Professional Documents
Culture Documents
Results
Results
Uploaded by
Alejandro OsunaOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Results
Results
Uploaded by
Alejandro OsunaCopyright:
Available Formats
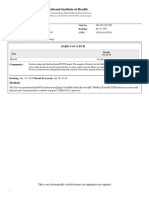
Report Lab Director: Yo-Chan Jeong, PhD
CLIA Number: 21D2077230
Name Req Form # Collected Client
Eduardo Osuna CDR09220094 02/18/2022 Ed Smith Stadium
Address Accession # Received Address
12007 Cypress Run Drive Orlando, FL, 32836 2229920 02/19/2022 2700 12th St. Sarasota, FL, 34237
Patient ID Gender Date of Birth Race Report Date Ordering Provider
1253039 Male 04/04/1962 Unknown 02/19/2022 Narendra Kini, MD
Ordered Test(s): PCR COVID-19 Collection Device: Nasopharyngeal swab Collection Medium: UTM
PCR COVID-19: Negative
Comments
Covid-19 Negative: No Covid-19 markers detected from valid specimen.
Real Time PCR Pathogen Results
COVID-19 genes Results Control Results
N_gene Negative MS2 Detected
ORF_1ab Negative
S_gene Negative
MS2 is an internal control. A Detected result for MS2 is normal and indicates sample integrity. A Not Detected result indicates sample integrity failed and the sample is Invalid.
Disclaimer: This assay is designed to detect the Orf-1ab, S, and N genes of COVID-19 using RNA reverse transcription and RT-PCR. A Negative result does not preclude the
possibility of COVID-19 infection since the adequacy of sample collection and/or low viral burden may result in the presence of viral nucleic acids below the analytical
sensitivity of this test method. Test results should be used along with other clinical and laboratory data in making the diagnosis. This test was developed and its performance
characteristics determined and validated by Real Diagnostics. The FDA has not approved or cleared this test. However, FDA clearance or approval is not currently required for
clinical use. The results are intended to be used only for clinical diagnosis.
If you have any questions regarding this report please contact your provider.
Paitent phone: (786) 825-0914 Patient email: osunaedu@gmail.com
Osuna, Eduardo 200 Business Center Dr , Reisterstown, Maryland 21136 Page 1 / 1
You might also like
- Test Bank For Introduction To Maternity and Pediatric Nursing 8th Edition by LeiferDocument36 pagesTest Bank For Introduction To Maternity and Pediatric Nursing 8th Edition by Leiferpatnesspertain.e2nd2o100% (48)
- Negative: What Does It Mean To Have A Test Result?Document2 pagesNegative: What Does It Mean To Have A Test Result?robertoNo ratings yet
- Margaret McWilliams-Fundamentals of Meal ManagementDocument361 pagesMargaret McWilliams-Fundamentals of Meal ManagementSaher Yasin89% (9)
- Covid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedDocument1 pageCovid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedRodel OrtegaNo ratings yet
- MrsSNIGDHA 43Y FemaleDocument3 pagesMrsSNIGDHA 43Y FemalePathkind LabNo ratings yet
- Lab Literacy for Doctors: A Guide to Ordering the Right Tests for Better Patient CareFrom EverandLab Literacy for Doctors: A Guide to Ordering the Right Tests for Better Patient CareRating: 3.5 out of 5 stars3.5/5 (3)
- Hypervibe BookDocument66 pagesHypervibe BookHypervibe1No ratings yet
