Professional Documents
Culture Documents
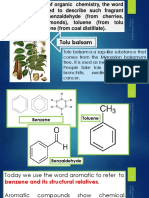
Aromatic Hydrocarbons
Aromatic Hydrocarbons
Uploaded by
Princess Mae EstabilloCopyright:
Available Formats
You might also like
- Enzyme Assays A Practical Approach by Robert Eisenthal, Michael DansonDocument304 pagesEnzyme Assays A Practical Approach by Robert Eisenthal, Michael DansonSara OchoaNo ratings yet
- BenzeneDocument14 pagesBenzeneJueeli More100% (1)
- Aromatic Hydrocarbons Pharmaceutical Organic Chemistry Porg111Document9 pagesAromatic Hydrocarbons Pharmaceutical Organic Chemistry Porg111AnnaGueseNo ratings yet
- Organic Chemistry Module 4Document5 pagesOrganic Chemistry Module 4Josephine TeroNo ratings yet
- Unsaturated Hydrocarbons Alkenes and Alkynes Aromatic CompoundDocument4 pagesUnsaturated Hydrocarbons Alkenes and Alkynes Aromatic CompoundGNo ratings yet
- Geometrical Isomerism: It is known that a carbon-carbon double bond is made up of one σ bond and one π-bond. The π-bond presents free rotation about the double bond.ThisDocument3 pagesGeometrical Isomerism: It is known that a carbon-carbon double bond is made up of one σ bond and one π-bond. The π-bond presents free rotation about the double bond.ThisKisha KhuranaNo ratings yet
- Chemistry Doc 3: Hydrocarbons Lewis Structures, Ball-And-Stick Models, and Space-Filling Models For MoleculesDocument3 pagesChemistry Doc 3: Hydrocarbons Lewis Structures, Ball-And-Stick Models, and Space-Filling Models For MoleculesKrishia Mae IlardeNo ratings yet
- Hydrocarbon S: Group 3: Leader: Angela Dela Cruz Daphne Sy Sofia Anne Caoagas Alen Gem Red Christian Faith ColisDocument16 pagesHydrocarbon S: Group 3: Leader: Angela Dela Cruz Daphne Sy Sofia Anne Caoagas Alen Gem Red Christian Faith ColisDaphne SyNo ratings yet
- Aromatic Hydrocarbons ReportDocument15 pagesAromatic Hydrocarbons ReportLyssa LimNo ratings yet
- UNIT 10 Organic ChemistryDocument51 pagesUNIT 10 Organic ChemistryTristan PereyNo ratings yet
- 8.1 - Learning Material-2Document49 pages8.1 - Learning Material-2Erica PagulayanNo ratings yet
- Organic 23 24Document18 pagesOrganic 23 24VigneshNo ratings yet
- (8.1) Learning MaterialDocument41 pages(8.1) Learning MaterialLuna VasquezNo ratings yet
- Topic 8 AlkenesDocument17 pagesTopic 8 Alkenesmark regino TuyayNo ratings yet
- Term Report: Submitted byDocument11 pagesTerm Report: Submitted byMUHAMMAD OMAIR AAMIR RAJANo ratings yet
- Nomenclature of Organic CompoundDocument10 pagesNomenclature of Organic CompounddeepashriguddaNo ratings yet
- Hydrocarbons NotesDocument15 pagesHydrocarbons Notesarjunrkumar2024No ratings yet
- ALKENEDocument11 pagesALKENEPrincess Mae EstabilloNo ratings yet
- Benzene: Milton Biswas Student Madhav University, Abu RoadDocument23 pagesBenzene: Milton Biswas Student Madhav University, Abu RoadMilton BiswasNo ratings yet
- W4 BENZENE Nomenclature Stability Physical & Chemical Properties PDFDocument22 pagesW4 BENZENE Nomenclature Stability Physical & Chemical Properties PDFathyrahNo ratings yet
- WEEK 10 Aromatic HydrocarbonDocument26 pagesWEEK 10 Aromatic HydrocarbonChris Angelo De GuzmanNo ratings yet
- Aromatic CompoundsDocument16 pagesAromatic CompoundsJotillnaimNo ratings yet
- Carbon Compounds: Presenter: Shannon SmithDocument61 pagesCarbon Compounds: Presenter: Shannon SmithShannon SmithNo ratings yet
- Cape Chemistry Unit 2 ReviewDocument7 pagesCape Chemistry Unit 2 Reviewkayanna100% (1)
- Assingment On Nomenclature OF Organic Compound: Submitted To: DR - Shobha Thakur Submitted By: Ritik Dwivedi 18BSPCM020Document30 pagesAssingment On Nomenclature OF Organic Compound: Submitted To: DR - Shobha Thakur Submitted By: Ritik Dwivedi 18BSPCM020ritik dwivediNo ratings yet
- AttachmentDocument14 pagesAttachmentaberaendale334No ratings yet
- Organic ChemistryDocument94 pagesOrganic ChemistryVrisanNo ratings yet
- +++fictionOrganic U-2 classes and name.docxDocument30 pages+++fictionOrganic U-2 classes and name.docxaberaendale334No ratings yet
- Lecture Nine Organic ChemistryDocument11 pagesLecture Nine Organic Chemistryabdihakimhasssan344No ratings yet
- Hydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSEDocument1 pageHydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSEjaindhruv850No ratings yet
- Organic CompoundsDocument22 pagesOrganic CompoundsMary Rose AguilaNo ratings yet
- Organic Chem U-2Document19 pagesOrganic Chem U-2sinte beyuNo ratings yet
- Organic ChemistryDocument70 pagesOrganic ChemistryGhulam Rabbani100% (1)
- organic chemistryDocument27 pagesorganic chemistryAnn SunilNo ratings yet
