Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
36 viewsEjercicio Cationes y Aniones
Ejercicio Cationes y Aniones
Uploaded by
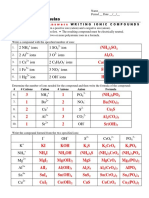
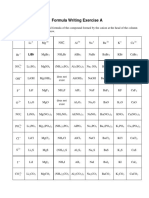
Manuel BarreraThis document lists common cations and anions that form ionic compounds, including metal cations such as Cu+, Cu2+, Fe2+, Fe3+, Cr3+, Al3+, Ti4+, Pb2+, Pb4+ and non-metal anions such as F-, Cl-, H-, O2-, S2-, N3-, P3-, C4-, (OH)-, (CN)-, (NO3)-, (CO3)2-, (SO4)2-, (PO4)3- and (NH4)+. The cations and anions shown can combine to form various ionic compounds through the transfer of electrons from metals to nonmetals.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- List of Common Polyatomic IonsDocument1 pageList of Common Polyatomic Ionsvia_alzahra100% (1)
- Chemical Formula Writing WorksheetDocument4 pagesChemical Formula Writing Worksheetprabhu4321100% (1)
- Ions - MemorizeDocument1 pageIons - MemorizeThea Clarice AmlonNo ratings yet
- Polyatomic Ions: +1 Cations - 2 AnionsDocument2 pagesPolyatomic Ions: +1 Cations - 2 AnionsAngelica Nehmyah CuevasNo ratings yet
- Naming & Balancing Chemical Formula - Sheet1Document1 pageNaming & Balancing Chemical Formula - Sheet1arseniy kraschenkoNo ratings yet
- Anion Schematic DiagramDocument1 pageAnion Schematic DiagramAlfie16No ratings yet
- IonsDocument1 pageIonsurbiztondotoniNo ratings yet
- TUpload 2Document1 pageTUpload 2Burikaw GamingNo ratings yet
- Chemical Formula Writing Worksheet PDFDocument4 pagesChemical Formula Writing Worksheet PDFkezia0% (1)
- Valence of Common Ions and RadicalsDocument3 pagesValence of Common Ions and RadicalsFrederick FranciscoNo ratings yet
- W3 02 Naming Chemical Formulas of CompoundsDocument20 pagesW3 02 Naming Chemical Formulas of CompoundsResmiel IrishNo ratings yet
- Cajepe, Cherry May F. Bses 1a ChemistryDocument4 pagesCajepe, Cherry May F. Bses 1a ChemistryNilda FranciscoNo ratings yet
- Ion Old Iupac Ion Old Name IupacDocument1 pageIon Old Iupac Ion Old Name IupacchelliNo ratings yet
- Polyatomic Ion Master ListDocument1 pagePolyatomic Ion Master ListAldrin Morallos MaglahusNo ratings yet
- Ion Memorization ListDocument2 pagesIon Memorization Listdchao94No ratings yet
- 7 - Chemical Formulas: AnswersDocument1 page7 - Chemical Formulas: AnswersAlyssa Mae MayonadoNo ratings yet
- Advanced-Chem Q1 LP7Document6 pagesAdvanced-Chem Q1 LP7Francesca BuenoNo ratings yet
- Common Ion ChargesDocument1 pageCommon Ion ChargesMoganan SubramaniamNo ratings yet
- Common Elements FFF Edit 11-12-09Document1 pageCommon Elements FFF Edit 11-12-09FFFNo ratings yet
- AP/Dual Ion Chart: Monatomic IonsDocument1 pageAP/Dual Ion Chart: Monatomic IonsReid SullivanNo ratings yet
- Ion Old Iupac Ion Old Name IupacDocument1 pageIon Old Iupac Ion Old Name IupacjenduekieNo ratings yet
- Polyatomic IonsDocument1 pagePolyatomic IonsJames PerriamNo ratings yet
- Ions and Their Charges: Metals With Variable Oxidation NumbersDocument1 pageIons and Their Charges: Metals With Variable Oxidation Numbers123 123No ratings yet
- JRS Tutorials: Chemistry IITDocument58 pagesJRS Tutorials: Chemistry IITtusharr11.mobNo ratings yet
- Information - Sheet - Merge 2Document3 pagesInformation - Sheet - Merge 2Frederick NakosNo ratings yet
- Charges 2Document2 pagesCharges 2athenaNo ratings yet
- Chemical Bonding: Why Bond Anyway?Document45 pagesChemical Bonding: Why Bond Anyway?PutRi Charolin GintingNo ratings yet
- Formula Writing - CambridgeDocument5 pagesFormula Writing - CambridgeQusai Saify100% (3)
- Formulas of Compounds Polyatomics KEYDocument2 pagesFormulas of Compounds Polyatomics KEYJewel Emerald C. CudiamatNo ratings yet
