Professional Documents
Culture Documents
Tutorial Q1
Tutorial Q1
Uploaded by
Mihai Mircea0 ratings0% found this document useful (0 votes)
3 views2 pagesThe document discusses the thermal equation of state for perfect gases. It provides 6 variations of the equation:
1) pV = mRT, relating pressure, volume, mass, temperature
2) pv = RT, relating pressure, specific volume, temperature
3) p = ρRT, relating pressure, density, temperature
4) pV = nRT, relating pressure, volume, number of moles, temperature
5) pVM = RT, relating pressure, molar volume, temperature
6) pNVN = mRTN, relating pressure, number of moles, molar volume, mass, temperature at normal conditions.
Original Description:
Original Title
Tutorial_Q1
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document discusses the thermal equation of state for perfect gases. It provides 6 variations of the equation:
1) pV = mRT, relating pressure, volume, mass, temperature
2) pv = RT, relating pressure, specific volume, temperature
3) p = ρRT, relating pressure, density, temperature
4) pV = nRT, relating pressure, volume, number of moles, temperature
5) pVM = RT, relating pressure, molar volume, temperature
6) pNVN = mRTN, relating pressure, number of moles, molar volume, mass, temperature at normal conditions.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
3 views2 pagesTutorial Q1
Tutorial Q1
Uploaded by
Mihai MirceaThe document discusses the thermal equation of state for perfect gases. It provides 6 variations of the equation:
1) pV = mRT, relating pressure, volume, mass, temperature
2) pv = RT, relating pressure, specific volume, temperature
3) p = ρRT, relating pressure, density, temperature
4) pV = nRT, relating pressure, volume, number of moles, temperature
5) pVM = RT, relating pressure, molar volume, temperature
6) pNVN = mRTN, relating pressure, number of moles, molar volume, mass, temperature at normal conditions.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 2
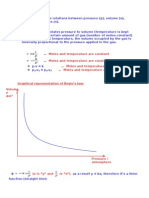
Thermal Equation of Stat for perfect gases
Theory Recap
Analytical expression of the thermal equation of state for
perfect gases:
a) pV = mRT , valid for m [kg] of gas
where p = absolute pressure of the gas [Pa];
V = volume of the gas [m3];
T = temperature of the gas [K];
m = mass of the gas [kg];
RM J
R= is the specific or gas constant
M kgK
J
with - RM = 8314,23 – universal (molar) constant
kmolK
- M – molar mass of the gas, i.e.:
kg kg kg
M O2 = 32 M = 44 M = 18
kmol ; kmol ; kmol ; etc.
CO2 H 2O
OBS: we will consider RM = 8315 J/(kmol K))
b) pv = RT , valid for 1 kg of gas
𝑉 𝑚3
where 𝑣 = 𝑚 [ 𝑘𝑔 ] = specific volume of the gas.
c) p = RT , equivalent of c), but expressed with density
m kg
where = = gas density.
V m 3
d) pV = n M RM T , valid for nM [kmol] of gas
m
where nM = kmol = number of kmol.
M
e) pVM = RM T
V m3
where VM = = molar volume.
