Professional Documents
Culture Documents
Module 20 - UNIT IV - Chemistry of The Soil (Part 2)
Module 20 - UNIT IV - Chemistry of The Soil (Part 2)
Uploaded by
Jhess GaliciaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Module 20 - UNIT IV - Chemistry of The Soil (Part 2)
Module 20 - UNIT IV - Chemistry of The Soil (Part 2)
Uploaded by
Jhess GaliciaCopyright:
Available Formats
CAGAYAN STATE UNIVERSITY
Carig Campus
College of Engineering
CHEMISTRY FOR ENGINEERS
CHEM111
UNIT IV
ENVIRONMENTAL CHEMISTRY: SOIL
Learning Objectives:
At the end of this unit, the students will be able to:
1. Enumerate and describe the different macro and micronutrients in soil;
2. Enumerate the basic fertilizer components and their significance;
3. Have a basic understanding of the organic contaminants present in soil; and
4. Understand the mechanisms of biological contaminant degradation.
CONTENTS:
III. Macronutrients and Micronutrients in Soil
IV. Fertilizers
V. Organic Contaminants in Soil
III. MACRO AND MICRONUTRIENTS IN SOIL
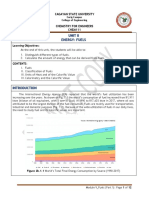
Soil supports plant growth by providing plants with essential nutrients: macronutrients and
micronutrients. Macronutrients are the elements that are present in the plant material/inside the
plant in significant amounts. Micronutrients on the other hand are essential only at very small
amounts. They are usually required for certain enzymes to function properly.
MACRONUTRIENTS
Plant macronutrients are carbon, hydrogen, oxygen, nitrogen, phosphorus, potassium,
calcium, magnesium, and sulfur.
• C, H, and O are obtained by plants directly from the atmosphere while the other
macronutrients must be obtained from the soil.
• The macronutrients N, P, and K are commonly lacking in soil so they are usually incorporated
into the soil mix thru fertilizers. They will be discussed more in detail later on.
• Calcium-deficient soils are relatively uncommon. Liming, or the application of lime provides a
more than adequate calcium supply for plants. However, soils can still become calcium
deficient due to calcium uptake by plants and leaching by carbonic acid.
Acidic soils sometimes make any calcium in the soil unavailable to plants due to hydrogen
ions competing with calcium ions (see cation exchange). Making soil more neutral generally treats
the calcium deficiency.
In basic (alkaline) soils, the high amounts of sodium, magnesium, and potassium sometimes
produces calcium deficiency because these ions compete with calcium for availability to plants.
• Magnesium is one of the most abundant elements in the earth’s crust, but most of it is strongly
bound in minerals. The magnesium available to plants is made up of Mg ions in organic matter
or clays. The availability of magnesium to plants depends upon the calcium/magnesium ratio.
If this ratio is too high (if there is too much calcium compared to magnesium), magnesium may
Module 20_Chemistry of the Soil (Part 2): Page 1 of 11
not be available to plants. This causes magnesium deficiency. Excessive levels of potassium or
sodium may also cause magnesium deficiency.
• Sulfur is assimilated by plants as the sulfate ion (SO42-). If there are large amounts of SO2 in the
air, plants may absorb the gas thru the leaves (SO2 pollution can kill vegetation in some
instances).
Sulfur is a component of some essential amino acids and of thiamin and biotin, which is why
sulfur-deficient soils do not support plant growth well. Whereas most fertilizers used to contain
sulfur, its use in commercial fertilizers has declined.
As mentioned in the previous module, toxic H2S can be produced when sulfate ions are
reduced. This can be inhibited by treatment with oxidizing compounds such as KNO3.
A. NITROGEN (N)
In most soils, over 90% of the nitrogen content is organic - the product of the biodegradation
of dead plants and animals. The organic nitrogen is eventually hydrolyzed into NH4+, which can then
be oxidized to NO3- by bacterial action.
Figure 4c.3.1 Nitrogen sinks and pathways in soil
Nitrogen, unlike potassium or phosphates, rarely comes from the weathering of minerals or
sediments. Nitrogen-fixing organisms cannot usually supply sufficient nitrogen to meet peak demand.
Inorganic nitrogen from fertilizers and rainwater is often largely lost by leaching. Soil humus,
however, contains significant amounts of nitrogen. It serves as an important reservoir of nitrogen. It
has the additional advantage that its rate of decay (its rate of nitrogen release to plants) roughly
parallels plant growth—rapid during the warm growing season, slow during the winter months.
Nitrogen is an essential component of proteins and other constituents of living matter. Plants
and cereals that are grown on nitrogen-rich soils provide higher yields; they are also more nutritious
as they are substantially richer in protein.
Generally, nitrogen is available to plants as nitrate ion (NO3-). When nitrogen is added to soils
in the ammonium form (NH4+), nitrifying bacteria converts it to the nitrate ion in a process called
nitrification. This is an essential process because while some plants (like rice) may utilize ammonium
nitrogen, it is toxic to other plants.
When soil is heavily fertilized in drought conditions, plants may absorb excessive amounts of
nitrogen (in nitrate form) from the soil. Forage crops containing excessive amounts of nitrate can
poison the ruminant animals who eat them. When these nitrate-rich plants are used for ensilage
(storing animal fodder in silos), the fermentation process that occurs reduce the nitrate ions into NO2
Module 20_Chemistry of the Soil (Part 2): Page 2 of 11
gas. This gas can accumulate inside the enclosed silos to toxic concentrations and humans have been
known to experience poisoning from such conditions.