- CW 4 Unit 5 - Chemical FormulaDocument1 pageCW 4 Unit 5 - Chemical Formulamohammad hasanNo ratings yet
- List of Cations and AnionsDocument3 pagesList of Cations and Anionsrabihchahal1No ratings yet
- Ion and AnionDocument1 pageIon and AnionciciNo ratings yet
- Common Ions and Their ChargesDocument1 pageCommon Ions and Their ChargesNonbinaryBroadwayNo ratings yet
- Activity 8 NomenclatureDocument2 pagesActivity 8 NomenclatureCyruss MeranoNo ratings yet
- Common Ions ListDocument2 pagesCommon Ions Listms.makaylacelineNo ratings yet
- Cations & Anions WorksheetDocument1 pageCations & Anions WorksheetGapor examNo ratings yet
- Formula WritingDocument20 pagesFormula Writinggian manlangitNo ratings yet
- AP Chemistry Summer Assignment 2017Document44 pagesAP Chemistry Summer Assignment 2017John SmithNo ratings yet
- Write The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Document2 pagesWrite The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Bea Lha Zandra BesingaNo ratings yet
- GenChem Nomenclature Updated PDFDocument2 pagesGenChem Nomenclature Updated PDFCamille AquinoNo ratings yet
- Ox. No & StateDocument2 pagesOx. No & StateajaxNo ratings yet
- Balancing PracticeDocument4 pagesBalancing PracticehtyhongNo ratings yet
- ANACHEMLABDocument3 pagesANACHEMLABPrincessNo ratings yet
- Chem HS HandOutsDocument2 pagesChem HS HandOutsKristianne Mae EchavezNo ratings yet
- Topic 3-Chemical Bonding-Exercise 1.2Document2 pagesTopic 3-Chemical Bonding-Exercise 1.2abd rahmanNo ratings yet
- Daftar MineralDocument8 pagesDaftar Mineraliky taniaNo ratings yet
- Cations and AnionsDocument2 pagesCations and AnionsG2 Atacador, Channa Keavy B.No ratings yet
- Elements (Anions) Symbol Oxidation NO. Elements (Anions) Symbol Oxidation NODocument5 pagesElements (Anions) Symbol Oxidation NO. Elements (Anions) Symbol Oxidation NOJims Cudinyerah100% (1)
- Common Ion and Their ChargesDocument1 pageCommon Ion and Their ChargesMika CossidNo ratings yet
- Naming Compounds: College of Arts and SciencesDocument2 pagesNaming Compounds: College of Arts and SciencesJalina, Emmanuel JoseNo ratings yet
- COLOUROf IONICCOMPOUNDSDocument2 pagesCOLOUROf IONICCOMPOUNDSkrutika goharkarNo ratings yet
- Chemistry ReviewerDocument4 pagesChemistry ReviewerBhel San Pedro MarzanNo ratings yet
- Polyatomic Ions ListDocument2 pagesPolyatomic Ions Listapi-256236481No ratings yet
- UntitledDocument2 pagesUntitledapi-233404189No ratings yet
- List of Cations and AnionsDocument2 pagesList of Cations and AnionsArvin MagtotoNo ratings yet
- NamingCmpdsHandoutKey_013158Document4 pagesNamingCmpdsHandoutKey_013158natashalouisse103No ratings yet
- ChemistryDocument4 pagesChemistrySaubhagyaAroraNo ratings yet
- Sarah Compound Dice ActivityDocument3 pagesSarah Compound Dice ActivityAnaria ManojNo ratings yet
- Ionic Compounds - Working Out The Formula and NameDocument4 pagesIonic Compounds - Working Out The Formula and NameHakim AbbasNo ratings yet
Ejercicio Cationes y Aniones
Ejercicio Cationes y Aniones
Uploaded by
Manuel Barrera0 ratings0% found this document useful (0 votes)