nM kmol
f)Thermal equation of state applied in the normal state:
pNVN = mRTN
The normal state is defined by reference values of pressure
and temperature:
𝑝𝑁 = 760𝑚𝑚𝐻𝑔 = 1.013𝑏𝑎𝑟 = 1.013 × 105 𝑃𝑎
{
𝑇𝑁 = 273.15𝐾
The molar volume in the normal state has the same
value for any gas:
𝑅𝑀 𝑇𝑁 8315 ⋅ 273.15
𝑝𝑁 𝑉𝑀,𝑁 = 𝑅𝑀 𝑇𝑁 ⇒ 𝑉𝑀,𝑁 = =
𝑝𝑁 1.013 ⋅ 105
3
𝑚𝑁
= 22.414
𝑘𝑚𝑜𝑙
You might also like
- Homogeneous Equilibrium: John Paul S. TulaoDocument9 pagesHomogeneous Equilibrium: John Paul S. Tulaokatherine corvera100% (1)
- Module 3 Ideal Gases and Ideal Gas LawDocument12 pagesModule 3 Ideal Gases and Ideal Gas LawHazel AdoNo ratings yet
- Chapter 4 States of Matter 2021Document24 pagesChapter 4 States of Matter 2021suh mey chongNo ratings yet
- Ideal Gas Law (Part 4)Document5 pagesIdeal Gas Law (Part 4)asapamore100% (1)
- Topic 11-4 Ideal GasDocument3 pagesTopic 11-4 Ideal GasHinata CosaNo ratings yet
- Chapter Three Working Fluids (A) Ideal Gas (Perfect Gas) :: Statement No. (1)Document16 pagesChapter Three Working Fluids (A) Ideal Gas (Perfect Gas) :: Statement No. (1)Bonifacio MifañaNo ratings yet
- Ideal Gas Law Derivation For The Equations of The Ideal Gas LawDocument4 pagesIdeal Gas Law Derivation For The Equations of The Ideal Gas LawIan Kirk VillanuevaNo ratings yet
- Properties of Gases - 220308 - 154934Document28 pagesProperties of Gases - 220308 - 154934Dhruvi PadmaniNo ratings yet
- Internal Energy of An Ideal GasDocument15 pagesInternal Energy of An Ideal GasMD Al Fahad NirobNo ratings yet
- The Ideal - Gas Equation of StateDocument13 pagesThe Ideal - Gas Equation of StateAudu SanusiNo ratings yet
- Lesson Plan 7Document13 pagesLesson Plan 7khandaker raiyanNo ratings yet
- E233 - Thermofluids: The Perfect GasDocument16 pagesE233 - Thermofluids: The Perfect GasYingyote LubphooNo ratings yet
- Chapter 2 Perfect GasDocument25 pagesChapter 2 Perfect GasDaniel EssayuuNo ratings yet
- Physical ChemistryDocument5 pagesPhysical ChemistryLynnden CastilloNo ratings yet
- Lecture Notes On ThermoDocument6 pagesLecture Notes On ThermoPrasad V. JoshiNo ratings yet
- Meterological ParametersDocument47 pagesMeterological ParametersRDWSD SedamNo ratings yet
- Unit 2 Gas Laws and Power CyclesDocument103 pagesUnit 2 Gas Laws and Power CyclesNishad BhavsarNo ratings yet
- 5 Temp Ideal Gas-Fall 2022Document22 pages5 Temp Ideal Gas-Fall 2022asakr8481No ratings yet
- A2 53b GasesDocument50 pagesA2 53b GasesHany ElGezawyNo ratings yet
- ME 161: Introduction To Mechanical Engineering: Asif KabirDocument21 pagesME 161: Introduction To Mechanical Engineering: Asif KabirMohammad Asif KabirNo ratings yet
- 1 23 Gas Calculations PDFDocument6 pages1 23 Gas Calculations PDFschool adressNo ratings yet
- NCERT Solutions For Class 11 Physics 15may Chapter 13 Kinetic TheoryDocument15 pagesNCERT Solutions For Class 11 Physics 15may Chapter 13 Kinetic TheoryGSN KISHORENo ratings yet
- L-9 Pure Substance Ideal Gases-IDocument25 pagesL-9 Pure Substance Ideal Gases-IShailin SequeiraNo ratings yet
- Ideal Gas Law. Application To AirDocument11 pagesIdeal Gas Law. Application To AirvyrgoNo ratings yet
- Phy Kinetic Theory of GasesDocument16 pagesPhy Kinetic Theory of GasesSachinNo ratings yet
- For 2nd Year CK&EC Chapter 4 Final PDFDocument51 pagesFor 2nd Year CK&EC Chapter 4 Final PDFbahru demekeNo ratings yet
- Physics PreparationDocument85 pagesPhysics Preparationarafatahmmed51No ratings yet
- Specific Heat Capacities of GasesDocument3 pagesSpecific Heat Capacities of GasesVenu GopalNo ratings yet
- Problem Set 12 Key - Physical Chemistry For Engineers (Book Work)Document6 pagesProblem Set 12 Key - Physical Chemistry For Engineers (Book Work)krymxenNo ratings yet