- Organic Compounds EditedDocument52 pagesOrganic Compounds EditedbrendonNo ratings yet
- Organic Chemistry LecturesDocument32 pagesOrganic Chemistry LecturesAbdulHameedNo ratings yet
- Corrected Fundamentals of Organic ChemistryDocument71 pagesCorrected Fundamentals of Organic ChemistryDAM2120No ratings yet
- Aromatic It yDocument8 pagesAromatic It ybalweg mackyNo ratings yet
- Organic ChemistryDocument30 pagesOrganic ChemistryMukhilNo ratings yet
- Nouman (CHEM 11)Document6 pagesNouman (CHEM 11)Junaid AkhtarNo ratings yet
- Pharmaceutical Chemistry Chapter 4 Organic Chemisty NotesDocument10 pagesPharmaceutical Chemistry Chapter 4 Organic Chemisty Notesbf327626No ratings yet
- Chapter 4 Aromatic HydrocarbonsDocument34 pagesChapter 4 Aromatic HydrocarbonsAbdirashid Adam IsakNo ratings yet
- Benzene & Its Derivatives: (The Class of Compounds, Which Contains Benzene or Substituted Derivative of Benzene)Document17 pagesBenzene & Its Derivatives: (The Class of Compounds, Which Contains Benzene or Substituted Derivative of Benzene)justin youngNo ratings yet
- Benzene Aromatic CompoundsDocument14 pagesBenzene Aromatic CompoundsBook of Life fgfhfghfghfghNo ratings yet
- Publication 10 22989 1587Document36 pagesPublication 10 22989 1587Ahsajib MatubberNo ratings yet
- Organic Chem U-2 Functional GroupDocument34 pagesOrganic Chem U-2 Functional Groupsinte beyuNo ratings yet
- Aromatic It yDocument9 pagesAromatic It yBentenBentenNo ratings yet
- Organic Chemistry Lecture 2 2022 Modif.Document60 pagesOrganic Chemistry Lecture 2 2022 Modif.Fady FadyNo ratings yet
- Chapter 4Document38 pagesChapter 4iB13eNo ratings yet
- Aromatic CompoundsDocument7 pagesAromatic Compoundsmanifest4godNo ratings yet
- Module 1. The Chemistry of Carbon Compounds TOPIC: Structure and FormulaeDocument38 pagesModule 1. The Chemistry of Carbon Compounds TOPIC: Structure and FormulaeOrlanda EllisNo ratings yet
- Organic Chemistry: Alkenes, Alkynes, and Aromatic CompoundsDocument43 pagesOrganic Chemistry: Alkenes, Alkynes, and Aromatic CompoundsDarius Gan100% (1)
- Alkenes and AlkynesDocument28 pagesAlkenes and AlkynesDorama AikaNo ratings yet
- Hydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSEDocument16 pagesHydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSERishabh Singh RajputNo ratings yet
- Organic ChemistryDocument55 pagesOrganic ChemistryKim Taeha BTSNo ratings yet
- 1) Reading Material by Nvs TeacherDocument34 pages1) Reading Material by Nvs Teacherjkc collegeNo ratings yet
- BENZENEDocument37 pagesBENZENEMaureen Galingan100% (1)
- LEC 19 Nomen-Culture and Structure of BenzeneDocument16 pagesLEC 19 Nomen-Culture and Structure of BenzeneIftikhar Ud DinNo ratings yet
- What is Organic Chemistry? Chemistry Book 4th Grade | Children's Chemistry BooksFrom EverandWhat is Organic Chemistry? Chemistry Book 4th Grade | Children's Chemistry BooksNo ratings yet
- Multi-Sulfur and Sulfur and Oxygen Five- and Six-Membered Heterocycles, Part 1From EverandMulti-Sulfur and Sulfur and Oxygen Five- and Six-Membered Heterocycles, Part 1David S. BreslowNo ratings yet
- Alkyl HalideDocument8 pagesAlkyl HalidePrincess Mae EstabilloNo ratings yet
- ALKYNEDocument6 pagesALKYNEPrincess Mae EstabilloNo ratings yet
- Forensic ChemistryDocument6 pagesForensic ChemistryPrincess Mae EstabilloNo ratings yet
- ALKENEDocument11 pagesALKENEPrincess Mae EstabilloNo ratings yet
- Ethics PrelimDocument36 pagesEthics PrelimPrincess Mae EstabilloNo ratings yet
- Forensic Chemistry and ToxicologyDocument31 pagesForensic Chemistry and ToxicologyPrincess Mae EstabilloNo ratings yet
- Aseptic Material Resistance TestDocument2 pagesAseptic Material Resistance TestFelipeAndrésLondoñoGrandaNo ratings yet
- Half Yearly Paper Class Xi ChemistryDocument5 pagesHalf Yearly Paper Class Xi ChemistryNeelotpal TripathiNo ratings yet
- (WORKSHEET) General Biology 1 2nd Quarter Activity 2 - LipidsDocument2 pages(WORKSHEET) General Biology 1 2nd Quarter Activity 2 - Lipidsaltheatoledo15No ratings yet
- Forced Degradation - Mass BalanceDocument8 pagesForced Degradation - Mass BalanceppiccoliniNo ratings yet
- Udvashito Mukh SSC22Document13 pagesUdvashito Mukh SSC22Showmajit SahaNo ratings yet
- CBSE Test Paper - 9 (Chemistry)Document7 pagesCBSE Test Paper - 9 (Chemistry)Suva lalNo ratings yet
- Food-PackagingDocument48 pagesFood-PackagingTrishitman DasNo ratings yet
- Carbonyl Compounds: Organic Chemistry Daily Practice ProblemsDocument28 pagesCarbonyl Compounds: Organic Chemistry Daily Practice ProblemsSiddhantNo ratings yet
