Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
19 viewsPages From FeC
Pages From FeC
Uploaded by
Fuhad HasanThe document discusses the iron-carbon phase diagram and the microstructures of steels. It describes the different phases in the diagram, including α-ferrite, γ-austenite, δ-ferrite, and cementite. It explains the eutectic and eutectoid reactions, and how the microstructure of hypoeutectoid, eutectoid, and hypereutectoid steels depends on their carbon content and heat treatment. Specifically, it discusses the formation of proeutectoid phases and pearlite, and how to calculate the relative amounts using the lever rule.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- Materials Data for Cyclic Loading: Aluminium and Titanium AlloysFrom EverandMaterials Data for Cyclic Loading: Aluminium and Titanium AlloysRating: 1 out of 5 stars1/5 (1)
- A Project Report On Microstructure Analysis of Gray Cast Iron, Aluminium and Brass Using Optical MicrographsDocument26 pagesA Project Report On Microstructure Analysis of Gray Cast Iron, Aluminium and Brass Using Optical MicrographsManvendra Pratap Singh Bisht100% (8)
- Iron-Carbon Phase Diagram (A Review) See Callister Chapter 9Document13 pagesIron-Carbon Phase Diagram (A Review) See Callister Chapter 9babiker mortadaNo ratings yet
- Fe-C Phase DiagramDocument34 pagesFe-C Phase DiagramYoung-long Choi100% (1)
- Iron-Carbon Phase Diagram (A Review) See Callister Chapter 9Document34 pagesIron-Carbon Phase Diagram (A Review) See Callister Chapter 9Zefa Erliana YullahNo ratings yet
- Fe CDocument34 pagesFe CZaza ArifinNo ratings yet
- Material Science and MettalurgyDocument21 pagesMaterial Science and MettalurgySR-71 BLACKBIRDNo ratings yet
- Iron-Iron Carbide (Fe-Fe3C) Phase Diagram: M. Tech. (FFT) Technology of Ferrous CastingDocument7 pagesIron-Iron Carbide (Fe-Fe3C) Phase Diagram: M. Tech. (FFT) Technology of Ferrous CastingRajulapati Sunil KumarNo ratings yet
- Fec DiagramDocument15 pagesFec DiagramShankarNo ratings yet
- Phase Diagrams - 040823Document23 pagesPhase Diagrams - 040823Anthony MubangaNo ratings yet
- 9 Engineering AlloysDocument17 pages9 Engineering AlloysdavidtomyNo ratings yet
- University of Babylon, College of Engineering, Engineering Metallurgy, Maithem H-RasheedDocument13 pagesUniversity of Babylon, College of Engineering, Engineering Metallurgy, Maithem H-RasheedAris BulaongNo ratings yet
- Iron Carbon DiagramDocument10 pagesIron Carbon DiagramsivakumarNo ratings yet
- Seminarski Rad Fe Fe3C DijagramDocument10 pagesSeminarski Rad Fe Fe3C DijagramAlina OmerovicNo ratings yet
- TTT 11Document55 pagesTTT 11MrDOTNo ratings yet
- 8.fe - Fe3C Phase DiagramDocument27 pages8.fe - Fe3C Phase DiagramMhackSahuNo ratings yet
- 18bem0046 Materials Da 2Document20 pages18bem0046 Materials Da 2QUANTUM SOLEOSNo ratings yet
- Iron Carbon Equilibrium DiagramDocument11 pagesIron Carbon Equilibrium Diagramganesh82No ratings yet
- Research of Phase Transformation On Fe-8.7Al-28.3Mn-1C-5.5Cr AlloyDocument4 pagesResearch of Phase Transformation On Fe-8.7Al-28.3Mn-1C-5.5Cr AlloyMarina PiermannNo ratings yet
- Fracture Dependence On Heat Treatment: Table 1-Dependence of Absorbed Energy (For Crack Propagation) Over TemperatureDocument4 pagesFracture Dependence On Heat Treatment: Table 1-Dependence of Absorbed Energy (For Crack Propagation) Over Temperaturelokomoko1No ratings yet
- Chapter Outline:: Heat Treatment (And Temperature)Document24 pagesChapter Outline:: Heat Treatment (And Temperature)DSGNo ratings yet
- 3 Fe-Fe3C Phase DiagramDocument33 pages3 Fe-Fe3C Phase DiagramRajat Mishra100% (1)
- Fe CdiagramDocument36 pagesFe CdiagramGeorge SingerNo ratings yet
- Fe-C Phase Diagram Lecture 33Document15 pagesFe-C Phase Diagram Lecture 33Manish Gupta100% (1)
- FALLSEM2019-20 MEE1005 ETH VL2019201001078 Reference Material I 29-Aug-2019 Fe-Fe3C Phase DiagramDocument33 pagesFALLSEM2019-20 MEE1005 ETH VL2019201001078 Reference Material I 29-Aug-2019 Fe-Fe3C Phase DiagramFazal KhanNo ratings yet
- Unit 3Document113 pagesUnit 3Abhishek ChavanNo ratings yet
- Carbon Steels: ME 215/205:materials Science LaboratoryDocument9 pagesCarbon Steels: ME 215/205:materials Science Laboratoryfaizan997No ratings yet
