Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
7 viewsXI 21-22 Term 2 Practical Q Paper
XI 21-22 Term 2 Practical Q Paper
Uploaded by
Joshua ZongteThis document provides instructions for a Chemistry practical exam for Class 11 students at Kendriya Vidyalaya Mizoram University in Aizawl. The exam consists of 3 parts worth a total of 15 marks: salt analysis (8 marks), volumetric analysis (4 marks), and a content-based experiment (2 marks). Students must also be evaluated on their class record and project during a viva worth 5 marks. Calculators are not allowed and log tables can be used.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- CHEMISTRY Manual With BlanksDocument73 pagesCHEMISTRY Manual With BlankssohiytkhantwalNo ratings yet
- EngineeringChemistryLabManual2017 18Document53 pagesEngineeringChemistryLabManual2017 18Gabbar PatelNo ratings yet
- Industrial Production of Iron OreDocument19 pagesIndustrial Production of Iron OreChandrakumar87No ratings yet
- Distribution of Topics For Mid-Term, First-Term and Second-Term (From June 2016 0nwards) Subject: CHEMISTRYDocument5 pagesDistribution of Topics For Mid-Term, First-Term and Second-Term (From June 2016 0nwards) Subject: CHEMISTRYvishweshwara hollaNo ratings yet
- CBSE Class 11 Chemistry Practicals 2023 24Document3 pagesCBSE Class 11 Chemistry Practicals 2023 24emergents.17No ratings yet
- Bchy101p Engineering-Chemistry-Lab Lo 1.0 65 Bchy101pDocument2 pagesBchy101p Engineering-Chemistry-Lab Lo 1.0 65 Bchy101pMinaNo ratings yet
- CHEM F110 Chemistry Laboratory I Sem 2022-23HODocument2 pagesCHEM F110 Chemistry Laboratory I Sem 2022-23HOjohn doeNo ratings yet
- Thực Tập Phân TíchDocument128 pagesThực Tập Phân TíchTrần Trọng Dũng0% (1)
- MARISEAL 300 Potable Water Certification From SingaporeDocument6 pagesMARISEAL 300 Potable Water Certification From SingaporeVirender KumarNo ratings yet
- Inquiry Based Chem ExptDocument41 pagesInquiry Based Chem ExptchanchusangNo ratings yet
- B P of Class XII Practical Term I Term IIDocument6 pagesB P of Class XII Practical Term I Term IIajaleomkarNo ratings yet
- CEE 214L Engineering Materials Laboratory ManualDocument45 pagesCEE 214L Engineering Materials Laboratory ManualcheggNo ratings yet
- Practice Test 09 - Test Schedule & Syllabus (19 - 11 - 2023) - (Lakshya JEE 2024)Document2 pagesPractice Test 09 - Test Schedule & Syllabus (19 - 11 - 2023) - (Lakshya JEE 2024)gpt1410No ratings yet
- Chemistry OlympiadDocument111 pagesChemistry OlympiadRitvik AroraNo ratings yet
- Sample 12436Document16 pagesSample 12436Adhi AdhiNo ratings yet
- CY 111 COURSE EVALUATION PLAN Jan 2024Document3 pagesCY 111 COURSE EVALUATION PLAN Jan 2024Siddharth SinghNo ratings yet
- PU 5th SEM SYLLABUSDocument22 pagesPU 5th SEM SYLLABUSSudeep KhadkaNo ratings yet
- Federal Board of Intermediate and Secondary Education H-8/4, IslamabadDocument5 pagesFederal Board of Intermediate and Secondary Education H-8/4, IslamabadhassanjoehanamNo ratings yet
- 5.basics of Electrical & Electronics Engg. LabDocument11 pages5.basics of Electrical & Electronics Engg. LabNIKHIL ASNo ratings yet
- Materials Testing LabDocument3 pagesMaterials Testing LabBenNo ratings yet
- Laboratory Manual PDFDocument81 pagesLaboratory Manual PDFaman badashaalam100% (1)
- Chemistry Pre-1st YearT1 Chapter 1Document1 pageChemistry Pre-1st YearT1 Chapter 1Muhammad RizwanNo ratings yet
- GATE 2020 New PDFDocument32 pagesGATE 2020 New PDFmayankNo ratings yet
- Mos - Lab ManualDocument80 pagesMos - Lab ManualDr. Ashok Kr DargarNo ratings yet