- Silica and Silicon in TextilesDocument7 pagesSilica and Silicon in Textilespuja_14No ratings yet
- Astm D 511Document9 pagesAstm D 511Daniel HernandezNo ratings yet
- Chemistry 2Document14 pagesChemistry 2Marianne0% (1)
- The Kinetics of The Adsorption Process of CR (VI) in Aqueous Solution Using Neem Seed Husk (Azadirachta Indica) Activated CarbonDocument13 pagesThe Kinetics of The Adsorption Process of CR (VI) in Aqueous Solution Using Neem Seed Husk (Azadirachta Indica) Activated CarbonNórida Pájaro GómezNo ratings yet
- Battery Recycling Steps in Different Kinds of Battery, Advantages and Disadvantages - Natural Energy HubDocument7 pagesBattery Recycling Steps in Different Kinds of Battery, Advantages and Disadvantages - Natural Energy HubNajihatul SyahirahNo ratings yet
- 8201 GapDocument4 pages8201 GapKrishna N HNo ratings yet
- Experiment 10 Synthesis of Metal-Ligand Complexes Containing The Ligand AcetylacetoneDocument4 pagesExperiment 10 Synthesis of Metal-Ligand Complexes Containing The Ligand AcetylacetoneSantino MusaNo ratings yet
- 2545 PracticalDocument10 pages2545 PracticalYared Genene100% (1)
- Galvanizing - 2022: Continuous Hot-Dip Galvanizing - Process and ProductsDocument36 pagesGalvanizing - 2022: Continuous Hot-Dip Galvanizing - Process and ProductsHerdisNo ratings yet
- Manual Service Vitodens 200Document77 pagesManual Service Vitodens 200MariusMNo ratings yet
- Gen ChemDocument43 pagesGen ChemhelloNo ratings yet
- Beach Babies WO Sunscreen SPF50+ SC-587-1Document1 pageBeach Babies WO Sunscreen SPF50+ SC-587-1ajitbadboy2No ratings yet
- Soldering Basic GuideDocument6 pagesSoldering Basic GuidechchuanNo ratings yet
- SAES-Q-001 (Criteria For The Design & Construction of Concrete Structures)Document5 pagesSAES-Q-001 (Criteria For The Design & Construction of Concrete Structures)Nino Celso Astillero100% (1)
- SDFSDDocument8 pagesSDFSDDavin AngwynNo ratings yet
- SMB 104 Introduction 1Document29 pagesSMB 104 Introduction 1Shem Peter Mutua MutuiriNo ratings yet
- Use of Partial Dissolution Techniques in Geochemical ExplorationDocument35 pagesUse of Partial Dissolution Techniques in Geochemical ExplorationMiguel Angel Catunta ZarateNo ratings yet
- Listening Project (Students Version)Document3 pagesListening Project (Students Version)Karoll Silvana GonzálezNo ratings yet
- PLC Based System For Measuring Concentration of Oxygen in Mines For Miners Safety With O2 SensorDocument7 pagesPLC Based System For Measuring Concentration of Oxygen in Mines For Miners Safety With O2 SensorIJAERS JOURNALNo ratings yet
- Chemical Bonding Mcqs Mdcat Nums Paid Test 2023 by Study CornerDocument7 pagesChemical Bonding Mcqs Mdcat Nums Paid Test 2023 by Study Cornerststa.2022No ratings yet
- X To XI Moving (JEE) Student School Assessment TestDocument11 pagesX To XI Moving (JEE) Student School Assessment TestAbhishek ChandraNo ratings yet
Aromatic Hydrocarbons
Aromatic Hydrocarbons
Uploaded by
Princess Mae EstabilloOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Aromatic Hydrocarbons
Aromatic Hydrocarbons
Uploaded by
Princess Mae EstabilloCopyright:
Available Formats
Page|1
Republic of the Philippines
ISABELA STATE UNIVERSITY
Jones, Isabela
COLLEGE OF CRIMINAL JUSTICE EDUCATION
Second Semester, School Year 2021-2022
ORGANIC CHEMISTRY
I. TITLE OF THE MODULE
Chapter FIVE (5): AROMATIC HYDROCARBONS
II. INTRODUCTION
Aromatic hydrocarbons are the third class of unsaturated hydrocarbons; the alkenes and
alkynes (previously considered) are the other two classes. An aromatic hydrocarbon is an
unsaturated cyclic hydrocarbon that does not readily undergo addition reactions. This reaction
behavior, which is very different from that of alkenes and alkynes, explains the separate
classification for aromatic hydrocarbons.
The fact that, even though they are unsaturated compounds, aromatic hydrocarbons do not
readily undergo addition reactions suggests that the bonding present in this type of compound
must differ significantly from that in alkenes and alkynes. Such is indeed the case
Let’s look at the bonding present in benzene, the simplest aromatic hydrocarbon, to explore
this new type of bonding situation and to also characterize the aromatic hydrocarbon functional
group. Benzene, a flat, symmetrical molecule with a molecular formula of C6H6, has a structural
formula that is often formalized as that of a cyclohexatriene—in other words, as a structural
formula that involves a six-membered carbon ring in which three double bonds are present.
