Professional Documents
Culture Documents
PH Vs Logarithm Curve
PH Vs Logarithm Curve
Uploaded by
Sabarno Saha0 ratings0% found this document useful (0 votes)
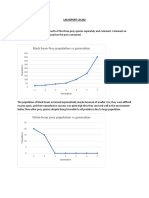
13 views1 pageThis graph shows the relationship between pH and the logarithm scale. The x-axis represents pH from 4 to 5.5, while the y-axis likely represents the logarithm of the hydrogen ion concentration or hydronium ion concentration. There appears to be an inverse relationship between pH and the logarithm, as pH increases the logarithm decreases in a logarithmic fashion.
Original Description:
Original Title
Untitled
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis graph shows the relationship between pH and the logarithm scale. The x-axis represents pH from 4 to 5.5, while the y-axis likely represents the logarithm of the hydrogen ion concentration or hydronium ion concentration. There appears to be an inverse relationship between pH and the logarithm, as pH increases the logarithm decreases in a logarithmic fashion.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
13 views1 pagePH Vs Logarithm Curve
PH Vs Logarithm Curve
Uploaded by
Sabarno SahaThis graph shows the relationship between pH and the logarithm scale. The x-axis represents pH from 4 to 5.5, while the y-axis likely represents the logarithm of the hydrogen ion concentration or hydronium ion concentration. There appears to be an inverse relationship between pH and the logarithm, as pH increases the logarithm decreases in a logarithmic fashion.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
pH vs logarithm curve
.5 4 4.5 5 5.5
pH
You might also like
- Sa&Vol FormulaeDocument1 pageSa&Vol FormulaeComputer ioNo ratings yet
- Formulas For Perimeter, Area, and Volume: 5 2l 5 2w 5 LW 5 4s 5 SDocument1 pageFormulas For Perimeter, Area, and Volume: 5 2l 5 2w 5 LW 5 4s 5 SRediet UtteNo ratings yet
- 2021 AN PH EquivalenceDocument4 pages2021 AN PH EquivalenceTrần Văn ViễnNo ratings yet
- Serie ProteínasDocument4 pagesSerie Proteínas316223744No ratings yet
- Biokimia Sel: Dr. Retno Sintowati, M.SCDocument99 pagesBiokimia Sel: Dr. Retno Sintowati, M.SCshella arlitaNo ratings yet
- Eco380 A2 Q2Document2 pagesEco380 A2 Q2rachelyuningliuNo ratings yet
- Sianvar - Virtual Vain (Trae)Document2 pagesSianvar - Virtual Vain (Trae)Biswajith ManimaranNo ratings yet
- Principles - Of.Foundation - Engineering. - 9th - Ed - Braja.M.Das 843 PDFDocument1 pagePrinciples - Of.Foundation - Engineering. - 9th - Ed - Braja.M.Das 843 PDFjcvalenciaNo ratings yet
- UntitledDocument1 pageUntitledSabarno SahaNo ratings yet
- Conductance Vs Amount of NaohDocument1 pageConductance Vs Amount of NaohSabarno SahaNo ratings yet
- Relative Abundance Vs Abundance RankDocument1 pageRelative Abundance Vs Abundance RankSabarno SahaNo ratings yet
- Distant Integer Signals: Identity, Maths Club of IISER KolkataDocument13 pagesDistant Integer Signals: Identity, Maths Club of IISER KolkataSabarno SahaNo ratings yet
- Black Bean Prey Population Vs GenerationDocument5 pagesBlack Bean Prey Population Vs GenerationSabarno SahaNo ratings yet
- PH Vs Logarithm CurveDocument1 pagePH Vs Logarithm CurveSabarno SahaNo ratings yet