- CHM131 - Chapter 6 - The Gaseous StateDocument37 pagesCHM131 - Chapter 6 - The Gaseous StateNotes NotesNo ratings yet
- States of MatterDocument50 pagesStates of Mattershivakafle039No ratings yet
- CHM 101 Lecture Note-Gas LawsDocument11 pagesCHM 101 Lecture Note-Gas LawsMichael DanielsNo ratings yet
- THE IDEAL GAS (Topic6)Document18 pagesTHE IDEAL GAS (Topic6)Ted Mana-ayNo ratings yet
- Ideal Gas LawDocument21 pagesIdeal Gas Lawjervis GrajoNo ratings yet
- Perfect GasesDocument15 pagesPerfect GasesellayuslianaNo ratings yet
- 5-State of MatterDocument26 pages5-State of MatterAbhinav VermaNo ratings yet
- Order 1771128Document4 pagesOrder 1771128Nahshon M. ObiriNo ratings yet
- Perhitungan Volum Molar GasDocument4 pagesPerhitungan Volum Molar GasRezty DulipatNo ratings yet
- Chem 181 Chemistry of GasesDocument15 pagesChem 181 Chemistry of GasesJoey PooleNo ratings yet
- 1.10 Ideal Gas EquationDocument14 pages1.10 Ideal Gas Equationjackmclaughlin21072No ratings yet
- 1.23 Gas Calculations: Molar Gas Volume (OCR +EDEXCEL)Document7 pages1.23 Gas Calculations: Molar Gas Volume (OCR +EDEXCEL)SunnyNo ratings yet
- PV RT: Equations of StateDocument11 pagesPV RT: Equations of StateJeff HardyNo ratings yet
- Gas PropertiesDocument54 pagesGas PropertiesAli AhmedNo ratings yet
- Ch01-Slides-1 Gas LawsDocument60 pagesCh01-Slides-1 Gas LawsDoreen BenezethNo ratings yet
- Measurement 16Document16 pagesMeasurement 16Gaurav ShekharNo ratings yet
- The Ideal Gas EquationDocument10 pagesThe Ideal Gas EquationBilly Jake CorpuzNo ratings yet
- 5.1 Pressure: Chapter 5: GasesDocument4 pages5.1 Pressure: Chapter 5: GasesSam ChungNo ratings yet
- 01 1stlaw ExessolsDocument29 pages01 1stlaw Exessolsblanca.pegueraNo ratings yet
- Revision Notes On Kinetic Theory of GasesDocument6 pagesRevision Notes On Kinetic Theory of Gasesvermarashmi2780No ratings yet
- Note Ideal Gas TutorialDocument5 pagesNote Ideal Gas TutorialGnabryNo ratings yet
- Gases and Their PropertiesDocument27 pagesGases and Their PropertiesMansonNo ratings yet
- Gas Mixtures Study Guide in Powerpoint: To AccompanyDocument21 pagesGas Mixtures Study Guide in Powerpoint: To AccompanyDon HoNo ratings yet
- Hints S 20Document18 pagesHints S 20ShivamNo ratings yet
- 1-Ideal Gas LectureDocument60 pages1-Ideal Gas LecturemahmoudNo ratings yet
- Ideal Gas Law LectureDocument46 pagesIdeal Gas Law LectureIkooyyNo ratings yet
- Gas Laws:: P V K VDocument18 pagesGas Laws:: P V K VFarah Zu'biNo ratings yet
- Properties of Mixtures Mixture of Perfect GasesDocument7 pagesProperties of Mixtures Mixture of Perfect GasesAudu SanusiNo ratings yet
- Ideal Gas LawDocument4 pagesIdeal Gas LawAbdul RaufNo ratings yet
- Victorio Oriel - Ideal Gas Law and Molar Mass EquationDocument5 pagesVictorio Oriel - Ideal Gas Law and Molar Mass Equationapi-233267698No ratings yet
- Lecture - 12 - High Speed Gas DynamicsDocument9 pagesLecture - 12 - High Speed Gas DynamicsMihai MirceaNo ratings yet
- Lecture - 11 - Second LawDocument10 pagesLecture - 11 - Second LawMihai MirceaNo ratings yet
- Lecture - 10 - Second LawDocument10 pagesLecture - 10 - Second LawMihai MirceaNo ratings yet
- First Law 1Document9 pagesFirst Law 1Mihai MirceaNo ratings yet
- Lecture - 7 - First LawDocument9 pagesLecture - 7 - First LawMihai MirceaNo ratings yet
- First Law 2Document9 pagesFirst Law 2Mihai MirceaNo ratings yet
- Lecture - 6 - First LawDocument9 pagesLecture - 6 - First LawMihai MirceaNo ratings yet
- Lecture 2 EDocument8 pagesLecture 2 EMihai MirceaNo ratings yet
- Gas MixtureDocument9 pagesGas MixtureMihai MirceaNo ratings yet
- Lecture 1Document5 pagesLecture 1Mihai MirceaNo ratings yet