This structure is one of two equivalent structures that can be drawn for benzene that differ
only in the locations of the double bonds (1,3,5 positions versus 2,4,6 positions):
Neither of these conventional structures, however, is totally correct. Experimental evidence
indicates that all of the carbon–carbon bonds in benzene are equivalent (identical), and these
structures imply three bonds of one type (double bonds) and three bonds of a different type
(single bonds). The equivalent nature of the carbon–carbon bonds in benzene is addressed by
considering the correct bonding structure for benzene to be an average of the two “triene”
structures. Related to this “average”-structure situation is the concept that electrons associated
with the ring double bonds are not held between specific carbon atoms; instead, they are free to
move “around” the carbon ring. Thus the true structure for benzene, an intermediate between that
represented by the two “triene” structures, is a situation in which all carbon–carbon bonds are
equivalent; they are neither single nor double bonds but something in between. Placing a double-
headed arrow between the conventional structures that are averaged to obtain the true structure is
one way to denote the average structure.
General Chemistry (Organic Chemistry) - Crim AdGE
Isabela State University – Jones Campus
Page|2
An alternative notation for denoting the bonding in benzene—a notation that involves a
single structure—is
In this “circle-in-the-ring” structure for benzene, the circle denotes the electrons associated
with the double bonds that move “around” the ring. Each carbon atom in the ring can be
considered to participate in three conventional (localized) bonds (two C—C bonds and one C—H
bond) and in one delocalized bond (the circle) that involves all six carbon atoms. A delocalized
bond is a covalent bond in which electrons are shared among more than two atoms. This
delocalized bond is what causes benzene and its derivatives to be resistant to addition reactions, a
property normally associated with unsaturation in a molecule
The structure represented by the notation is called an aromatic ring system, and it is the
functional group present in aromatic compounds. An aromatic ring system is a highly unsaturated
carbon ring system in which both localized and delocalized bonds are present.
III. TABLE OF CONTENT
i. NOMENCLATURE OF AROMATIC HYDROCARBONS
ii. AROMATIC HYDROCARBONS: PHYSICAL PROPERTIES AND SOURCES
iii. CHEMICAL REACTIONS OF AROMATIC HYDROCARBONS
iv. FUSED-RING AROMATIC HYDROCARBONS
v. USES OF BENZENE AND BENZENE DERIVATIVES
IV. LEARNING OUTCOME
At the end of this module, the students are expected to:
Recognize the structure of benzene;
Name various substitution of benzene; and
Recognize the structures of various aromatic compounds.
V. LEARNING CONTENT
i. NOMENCLATURE OF AROMATIC HYDROCARBONS
Replacement of one or more of the hydrogen atoms on benzene with other groups
produces benzene derivatives. Compounds with alkyl groups or halogen atoms attached
to the benzene ring are commonly encountered. We consider first the naming of benzene
derivatives with one substituent, then the naming of those with two substituents, and
finally the naming of those with three or more substituents.
Benzene Derivatives with One Substituent
The IUPAC system of naming monosubstituted benzene derivatives uses the name of
the substituent as a prefix to the name benzene. Examples of this type of nomenclature
include
A few monosubstituted benzenes have names wherein the substituent and the benzene
ring taken together constitute a new parent name. Two important examples of such
nomenclature with hydrocarbon substituents are
Both of these compounds are industrially important chemicals.
General Chemistry (Organic Chemistry) - Crim AdGE
Isabela State University – Jones Campus
Page|3
Monosubstituted benzene structures are often drawn with the substituent at the “12
o’clock” position, as in the previous structures. However, because all the hydrogen atoms
in benzene are equivalent, it does not matter at which carbon of the ring the substituted
group is located. Each of the following formulas represents chlorobenzene.
For monosubstituted benzene rings that have a group attached that is not easily named
as a substituent, the benzene ring is often treated as a group attached to this substituent. In
this reversed approach, the benzene ring attachment is called a phenyl group, and the
compound is named according to the rules for naming alkanes, alkenes, and alkynes.
Benzene Derivatives with Two Substituent
When two substituents, either the same or different, are attached to a benzene ring,
three isomeric structures are possible.
To distinguish among these three isomers, we must specify the positions of the
substituents relative to one another.
This can be done in either of two ways: by using numbers or by using non-numerical
prefixes. When numbers are used, the three isomeric dichlorobenzenes have the first-
listed set of names
The prefix system uses the prefixes ortho-, meta-, and para- (abbreviated o-, m-, and p-).
Ortho- means 1,2 di-substitution; the substituents are on adjacent carbon
atoms.
Meta- means 1,3 di-substitution; the substituents are one carbon removed
from each other.
Para- means 1,4 di-substitution; the substituents are two carbons removed
from each other (on opposite sides of the ring).
When prefixes are used, the three isomeric dichlorobenzenes have the second-listed
set of names above.
When one of the two substituents in a di-substituted benzene imparts a special name
to the compound (as, for example, toluene), the compound is named as a derivative of
that parent molecule. The special substituent is assumed to be at ring position 1.
When neither substituent group imparts a special name, the substituents are cited in
alphabetical order before the ending -benzene. The carbon of the benzene ring bearing the
substituent with alphabetical priority becomes carbon 1.
General Chemistry (Organic Chemistry) - Crim AdGE
Isabela State University – Jones Campus
Page|4
A benzene ring bearing two methyl groups is a situation that generates a new special
base name. Such compounds (there are three isomers) are not named as
dimethylbenzenes or as methyl toluenes. They are called xylenes.