36 views2 pagesThis document lists common cations and anions that form ionic compounds, including metal cations such as Cu+, Cu2+, Fe2+, Fe3+, Cr3+, Al3+, Ti4+, Pb2+, Pb4+ and non-metal anions such as F-, Cl-, H-, O2-, S2-, N3-, P3-, C4-, (OH)-, (CN)-, (NO3)-, (CO3)2-, (SO4)2-, (PO4)3- and (NH4)+. The cations and anions shown can combine to form various ionic compounds through the transfer of electrons from metals to nonmetals.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document lists common cations and anions that form ionic compounds, including metal cations such as Cu+, Cu2+, Fe2+, Fe3+, Cr3+, Al3+, Ti4+, Pb2+, Pb4+ and non-metal anions such as F-, Cl-, H-, O2-, S2-, N3-, P3-, C4-, (OH)-, (CN)-, (NO3)-, (CO3)2-, (SO4)2-, (PO4)3- and (NH4)+. The cations and anions shown can combine to form various ionic compounds through the transfer of electrons from metals to nonmetals.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
36 views2 pagesEjercicio Cationes y Aniones
Ejercicio Cationes y Aniones
Uploaded by
Manuel BarreraThis document lists common cations and anions that form ionic compounds, including metal cations such as Cu+, Cu2+, Fe2+, Fe3+, Cr3+, Al3+, Ti4+, Pb2+, Pb4+ and non-metal anions such as F-, Cl-, H-, O2-, S2-, N3-, P3-, C4-, (OH)-, (CN)-, (NO3)-, (CO3)2-, (SO4)2-, (PO4)3- and (NH4)+. The cations and anions shown can combine to form various ionic compounds through the transfer of electrons from metals to nonmetals.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 2
Aniones
Cationes F-1 Cl-1 H-1 O-2 S-2 N-3 P-3 C-4
Cu+1
Cu+2
Fe+2
Fe+3
Cr+3
Al+3
Ti+4
Pb+2
Pb+4
Aniones
Cationes (OH)-1 (CN)-1 (NO3)-1 (CO3)-2 (SO4)-2 (PO4)-3
Cu+1
Cu+2
Fe+2
Fe+3
Cr+3
Al+3
Ti+4
Pb+2
Pb+4
(NH4)+1
You might also like
- List of Common Polyatomic IonsDocument1 pageList of Common Polyatomic Ionsvia_alzahra100% (1)
- Chemical Formula Writing WorksheetDocument4 pagesChemical Formula Writing Worksheetprabhu4321100% (1)
- Ions - MemorizeDocument1 pageIons - MemorizeThea Clarice AmlonNo ratings yet
- Polyatomic Ions: +1 Cations - 2 AnionsDocument2 pagesPolyatomic Ions: +1 Cations - 2 AnionsAngelica Nehmyah CuevasNo ratings yet
- Naming & Balancing Chemical Formula - Sheet1Document1 pageNaming & Balancing Chemical Formula - Sheet1arseniy kraschenkoNo ratings yet
- Anion Schematic DiagramDocument1 pageAnion Schematic DiagramAlfie16No ratings yet
- IonsDocument1 pageIonsurbiztondotoniNo ratings yet
- TUpload 2Document1 pageTUpload 2Burikaw GamingNo ratings yet
- Chemical Formula Writing Worksheet PDFDocument4 pagesChemical Formula Writing Worksheet PDFkezia0% (1)
- Valence of Common Ions and RadicalsDocument3 pagesValence of Common Ions and RadicalsFrederick FranciscoNo ratings yet
- W3 02 Naming Chemical Formulas of CompoundsDocument20 pagesW3 02 Naming Chemical Formulas of CompoundsResmiel IrishNo ratings yet
- Cajepe, Cherry May F. Bses 1a ChemistryDocument4 pagesCajepe, Cherry May F. Bses 1a ChemistryNilda FranciscoNo ratings yet
- Ion Old Iupac Ion Old Name IupacDocument1 pageIon Old Iupac Ion Old Name IupacchelliNo ratings yet
- Polyatomic Ion Master ListDocument1 pagePolyatomic Ion Master ListAldrin Morallos MaglahusNo ratings yet
- Ion Memorization ListDocument2 pagesIon Memorization Listdchao94No ratings yet
- 7 - Chemical Formulas: AnswersDocument1 page7 - Chemical Formulas: AnswersAlyssa Mae MayonadoNo ratings yet
- Advanced-Chem Q1 LP7Document6 pagesAdvanced-Chem Q1 LP7Francesca BuenoNo ratings yet
- Common Ion ChargesDocument1 pageCommon Ion ChargesMoganan SubramaniamNo ratings yet
- Common Elements FFF Edit 11-12-09Document1 pageCommon Elements FFF Edit 11-12-09FFFNo ratings yet
- AP/Dual Ion Chart: Monatomic IonsDocument1 pageAP/Dual Ion Chart: Monatomic IonsReid SullivanNo ratings yet
- Ion Old Iupac Ion Old Name IupacDocument1 pageIon Old Iupac Ion Old Name IupacjenduekieNo ratings yet
- Polyatomic IonsDocument1 pagePolyatomic IonsJames PerriamNo ratings yet
- Ions and Their Charges: Metals With Variable Oxidation NumbersDocument1 pageIons and Their Charges: Metals With Variable Oxidation Numbers123 123No ratings yet