- Board Practical QP PDFDocument1 pageBoard Practical QP PDFkeerthivarmanbaranirajNo ratings yet
- Meta Lab ManualDocument66 pagesMeta Lab Manualharshal wasnik100% (1)
- Mandvi Rice MillsDocument1 pageMandvi Rice MillsGyaan Disha To choose right pathNo ratings yet
- CE 431 Construction Materials Laboratory - 3Document5 pagesCE 431 Construction Materials Laboratory - 3Hasib DoskeNo ratings yet
- Som Lab Manual Nnce PDFDocument39 pagesSom Lab Manual Nnce PDFObakoma JosiahNo ratings yet
- Gate CEDocument33 pagesGate CENayan jain0% (1)
- Student Hand Book Chemistry LabDocument9 pagesStudent Hand Book Chemistry LabpradyumnatpoNo ratings yet
- 14Cv35 - Building Materials and Construction (Lab Component)Document30 pages14Cv35 - Building Materials and Construction (Lab Component)Bishwombhar Prakash Pandit100% (3)
- CCIL Requirements For Aggregate Laboratory Technican GL Final May 11 2017Document1 pageCCIL Requirements For Aggregate Laboratory Technican GL Final May 11 2017CedricChanNo ratings yet
- Syllabus: PhysicsDocument2 pagesSyllabus: PhysicssannosxnNo ratings yet
- AISSCE PRACTICAL EXAMINATIO1 Final 2Document1 pageAISSCE PRACTICAL EXAMINATIO1 Final 2pateljomi6No ratings yet
- Experiment 8 Data SheetDocument8 pagesExperiment 8 Data SheetCrecia Bullecer2No ratings yet
- Engineering Chemistry Lab: Page 1 of 1Document1 pageEngineering Chemistry Lab: Page 1 of 1asha_989529794No ratings yet
- Syllabus AIML 2nd SemDocument9 pagesSyllabus AIML 2nd Semhr71carNo ratings yet
- 18CVL38 Building Materials Testing LaboratoryDocument71 pages18CVL38 Building Materials Testing LaboratoryKarthik. HNo ratings yet
- School of Chemistry CHM1051 Lab Manual Chemistry I Advanced 2020Document68 pagesSchool of Chemistry CHM1051 Lab Manual Chemistry I Advanced 2020flat furNo ratings yet
- Revised & Final Engg. Chemistry Lab Manual II SEM April 2023Document34 pagesRevised & Final Engg. Chemistry Lab Manual II SEM April 2023surakantianuragNo ratings yet
- CHEMISTRY PRACTICAL SSC-23 RevisedDocument4 pagesCHEMISTRY PRACTICAL SSC-23 Revisedtalhaaslam7516034No ratings yet
- Practice Test 01 - Test Schedule & Syllabus (02 - 07 - 2023) - Arjuna JEE 2.0 2024Document2 pagesPractice Test 01 - Test Schedule & Syllabus (02 - 07 - 2023) - Arjuna JEE 2.0 2024Subhankar BorahNo ratings yet
- Session 2017 - Particle Technology (Chemical Engineering) PDFDocument5 pagesSession 2017 - Particle Technology (Chemical Engineering) PDFahmadNo ratings yet
- Overview of Engg Chemistry: Assistant Professor, Dept of Applied Chemistry, Aias, Amity University, NoidaDocument14 pagesOverview of Engg Chemistry: Assistant Professor, Dept of Applied Chemistry, Aias, Amity University, Noidagaurav toppoNo ratings yet
- LTC Vaidyah-24 - Set - 1 & 2 - Expt Skills SyllabusDocument1 pageLTC Vaidyah-24 - Set - 1 & 2 - Expt Skills Syllabussai982005No ratings yet
- sampletask-scicom_chem-e-200110Document17 pagessampletask-scicom_chem-e-200110Oditor MwaangaNo ratings yet
- 2nd Year Scheme Syllabus EEEDocument62 pages2nd Year Scheme Syllabus EEENikhil KumarNo ratings yet
- Ade Final Manual PDFDocument59 pagesAde Final Manual PDFRam Krishna JhaNo ratings yet
- Class 12Document3 pagesClass 12Management HODNo ratings yet
- Chemistry Lab Manual VTU 1st YearDocument53 pagesChemistry Lab Manual VTU 1st YearRitvik RaiNo ratings yet
- PCT-03 PDocument29 pagesPCT-03 PcrezsathishNo ratings yet
- Scheme of Chemistry Practical ExaminationDocument5 pagesScheme of Chemistry Practical ExaminationSubhasish ChakrabortyNo ratings yet
- Experiments ListDocument4 pagesExperiments ListYukiyo LangevinNo ratings yet