- Ch-27.5 Iron Carbon Equilibrium DiagramDocument52 pagesCh-27.5 Iron Carbon Equilibrium DiagramManojNo ratings yet
- Chapter 5, THE IRON-CARBON EQUILIBRIUM DIAGRAMDocument13 pagesChapter 5, THE IRON-CARBON EQUILIBRIUM DIAGRAMPAUL NDIRITUNo ratings yet
- What Is PearliteDocument4 pagesWhat Is Pearliteardy cornettoNo ratings yet
- The Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalDocument38 pagesThe Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalYogesh KumbharNo ratings yet
- Chapter Outline: Phase Transformations. KineticsDocument23 pagesChapter Outline: Phase Transformations. Kineticsmpcd07No ratings yet
- BG2802 Heat Treatment and Mechanical Properties of SteelsDocument11 pagesBG2802 Heat Treatment and Mechanical Properties of SteelsVenus LimNo ratings yet
- Iron-Carbon Phase Diagram: By: Awad Elaraby ID:052022009Document33 pagesIron-Carbon Phase Diagram: By: Awad Elaraby ID:052022009Mahmoud RefaatNo ratings yet
- Weldability of Metals - NPTELDocument18 pagesWeldability of Metals - NPTELKaushal Gandhi0% (1)
- Iron - Carbon Phase DiagramDocument19 pagesIron - Carbon Phase DiagramGaurav DograNo ratings yet
- Foundations of Materials Science and Engineering 5th Edition Smith Solutions ManualDocument39 pagesFoundations of Materials Science and Engineering 5th Edition Smith Solutions Manualcacoonnymphaea6wgyct100% (16)
- Foundations of Materials Science and Engineering 5th Edition Smith Solutions ManualDocument79 pagesFoundations of Materials Science and Engineering 5th Edition Smith Solutions Manualdextrermachete4amgqgNo ratings yet
- Dokumen - Tips - Iron Iron Carbide Phase Diagram 58ac3a092bd8dDocument16 pagesDokumen - Tips - Iron Iron Carbide Phase Diagram 58ac3a092bd8dAfrizal Adithya PNo ratings yet
- Ch-27.3 Iron Carbon Equilibrium DiagramDocument58 pagesCh-27.3 Iron Carbon Equilibrium DiagramasjfgauojfgfNo ratings yet
- Iron Carbon Phase DiagramDocument4 pagesIron Carbon Phase DiagramMizanur RahmanNo ratings yet
- 3 Iron Carbon DiaDocument21 pages3 Iron Carbon DiaChhavi SharmaNo ratings yet
- Sudipta Nath: Materials EngineeringDocument19 pagesSudipta Nath: Materials EngineeringSudipta NathNo ratings yet
- Industrial Materials (B)Document15 pagesIndustrial Materials (B)Muhammad Bilal SahiNo ratings yet
- Introduction To Fe-C Equilibrium Phase Diagram: Chia-Chang ShihDocument42 pagesIntroduction To Fe-C Equilibrium Phase Diagram: Chia-Chang ShihTuấnPhạmNo ratings yet
- Phase Diagram 3Document26 pagesPhase Diagram 3Tony StarkNo ratings yet
- Engineering Materials 27-29Document40 pagesEngineering Materials 27-29Sanu SouravNo ratings yet
- Materials of Construction and Selection: Faculty of Chemical Engineering Universiti Teknologi MaraDocument80 pagesMaterials of Construction and Selection: Faculty of Chemical Engineering Universiti Teknologi MaraAisyah Addia AzizanNo ratings yet
- Capili Jefferson 10Document5 pagesCapili Jefferson 10Christian Al EncarnacionNo ratings yet
- Pure Iron, Allotropy:: Steels Are Alloys of Iron and Carbon With Carbon Content Up To 2%Document13 pagesPure Iron, Allotropy:: Steels Are Alloys of Iron and Carbon With Carbon Content Up To 2%Abhishek SakatNo ratings yet
- Iron Carbon Note 1 2023Document23 pagesIron Carbon Note 1 2023gerrard samuelNo ratings yet
- 8.fe - Fe3C Phase Diagram PDFDocument27 pages8.fe - Fe3C Phase Diagram PDF13311A0341 S SHIVA SAI KIRANNo ratings yet
- How The Material Built For Special PurposeDocument43 pagesHow The Material Built For Special PurposeDidi SudiprayitnaNo ratings yet
- Iron Carbon Diagram of Steel PDFDocument6 pagesIron Carbon Diagram of Steel PDFshihabscb1971100% (1)
- Iron-Carbon DiagramDocument3 pagesIron-Carbon DiagramnaniNo ratings yet
- Heat TreatmentDocument4 pagesHeat Treatment정석원No ratings yet
- Control and Analysis in Iron and SteelmakingFrom EverandControl and Analysis in Iron and SteelmakingRating: 3 out of 5 stars3/5 (2)
- Proceedings of the 2014 Energy Materials Conference: Xi'an, Shaanxi Province, China, November 4 - 6, 2014From EverandProceedings of the 2014 Energy Materials Conference: Xi'an, Shaanxi Province, China, November 4 - 6, 2014No ratings yet
- Expansionism and Religion - The Fatal Flaws of The Aztec EmpireDocument27 pagesExpansionism and Religion - The Fatal Flaws of The Aztec EmpireFuhad HasanNo ratings yet
