 1907Document21 pagesInstallation Instructions SW CRE - 1907КасымКаташовNo ratings yet
- SectionA IntroductionDocument7 pagesSectionA IntroductionPaweł KopyśćNo ratings yet
- DS0000101 User's Manual - X-View2 SoftwareDocument53 pagesDS0000101 User's Manual - X-View2 SoftwareThet Tun SanNo ratings yet
- Checkpoint NGX R65 ReleasenotesDocument42 pagesCheckpoint NGX R65 Releasenotesalexander_couceiroNo ratings yet
- Security Center 5 Reference Architecture With Pivot3 VSTACDocument14 pagesSecurity Center 5 Reference Architecture With Pivot3 VSTACmrkaspertytNo ratings yet
- CAMERADEBUGDOCDocument11 pagesCAMERADEBUGDOCRohit YadavNo ratings yet
- Advanced Penetration Testing for Highly-Secured Environments: The Ultimate Security GuideFrom EverandAdvanced Penetration Testing for Highly-Secured Environments: The Ultimate Security GuideRating: 4.5 out of 5 stars4.5/5 (6)
- Getting Started with Tiva ARM Cortex M4 Microcontrollers: A Lab Manual for Tiva LaunchPad Evaluation KitFrom EverandGetting Started with Tiva ARM Cortex M4 Microcontrollers: A Lab Manual for Tiva LaunchPad Evaluation KitNo ratings yet
- BE2000E BrochureDocument4 pagesBE2000E BrochureOrlando VeladoNo ratings yet
- Student Performance Analysis System With Graph & Academic Project ManagementDocument77 pagesStudent Performance Analysis System With Graph & Academic Project ManagementArjunNo ratings yet
- Midterm PracticeDocument7 pagesMidterm PracticeZhang YoudanNo ratings yet
- MAESTRO 100 20 User Guide Rev02Document17 pagesMAESTRO 100 20 User Guide Rev02wahyu abakusNo ratings yet
- Microstrip and Stripline DesignDocument7 pagesMicrostrip and Stripline DesignDurbha RaviNo ratings yet
- Nitin SivakumarDocument27 pagesNitin SivakumarnitinsivaNo ratings yet
- Introduction To Computer SoftwareDocument21 pagesIntroduction To Computer SoftwareEdwin MilogNo ratings yet
- ATA IT JD - Senior QA Engineer (Software Development in Test)Document2 pagesATA IT JD - Senior QA Engineer (Software Development in Test)The Generation's CodeNo ratings yet
- Tle TVL 9-12ia - CSS - Q1 - Wk6day1 - 4Document4 pagesTle TVL 9-12ia - CSS - Q1 - Wk6day1 - 4Lhai Diaz PoloNo ratings yet
- Ict Windows Installation AutosavedDocument34 pagesIct Windows Installation AutosavedTimeless GamingNo ratings yet
- Binary To Others Number Systems ConversionsDocument41 pagesBinary To Others Number Systems Conversionsi6apparNo ratings yet
- Installing Linux - The Easy WayDocument13 pagesInstalling Linux - The Easy WayVinay HegdeNo ratings yet
- P1260ATF: N-Channel Enhancement Mode MOSFETDocument5 pagesP1260ATF: N-Channel Enhancement Mode MOSFETGioVoTamNo ratings yet
- ESI Study Guide For Exam AZ-104Document8 pagesESI Study Guide For Exam AZ-104albertotecaNo ratings yet
- IC3 TeacherDocument46 pagesIC3 TeachertechnetvnNo ratings yet
- Event Handling 2Document41 pagesEvent Handling 2PratyushNo ratings yet
- Computer Security Chapter 5Document8 pagesComputer Security Chapter 5rh_rathodNo ratings yet
- Evolution, Challenge, and Outlook of TSV 3D IC - ICEP April 2011 KeynoteDocument16 pagesEvolution, Challenge, and Outlook of TSV 3D IC - ICEP April 2011 Keynoteice_speed2006No ratings yet
- RT8870ADocument12 pagesRT8870AFrastyan FazzaNo ratings yet
- Using OMICRON MBX1 For GOOSE Signal Switchboard TestingDocument47 pagesUsing OMICRON MBX1 For GOOSE Signal Switchboard TestingGian ChiminelliNo ratings yet
- Cheat Sheet CollectionDocument35 pagesCheat Sheet CollectionwerdegastNo ratings yet
- 12-Bit ADCDocument24 pages12-Bit ADCSamvel YanNo ratings yet
- Iot Based Underground Optical Fiber Cable Fault Detection SystemDocument5 pagesIot Based Underground Optical Fiber Cable Fault Detection SystemDamini RNo ratings yet
- ACS880 DC/DC Converter Control Program: Firmware ManualDocument210 pagesACS880 DC/DC Converter Control Program: Firmware ManualKechaouNo ratings yet
- Lab Manual 1 of Digital ElectronicsDocument4 pagesLab Manual 1 of Digital ElectronicsQuqan TahirNo ratings yet
- An Outline of The Problems and Potential Solutions For Cloud Computing SecurityDocument13 pagesAn Outline of The Problems and Potential Solutions For Cloud Computing SecurityInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- How To Reflow A Laptop Motherboard in A Household Oven - Computer Repair TipsDocument15 pagesHow To Reflow A Laptop Motherboard in A Household Oven - Computer Repair TipsAdnan KhAn100% (2)
- Inversor Yaskawa GPD 315-V7Document44 pagesInversor Yaskawa GPD 315-V7reinaldopf2012No ratings yet
- C++ Worksheet 2Document2 pagesC++ Worksheet 2Hanan FuadNo ratings yet