- Final ReportDocument7 pagesFinal ReportDinesh SaiNo ratings yet
- Ochem Lab SyllabusDocument6 pagesOchem Lab SyllabuslolaNo ratings yet
- Key Comparison CCQM-K34 Assay of Potassium Hydrogen PhthalateDocument12 pagesKey Comparison CCQM-K34 Assay of Potassium Hydrogen PhthalatemariassyNo ratings yet
- Met 312 NDTDocument8 pagesMet 312 NDTJanit JijiNo ratings yet
- Xi PT Phy 2021 22Document3 pagesXi PT Phy 2021 22Joshua ZongteNo ratings yet
- XII - Autumn Break 2022 - Holiday HomeworkDocument1 pageXII - Autumn Break 2022 - Holiday HomeworkJoshua ZongteNo ratings yet
- Class XI PH - Edu Term 2 PaperDocument1 pageClass XI PH - Edu Term 2 PaperJoshua ZongteNo ratings yet
- Xii Phy Ab HW 2022 23Document1 pageXii Phy Ab HW 2022 23Joshua ZongteNo ratings yet
- Xi See Bio 2022 QP Set 1 Surender SharmaDocument4 pagesXi See Bio 2022 QP Set 1 Surender SharmaJoshua ZongteNo ratings yet
- Xi - Chemistry - QPDocument3 pagesXi - Chemistry - QPJoshua ZongteNo ratings yet
- Chemistry ProjectDocument18 pagesChemistry ProjectJoshua ZongteNo ratings yet
- Class Xi Biology Ptii QuestionDocument3 pagesClass Xi Biology Ptii QuestionJoshua ZongteNo ratings yet
- Chemistry Investigatory Project 2020.pdf by ChirayuDocument13 pagesChemistry Investigatory Project 2020.pdf by ChirayuJoshua ZongteNo ratings yet
- Chemistry XI PT-2 Final PAPERDocument3 pagesChemistry XI PT-2 Final PAPERJoshua ZongteNo ratings yet
XI 21-22 Term 2 Practical Q Paper
XI 21-22 Term 2 Practical Q Paper
Uploaded by
Joshua Zongte0 ratings0% found this document useful (0 votes)
7 views1 pageThis document provides instructions for a Chemistry practical exam for Class 11 students at Kendriya Vidyalaya Mizoram University in Aizawl. The exam consists of 3 parts worth a total of 15 marks: salt analysis (8 marks), volumetric analysis (4 marks), and a content-based experiment (2 marks). Students must also be evaluated on their class record and project during a viva worth 5 marks. Calculators are not allowed and log tables can be used.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides instructions for a Chemistry practical exam for Class 11 students at Kendriya Vidyalaya Mizoram University in Aizawl. The exam consists of 3 parts worth a total of 15 marks: salt analysis (8 marks), volumetric analysis (4 marks), and a content-based experiment (2 marks). Students must also be evaluated on their class record and project during a viva worth 5 marks. Calculators are not allowed and log tables can be used.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
7 views1 pageXI 21-22 Term 2 Practical Q Paper
XI 21-22 Term 2 Practical Q Paper
Uploaded by
Joshua ZongteThis document provides instructions for a Chemistry practical exam for Class 11 students at Kendriya Vidyalaya Mizoram University in Aizawl. The exam consists of 3 parts worth a total of 15 marks: salt analysis (8 marks), volumetric analysis (4 marks), and a content-based experiment (2 marks). Students must also be evaluated on their class record and project during a viva worth 5 marks. Calculators are not allowed and log tables can be used.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
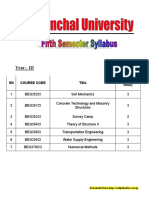
KENDRIYA VIDYALAYA MIZORAM UNIVERSITY AIZAWL
AISSSC (CBSE) Practical Exam-2021-22 TERM 2
SUBJECT- CHEMISTRY (043)
Time- 03 hrs. CLASS- XI Max. Marks 15
Evaluation Scheme for examination Salt Analysis- 08 Marks
Content Based Experiment- 02 Marks
Class Record & Project, Viva- 05 Marks
General Instructions
: -Attempt one question from each part.