The xylenes are good solvents for grease and oil and are used for cleaning microscope
slides and optical lenses and for removing wax from skis.
Benzene Derivatives with Three Substituent
When more than two groups are present on the benzene ring, their positions are
indicated with numbers. The ring is numbered in such a way as to obtain the lowest
possible numbers for the carbon atoms that have substituents. If there is a choice of
numbering systems (two systems give the same lowest set), then the group that comes
first alphabetically is given the lower number.
ii. AROMATIC HYDROCARBONS: PHYSICAL PROPERTIES AND SOURCES
In general, aromatic hydrocarbons resemble other hydrocarbons in physical
properties. They are insoluble in water, are good solvents for other nonpolar materials,
and are less dense than water.
Benzene, monosubstituted benzenes, and many di-substituted benzenes are liquids at
room temperature. Benzene itself is a colorless, flammable liquid that burns with a sooty
flame because of incomplete combustion.
At one time, coal tar was the main source of aromatic hydrocarbons. Petroleum is
now the primary source of such compounds. At high temperatures, with special catalysts,
saturated hydrocarbons obtained from petroleum can be converted to aromatic
hydrocarbons. The production of toluene from heptane is representative of such a
conversion.
Benzene was once widely used as an organic solvent. Such use has been discontinued
because benzene’s short- and long-term toxic effects are now recognized. Benzene
inhalation can cause nausea and respiratory problems.
iii. CHEMICAL REACTIONS OF AROMATIC HYDROCARBONS
We have noted that aromatic hydrocarbons do not readily undergo the addition
reactions characteristic of other unsaturated hydrocarbons. An addition reaction would
require breaking up the delocalized bonding present in the ring system.
If benzene is so unresponsive to addition reactions, what reactions does it undergo?
Benzene undergoes substitution reactions. As you recall, substitution reactions are
characterized by different atoms or groups of atoms replacing hydrogen atoms in a
hydrocarbon molecule. Two important types of substitution reactions for benzene and
other aromatic hydrocarbons are alkylation and halogenation.
1. Alkylation: An alkyl group (R—) from an alkyl chloride (R—Cl) substitutes for a
hydrogen atom on the benzene ring. A catalyst, AlCl3, is needed for alkylation.
In general terms, the alkylation of benzene can be written as
General Chemistry (Organic Chemistry) - Crim AdGE
Isabela State University – Jones Campus
Page|5
Alkylation is the most important industrial reaction of benzene.
2. Halogenation (bromination or chlorination): A hydrogen atom on a benzene ring
can be replaced by bromine or chlorine if benzene is treated with Br2 or Cl2 in the
presence of a catalyst. The catalyst is usually FeBr3 for bromination and FeCl3
for chlorination.
Aromatic halogenation differs from alkane halogenation (Section 12.17) in that
light is not required to initiate aromatic halogenation.
iv. FUSED-RING AROMATIC HYDROCARBONS
Benzene and its substituted derivatives are not the only type of aromatic hydrocarbon
that exists. Another large class of aromatic hydrocarbons is the fused-ring aromatic
hydrocarbons. A fused-ring aromatic hydrocarbon is an aromatic hydrocarbon whose
structure contains two or more rings fused together. Two carbon rings that share a pair of
carbon atoms are said to be fused.
The three simplest fused-ring aromatic compounds are naphthalene, anthracene, and
phenanthrene. All three are solids at room temperature.
v. USES OF BENZENE AND BENZENE DERIVATIVES
Benzene is formed from both natural processes and human activities. Natural sources of
benzene include volcanoes and forest fires. Benzene is also a natural part of crude oil,
gasoline, and cigarette smoke. In fact, it ranks in the top 20 chemicals for production volume.
Early Uses of Benzene
In the 19th and early-20th centuries, benzene was used as an after-shave lotion because of
its pleasant smell. Prior to the 1920s, benzene was frequently used as an industrial solvent,
especially for degreasing metal. As its toxicity became obvious, benzene was supplanted by
other solvents, especially toluene (methyl benzene), which has similar physical properties but
is not as carcinogenic.
In 1903, Ludwig Roselius popularized the use of benzene to decaffeinate coffee. This
discovery led to the production of Sanka (the letters "ka" in the brand name stand for kaffein).
This process was later discontinued.
As a petrol additive, benzene increases the octane rating and reduces knocking.
Consequently, petrol often contained several percent benzene before the 1950s, when
tetraethyl lead replaced it as the most widely-used antiknock additive. With the global
phaseout of leaded petrol, benzene has made a comeback as a gasoline additive in some
nations. In the United States, concern over its negative health effects and the possibility of
benzene entering the groundwater have led to stringent regulation of petrol's benzene content,
with limits typically around 1%. European petrol specifications now contain the same 1%
limit on benzene content. The US EPA has new regulations that will lower the benzene
content in gasoline to 0.62% in 2011.
Current Uses of Benzene
Today benzene is mainly used as an intermediate to make other chemicals. Its most widely-
produced derivatives include styrene, which is used to make polymers and plastics, phenol for
resins and adhesives (via cumene), and cyclohexane, which is used in the manufacture of
General Chemistry (Organic Chemistry) - Crim AdGE
Isabela State University – Jones Campus
Page|6
Nylon. Smaller amounts of benzene are used to make some types of rubbers, lubricants, dyes,
detergents, drugs, explosives, napalm and pesticides.
In laboratory research, toluene is now often used as a substitute for benzene. The solvent-
properties of the two are similar but toluene is less toxic and has a wider liquid range.
Benzene has been used as a basic research tool in a variety of experiments including
analysis of a two-dimensional gas.