Nitrogen fixation is the process by which atmospheric N2 is converted to available nitrogen
for the use of plants. Human activities are resulting in the fixation of larger than normal amounts
of nitrogen. These activities include chemical fertilizer manufacture, combustion of nitrogen
containing fuels, and the increased cultivation of nitrogen-fixing legumes. This is concerning because
it may have adverse effects to the ozone layer since N2O is released during the denitrification of
fixed nitrogen.
In the past, before widespread use of nitrogen fertilizers, soil nitrogen was provided primarily
by legumes (plants like soybeans, alfalfa, clover, etc.). These plants have root structures that
contain bacteria capable of fixing atmospheric nitrogen thru nitrification. Legumes may add up to
10 pounds per acre per year of nitrogen to soil - comparable to amounts commonly added as
synthetic fertilizers. Soil fertility with respect to nitrogen can be maintained by rotating plantings of
nitrogen-consuming plants with plantings of legumes, something that has been in practice since the
Roman era.
The nitrogen-fixing bacteria in legumes exist
in special structures on the roots called root
nodules. These rod-shaped bacteria are members
of a special genus, Rhizobium.
These bacteria can exist independently, but
they cannot fix nitrogen except when they are in
the root nodules.
All species of Rhizobium appear to be very
similar, but they exhibit a great deal of specificity
in their choice of host plants.
Legume root nodules also contain a form of
hemoglobin, which must somehow be involved in
the nitrogen-fixation process.
In some agricultural areas, nitrate pollution in water bodies has become a problem. The major
cause is the growing of livestock. The sheer amount of manure produced by livestock animals pollute
the waters to the extent that water bodies in some agricultural areas are just as polluted as those in
populated and highly industrialized areas. In addition to these, the acidic stomach contents of
ruminants contain bacteria that are capable of reducing nitrate ion into the toxic nitrite ion:
𝑁𝑂3− (𝑛𝑖𝑡𝑟𝑎𝑡𝑒 𝑖𝑜𝑛) + 2𝐻 + + 2𝑒 − → 𝑁𝑂2− (𝑛𝑖𝑡𝑟𝑖𝑡𝑒 𝑖𝑜𝑛) + 𝐻2 𝑂
Nitrate ions are not strongly bound to soil. It can be readily carried through soil formations by water.
B. PHOSPHORUS (P)
Although the percentage of phosphorus in plant material is relatively low, it is an essential
component of plants. Phosphorus, like nitrogen, must be present in a simple inorganic form before it
can be taken up by plants. (Manahan, et al.) The utilizable phosphorus species is some form of
orthophosphate ion 𝑯𝟐 𝑷𝑶− 𝟐−
𝟒 . and 𝑯𝑷𝑶𝟒 are the predominant orthophosphate species that exist in
the pH range of most soils.
Orthophosphate is most available to plants at near neutral pH values. In relatively acidic soils,
orthophosphate ions are precipitated or sorbed by species of Al (III) and Fe (III). In basic (alkaline)
soils, orthophosphates may react with calcium carbonate to form hydroxyapatite which is relatively
insoluble:
3𝐻𝑃𝑂42− + 5𝐶𝑎𝐶𝑂3 (𝑠) + 2𝐻2 𝑂 → 𝐶𝑎5 (𝑃𝑂4 )3 (𝑂𝐻)(𝑠) + 5𝐻𝐶𝑂3− + 𝑂𝐻 −
Module 20_Chemistry of the Soil (Part 2): Page 3 of 11
C. POTASSIUM (K)
Potassium is needed in the activation of some plant enzymes and it also plays a role in the
water balance in plants as well as in some carbohydrate transformations. This is why relatively high
levels of potassium are needed by growing plants. The higher the productivity of the crop, the
more potassium is removed from soil. The addition of nitrogen fertilizers also enhances the
removal of potassium.
Potassium is one of the most abundant elements in the earth’s crust but, like magnesium, much
of this potassium is not easily available to plants. For example, some silicate minerals such as leucite
𝐾2 𝑂 • 𝐴𝑙2 𝑂3 • 4𝑆𝑖𝑂2 contain strongly bound potassium. Potassium that is available to plants is usually
found as exchangeable potassium in clay minerals.
MICRONUTRIENTS
Boron, chlorine, copper, iron, manganese, molybdenum (for N-fixation), and zinc are
considered essential plant micronutrients. Plants only need them at very low levels and large
amounts may be toxic to plants. These elements usually function as components of essential
enzymes. Manganese, iron, chlorine, and zinc may be involved in photosynthesis. Sodium, silicon,
and cobalt may also be essential plant nutrients.
Iron and manganese are present in a number of soil minerals. Sodium and chlorine (as chloride)
occur naturally in soil. Some other micronutrients and trace elements are found in unweathered
minerals in soil.
Some plants are capable of accumulating extremely high levels of specific trace metals in their
tissues. Those that can accumulate more than 1.00 mg/g of dry weight are called
hyperaccumulators. The hyperaccumulation of metals has led to the idea of phytoremediation in
which plants growing on contaminated ground accumulate metals, which are then removed with
the plant biomass. Brassica juncea and Brassica chinensis (Chinese cabbage) have been shown to
hyperaccumulate as much as 5 grams of uranium/kg plant dry weight when grown on uranium-
contaminated soil. Uranium accumulation in the plants was enhanced by the addition of citrate,
which complexes uranium and makes it more soluble. This is a potential solution to toxic soils.
IV. FERTILIZERS
The major components of fertilizers are the elements N, P, and K. They are designated number
combinations like 6-12-8, which shows the percentages of N (6%), P as 𝑃2 𝑂5 (12%), and K as 𝐾2 𝑂 (8%).
Micronutrients such as magnesium and sulfate may also be added to the formula.