:- Use log tables if necessary, use of calculator is not allowed.
Part- A (Salt Analysis) 8 Marks
1. Identify the cation and anion in the given salt sample.
Part- B (Volumetric Analysis) 4 Marks
2. Prepare a crystal of pure copper sulphate from given impure sample of Blue vitriol.
Part- C (Content Based Experiment) 2 Marks
3. Class Record & Viva
Examiner
ANOOP KUMAR
PGT CHEMISTRY
KV MZU AIZAWL
You might also like
- CHEMISTRY Manual With BlanksDocument73 pagesCHEMISTRY Manual With BlankssohiytkhantwalNo ratings yet
- EngineeringChemistryLabManual2017 18Document53 pagesEngineeringChemistryLabManual2017 18Gabbar PatelNo ratings yet
- Industrial Production of Iron OreDocument19 pagesIndustrial Production of Iron OreChandrakumar87No ratings yet
- Distribution of Topics For Mid-Term, First-Term and Second-Term (From June 2016 0nwards) Subject: CHEMISTRYDocument5 pagesDistribution of Topics For Mid-Term, First-Term and Second-Term (From June 2016 0nwards) Subject: CHEMISTRYvishweshwara hollaNo ratings yet
- CBSE Class 11 Chemistry Practicals 2023 24Document3 pagesCBSE Class 11 Chemistry Practicals 2023 24emergents.17No ratings yet
- Bchy101p Engineering-Chemistry-Lab Lo 1.0 65 Bchy101pDocument2 pagesBchy101p Engineering-Chemistry-Lab Lo 1.0 65 Bchy101pMinaNo ratings yet
- CHEM F110 Chemistry Laboratory I Sem 2022-23HODocument2 pagesCHEM F110 Chemistry Laboratory I Sem 2022-23HOjohn doeNo ratings yet
- Thực Tập Phân TíchDocument128 pagesThực Tập Phân TíchTrần Trọng Dũng0% (1)
- MARISEAL 300 Potable Water Certification From SingaporeDocument6 pagesMARISEAL 300 Potable Water Certification From SingaporeVirender KumarNo ratings yet
- Inquiry Based Chem ExptDocument41 pagesInquiry Based Chem ExptchanchusangNo ratings yet
- B P of Class XII Practical Term I Term IIDocument6 pagesB P of Class XII Practical Term I Term IIajaleomkarNo ratings yet
- CEE 214L Engineering Materials Laboratory ManualDocument45 pagesCEE 214L Engineering Materials Laboratory ManualcheggNo ratings yet
- Practice Test 09 - Test Schedule & Syllabus (19 - 11 - 2023) - (Lakshya JEE 2024)Document2 pagesPractice Test 09 - Test Schedule & Syllabus (19 - 11 - 2023) - (Lakshya JEE 2024)gpt1410No ratings yet
- Chemistry OlympiadDocument111 pagesChemistry OlympiadRitvik AroraNo ratings yet
- Sample 12436Document16 pagesSample 12436Adhi AdhiNo ratings yet
- CY 111 COURSE EVALUATION PLAN Jan 2024Document3 pagesCY 111 COURSE EVALUATION PLAN Jan 2024Siddharth SinghNo ratings yet
- PU 5th SEM SYLLABUSDocument22 pagesPU 5th SEM SYLLABUSSudeep KhadkaNo ratings yet
- Federal Board of Intermediate and Secondary Education H-8/4, IslamabadDocument5 pagesFederal Board of Intermediate and Secondary Education H-8/4, IslamabadhassanjoehanamNo ratings yet