- JRS Tutorials: Chemistry IITDocument58 pagesJRS Tutorials: Chemistry IITtusharr11.mobNo ratings yet
- Information - Sheet - Merge 2Document3 pagesInformation - Sheet - Merge 2Frederick NakosNo ratings yet
- Charges 2Document2 pagesCharges 2athenaNo ratings yet
- Chemical Bonding: Why Bond Anyway?Document45 pagesChemical Bonding: Why Bond Anyway?PutRi Charolin GintingNo ratings yet
- Formula Writing - CambridgeDocument5 pagesFormula Writing - CambridgeQusai Saify100% (3)
- Formulas of Compounds Polyatomics KEYDocument2 pagesFormulas of Compounds Polyatomics KEYJewel Emerald C. CudiamatNo ratings yet
- CW 4 Unit 5 - Chemical FormulaDocument1 pageCW 4 Unit 5 - Chemical Formulamohammad hasanNo ratings yet
- List of Cations and AnionsDocument3 pagesList of Cations and Anionsrabihchahal1No ratings yet
- Ion and AnionDocument1 pageIon and AnionciciNo ratings yet
- Common Ions and Their ChargesDocument1 pageCommon Ions and Their ChargesNonbinaryBroadwayNo ratings yet
- Activity 8 NomenclatureDocument2 pagesActivity 8 NomenclatureCyruss MeranoNo ratings yet
- Common Ions ListDocument2 pagesCommon Ions Listms.makaylacelineNo ratings yet
- Cations & Anions WorksheetDocument1 pageCations & Anions WorksheetGapor examNo ratings yet
- Formula WritingDocument20 pagesFormula Writinggian manlangitNo ratings yet
- AP Chemistry Summer Assignment 2017Document44 pagesAP Chemistry Summer Assignment 2017John SmithNo ratings yet
- Write The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Document2 pagesWrite The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Bea Lha Zandra BesingaNo ratings yet
- GenChem Nomenclature Updated PDFDocument2 pagesGenChem Nomenclature Updated PDFCamille AquinoNo ratings yet
- Ox. No & StateDocument2 pagesOx. No & StateajaxNo ratings yet
- Balancing PracticeDocument4 pagesBalancing PracticehtyhongNo ratings yet
- ANACHEMLABDocument3 pagesANACHEMLABPrincessNo ratings yet
- Chem HS HandOutsDocument2 pagesChem HS HandOutsKristianne Mae EchavezNo ratings yet
- Topic 3-Chemical Bonding-Exercise 1.2Document2 pagesTopic 3-Chemical Bonding-Exercise 1.2abd rahmanNo ratings yet
- Daftar MineralDocument8 pagesDaftar Mineraliky taniaNo ratings yet
- Cations and AnionsDocument2 pagesCations and AnionsG2 Atacador, Channa Keavy B.No ratings yet
- Elements (Anions) Symbol Oxidation NO. Elements (Anions) Symbol Oxidation NODocument5 pagesElements (Anions) Symbol Oxidation NO. Elements (Anions) Symbol Oxidation NOJims Cudinyerah100% (1)
- Common Ion and Their ChargesDocument1 pageCommon Ion and Their ChargesMika CossidNo ratings yet
- Naming Compounds: College of Arts and SciencesDocument2 pagesNaming Compounds: College of Arts and SciencesJalina, Emmanuel JoseNo ratings yet
- COLOUROf IONICCOMPOUNDSDocument2 pagesCOLOUROf IONICCOMPOUNDSkrutika goharkarNo ratings yet
- Chemistry ReviewerDocument4 pagesChemistry ReviewerBhel San Pedro MarzanNo ratings yet
- Polyatomic Ions ListDocument2 pagesPolyatomic Ions Listapi-256236481No ratings yet
- UntitledDocument2 pagesUntitledapi-233404189No ratings yet
- List of Cations and AnionsDocument2 pagesList of Cations and AnionsArvin MagtotoNo ratings yet
- NamingCmpdsHandoutKey_013158Document4 pagesNamingCmpdsHandoutKey_013158natashalouisse103No ratings yet
- ChemistryDocument4 pagesChemistrySaubhagyaAroraNo ratings yet
- Sarah Compound Dice ActivityDocument3 pagesSarah Compound Dice ActivityAnaria ManojNo ratings yet
- Ionic Compounds - Working Out The Formula and NameDocument4 pagesIonic Compounds - Working Out The Formula and NameHakim AbbasNo ratings yet