Fertilizer from manure is approx. equivalent to a 0.5-0.24-0.5 NPK fertilizer. Such organic
fertilizers, including compost, must first undergo biodegradation in order for it to release the simple
inorganic species (𝑁𝑂3− , 𝐻𝑥 𝑃𝑂4 𝑥−3 , 𝐾 + ) which can be used by plants.
i. Most modern nitrogen fertilizers are made by the Haber process. Here, 𝑁2 and 𝐻2 are
combined in the presence of a catalyst at approximately 500°C and pressures up to 1000 atm:
𝑁2 + 3𝐻2 → 2𝑁𝐻3
The anhydrous ammonia gas product has a nitrogen content (82% by weight) but is toxic so special
equipment is needed in the process. Anhydrous ammonia can then be added directly to the soil,
for which it has a strong affinity. This is because it is water-soluble and forms ammonium ions:
𝑁𝐻3 (𝑔) 𝑖𝑛 (𝑤𝑎𝑡𝑒𝑟) → 𝑁𝐻3 (𝑎𝑞) (𝑎)
𝑁𝐻3 (𝑔) + 𝐻2 𝑂 𝑖𝑛 𝑠𝑜𝑖𝑙 → 𝑁𝐻4+ + 𝑂𝐻 − (𝑏)
Module 20_Chemistry of the Soil (Part 2): Page 4 of 11
Ammonia gas reacts quickly with some substances on top of its toxicity so care must be made
in its handling, storage, and disposal. For this reason, aqua ammonia, a 30% solution of NH3 in
water, is a safer alternative to ammonia gas and is sometimes added directly to irrigation water.
Another common solid nitrogen fertilizer is ammonium nitrate (𝑁𝐻4 𝑁𝑂3 ). It is made by
oxidizing ammonia over a platinum catalyst, converting the nitric oxide product to nitric acid,
and reacting the nitric acid with ammonia. The molten ammonium nitrate product is forced
through nozzles at the top of a prilling tower and solidifies to form small pellets while falling
through the tower. The particles are coated with a water repellent. (Manahan, et al.) Ammonium
nitrate contains 33.5% nitrogen by weight. It is easy to apply to soil, but it poses some hazards.
Since it is explosive, much care is needed during manufacture and storage.
Urea (CO(NH2)2) is easier to manufacture and handle than ammonium nitrate and is the most
popular solid nitrogen fertilizer. The overall reaction for urea synthesis is:
𝐶𝑂2 + 2𝑁𝐻3 → 𝐶𝑂(𝑁𝐻2 )2 + 𝐻2 𝑂
Other compounds used as nitrogen fertilizers are sodium nitrate, calcium nitrate,
potassium nitrate, and ammonium phosphates. Ammonium sulfate used to be widely applied
as fertilizer. Whereas alkali metal nitrates tend to make soil alkaline, ammonium sulfate leaves
an acidic residue.
ii. The principal mineral used as a source of phosphate is fluorapatite 𝐶𝑎5 (𝑃𝑂4 )3 𝐹. The phosphate
from fluorapatite bound strongly to the mineral so it is largely unavailable to plants. Fluorapatite
must first be treated with phosphoric or sulfuric acids to produce superphosphates which are
much more soluble than the parent phosphate minerals:
2𝐶𝑎5 (𝑃𝑂4 )3 𝐹(𝑠) + 14𝐻3 𝑃𝑂4 + 10𝐻2 𝑂 → 2𝐻𝐹(𝑔) + 10𝐶𝑎(𝐻2 𝑃𝑂4 )2 • 𝐻2 𝑂
2𝐶𝑎5 (𝑃𝑂4 )3 𝐹(𝑠) + 𝐻2 𝑆𝑂4 + 3𝐻2 𝑂 → 2𝐻𝐹(𝑔) + 3𝐶𝑎(𝐻2 𝑃𝑂4 )2 • 𝐻2 𝑂 + 7𝐶𝑎𝑆𝑂4
Note that the HF gas produced in superphosphate production can create air pollution problems.
Phosphate minerals contain various trace elements necessary for good plant growth, such
as B, Cu, Mn, Mo and Zn. Ironically, a large percentage of these elements are lost upon conversion
of the phosphate minerals to a more soluble phosphate form.
Ammonium phosphates are excellent, soluble phosphate fertilizers. They are usually made
in a liquid polyphosphate form: ammonium salts of pyrophosphate, triphosphate, and small
quantities of higher polymeric phosphate anions in aqueous solution. These polyphosphates can
chelate metal ions and make them more available to plants.
iii. Potassium fertilizers consist of potassium salts, generally KCl. These salts are all soluble in water
and may be obtained from ground deposits or from some brines.
One consideration in the use of potassium fertilizers is the luxury uptake of potassium by
some crops. These crops absorb more potassium than is needed for their maximum growth. In
crops where only the grain is harvested, this is not a concern since most of the potassium is
returned to the soil with the dead plant. However, for crops where the entire plant is taken away,
such as when hay/forage is harvested, the potassium contained in the plant is completely lost
from the soil.
V. ORGANIC CONTAMINANTS IN SOIL
Soil receives large quantities of waste products from a wide range of sources. Soils in different
locations exhibit different degrees of contamination depending on a lot of factors. Soil is more
capable of retaining pollutants due to its predominantly solid nature. These retention (sorption)
mechanisms cause soil to act as sinks for contaminants and as a kind of filter, reducing/preventing
contaminants reaching the groundwater. The various biological processes happening in soil may also
Module 20_Chemistry of the Soil (Part 2): Page 5 of 11
help degrade contaminants. However, contaminants which are not degraded or are sorbed can
accumulate to reach dangerous levels.