- 5.basics of Electrical & Electronics Engg. LabDocument11 pages5.basics of Electrical & Electronics Engg. LabNIKHIL ASNo ratings yet
- Materials Testing LabDocument3 pagesMaterials Testing LabBenNo ratings yet
- Laboratory Manual PDFDocument81 pagesLaboratory Manual PDFaman badashaalam100% (1)
- Chemistry Pre-1st YearT1 Chapter 1Document1 pageChemistry Pre-1st YearT1 Chapter 1Muhammad RizwanNo ratings yet
- GATE 2020 New PDFDocument32 pagesGATE 2020 New PDFmayankNo ratings yet
- Mos - Lab ManualDocument80 pagesMos - Lab ManualDr. Ashok Kr DargarNo ratings yet
- Board Practical QP PDFDocument1 pageBoard Practical QP PDFkeerthivarmanbaranirajNo ratings yet
- Meta Lab ManualDocument66 pagesMeta Lab Manualharshal wasnik100% (1)
- Mandvi Rice MillsDocument1 pageMandvi Rice MillsGyaan Disha To choose right pathNo ratings yet
- CE 431 Construction Materials Laboratory - 3Document5 pagesCE 431 Construction Materials Laboratory - 3Hasib DoskeNo ratings yet
- Som Lab Manual Nnce PDFDocument39 pagesSom Lab Manual Nnce PDFObakoma JosiahNo ratings yet
- Gate CEDocument33 pagesGate CENayan jain0% (1)
- Student Hand Book Chemistry LabDocument9 pagesStudent Hand Book Chemistry LabpradyumnatpoNo ratings yet
- 14Cv35 - Building Materials and Construction (Lab Component)Document30 pages14Cv35 - Building Materials and Construction (Lab Component)Bishwombhar Prakash Pandit100% (3)
- CCIL Requirements For Aggregate Laboratory Technican GL Final May 11 2017Document1 pageCCIL Requirements For Aggregate Laboratory Technican GL Final May 11 2017CedricChanNo ratings yet
- Syllabus: PhysicsDocument2 pagesSyllabus: PhysicssannosxnNo ratings yet
- AISSCE PRACTICAL EXAMINATIO1 Final 2Document1 pageAISSCE PRACTICAL EXAMINATIO1 Final 2pateljomi6No ratings yet
- Experiment 8 Data SheetDocument8 pagesExperiment 8 Data SheetCrecia Bullecer2No ratings yet
- Engineering Chemistry Lab: Page 1 of 1Document1 pageEngineering Chemistry Lab: Page 1 of 1asha_989529794No ratings yet
- Syllabus AIML 2nd SemDocument9 pagesSyllabus AIML 2nd Semhr71carNo ratings yet
- 18CVL38 Building Materials Testing LaboratoryDocument71 pages18CVL38 Building Materials Testing LaboratoryKarthik. HNo ratings yet
- School of Chemistry CHM1051 Lab Manual Chemistry I Advanced 2020Document68 pagesSchool of Chemistry CHM1051 Lab Manual Chemistry I Advanced 2020flat furNo ratings yet
- Revised & Final Engg. Chemistry Lab Manual II SEM April 2023Document34 pagesRevised & Final Engg. Chemistry Lab Manual II SEM April 2023surakantianuragNo ratings yet
- CHEMISTRY PRACTICAL SSC-23 RevisedDocument4 pagesCHEMISTRY PRACTICAL SSC-23 Revisedtalhaaslam7516034No ratings yet
- Practice Test 01 - Test Schedule & Syllabus (02 - 07 - 2023) - Arjuna JEE 2.0 2024Document2 pagesPractice Test 01 - Test Schedule & Syllabus (02 - 07 - 2023) - Arjuna JEE 2.0 2024Subhankar BorahNo ratings yet