- Sugar Cane JuiceDocument17 pagesSugar Cane JuiceFuhad HasanNo ratings yet
- Date JuiceDocument8 pagesDate JuiceFuhad HasanNo ratings yet
- Computational Simulation of Concentration by OsmotDocument15 pagesComputational Simulation of Concentration by OsmotFuhad HasanNo ratings yet
- Iron Carbon Phase DiagramDocument4 pagesIron Carbon Phase DiagramFuhad HasanNo ratings yet
- Phase Diagram of Fe-Fe3CDocument25 pagesPhase Diagram of Fe-Fe3CIram MustaviNo ratings yet
- Sub Module-1 Aircraft-Ferrous Materials 1.1 Ferrous Aircraft MetalsDocument22 pagesSub Module-1 Aircraft-Ferrous Materials 1.1 Ferrous Aircraft MetalsDeepak Choudhary DCNo ratings yet
- Outokumpu Handbook of Stainless Steel 2017 EditionDocument92 pagesOutokumpu Handbook of Stainless Steel 2017 EditionRavindra BarangeNo ratings yet
- Fabrication Requirements of Pressure Vessel Coils and JumpovDocument7 pagesFabrication Requirements of Pressure Vessel Coils and JumpovAleem QureshiNo ratings yet
- Full-Range Stress-Strain Curves For Stainless SteelDocument15 pagesFull-Range Stress-Strain Curves For Stainless SteelNurul HudaNo ratings yet
- Resistance Upset Butt Welding of AusteniDocument7 pagesResistance Upset Butt Welding of AusteniRamzi BEN AHMEDNo ratings yet
- Properties of Engineering Materials Lesson 3 (I)Document11 pagesProperties of Engineering Materials Lesson 3 (I)Douglas Kufre-Abasi GilbertNo ratings yet
- Materials and Processes Quiz 1Document6 pagesMaterials and Processes Quiz 1RuthNo ratings yet
- A 488 - A 488M - 99 Qtq4oc05oq - PDFDocument15 pagesA 488 - A 488M - 99 Qtq4oc05oq - PDFMarceloNo ratings yet
- Materials For Ultra Supercritical Fossil Power Plants by R. ViswanathanDocument66 pagesMaterials For Ultra Supercritical Fossil Power Plants by R. ViswanathanHASAN IZHAR KHANNo ratings yet
- ME692 - Welding - Lecture Till 6 FebDocument107 pagesME692 - Welding - Lecture Till 6 Febpramrahar20No ratings yet
- Ese 2021: Prelims Exam: MechanicalDocument22 pagesEse 2021: Prelims Exam: MechanicalRamesh TammineniNo ratings yet
- Welding Journal 1960 4Document219 pagesWelding Journal 1960 4AlexeyNo ratings yet
- Dormer AplicacionesDocument188 pagesDormer Aplicacionesjserna_22No ratings yet
- A Comparative Evaluation of Welding Consumables For Dissimilar Welds Between 316LN Austenitic Stainless Steel and Alloy 800Document12 pagesA Comparative Evaluation of Welding Consumables For Dissimilar Welds Between 316LN Austenitic Stainless Steel and Alloy 800Élida AlvesNo ratings yet
- 1 Dec2008Document9 pages1 Dec2008el_sharkawy2011No ratings yet
- Study of Microstructure Degradation of Boiler Tubes Due To Creep For Remaining Life AnalysisDocument7 pagesStudy of Microstructure Degradation of Boiler Tubes Due To Creep For Remaining Life AnalysisRaka DaffaNo ratings yet
- Chapter 8 - Stainless Steels - 2016 - Applied Welding EngineeringDocument8 pagesChapter 8 - Stainless Steels - 2016 - Applied Welding EngineeringJhair Jhamidh Aquino PortocarreroNo ratings yet
- Introduction-Iron Carbon Phase DiagramDocument31 pagesIntroduction-Iron Carbon Phase DiagramTHE BBEASTNo ratings yet
- Nsba Night School Course B1 Session 3 PDFDocument51 pagesNsba Night School Course B1 Session 3 PDFLuis CortesNo ratings yet
- Steel Casting MetallurgyDocument258 pagesSteel Casting MetallurgySiddharth Gupta86% (7)
- Lec 4. Solid Solution - Phase DiagramsDocument27 pagesLec 4. Solid Solution - Phase DiagramsTrí Tạ MinhNo ratings yet
- BME (Steel)Document8 pagesBME (Steel)Mohil JainNo ratings yet
- Low Temperature Embrittlement Studies On Stainless Steel 304 LN TIG WeldsDocument10 pagesLow Temperature Embrittlement Studies On Stainless Steel 304 LN TIG WeldsRajmchzNo ratings yet
- Ferro Fundido Nodular Normas PortDocument5 pagesFerro Fundido Nodular Normas PortCarlos LaoNo ratings yet
- EN380 Homework #6 Solution: 1084 C 1455 C 70% N i L + α C = 61% C = 73% W = = = 25% W = = = 75%Document4 pagesEN380 Homework #6 Solution: 1084 C 1455 C 70% N i L + α C = 61% C = 73% W = = = 25% W = = = 75%roseNo ratings yet
- M GN 365Document46 pagesM GN 365reza329329No ratings yet
- Austenitic Stainless SteelsDocument4 pagesAustenitic Stainless Steelsandy1036No ratings yet