- PCR COVID-19: Negative: If You Have Any Questions Regarding This Report Please Contact Your ProviderDocument1 pagePCR COVID-19: Negative: If You Have Any Questions Regarding This Report Please Contact Your ProviderLuis ReyesNo ratings yet
- Covid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedDocument1 pageCovid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedGEr JrvillaruElNo ratings yet
- Covid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not DetectedDocument1 pageCovid-19 Test Result Summary: Sars-Cov-2 Viral Rna - Not Detectedpogito ramosNo ratings yet
- Roshan Singh-Male28 Years-195636Document2 pagesRoshan Singh-Male28 Years-195636govind samantNo ratings yet
- RTPCR of Pramod YadavDocument2 pagesRTPCR of Pramod YadavPramod YadavNo ratings yet
- Report CXX220621001 1655831565220Document2 pagesReport CXX220621001 1655831565220CKNo ratings yet
- Molecular Diagnostics Molecular Diagnostics Molecular Diagnostics Molecular DiagnosticsDocument3 pagesMolecular Diagnostics Molecular Diagnostics Molecular Diagnostics Molecular DiagnosticsNiraj DeyNo ratings yet
- Molecular Diagnostics Molecular Diagnostics Molecular Diagnostics Molecular DiagnosticsDocument3 pagesMolecular Diagnostics Molecular Diagnostics Molecular Diagnostics Molecular DiagnosticsNiraj DeyNo ratings yet
- Molecular Diagnostics: Report Status - Final Test Name Result Biological Ref. Interval UnitDocument3 pagesMolecular Diagnostics: Report Status - Final Test Name Result Biological Ref. Interval UnitARVIND SINGHNo ratings yet
- Satyendra Kumar Sharma-Male31 Years-119413Document2 pagesSatyendra Kumar Sharma-Male31 Years-119413Firoz KhanNo ratings yet
- Take Care Sa GensanDocument1 pageTake Care Sa GensanAya BeeNo ratings yet
- Molecular Diagnostics: Report Status - Final Test Name Result Biological Ref. Interval UnitDocument3 pagesMolecular Diagnostics: Report Status - Final Test Name Result Biological Ref. Interval UnitARVIND SINGHNo ratings yet
- ResultDocument1 pageResultNandini Pritesh PatelNo ratings yet
- Department of Serology Covid-19 Antigen Test: Covid 19 Ag Test, Test Name Result Unit Bio. Ref. Range MethodDocument1 pageDepartment of Serology Covid-19 Antigen Test: Covid 19 Ag Test, Test Name Result Unit Bio. Ref. Range MethodPradeep VunnamNo ratings yet
- Corona Report-26 DecDocument1 pageCorona Report-26 DecRana BasitNo ratings yet
- FrmPatientViewAllReport - 2021-05-14T213255.543Document3 pagesFrmPatientViewAllReport - 2021-05-14T213255.543Manoj KumarNo ratings yet
- Report of F - CH Maribel Snigitha CiceroDocument2 pagesReport of F - CH Maribel Snigitha CiceroR.Pearlsis SophiNo ratings yet
- Navdeep Madheshiya Report - 2022-01-15t183422.725Document1 pageNavdeep Madheshiya Report - 2022-01-15t183422.725navdeep madheshiyaNo ratings yet
- COVID 19 PCR Sample ReportDocument1 pageCOVID 19 PCR Sample ReportRamanamma PosaNo ratings yet
- C284146 Covidtempl Abdul 050821164842Document1 pageC284146 Covidtempl Abdul 050821164842Sarhm SevenNo ratings yet
- Department of Molecular Biology. Covid 19 RTPCR With Home Collection Test Name Result Unit Bio. Ref. Range MethodDocument2 pagesDepartment of Molecular Biology. Covid 19 RTPCR With Home Collection Test Name Result Unit Bio. Ref. Range MethodPritam JanaNo ratings yet