- Session 2017 - Particle Technology (Chemical Engineering) PDFDocument5 pagesSession 2017 - Particle Technology (Chemical Engineering) PDFahmadNo ratings yet
- Overview of Engg Chemistry: Assistant Professor, Dept of Applied Chemistry, Aias, Amity University, NoidaDocument14 pagesOverview of Engg Chemistry: Assistant Professor, Dept of Applied Chemistry, Aias, Amity University, Noidagaurav toppoNo ratings yet
- LTC Vaidyah-24 - Set - 1 & 2 - Expt Skills SyllabusDocument1 pageLTC Vaidyah-24 - Set - 1 & 2 - Expt Skills Syllabussai982005No ratings yet
- sampletask-scicom_chem-e-200110Document17 pagessampletask-scicom_chem-e-200110Oditor MwaangaNo ratings yet
- 2nd Year Scheme Syllabus EEEDocument62 pages2nd Year Scheme Syllabus EEENikhil KumarNo ratings yet
- Ade Final Manual PDFDocument59 pagesAde Final Manual PDFRam Krishna JhaNo ratings yet
- Class 12Document3 pagesClass 12Management HODNo ratings yet
- Chemistry Lab Manual VTU 1st YearDocument53 pagesChemistry Lab Manual VTU 1st YearRitvik RaiNo ratings yet
- PCT-03 PDocument29 pagesPCT-03 PcrezsathishNo ratings yet
- Scheme of Chemistry Practical ExaminationDocument5 pagesScheme of Chemistry Practical ExaminationSubhasish ChakrabortyNo ratings yet
- Experiments ListDocument4 pagesExperiments ListYukiyo LangevinNo ratings yet
- Final ReportDocument7 pagesFinal ReportDinesh SaiNo ratings yet
- Ochem Lab SyllabusDocument6 pagesOchem Lab SyllabuslolaNo ratings yet
- Key Comparison CCQM-K34 Assay of Potassium Hydrogen PhthalateDocument12 pagesKey Comparison CCQM-K34 Assay of Potassium Hydrogen PhthalatemariassyNo ratings yet
- Met 312 NDTDocument8 pagesMet 312 NDTJanit JijiNo ratings yet
- Xi PT Phy 2021 22Document3 pagesXi PT Phy 2021 22Joshua ZongteNo ratings yet
- XII - Autumn Break 2022 - Holiday HomeworkDocument1 pageXII - Autumn Break 2022 - Holiday HomeworkJoshua ZongteNo ratings yet
- Class XI PH - Edu Term 2 PaperDocument1 pageClass XI PH - Edu Term 2 PaperJoshua ZongteNo ratings yet
- Xii Phy Ab HW 2022 23Document1 pageXii Phy Ab HW 2022 23Joshua ZongteNo ratings yet
- Xi See Bio 2022 QP Set 1 Surender SharmaDocument4 pagesXi See Bio 2022 QP Set 1 Surender SharmaJoshua ZongteNo ratings yet
- Xi - Chemistry - QPDocument3 pagesXi - Chemistry - QPJoshua ZongteNo ratings yet
- Chemistry ProjectDocument18 pagesChemistry ProjectJoshua ZongteNo ratings yet
- Class Xi Biology Ptii QuestionDocument3 pagesClass Xi Biology Ptii QuestionJoshua ZongteNo ratings yet
- Chemistry Investigatory Project 2020.pdf by ChirayuDocument13 pagesChemistry Investigatory Project 2020.pdf by ChirayuJoshua ZongteNo ratings yet
- Chemistry XI PT-2 Final PAPERDocument3 pagesChemistry XI PT-2 Final PAPERJoshua ZongteNo ratings yet