- Wrought 18Chromium-14Nickel-2.5Molybdenum Stainless Steel Sheet and Strip For Surgical Implants (UNS S31673)Document4 pagesWrought 18Chromium-14Nickel-2.5Molybdenum Stainless Steel Sheet and Strip For Surgical Implants (UNS S31673)Angel ChangNo ratings yet
Pages From FeC
Pages From FeC
Uploaded by
Fuhad Hasan0 ratings0% found this document useful (0 votes)
19 views13 pagesThe document discusses the iron-carbon phase diagram and the microstructures of steels. It describes the different phases in the diagram, including α-ferrite, γ-austenite, δ-ferrite, and cementite. It explains the eutectic and eutectoid reactions, and how the microstructure of hypoeutectoid, eutectoid, and hypereutectoid steels depends on their carbon content and heat treatment. Specifically, it discusses the formation of proeutectoid phases and pearlite, and how to calculate the relative amounts using the lever rule.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document discusses the iron-carbon phase diagram and the microstructures of steels. It describes the different phases in the diagram, including α-ferrite, γ-austenite, δ-ferrite, and cementite. It explains the eutectic and eutectoid reactions, and how the microstructure of hypoeutectoid, eutectoid, and hypereutectoid steels depends on their carbon content and heat treatment. Specifically, it discusses the formation of proeutectoid phases and pearlite, and how to calculate the relative amounts using the lever rule.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
19 views13 pagesPages From FeC
Pages From FeC
Uploaded by
Fuhad HasanThe document discusses the iron-carbon phase diagram and the microstructures of steels. It describes the different phases in the diagram, including α-ferrite, γ-austenite, δ-ferrite, and cementite. It explains the eutectic and eutectoid reactions, and how the microstructure of hypoeutectoid, eutectoid, and hypereutectoid steels depends on their carbon content and heat treatment. Specifically, it discusses the formation of proeutectoid phases and pearlite, and how to calculate the relative amounts using the lever rule.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 13
MSE 300 Materials Laboratory Procedures
Iron-Carbon Phase
Diagram (a review) see
Callister Chapter 9
University of Tennessee, Dept. of Materials Science and Engineering 1
MSE 300 Materials Laboratory Procedures
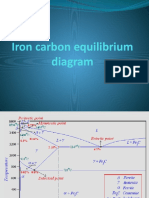
The Iron–Iron Carbide (Fe–Fe3C) Phase Diagram
In their simplest form, steels are alloys of Iron (Fe) and
Carbon (C). The Fe-C phase diagram is a fairly complex
one, but we will only consider the steel part of the diagram,
up to around 7% Carbon.
University of Tennessee, Dept. of Materials Science and Engineering 2
MSE 300 Materials Laboratory Procedures
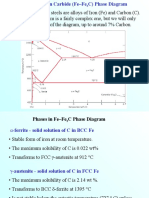
Phases in Fe–Fe3C Phase Diagram
¾ α-ferrite - solid solution of C in BCC Fe
• Stable form of iron at room temperature.
• The maximum solubility of C is 0.022 wt%
• Transforms to FCC γ-austenite at 912 °C
¾ γ-austenite - solid solution of C in FCC Fe
• The maximum solubility of C is 2.14 wt %.
• Transforms to BCC δ-ferrite at 1395 °C
• Is not stable below the eutectic temperature
(727 ° C) unless cooled rapidly (Chapter 10)
¾ δ-ferrite solid solution of C in BCC Fe
• The same structure as α-ferrite
• Stable only at high T, above 1394 °C
• Melts at 1538 °C
¾ Fe3C (iron carbide or cementite)
• This intermetallic compound is metastable, it
remains as a compound indefinitely at room T, but
decomposes (very slowly, within several years)
into α-Fe and C (graphite) at 650 - 700 °C
¾ Fe-C liquid solution
University of Tennessee, Dept. of Materials Science and Engineering 3
MSE 300 Materials Laboratory Procedures
A few comments on Fe–Fe3C system
C is an interstitial impurity in Fe. It forms a solid solution
with α, γ, δ phases of iron
Maximum solubility in BCC α-ferrite is limited (max.
0.022 wt% at 727 °C) - BCC has relatively small interstitial
positions
Maximum solubility in FCC austenite is 2.14 wt% at 1147
°C - FCC has larger interstitial positions
Mechanical properties: Cementite is very hard and brittle -
can strengthen steels. Mechanical properties also depend
on the microstructure, that is, how ferrite and cementite are
mixed.