Table 4C.5.1. Sample sources of soil contaminants
Atmospheric fallout —fossil fuel combustion (oxides/acid anions of S and N; polycyclic aromatic HCs
(PAHs), heavy metals)
—metal smelting operations (As, Cd, Cu, Cr, Hg, Ni, Pb, Sb, Tl, Zn)
—metal-using industries, including foundries (Cd, Cu, Pb, Zn)
—chemical industries (organic micropollutants, Hg)
—waste disposal by incineration (Cd, dioxins)
—radionuclides from reactor accidents/ atmospheric nuclear testing
—large fires (PAHs; Pb, Cr, etc. from paint; dioxins)
Agricultural chemicals —herbicides (organic molecules; B, and As compounds)
—insecticides (chlorinated hydrocarbons, e.g. DDT, BHC)
—fungicides (Cu, Zn, Hg, and organic molecules)
—acaricides {e.g. Tar Oil')
—fertilizers {e.g. Cd and U impurities in phosphates)
Waste Disposal —farm manures (As, Cu, and Zn in pig and poultry manures)
(intentional/unintentional —sewage sludges (heavy metals; organic pollutants; PAHs; pathogens)
input to soil): —composts from domestic wastes (metals; organics)
—mine wastes (SO4~; As, Cd, Cu, Pb, Zn, Ba, U, etc.)
—seepage of leachate from landfills (metals; Cl""; PCBs; etc.)
—ash from combustion, incinerators, bonfires/other fires (PAHs; TCDDs; metals;
etc.)
—burial of diseased livestock on farmland
Incidental Accumulation —corrosion of metal in contact with soil (e.g. Zn from galvanized metal; Cu and Pb
of contaminants from roofing, scrapyards, etc.)
—wood preservatives (PCP; PAHs; As; Cr; Cu)
—leakage from underground storage tanks (petroleum; solvents)
—warfare and military training (HCs; PAHs; explosives; metals)
—sports and leisure activities (Pb, Sb, and As from shotgun pellets; Pb from fishing
weights; Pb, Cd, Ni, and Hg from discarded batteries; HCs from spilt petrol and
lubricating oil, etc.)
Derelict industrial sites —wide range of contaminants from production, waste disposal, and building
demolition —Gas works—phenols; tars (PAHs); cyanides; and As
—Electrical industries—Cu, Pb, Zn; PCBs; solvents
—Tanneries—Cr
—Scrapyards—metals; PCBs; hydrocarbons
Some choose to differentiate between 'pollution' and 'contamination'. 'Contamination' is
sometimes used when a substance resulting from human activity is present in the environment but
not causing any obvious harmful effects. In contrast, the more pejorative term 'pollution' is often
used when a substance is present and having a harmful effect. However, it is the convention to refer
to all cases of pollution or contamination of land as contamination.
As shown in Table 4C.5.1, the range of chemical contaminants is vast and can include, for
example: heavy metals, radionuclides, polycyclic aromatic hydrocarbons (PAHs) formed during
incomplete combustion, synthesized organic solvents and pesticides, and contaminants (such as Cd)
reaching agricultural land as impurities in fertilizers and manures.
Organic contaminants are those that have a carbon skeleton, usually associated with atoms
of H, O, N, P and S. These atoms may form an integral part of the molecule or they can be present
in functional groups. (Andrews, et al.) Functional groups are chemical structures that impart specific
chemical properties on a molecule. For example, hydroxyl (-OH) and carboxyl (-COOH) functional
groups makes a molecule more soluble by increasing its polarity. Carboxyl groups also make the
molecule acidic due to dissociation of H+. The various structural forms of organic molecules -
Module 20_Chemistry of the Soil (Part 2): Page 6 of 11
saturated and unsaturated chains and rings -and of functional groups, result in a diverse range
of organic contaminants.
Once the organic contaminants are in the soil, they may either dissipate by moving
in/interacting with the soil atmosphere, soil solution, mineral fractions and organic matter, or they
could persist and accumulate.
Volatile organic compounds (VOCs) like benzene, toluene, xylenes, dichloromethane, and
more, may contaminate soil in industrialized and commercialized areas, particularly in countries with
lax enforcement of regulations. One of the more common sources of these contaminants is leaking
underground storage tanks, improperly discarded solvents, and landfills.
Figure 3c.5.1. A few examples of organic contaminants
Organic contaminants interact mainly with either the mineral or organic components of soils.
Two types of non-reactive interactions are possible:
i) adsorption (a surface phenomenon); and
ii) entrapment within the soil minerals or components.
The interactions that can happen are dependent on the concentration, the properties of the
molecule — solubility, vapor pressure, and hydrophobicity — and the properties of the soil. The
combined result of adsorption and entrapment is the decrease in the availability of soil-associated
contaminants with time. Ageing, the decrease of the amount of compound in the ‘available’
fraction and the increase of the amount of compound in the ‘non-available’ fraction as time
passes, may cause the degree and speed of degradation of an organic contaminant to lessen (i.e. the
lessening of reactants, in this case the ‘available’ contaminants causes the speed of conversion to
‘non-available’ contaminants to slow down).
Organic contaminants attached to soil particles exist in four fractions which differ in the ease
with which they can be released/desorbed from the particles:
i) a rapidly desorbable fraction (‘detaches’ quickly from soil particles);
ii) a slowly desorbable fraction (‘detaches’ slowly from soil particles);
iii) a very slowly desorbable fraction (‘detaches’ very slowly from soil particles); and
iv) non-extractable (bound) residues.