- De La Cuesta, Joseph Adrian - Negative Covid TestDocument3 pagesDe La Cuesta, Joseph Adrian - Negative Covid TestRegi PonferradaNo ratings yet
- Alauddin Ansari Male53 Years 119412Document2 pagesAlauddin Ansari Male53 Years 119412Firoz KhanNo ratings yet
- Prueba Antigenos JesusDocument1 pagePrueba Antigenos JesusJesus RicaurteNo ratings yet
- Test Name Result Ref. Range Unit: COVID 19 PCR FacilityDocument2 pagesTest Name Result Ref. Range Unit: COVID 19 PCR Facilitybikramjit debNo ratings yet
- BooksDocument1 pageBooksCollin LongNo ratings yet
- Negative: What Does It Mean To Have A Test Result?Document2 pagesNegative: What Does It Mean To Have A Test Result?PerlaNo ratings yet
- Covid ReportDocument1 pageCovid ReportGourima BabbarNo ratings yet
- Testing Lab: Quality Healthcare Is A Human RightDocument1 pageTesting Lab: Quality Healthcare Is A Human RightNikhil JainNo ratings yet
- JOUD SAMER DALLOUL-10052021 (1) (1) - For MergeDocument2 pagesJOUD SAMER DALLOUL-10052021 (1) (1) - For MergeJalal El DeanNo ratings yet
- Testing LabDocument1 pageTesting LabKavita AryaNo ratings yet
- Buttler, Irene Dr. Thornton, Karen 17851920Document1 pageButtler, Irene Dr. Thornton, Karen 17851920ahmedNo ratings yet
- Sars Cov2 (Covid-19) RT-PCR Test: Molecular BiologyDocument1 pageSars Cov2 (Covid-19) RT-PCR Test: Molecular BiologyBina MistryNo ratings yet
- RT 4Document1 pageRT 4Bhumika Desai It's very creative.No ratings yet
- ArvindDocument2 pagesArvindSukhmeet SinghNo ratings yet
- Nusaim 2rtpcrDocument1 pageNusaim 2rtpcrNawfal ShaikNo ratings yet
- National Institute of Health: Sars-Cov-2 PCRDocument1 pageNational Institute of Health: Sars-Cov-2 PCRSardar Shohaib SadiqNo ratings yet
- rtpcr2022 PDFDocument3 pagesrtpcr2022 PDFf20202449No ratings yet
- Check Out This File: COV-350851-1-SARS-CoV-2 - 2019-nCoV-1632039566Document1 pageCheck Out This File: COV-350851-1-SARS-CoV-2 - 2019-nCoV-1632039566Joana Marie DomingoNo ratings yet
- Patient Report Direct ViewDocument1 pagePatient Report Direct ViewafdsaNo ratings yet
- CDMDL 22 024227Document1 pageCDMDL 22 024227aleeNo ratings yet
- Covid-19 RT-PCR Laboratory Result Form: City of Dasmariñas Molecular Diagnostic LaboratoryDocument1 pageCovid-19 RT-PCR Laboratory Result Form: City of Dasmariñas Molecular Diagnostic LaboratoryCelineNo ratings yet
- 11201501261RTPCR Rakesh PatelDocument1 page11201501261RTPCR Rakesh PatelRakesh PatelNo ratings yet
- Testing LabDocument1 pageTesting LabVijay ThakurNo ratings yet
- SMSHLD tPEkEzDocument1 pageSMSHLD tPEkEzPranjal JindalNo ratings yet
- Vargas, Juana Dr. Thornton, Karen 17675278Document1 pageVargas, Juana Dr. Thornton, Karen 17675278ahmedNo ratings yet
- Nishi Kumari RTPCRDocument2 pagesNishi Kumari RTPCRvikasNo ratings yet
- Sajal AgarwalDocument1 pageSajal AgarwalMayank JunejaNo ratings yet
- KSD 2021072142Document1 pageKSD 2021072142Akshaya kunjthurNo ratings yet
- Patients Profile: Not Detected NegativeDocument2 pagesPatients Profile: Not Detected NegativeELLIE JAMES PLACIONo ratings yet
- Covid-19 RT-PCR: Test Results PanelDocument1 pageCovid-19 RT-PCR: Test Results PanelPatricia Cottle-SalyerNo ratings yet
- Microbiology Test Result Microbiology RT PCR Sars Cov-2Document1 pageMicrobiology Test Result Microbiology RT PCR Sars Cov-2Anita Rezeki CarolinaNo ratings yet