Magnetic properties: α -ferrite is magnetic below 768 °C,
austenite is non-magnetic
Classification. Three types of ferrous alloys:
• Iron: less than 0.008 wt % C in α−ferrite at room T
• Steels: 0.008 - 2.14 wt % C (usually < 1 wt % )
α-ferrite + Fe3C at room T (Chapter 12)
• Cast iron: 2.14 - 6.7 wt % (usually < 4.5 wt %)
University of Tennessee, Dept. of Materials Science and Engineering 4
MSE 300 Materials Laboratory Procedures
Eutectic and eutectoid reactions in Fe–Fe3C
Eutectic: 4.30 wt% C, 1147 °C
L ↔ γ + Fe3C
Eutectoid: 0.76 wt%C, 727 °C
γ(0.76 wt% C) ↔ α (0.022 wt% C) + Fe3C
Eutectic and eutectoid reactions are very important in
heat treatment of steels
University of Tennessee, Dept. of Materials Science and Engineering 5
MSE 300 Materials Laboratory Procedures
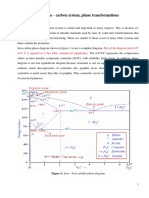
Development of Microstructure in Iron - Carbon alloys
Microstructure depends on composition (carbon
content) and heat treatment. In the discussion below we
consider slow cooling in which equilibrium is maintained.
Microstructure of eutectoid steel (I)
University of Tennessee, Dept. of Materials Science and Engineering 6
MSE 300 Materials Laboratory Procedures
Microstructure of eutectoid steel (II)
When alloy of eutectoid composition (0.76 wt % C) is
cooled slowly it forms perlite, a lamellar or layered
structure of two phases: α-ferrite and cementite (Fe3C)
The layers of alternating phases in pearlite are formed for
the same reason as layered structure of eutectic structures:
redistribution C atoms between ferrite (0.022 wt%) and
cementite (6.7 wt%) by atomic diffusion.
Mechanically, pearlite has properties intermediate to soft,
ductile ferrite and hard, brittle cementite.
In the micrograph, the dark areas are
Fe3C layers, the light phase is α-
ferrite
University of Tennessee, Dept. of Materials Science and Engineering 7
MSE 300 Materials Laboratory Procedures
Microstructure of hypoeutectoid steel (I)
Compositions to the left of eutectoid (0.022 - 0.76 wt % C)
hypoeutectoid (less than eutectoid -Greek) alloys.
γ → α + γ → α + Fe3C
University of Tennessee, Dept. of Materials Science and Engineering 8
MSE 300 Materials Laboratory Procedures
Microstructure of hypoeutectoid steel (II)
Hypoeutectoid alloys contain proeutectoid ferrite (formed
above the eutectoid temperature) plus the eutectoid perlite
that contain eutectoid ferrite and cementite.
University of Tennessee, Dept. of Materials Science and Engineering 9
MSE 300 Materials Laboratory Procedures
Microstructure of hypereutectoid steel (I)
Compositions to the right of eutectoid (0.76 - 2.14 wt % C)
hypereutectoid (more than eutectoid -Greek) alloys.
γ → γ + Fe3C → α + Fe3C
University of Tennessee, Dept. of Materials Science and Engineering 10
MSE 300 Materials Laboratory Procedures
Microstructure of hypereutectoid steel (II)
Hypereutectoid alloys contain proeutectoid cementite
(formed above the eutectoid temperature) plus perlite that
contain eutectoid ferrite and cementite.
University of Tennessee, Dept. of Materials Science and Engineering 11
MSE 300 Materials Laboratory Procedures
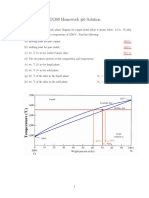
How to calculate the relative amounts of proeutectoid
phase (α or Fe3C) and pearlite?
Application of the lever rule with tie line that extends from
the eutectoid composition (0.76 wt% C) to α – (α + Fe3C)
boundary (0.022 wt% C) for hypoeutectoid alloys and to (α
+ Fe3C) – Fe3C boundary (6.7 wt% C) for hypereutectoid
alloys.