VI. ACTIVITY
4.1 Assign IUPAC names to the following benzene derivatives
VII. ASSESSMENT
1. A circle (ring) within a hexagon is often used to represent an aromatic carbon ring. What
does the circle represent?
2. Assign an IUPAC name to each of the following disubstituted benzenes. Use numbers
rather than prefixes to locate the substituents on the benzene ring.
General Chemistry (Organic Chemistry) - Crim AdGE
Isabela State University – Jones Campus
Page|7
3. Assign each of the compounds in item number 2 an IUPAC name in which the
substituents on the benzene ring are located using the ortho-, meta-, para- prefix system.
4. Write a structural formula for each of the following compounds.
a. 1,3-Diethylbenzene
b. o-Xylene
c. p-Ethyltoluene
d. Phenylbenzene
5. Indicate whether or not each of the following is a general physical property of aromatic
hydrocarbons. Answers could range from a simple yes-no response or as detailed as you
could.
a. Soluble in water
b. Less dense than water
c. Good solvent for nonpolar substances
d. All solids at room temperatures
6. What is currently the primary source for aromatic hydrocarbons and what was the
previous primary source of aromatic hydrocarbon?
7. Why is this functioned called aromatic hydrocarbons, in the first place?
VIII. RESOURCES
Sachkheim, George I. and Lehman Denis D. Chemistry for the Health Sciences. 8th
Edition. Prentice-Hall, Inc.
Stoker, Stephen H. General, Organic, and Biological Chemistry. 5th Edition.
Brooks//Cole, Cenage Learning. 2010
Holm, Johanna Forgotten Chemistry. Barron’s Educational Series, Inc. 2006
Vollhardt, Peter, and Neil E. Schore. Organic Chemistry: Structure and Function. 5th
Edition. New York: W. H. Freeman & Company, 2007.
General Chemistry (Organic Chemistry) - Crim AdGE
Isabela State University – Jones Campus
You might also like
- Enzyme Assays A Practical Approach by Robert Eisenthal, Michael DansonDocument304 pagesEnzyme Assays A Practical Approach by Robert Eisenthal, Michael DansonSara OchoaNo ratings yet
- BenzeneDocument14 pagesBenzeneJueeli More100% (1)
- Aromatic Hydrocarbons Pharmaceutical Organic Chemistry Porg111Document9 pagesAromatic Hydrocarbons Pharmaceutical Organic Chemistry Porg111AnnaGueseNo ratings yet
- Organic Chemistry Module 4Document5 pagesOrganic Chemistry Module 4Josephine TeroNo ratings yet
- Unsaturated Hydrocarbons Alkenes and Alkynes Aromatic CompoundDocument4 pagesUnsaturated Hydrocarbons Alkenes and Alkynes Aromatic CompoundGNo ratings yet
- Geometrical Isomerism: It is known that a carbon-carbon double bond is made up of one σ bond and one π-bond. The π-bond presents free rotation about the double bond.ThisDocument3 pagesGeometrical Isomerism: It is known that a carbon-carbon double bond is made up of one σ bond and one π-bond. The π-bond presents free rotation about the double bond.ThisKisha KhuranaNo ratings yet
- Chemistry Doc 3: Hydrocarbons Lewis Structures, Ball-And-Stick Models, and Space-Filling Models For MoleculesDocument3 pagesChemistry Doc 3: Hydrocarbons Lewis Structures, Ball-And-Stick Models, and Space-Filling Models For MoleculesKrishia Mae IlardeNo ratings yet
- Hydrocarbon S: Group 3: Leader: Angela Dela Cruz Daphne Sy Sofia Anne Caoagas Alen Gem Red Christian Faith ColisDocument16 pagesHydrocarbon S: Group 3: Leader: Angela Dela Cruz Daphne Sy Sofia Anne Caoagas Alen Gem Red Christian Faith ColisDaphne SyNo ratings yet
- Aromatic Hydrocarbons ReportDocument15 pagesAromatic Hydrocarbons ReportLyssa LimNo ratings yet
- UNIT 10 Organic ChemistryDocument51 pagesUNIT 10 Organic ChemistryTristan PereyNo ratings yet
- 8.1 - Learning Material-2Document49 pages8.1 - Learning Material-2Erica PagulayanNo ratings yet
- Organic 23 24Document18 pagesOrganic 23 24VigneshNo ratings yet
- (8.1) Learning MaterialDocument41 pages(8.1) Learning MaterialLuna VasquezNo ratings yet
- Topic 8 AlkenesDocument17 pagesTopic 8 Alkenesmark regino TuyayNo ratings yet
- Term Report: Submitted byDocument11 pagesTerm Report: Submitted byMUHAMMAD OMAIR AAMIR RAJANo ratings yet
- Nomenclature of Organic CompoundDocument10 pagesNomenclature of Organic CompounddeepashriguddaNo ratings yet
- Hydrocarbons NotesDocument15 pagesHydrocarbons Notesarjunrkumar2024No ratings yet
- ALKENEDocument11 pagesALKENEPrincess Mae EstabilloNo ratings yet
- Benzene: Milton Biswas Student Madhav University, Abu RoadDocument23 pagesBenzene: Milton Biswas Student Madhav University, Abu RoadMilton BiswasNo ratings yet