The non-extractable/bound residues are the organic compounds (or its metabolites) that persists
in the soil matrix by ‘binding’ with humus during humification, largely through bacterial action. It is
Module 20_Chemistry of the Soil (Part 2): Page 7 of 11
very difficult to extract them using solvents and they are resistant to biological and chemical attack.
They represent an extreme end-member of ageing.
The amount of each fraction varies and is dependent on how long the contaminant and the soil
have been interacting.
Degradation of Organic Contaminants in Soils
The degradation of organic contaminants in soils occurs by either chemical (including
photochemical) or microbiological means. The effectiveness of degradation depends much on the
contaminant availability (i.e. how much is left ‘available’), although the degree of persistence is also
influenced by the contaminant’s chemical structure. If the chemical structure of the contaminant
is similar to that of a natural substance it is more likely to be degradable. In general, if the
structure is complex the rate of degradation is slower and is more likely to be incomplete.
Resistance to biodegradation is known as recalcitrance. It is caused by a number of factors:
1. Specific microbes or enzymes required for degradation may not be present in the soil.
2. Unusual or complex substitutions in a molecule (e.g. Cl, Br, or F, or any unusual bonds/bond
sequences) may ‘confuse’ microorganisms.
3. A high degree of aromaticity (the more benzene rings) results in molecules that are difficult to break
down.
4. Large, complex and heavy molecules tend to be less water soluble, and therefore are physically
unavailable to microorganisms that use intracellular degradation processes.
Biodegradation of organic contaminants is the result of the efforts of a mixture of microbes.
The degradation process ranges from only minor structural changes to the parent molecule
(primary degradation) to the complete conversion of the parent molecule into to mineral
constituents (mineralization), for example CO2 or H2O.
It is important to note that even the slightest structural molecular changes to the parent contaminant
molecule through biodegradation can significantly alter original contaminant’s mobility, toxicity, and
its affinity for soil surfaces.
MECHANISMS OF MICROBIAL DEGRADATION TRANSFORMATION:
1. Mineralization. the complete conversion of the parent molecule into to mineral constituents
2. Polymerization. A metabolic transformation that involves the coupling/bonding of small molecules
to form polymers.
Module 20_Chemistry of the Soil (Part 2): Page 8 of 11
3. Detoxification. Detoxification involves a chemical change to a molecule resulting to a less toxic
compound. Detoxification by one group of microorganisms often allows other organisms to continue
biodegradation.
Pentachlorophenol (PCP), a powerful biocide, undergoes O-methylation to produce a far less toxic
compound pentachloroanisole.
4. Co-metabolism. Although many aromatic compounds are subject to microbial degradation, the
presence of unusual substituents (e.g. Cl, Br, CH3, etc.) on the molecule can result in an increase in
their resistance to biodegradation. In these instances, microbes that would normally recognize the
molecule as a substrate get ‘confused’ and fail to do so because of the presence of the novel
substituents. Because of this, they do not produce enzymes to destroy the compounds. This co-
metabolism process occurring when organic compounds and substituted compounds account for the
degradation of many xenobiotic (foreign to life) compounds.
For example, biphenyl (unsubstituted) is readily degraded. However, polychlorinated biphenyls
(PCBs), for example 2,3,2’,4’-tetrachlorinated biphenyl shown above, do not trigger microorganisms
to degrade them.
Interestingly where BOTH biphenyls and PCBs are present together, both are degraded. This is
because the microbes recognize the biphenyl and produce enzymes to degrade it. The same enzymes
degrade the PCBs and thus both contaminants are removed.
5. Accumulation within microbes. Certain microorganisms, particularly those with high lipid (fat)
contents, can absorb water-insoluble chemicals. Although this is not strictly biodegradation, it is a
process that removes pollutants from the environment.
Module 20_Chemistry of the Soil (Part 2): Page 9 of 11
SUMMARY
• Macronutrients are the elements that are present in the plant material/inside the plant in
significant amounts: C, H, O, N, P, K, Ca, Mg, S. Micronutrients on the other hand are essential
only at very small amounts and may be toxic at high levels: B, Cl, Cu, Fe, Mn, Mo, Zn, etc.
• The major components of fertilizers are N, P, and K. They are designated number combinations
like x-y-z, which shows the percentages of N (x %), P as P2O5 (y %), and K2O (z %). Micronutrients
such as magnesium and sulfate may also be added to the formula.
• Soil receives large quantities of waste products and is more capable of retaining pollutants due
to its predominantly solid nature.
• Organic contaminants are those that have a carbon skeleton, usually associated with atoms of
H, O, N, P and S. These atoms may form an integral part of the molecule or they can be present
in functional groups.
• Once the organic contaminants are in the soil, they may either dissipate by moving
in/interacting with the soil atmosphere, soil solution, mineral fractions and organic matter, or
they could persist and accumulate.
• Organic contaminants can undergo two types of non-reactive interactions:
i. adsorption
ii. entrapment within the soil mineral/components
• Ageing, the decrease of the amount of compound in the ‘available’ fraction and the increase of
the amount of compound in the ‘non-available’ fraction as time passes, may cause the degree
and speed of degradation of an organic contaminant to slow down.
• Non-extractable/bound residues are the organic compounds (or its metabolites) that persists in
the soil matrix by ‘binding’ with humus during humification. They are resistant to biological and
chemical attack. They represent an extreme end-member of ageing.
• The degradation of organic contaminants in soils occurs by either chemical (including
photochemical) or microbiological means.
• If the chemical structure of the contaminant is similar to that of a natural substance it is more
likely to be degradable. Or, if the structure is complex the rate of degradation is slower and is
more likely to be incomplete.