Fraction of α phase is determined by application of the
lever rule across
University the entire
of Tennessee, Dept.(α + Fe3C)
of Materials phase
Science field:
and Engineering 12
MSE 300 Materials Laboratory Procedures
Example for hypereutectoid alloy with composition C1
Fraction of pearlite:
WP = X / (V+X) = (6.7 – C1) / (6.7 – 0.76)
Fraction of proeutectoid cementite:
WFe3C = V / (V+X) = (C1 – 0.76) / (6.7 – 0.76)
University of Tennessee, Dept. of Materials Science and Engineering 13
You might also like
- Materials Data for Cyclic Loading: Aluminium and Titanium AlloysFrom EverandMaterials Data for Cyclic Loading: Aluminium and Titanium AlloysRating: 1 out of 5 stars1/5 (1)
- A Project Report On Microstructure Analysis of Gray Cast Iron, Aluminium and Brass Using Optical MicrographsDocument26 pagesA Project Report On Microstructure Analysis of Gray Cast Iron, Aluminium and Brass Using Optical MicrographsManvendra Pratap Singh Bisht100% (8)
- Iron-Carbon Phase Diagram (A Review) See Callister Chapter 9Document13 pagesIron-Carbon Phase Diagram (A Review) See Callister Chapter 9babiker mortadaNo ratings yet
- Fe-C Phase DiagramDocument34 pagesFe-C Phase DiagramYoung-long Choi100% (1)
- Iron-Carbon Phase Diagram (A Review) See Callister Chapter 9Document34 pagesIron-Carbon Phase Diagram (A Review) See Callister Chapter 9Zefa Erliana YullahNo ratings yet
- Fe CDocument34 pagesFe CZaza ArifinNo ratings yet
- Material Science and MettalurgyDocument21 pagesMaterial Science and MettalurgySR-71 BLACKBIRDNo ratings yet
- Iron-Iron Carbide (Fe-Fe3C) Phase Diagram: M. Tech. (FFT) Technology of Ferrous CastingDocument7 pagesIron-Iron Carbide (Fe-Fe3C) Phase Diagram: M. Tech. (FFT) Technology of Ferrous CastingRajulapati Sunil KumarNo ratings yet
- Fec DiagramDocument15 pagesFec DiagramShankarNo ratings yet
- Phase Diagrams - 040823Document23 pagesPhase Diagrams - 040823Anthony MubangaNo ratings yet
- 9 Engineering AlloysDocument17 pages9 Engineering AlloysdavidtomyNo ratings yet
- University of Babylon, College of Engineering, Engineering Metallurgy, Maithem H-RasheedDocument13 pagesUniversity of Babylon, College of Engineering, Engineering Metallurgy, Maithem H-RasheedAris BulaongNo ratings yet
- Iron Carbon DiagramDocument10 pagesIron Carbon DiagramsivakumarNo ratings yet
- Seminarski Rad Fe Fe3C DijagramDocument10 pagesSeminarski Rad Fe Fe3C DijagramAlina OmerovicNo ratings yet
- TTT 11Document55 pagesTTT 11MrDOTNo ratings yet
- 8.fe - Fe3C Phase DiagramDocument27 pages8.fe - Fe3C Phase DiagramMhackSahuNo ratings yet
- 18bem0046 Materials Da 2Document20 pages18bem0046 Materials Da 2QUANTUM SOLEOSNo ratings yet
- Iron Carbon Equilibrium DiagramDocument11 pagesIron Carbon Equilibrium Diagramganesh82No ratings yet
- Research of Phase Transformation On Fe-8.7Al-28.3Mn-1C-5.5Cr AlloyDocument4 pagesResearch of Phase Transformation On Fe-8.7Al-28.3Mn-1C-5.5Cr AlloyMarina PiermannNo ratings yet
- Fracture Dependence On Heat Treatment: Table 1-Dependence of Absorbed Energy (For Crack Propagation) Over TemperatureDocument4 pagesFracture Dependence On Heat Treatment: Table 1-Dependence of Absorbed Energy (For Crack Propagation) Over Temperaturelokomoko1No ratings yet
- Chapter Outline:: Heat Treatment (And Temperature)Document24 pagesChapter Outline:: Heat Treatment (And Temperature)DSGNo ratings yet
- 3 Fe-Fe3C Phase DiagramDocument33 pages3 Fe-Fe3C Phase DiagramRajat Mishra100% (1)
- Fe CdiagramDocument36 pagesFe CdiagramGeorge SingerNo ratings yet
- Fe-C Phase Diagram Lecture 33Document15 pagesFe-C Phase Diagram Lecture 33Manish Gupta100% (1)
- FALLSEM2019-20 MEE1005 ETH VL2019201001078 Reference Material I 29-Aug-2019 Fe-Fe3C Phase DiagramDocument33 pagesFALLSEM2019-20 MEE1005 ETH VL2019201001078 Reference Material I 29-Aug-2019 Fe-Fe3C Phase DiagramFazal KhanNo ratings yet
- Unit 3Document113 pagesUnit 3Abhishek ChavanNo ratings yet
- Carbon Steels: ME 215/205:materials Science LaboratoryDocument9 pagesCarbon Steels: ME 215/205:materials Science Laboratoryfaizan997No ratings yet
- Ch-27.5 Iron Carbon Equilibrium DiagramDocument52 pagesCh-27.5 Iron Carbon Equilibrium DiagramManojNo ratings yet
- Chapter 5, THE IRON-CARBON EQUILIBRIUM DIAGRAMDocument13 pagesChapter 5, THE IRON-CARBON EQUILIBRIUM DIAGRAMPAUL NDIRITUNo ratings yet