- W4 BENZENE Nomenclature Stability Physical & Chemical Properties PDFDocument22 pagesW4 BENZENE Nomenclature Stability Physical & Chemical Properties PDFathyrahNo ratings yet
- WEEK 10 Aromatic HydrocarbonDocument26 pagesWEEK 10 Aromatic HydrocarbonChris Angelo De GuzmanNo ratings yet
- Aromatic CompoundsDocument16 pagesAromatic CompoundsJotillnaimNo ratings yet
- Carbon Compounds: Presenter: Shannon SmithDocument61 pagesCarbon Compounds: Presenter: Shannon SmithShannon SmithNo ratings yet
- Cape Chemistry Unit 2 ReviewDocument7 pagesCape Chemistry Unit 2 Reviewkayanna100% (1)
- Assingment On Nomenclature OF Organic Compound: Submitted To: DR - Shobha Thakur Submitted By: Ritik Dwivedi 18BSPCM020Document30 pagesAssingment On Nomenclature OF Organic Compound: Submitted To: DR - Shobha Thakur Submitted By: Ritik Dwivedi 18BSPCM020ritik dwivediNo ratings yet
- AttachmentDocument14 pagesAttachmentaberaendale334No ratings yet
- Organic ChemistryDocument94 pagesOrganic ChemistryVrisanNo ratings yet
- +++fictionOrganic U-2 classes and name.docxDocument30 pages+++fictionOrganic U-2 classes and name.docxaberaendale334No ratings yet
- Lecture Nine Organic ChemistryDocument11 pagesLecture Nine Organic Chemistryabdihakimhasssan344No ratings yet
- Hydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSEDocument1 pageHydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSEjaindhruv850No ratings yet
- Organic CompoundsDocument22 pagesOrganic CompoundsMary Rose AguilaNo ratings yet
- Organic Chem U-2Document19 pagesOrganic Chem U-2sinte beyuNo ratings yet
- Organic ChemistryDocument70 pagesOrganic ChemistryGhulam Rabbani100% (1)
- organic chemistryDocument27 pagesorganic chemistryAnn SunilNo ratings yet
- Organic Compounds EditedDocument52 pagesOrganic Compounds EditedbrendonNo ratings yet
- Organic Chemistry LecturesDocument32 pagesOrganic Chemistry LecturesAbdulHameedNo ratings yet
- Corrected Fundamentals of Organic ChemistryDocument71 pagesCorrected Fundamentals of Organic ChemistryDAM2120No ratings yet
- Aromatic It yDocument8 pagesAromatic It ybalweg mackyNo ratings yet
- Organic ChemistryDocument30 pagesOrganic ChemistryMukhilNo ratings yet
- Nouman (CHEM 11)Document6 pagesNouman (CHEM 11)Junaid AkhtarNo ratings yet
- Pharmaceutical Chemistry Chapter 4 Organic Chemisty NotesDocument10 pagesPharmaceutical Chemistry Chapter 4 Organic Chemisty Notesbf327626No ratings yet
- Chapter 4 Aromatic HydrocarbonsDocument34 pagesChapter 4 Aromatic HydrocarbonsAbdirashid Adam IsakNo ratings yet
- Benzene & Its Derivatives: (The Class of Compounds, Which Contains Benzene or Substituted Derivative of Benzene)Document17 pagesBenzene & Its Derivatives: (The Class of Compounds, Which Contains Benzene or Substituted Derivative of Benzene)justin youngNo ratings yet
- Benzene Aromatic CompoundsDocument14 pagesBenzene Aromatic CompoundsBook of Life fgfhfghfghfghNo ratings yet
- Publication 10 22989 1587Document36 pagesPublication 10 22989 1587Ahsajib MatubberNo ratings yet
- Organic Chem U-2 Functional GroupDocument34 pagesOrganic Chem U-2 Functional Groupsinte beyuNo ratings yet
- Aromatic It yDocument9 pagesAromatic It yBentenBentenNo ratings yet
- Organic Chemistry Lecture 2 2022 Modif.Document60 pagesOrganic Chemistry Lecture 2 2022 Modif.Fady FadyNo ratings yet
- Chapter 4Document38 pagesChapter 4iB13eNo ratings yet
- Aromatic CompoundsDocument7 pagesAromatic Compoundsmanifest4godNo ratings yet
- Module 1. The Chemistry of Carbon Compounds TOPIC: Structure and FormulaeDocument38 pagesModule 1. The Chemistry of Carbon Compounds TOPIC: Structure and FormulaeOrlanda EllisNo ratings yet
- Organic Chemistry: Alkenes, Alkynes, and Aromatic CompoundsDocument43 pagesOrganic Chemistry: Alkenes, Alkynes, and Aromatic CompoundsDarius Gan100% (1)
- Alkenes and AlkynesDocument28 pagesAlkenes and AlkynesDorama AikaNo ratings yet
- Hydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSEDocument16 pagesHydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSERishabh Singh RajputNo ratings yet
- Organic ChemistryDocument55 pagesOrganic ChemistryKim Taeha BTSNo ratings yet
- 1) Reading Material by Nvs TeacherDocument34 pages1) Reading Material by Nvs Teacherjkc collegeNo ratings yet
- BENZENEDocument37 pagesBENZENEMaureen Galingan100% (1)
- LEC 19 Nomen-Culture and Structure of BenzeneDocument16 pagesLEC 19 Nomen-Culture and Structure of BenzeneIftikhar Ud DinNo ratings yet
- What is Organic Chemistry? Chemistry Book 4th Grade | Children's Chemistry BooksFrom EverandWhat is Organic Chemistry? Chemistry Book 4th Grade | Children's Chemistry BooksNo ratings yet