• Factors that cause resistance to biodegradation (recalcitrance):
1. Specific microbes/enzymes required for degradation may not be present.
2. Unusual or complex substitutions in a molecule may ‘confuse’ microorganisms.
3. A high degree of aromaticity results in molecules that are difficult to break down.
4. Large, complex, and heavy molecules tend to be less water soluble, and therefore are
physically unavailable to microorganisms that use intracellular degradation processes.
• Biodegradation ranges from only minor structural changes to the parent molecule (primary
degradation) to the complete conversion of the parent molecule into to mineral constituents
(mineralization).
Module 20_Chemistry of the Soil (Part 2): Page 10 of 11
• MECHANISMS OF MICROBIAL DEGRADATION TRANSFORMATION:
1. Mineralization
2. Polymerization
3. Detoxification
4. Co-metabolism
5. Accumulation within microbes
Self-Assessment 22
1. In most soils, over 90% of the _______________ content is organic.
2. Legumes are plants that host bacteria on their roots capable of _______________.
3. The numbers in a notation for fertilizer such as 8-13-8 refers to, respectively,
_______________.
4. How many moles of H+ ion are consumed when 200 kilograms of NaNO3 undergo
denitrification in soil? (clue: check the chemical reaction for denitrification)
5. Of the following phenomena, the one that eventually makes soil more basic is
(a) removal of metal cations by roots
(b) leaching of soil with CO2-saturated water
(c) oxidation of soil pyrite
(d) fertilization with (NH4)2SO4
(e) fertilization with KNO3
6. How many metric tons of farm manure are equivalent to 100 kg of 10-5-10 fertilizer? Farm
manure is similar to 0.5-0.24-0.5 fertilizer.
References:
1. Andrews, et. al. An Introduction to Environmental Chemistry, 2nd Ed. Blackwell Science Ltd.
2004.
2. Harrison, Roy. Principles of Environmental Chemistry. The Royal Society of Chemistry.
Cambridge. 2007.
3. Manahan, Stanley. Fundamentals of Environmental Chemistry, 2nd Ed. Boca Raton: CRC Press
LLC. 2001.
Module 20_Chemistry of the Soil (Part 2): Page 11 of 11
You might also like
- Vat Receipt: MR Kyle BoydDocument1 pageVat Receipt: MR Kyle BoydNovak Brodsky83% (6)
- Cox-Murray - Apollo The Race of The MoonDocument329 pagesCox-Murray - Apollo The Race of The MoonHipatia50% (2)
- Concept Note For Trade FairDocument5 pagesConcept Note For Trade FairRalph Aldrin F. VallesterosNo ratings yet
- The Eyes of The Skin - Opinion Piece.Document2 pagesThe Eyes of The Skin - Opinion Piece.Disha RameshNo ratings yet
- Nitrogen Fixing PlantsDocument4 pagesNitrogen Fixing PlantsJayanth BvNo ratings yet
- Nitrogen in PlantsDocument10 pagesNitrogen in PlantsSusana CabotajeNo ratings yet
- Fertilizer PDFDocument9 pagesFertilizer PDFAmmr MahmoodNo ratings yet
- NitrogenDocument19 pagesNitrogenKayıtsız ŞartsızNo ratings yet
- 5-Role of Plant in Waste Management REVISEDDocument10 pages5-Role of Plant in Waste Management REVISEDommimishra0No ratings yet
- Plant Nutrition - WikipediaDocument87 pagesPlant Nutrition - WikipediaBashiir NuurNo ratings yet
- Soil and Plant NutritionDocument85 pagesSoil and Plant NutritionFarhadullah KhanNo ratings yet
- Nutrient CyclingDocument24 pagesNutrient CyclingSofia Dicen100% (1)
- NutrientsDocument37 pagesNutrientsarifismailbayrakNo ratings yet
- Chapter 5-7Document38 pagesChapter 5-7mekonnenNo ratings yet
- Fludizer ReportsDocument29 pagesFludizer Reportsکبری ادریس رسولNo ratings yet
- Biological Process in Soil: Mayrina FirdayatiDocument19 pagesBiological Process in Soil: Mayrina FirdayatiEster MelindaNo ratings yet
- What Are The Causes of Soil AcidityDocument7 pagesWhat Are The Causes of Soil AcidityReySimpleTuber renorNo ratings yet
- Essay Sythetic FertilizersDocument5 pagesEssay Sythetic FertilizersMichelle Francisca ArsjadNo ratings yet
- The Cycle of Nitrogen Sulphur & Carbon - Analysis by FahmidDocument5 pagesThe Cycle of Nitrogen Sulphur & Carbon - Analysis by FahmidFahmid Tousif KhanNo ratings yet
- Mikro I Makro Elementi ENGDocument14 pagesMikro I Makro Elementi ENGDusan MilosevicNo ratings yet
- Membrane UnitDocument15 pagesMembrane UnitHalo BashirNo ratings yet
- Assignment No 2 (Abdullah Tariq)Document5 pagesAssignment No 2 (Abdullah Tariq)Usman KhanNo ratings yet
- Lecture 10 SoilDocument3 pagesLecture 10 Soilayse0119No ratings yet
- Course WorkDocument4 pagesCourse WorkMuhindo ShamsuddiinNo ratings yet
- Nitrogen Cycle: Ecological FunctionDocument8 pagesNitrogen Cycle: Ecological FunctionApam BenjaminNo ratings yet