- What Is PearliteDocument4 pagesWhat Is Pearliteardy cornettoNo ratings yet
- The Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalDocument38 pagesThe Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalYogesh KumbharNo ratings yet
- Chapter Outline: Phase Transformations. KineticsDocument23 pagesChapter Outline: Phase Transformations. Kineticsmpcd07No ratings yet
- BG2802 Heat Treatment and Mechanical Properties of SteelsDocument11 pagesBG2802 Heat Treatment and Mechanical Properties of SteelsVenus LimNo ratings yet
- Iron-Carbon Phase Diagram: By: Awad Elaraby ID:052022009Document33 pagesIron-Carbon Phase Diagram: By: Awad Elaraby ID:052022009Mahmoud RefaatNo ratings yet
- Weldability of Metals - NPTELDocument18 pagesWeldability of Metals - NPTELKaushal Gandhi0% (1)
- Iron - Carbon Phase DiagramDocument19 pagesIron - Carbon Phase DiagramGaurav DograNo ratings yet
- Foundations of Materials Science and Engineering 5th Edition Smith Solutions ManualDocument39 pagesFoundations of Materials Science and Engineering 5th Edition Smith Solutions Manualcacoonnymphaea6wgyct100% (16)
- Foundations of Materials Science and Engineering 5th Edition Smith Solutions ManualDocument79 pagesFoundations of Materials Science and Engineering 5th Edition Smith Solutions Manualdextrermachete4amgqgNo ratings yet
- Dokumen - Tips - Iron Iron Carbide Phase Diagram 58ac3a092bd8dDocument16 pagesDokumen - Tips - Iron Iron Carbide Phase Diagram 58ac3a092bd8dAfrizal Adithya PNo ratings yet
- Ch-27.3 Iron Carbon Equilibrium DiagramDocument58 pagesCh-27.3 Iron Carbon Equilibrium DiagramasjfgauojfgfNo ratings yet
- Iron Carbon Phase DiagramDocument4 pagesIron Carbon Phase DiagramMizanur RahmanNo ratings yet
- 3 Iron Carbon DiaDocument21 pages3 Iron Carbon DiaChhavi SharmaNo ratings yet
- Sudipta Nath: Materials EngineeringDocument19 pagesSudipta Nath: Materials EngineeringSudipta NathNo ratings yet
- Industrial Materials (B)Document15 pagesIndustrial Materials (B)Muhammad Bilal SahiNo ratings yet
- Introduction To Fe-C Equilibrium Phase Diagram: Chia-Chang ShihDocument42 pagesIntroduction To Fe-C Equilibrium Phase Diagram: Chia-Chang ShihTuấnPhạmNo ratings yet
- Phase Diagram 3Document26 pagesPhase Diagram 3Tony StarkNo ratings yet
- Engineering Materials 27-29Document40 pagesEngineering Materials 27-29Sanu SouravNo ratings yet
- Materials of Construction and Selection: Faculty of Chemical Engineering Universiti Teknologi MaraDocument80 pagesMaterials of Construction and Selection: Faculty of Chemical Engineering Universiti Teknologi MaraAisyah Addia AzizanNo ratings yet
- Capili Jefferson 10Document5 pagesCapili Jefferson 10Christian Al EncarnacionNo ratings yet
- Pure Iron, Allotropy:: Steels Are Alloys of Iron and Carbon With Carbon Content Up To 2%Document13 pagesPure Iron, Allotropy:: Steels Are Alloys of Iron and Carbon With Carbon Content Up To 2%Abhishek SakatNo ratings yet
- Iron Carbon Note 1 2023Document23 pagesIron Carbon Note 1 2023gerrard samuelNo ratings yet
- 8.fe - Fe3C Phase Diagram PDFDocument27 pages8.fe - Fe3C Phase Diagram PDF13311A0341 S SHIVA SAI KIRANNo ratings yet
- How The Material Built For Special PurposeDocument43 pagesHow The Material Built For Special PurposeDidi SudiprayitnaNo ratings yet
- Iron Carbon Diagram of Steel PDFDocument6 pagesIron Carbon Diagram of Steel PDFshihabscb1971100% (1)
- Iron-Carbon DiagramDocument3 pagesIron-Carbon DiagramnaniNo ratings yet
- Heat TreatmentDocument4 pagesHeat Treatment정석원No ratings yet
- Control and Analysis in Iron and SteelmakingFrom EverandControl and Analysis in Iron and SteelmakingRating: 3 out of 5 stars3/5 (2)
- Proceedings of the 2014 Energy Materials Conference: Xi'an, Shaanxi Province, China, November 4 - 6, 2014From EverandProceedings of the 2014 Energy Materials Conference: Xi'an, Shaanxi Province, China, November 4 - 6, 2014No ratings yet
- Expansionism and Religion - The Fatal Flaws of The Aztec EmpireDocument27 pagesExpansionism and Religion - The Fatal Flaws of The Aztec EmpireFuhad HasanNo ratings yet
- Sugar Cane JuiceDocument17 pagesSugar Cane JuiceFuhad HasanNo ratings yet
- Date JuiceDocument8 pagesDate JuiceFuhad HasanNo ratings yet