- Multi-Sulfur and Sulfur and Oxygen Five- and Six-Membered Heterocycles, Part 1From EverandMulti-Sulfur and Sulfur and Oxygen Five- and Six-Membered Heterocycles, Part 1David S. BreslowNo ratings yet
- Alkyl HalideDocument8 pagesAlkyl HalidePrincess Mae EstabilloNo ratings yet
- ALKYNEDocument6 pagesALKYNEPrincess Mae EstabilloNo ratings yet
- Forensic ChemistryDocument6 pagesForensic ChemistryPrincess Mae EstabilloNo ratings yet
- ALKENEDocument11 pagesALKENEPrincess Mae EstabilloNo ratings yet
- Ethics PrelimDocument36 pagesEthics PrelimPrincess Mae EstabilloNo ratings yet
- Forensic Chemistry and ToxicologyDocument31 pagesForensic Chemistry and ToxicologyPrincess Mae EstabilloNo ratings yet
- Aseptic Material Resistance TestDocument2 pagesAseptic Material Resistance TestFelipeAndrésLondoñoGrandaNo ratings yet
- Half Yearly Paper Class Xi ChemistryDocument5 pagesHalf Yearly Paper Class Xi ChemistryNeelotpal TripathiNo ratings yet
- (WORKSHEET) General Biology 1 2nd Quarter Activity 2 - LipidsDocument2 pages(WORKSHEET) General Biology 1 2nd Quarter Activity 2 - Lipidsaltheatoledo15No ratings yet
- Forced Degradation - Mass BalanceDocument8 pagesForced Degradation - Mass BalanceppiccoliniNo ratings yet
- Udvashito Mukh SSC22Document13 pagesUdvashito Mukh SSC22Showmajit SahaNo ratings yet
- CBSE Test Paper - 9 (Chemistry)Document7 pagesCBSE Test Paper - 9 (Chemistry)Suva lalNo ratings yet
- Food-PackagingDocument48 pagesFood-PackagingTrishitman DasNo ratings yet
- Carbonyl Compounds: Organic Chemistry Daily Practice ProblemsDocument28 pagesCarbonyl Compounds: Organic Chemistry Daily Practice ProblemsSiddhantNo ratings yet
- Silica and Silicon in TextilesDocument7 pagesSilica and Silicon in Textilespuja_14No ratings yet
- Astm D 511Document9 pagesAstm D 511Daniel HernandezNo ratings yet
- Chemistry 2Document14 pagesChemistry 2Marianne0% (1)
- The Kinetics of The Adsorption Process of CR (VI) in Aqueous Solution Using Neem Seed Husk (Azadirachta Indica) Activated CarbonDocument13 pagesThe Kinetics of The Adsorption Process of CR (VI) in Aqueous Solution Using Neem Seed Husk (Azadirachta Indica) Activated CarbonNórida Pájaro GómezNo ratings yet
- Battery Recycling Steps in Different Kinds of Battery, Advantages and Disadvantages - Natural Energy HubDocument7 pagesBattery Recycling Steps in Different Kinds of Battery, Advantages and Disadvantages - Natural Energy HubNajihatul SyahirahNo ratings yet
- 8201 GapDocument4 pages8201 GapKrishna N HNo ratings yet
- Experiment 10 Synthesis of Metal-Ligand Complexes Containing The Ligand AcetylacetoneDocument4 pagesExperiment 10 Synthesis of Metal-Ligand Complexes Containing The Ligand AcetylacetoneSantino MusaNo ratings yet
- 2545 PracticalDocument10 pages2545 PracticalYared Genene100% (1)
- Galvanizing - 2022: Continuous Hot-Dip Galvanizing - Process and ProductsDocument36 pagesGalvanizing - 2022: Continuous Hot-Dip Galvanizing - Process and ProductsHerdisNo ratings yet
- Manual Service Vitodens 200Document77 pagesManual Service Vitodens 200MariusMNo ratings yet
- Gen ChemDocument43 pagesGen ChemhelloNo ratings yet
- Beach Babies WO Sunscreen SPF50+ SC-587-1Document1 pageBeach Babies WO Sunscreen SPF50+ SC-587-1ajitbadboy2No ratings yet
- Soldering Basic GuideDocument6 pagesSoldering Basic GuidechchuanNo ratings yet
- SAES-Q-001 (Criteria For The Design & Construction of Concrete Structures)Document5 pagesSAES-Q-001 (Criteria For The Design & Construction of Concrete Structures)Nino Celso Astillero100% (1)
- SDFSDDocument8 pagesSDFSDDavin AngwynNo ratings yet
- SMB 104 Introduction 1Document29 pagesSMB 104 Introduction 1Shem Peter Mutua MutuiriNo ratings yet
- Use of Partial Dissolution Techniques in Geochemical ExplorationDocument35 pagesUse of Partial Dissolution Techniques in Geochemical ExplorationMiguel Angel Catunta ZarateNo ratings yet
- Listening Project (Students Version)Document3 pagesListening Project (Students Version)Karoll Silvana GonzálezNo ratings yet
- PLC Based System For Measuring Concentration of Oxygen in Mines For Miners Safety With O2 SensorDocument7 pagesPLC Based System For Measuring Concentration of Oxygen in Mines For Miners Safety With O2 SensorIJAERS JOURNALNo ratings yet
- Chemical Bonding Mcqs Mdcat Nums Paid Test 2023 by Study CornerDocument7 pagesChemical Bonding Mcqs Mdcat Nums Paid Test 2023 by Study Cornerststa.2022No ratings yet
- X To XI Moving (JEE) Student School Assessment TestDocument11 pagesX To XI Moving (JEE) Student School Assessment TestAbhishek ChandraNo ratings yet