- Fertilizer Industry Handbook - October 2018 (With Notes)Document97 pagesFertilizer Industry Handbook - October 2018 (With Notes)stanley nyatondoNo ratings yet
- Ecology 2Document8 pagesEcology 2Cowboy XxNo ratings yet
- NMRG Soil Nutrients, Sources and Uptake PDFDocument9 pagesNMRG Soil Nutrients, Sources and Uptake PDFYosephNo ratings yet
- Chem Form 4, Fertilisers and NitrogenDocument3 pagesChem Form 4, Fertilisers and NitrogenshshbshshsjsjNo ratings yet
- FertilizerDocument15 pagesFertilizerLuckySharmaNo ratings yet
- Oral Presentation On Fertilizer by Group 3: Course Title: Inorganic Chemical Technology Course Code: CHE 515Document31 pagesOral Presentation On Fertilizer by Group 3: Course Title: Inorganic Chemical Technology Course Code: CHE 515mutencoNo ratings yet
- REAL WORK EMMA EDET THREEDocument60 pagesREAL WORK EMMA EDET THREEBless UgbongNo ratings yet
- 7 AnswerDocument4 pages7 AnswerTeamireab DestaNo ratings yet
- Agrochemical LCDocument7 pagesAgrochemical LCIndri SuciNo ratings yet
- 11.7 Nitrogen Transformations-FinalDocument6 pages11.7 Nitrogen Transformations-FinalPeter DindahNo ratings yet
- AGR 4303 Plant Nutrition - Topic2Document60 pagesAGR 4303 Plant Nutrition - Topic2Iain Choong WKNo ratings yet
- Macronutrients in Plant Tissue Culture (N, MG, S)Document32 pagesMacronutrients in Plant Tissue Culture (N, MG, S)bukankhadam100% (1)
- Nutrient Management in Nursery and Floriculture: Publication 8221Document9 pagesNutrient Management in Nursery and Floriculture: Publication 8221Viji ThulasiramanNo ratings yet
- Plants: Zinc Hyperaccumulation in Plants: A ReviewDocument22 pagesPlants: Zinc Hyperaccumulation in Plants: A Reviewbob krelNo ratings yet
- Nitrogen SourcesDocument3 pagesNitrogen SourcesRoque VirgilioNo ratings yet
- SSC AssignmentDocument6 pagesSSC Assignmentesomchi obioraNo ratings yet
- Macroelements - Michelle A. MorongDocument4 pagesMacroelements - Michelle A. MorongMarcelito MorongNo ratings yet
- The Nature and Properties of Soils (9-10)Document80 pagesThe Nature and Properties of Soils (9-10)Cristóbal GuerreroNo ratings yet
- Plant Nutrition and GrowthDocument7 pagesPlant Nutrition and GrowthSavannah Simone PetrachenkoNo ratings yet
- Biological N FixationDocument9 pagesBiological N Fixationtri cutNo ratings yet
- Chem of GardeningDocument13 pagesChem of GardeningCrystal DavidsonNo ratings yet
- Nitrogen CycleDocument3 pagesNitrogen CycleAliyaNo ratings yet
- So 402Document12 pagesSo 402Salome EtefiaNo ratings yet
- Plant Nutrition Is The Study of The: Emanuel EpsteinDocument24 pagesPlant Nutrition Is The Study of The: Emanuel EpsteinArkei FortalezaNo ratings yet
- Zinc in Crop Production and Interaction With PhosphorusDocument8 pagesZinc in Crop Production and Interaction With PhosphorusSebastian Garcia romeroNo ratings yet
- C.6 The Nitrogen and Phosphorus Cycles (AHL) : Essential Idea: Soil Cycles Are Subject To DisruptionDocument22 pagesC.6 The Nitrogen and Phosphorus Cycles (AHL) : Essential Idea: Soil Cycles Are Subject To DisruptionAna Sofia Guzman FernandezNo ratings yet
- Day 2 Soil OrganismsDocument16 pagesDay 2 Soil OrganismsBrían GohNo ratings yet
- Ciclos Elementos No SoloDocument18 pagesCiclos Elementos No SoloAnanda AntonioNo ratings yet
- Plant NutritionDocument19 pagesPlant NutritionOjok JodNo ratings yet
- Algae As A Soil Conditioner: Saurabh Prakash & Kumar NikhilDocument3 pagesAlgae As A Soil Conditioner: Saurabh Prakash & Kumar NikhilerpublicationNo ratings yet
- Lesson 3 PLANT AND ANIMAL NUTRITIONDocument6 pagesLesson 3 PLANT AND ANIMAL NUTRITIONNicoleNo ratings yet
- Nitrogen CycleDocument5 pagesNitrogen CycleVamsi K Kuchibhotla100% (1)
- Nutrient Cycles: Prof. Murray, Univ of Illinois at ChicagoDocument35 pagesNutrient Cycles: Prof. Murray, Univ of Illinois at Chicagomahesh nagarNo ratings yet
- Plant Nutrition 3 Plant ChapterDocument10 pagesPlant Nutrition 3 Plant ChapterRaihanda Dwimart MangaweNo ratings yet
- Crop Sci 2-Unit III-Soil Nutrient Cycling-SY 23-24Document6 pagesCrop Sci 2-Unit III-Soil Nutrient Cycling-SY 23-24Gaivereal TagabeNo ratings yet
- Plant NutrientsDocument10 pagesPlant NutrientsAdrian GligaNo ratings yet
- BNF by FrankiaDocument11 pagesBNF by FrankiaShalini MNo ratings yet
- Handbook of Fertilizers - Their Sources, Make-Up, Effects, and UseFrom EverandHandbook of Fertilizers - Their Sources, Make-Up, Effects, and UseRating: 4.5 out of 5 stars4.5/5 (4)