- Computational Simulation of Concentration by OsmotDocument15 pagesComputational Simulation of Concentration by OsmotFuhad HasanNo ratings yet
- Iron Carbon Phase DiagramDocument4 pagesIron Carbon Phase DiagramFuhad HasanNo ratings yet
- Phase Diagram of Fe-Fe3CDocument25 pagesPhase Diagram of Fe-Fe3CIram MustaviNo ratings yet
- Sub Module-1 Aircraft-Ferrous Materials 1.1 Ferrous Aircraft MetalsDocument22 pagesSub Module-1 Aircraft-Ferrous Materials 1.1 Ferrous Aircraft MetalsDeepak Choudhary DCNo ratings yet
- Outokumpu Handbook of Stainless Steel 2017 EditionDocument92 pagesOutokumpu Handbook of Stainless Steel 2017 EditionRavindra BarangeNo ratings yet
- Fabrication Requirements of Pressure Vessel Coils and JumpovDocument7 pagesFabrication Requirements of Pressure Vessel Coils and JumpovAleem QureshiNo ratings yet
- Full-Range Stress-Strain Curves For Stainless SteelDocument15 pagesFull-Range Stress-Strain Curves For Stainless SteelNurul HudaNo ratings yet
- Resistance Upset Butt Welding of AusteniDocument7 pagesResistance Upset Butt Welding of AusteniRamzi BEN AHMEDNo ratings yet
- Properties of Engineering Materials Lesson 3 (I)Document11 pagesProperties of Engineering Materials Lesson 3 (I)Douglas Kufre-Abasi GilbertNo ratings yet
- Materials and Processes Quiz 1Document6 pagesMaterials and Processes Quiz 1RuthNo ratings yet
- A 488 - A 488M - 99 Qtq4oc05oq - PDFDocument15 pagesA 488 - A 488M - 99 Qtq4oc05oq - PDFMarceloNo ratings yet
- Materials For Ultra Supercritical Fossil Power Plants by R. ViswanathanDocument66 pagesMaterials For Ultra Supercritical Fossil Power Plants by R. ViswanathanHASAN IZHAR KHANNo ratings yet
- ME692 - Welding - Lecture Till 6 FebDocument107 pagesME692 - Welding - Lecture Till 6 Febpramrahar20No ratings yet
- Ese 2021: Prelims Exam: MechanicalDocument22 pagesEse 2021: Prelims Exam: MechanicalRamesh TammineniNo ratings yet
- Welding Journal 1960 4Document219 pagesWelding Journal 1960 4AlexeyNo ratings yet
- Dormer AplicacionesDocument188 pagesDormer Aplicacionesjserna_22No ratings yet
- A Comparative Evaluation of Welding Consumables For Dissimilar Welds Between 316LN Austenitic Stainless Steel and Alloy 800Document12 pagesA Comparative Evaluation of Welding Consumables For Dissimilar Welds Between 316LN Austenitic Stainless Steel and Alloy 800Élida AlvesNo ratings yet
- 1 Dec2008Document9 pages1 Dec2008el_sharkawy2011No ratings yet
- Study of Microstructure Degradation of Boiler Tubes Due To Creep For Remaining Life AnalysisDocument7 pagesStudy of Microstructure Degradation of Boiler Tubes Due To Creep For Remaining Life AnalysisRaka DaffaNo ratings yet
- Chapter 8 - Stainless Steels - 2016 - Applied Welding EngineeringDocument8 pagesChapter 8 - Stainless Steels - 2016 - Applied Welding EngineeringJhair Jhamidh Aquino PortocarreroNo ratings yet
- Introduction-Iron Carbon Phase DiagramDocument31 pagesIntroduction-Iron Carbon Phase DiagramTHE BBEASTNo ratings yet
- Nsba Night School Course B1 Session 3 PDFDocument51 pagesNsba Night School Course B1 Session 3 PDFLuis CortesNo ratings yet
- Steel Casting MetallurgyDocument258 pagesSteel Casting MetallurgySiddharth Gupta86% (7)
- Lec 4. Solid Solution - Phase DiagramsDocument27 pagesLec 4. Solid Solution - Phase DiagramsTrí Tạ MinhNo ratings yet
- BME (Steel)Document8 pagesBME (Steel)Mohil JainNo ratings yet
- Low Temperature Embrittlement Studies On Stainless Steel 304 LN TIG WeldsDocument10 pagesLow Temperature Embrittlement Studies On Stainless Steel 304 LN TIG WeldsRajmchzNo ratings yet
- Ferro Fundido Nodular Normas PortDocument5 pagesFerro Fundido Nodular Normas PortCarlos LaoNo ratings yet
- EN380 Homework #6 Solution: 1084 C 1455 C 70% N i L + α C = 61% C = 73% W = = = 25% W = = = 75%Document4 pagesEN380 Homework #6 Solution: 1084 C 1455 C 70% N i L + α C = 61% C = 73% W = = = 25% W = = = 75%roseNo ratings yet
- M GN 365Document46 pagesM GN 365reza329329No ratings yet
- Austenitic Stainless SteelsDocument4 pagesAustenitic Stainless Steelsandy1036No ratings yet
- Wrought 18Chromium-14Nickel-2.5Molybdenum Stainless Steel Sheet and Strip For Surgical Implants (UNS S31673)Document4 pagesWrought 18Chromium-14Nickel-2.5Molybdenum Stainless Steel Sheet and Strip For Surgical Implants (UNS S31673)Angel ChangNo ratings yet