- Nitrogen Bacteria and Legumes: With special reference to red clover, cowpeas, soy beans, alfalfa, and sweet clover, on Illinois soilsFrom EverandNitrogen Bacteria and Legumes: With special reference to red clover, cowpeas, soy beans, alfalfa, and sweet clover, on Illinois soilsNo ratings yet
- Module 16 - UNIT IV - Chemistry of The AtmosphereDocument10 pagesModule 16 - UNIT IV - Chemistry of The AtmosphereJhess GaliciaNo ratings yet
- Module 19 - UNIT IV - Chemistry of The Soil (Part 1)Document8 pagesModule 19 - UNIT IV - Chemistry of The Soil (Part 1)Jhess GaliciaNo ratings yet
- Module 9 - UNIT II - Fuels (Part 1)Document12 pagesModule 9 - UNIT II - Fuels (Part 1)Jhess GaliciaNo ratings yet
- Module 18 - UNIT IV - Chemistry of Water (Part 2)Document8 pagesModule 18 - UNIT IV - Chemistry of Water (Part 2)Jhess GaliciaNo ratings yet
- Module 14 - UNIT III - MetalsDocument7 pagesModule 14 - UNIT III - MetalsJhess GaliciaNo ratings yet
- Module 10 - UNIT II - Fuels (Part 2)Document7 pagesModule 10 - UNIT II - Fuels (Part 2)Jhess GaliciaNo ratings yet
- MMW Module 1 Nature of MathematicsDocument6 pagesMMW Module 1 Nature of MathematicsJhess GaliciaNo ratings yet
- Triple IDocument1 pageTriple IJhess GaliciaNo ratings yet
- MMW Chapter 2 Language of Set-1Document25 pagesMMW Chapter 2 Language of Set-1Jhess GaliciaNo ratings yet
- Module 15 - UNIT III - PolymersDocument12 pagesModule 15 - UNIT III - PolymersJhess GaliciaNo ratings yet
- Mathematics For Engineers - Unit 3-1Document17 pagesMathematics For Engineers - Unit 3-1Jhess GaliciaNo ratings yet
- Loops and CountersDocument13 pagesLoops and CountersJhess GaliciaNo ratings yet
- Dr. Wifanto-Management Liver Metastasis CRCDocument46 pagesDr. Wifanto-Management Liver Metastasis CRCAfkar30No ratings yet
- Letter VirajDocument1 pageLetter VirajPratyushAgarwalNo ratings yet
- Auto Build ZDocument52 pagesAuto Build ZLittle AyaNo ratings yet
- Smart Battery Data Specification Revision 1.1 ErrataDocument5 pagesSmart Battery Data Specification Revision 1.1 ErrataredmsbatteryNo ratings yet
- Stage 7 SampleDocument48 pagesStage 7 SampleYug ChotaiNo ratings yet
- Paste Thickener PDFDocument13 pagesPaste Thickener PDFAfshin MirzaeiNo ratings yet
- Spare Parts Price List - DeluxeDocument13 pagesSpare Parts Price List - Deluxesrikanthsri4uNo ratings yet
- Personal Branding Workbook - Ext - 2nd EdDocument23 pagesPersonal Branding Workbook - Ext - 2nd EdMiguelito JeromeNo ratings yet
- BCG MatrixDocument18 pagesBCG Matrixsamm78992% (12)
- Avnet TP Sap Arp PL May14Document77 pagesAvnet TP Sap Arp PL May14Kathak DancerNo ratings yet
- Fire SafetyDocument31 pagesFire SafetyNaga FireballNo ratings yet
- Section Capacity Section Capacity: AbutmentDocument4 pagesSection Capacity Section Capacity: AbutmentGajendra SNo ratings yet
- 6 - Telephone and Cable Networks For Data TransmissionDocument31 pages6 - Telephone and Cable Networks For Data TransmissionpranjalcrackuNo ratings yet
- Wiley American Anthropological AssociationDocument3 pagesWiley American Anthropological AssociationSimeon Pol'shinNo ratings yet
- Attachment - 9387 - 63bd823872cf8 - 63bd81f56e412 - A Man Like None Other 101 110Document29 pagesAttachment - 9387 - 63bd823872cf8 - 63bd81f56e412 - A Man Like None Other 101 110BURAK DEMIRNo ratings yet
- FFC New Imp FormatDocument1 pageFFC New Imp FormatMalik Zaryab babarNo ratings yet
- Punjab Police SI Intelligence Officer Model PapersDocument33 pagesPunjab Police SI Intelligence Officer Model Papershk3987gmailcomNo ratings yet
- Orient Ship Management & Manning Pvt. LTD.: Licence No. RPSL-MUM-019 Validity: 15.02.20 21Document2 pagesOrient Ship Management & Manning Pvt. LTD.: Licence No. RPSL-MUM-019 Validity: 15.02.20 21anujNo ratings yet
- 0650-EDW-00009-02 - Seaking DST Parts List - Customer Parts ListDocument11 pages0650-EDW-00009-02 - Seaking DST Parts List - Customer Parts ListJosianeMacielNo ratings yet
- WiFi Troubleshooting CheatSheetDocument21 pagesWiFi Troubleshooting CheatSheetVee Pal100% (1)
- Cap Table Cheat SheetDocument5 pagesCap Table Cheat SheetNasrul SalmanNo ratings yet
- Coa MyobDocument4 pagesCoa Myobalthaf alfadliNo ratings yet
- 7th History Workbook English Medium Maharashtra BoardDocument12 pages7th History Workbook English Medium Maharashtra Boardpankarvi6100% (1)
- 3is FORMATDocument10 pages3is FORMATAMACIO, MARIAN JOY A.No ratings yet
- Work Shop Practice-Ii: Diploma in Mechanical EngineeringDocument36 pagesWork Shop Practice-Ii: Diploma in Mechanical EngineeringLEAGUE OF WARLORDSNo ratings yet