- Velasco, Crestita VelosoDocument1 pageVelasco, Crestita VelosoAdan NunungNo ratings yet
- Dinesh RamDocument1 pageDinesh RamchandanNo ratings yet
- This Is A Computer Generated Form and If Issued Without Any Alteration, This Does Not Require A SignatureDocument1 pageThis Is A Computer Generated Form and If Issued Without Any Alteration, This Does Not Require A SignatureIssa LlamasNo ratings yet
- CT1208835 2057029459 Files indivPDF 2020 7 2 2057029459-1Document1 pageCT1208835 2057029459 Files indivPDF 2020 7 2 2057029459-1M GeeNo ratings yet
- Physio Neuro Ax TemplateDocument8 pagesPhysio Neuro Ax TemplateHussain LaftaNo ratings yet
- Journal of Attention Disorders: Executive Function Impairments in High IQ Adults With ADHDDocument8 pagesJournal of Attention Disorders: Executive Function Impairments in High IQ Adults With ADHDLauren SerafiniNo ratings yet
- Professions and Occupations in BoliviaDocument7 pagesProfessions and Occupations in BoliviaMaribel Jarata JulianNo ratings yet
- Hospitals BolangirDocument16 pagesHospitals BolangirSam PattnaikNo ratings yet
- Sustainable Building Assessment ToolDocument0 pagesSustainable Building Assessment ToolEnd FernandesNo ratings yet
- Peptic Ulcer Disease: NCM 112 Laboratory Case StudyDocument6 pagesPeptic Ulcer Disease: NCM 112 Laboratory Case StudyClair DomingoNo ratings yet
- Which Career Pathway Is Right For You?Document6 pagesWhich Career Pathway Is Right For You?Kinelyn BaguioNo ratings yet
- Course Hero 1Document8 pagesCourse Hero 1sekhon.rajdeep27No ratings yet
- Essiac Tea Is A Cancer Cure Big Pharma Does Not Want You To Know AboutDocument3 pagesEssiac Tea Is A Cancer Cure Big Pharma Does Not Want You To Know Aboutwekesamadzimoyo1100% (1)
- Enema PDFDocument2 pagesEnema PDFSandy Jusi100% (1)
- Classification and Diagnosis of Hypertensive Disorders of PregnancyDocument6 pagesClassification and Diagnosis of Hypertensive Disorders of PregnancyZAHID AHMED KHANNo ratings yet
- Q3 COT - LP HEALTH7-WEEK2 (Kinds of Stress)Document4 pagesQ3 COT - LP HEALTH7-WEEK2 (Kinds of Stress)Lemuel Español CamusNo ratings yet
- Anticoagulation For Chronic Kidney DisiesesDocument9 pagesAnticoagulation For Chronic Kidney DisiesesorelglibNo ratings yet
- Senior Project Outline Salam ADocument3 pagesSenior Project Outline Salam Aapi-588486278No ratings yet
- Perdev: Personality Development SyllabusDocument10 pagesPerdev: Personality Development SyllabusJelly Marie Baya FloresNo ratings yet
- Irc CVDocument4 pagesIrc CVEmbay AmareNo ratings yet
- Sesion 3 Articulo Seminario 2 PsoriasisDocument16 pagesSesion 3 Articulo Seminario 2 PsoriasisQuispe Canares MariangelesNo ratings yet
- Fast Food Vs Home FoodDocument2 pagesFast Food Vs Home FoodMounir EimanNo ratings yet
- Orchitis 160406115741Document28 pagesOrchitis 160406115741Right VentricleNo ratings yet
- MSDS - Stanbio Kolesterol TotalDocument4 pagesMSDS - Stanbio Kolesterol TotalDoni Eka PrasetiyoNo ratings yet
- Implementing Culture Change in Health Care: Theory and PracticeDocument15 pagesImplementing Culture Change in Health Care: Theory and PracticeElvisa FitriNo ratings yet
- Case Study 1-1Document9 pagesCase Study 1-1api-242226130No ratings yet
- Acute Heart FailureDocument24 pagesAcute Heart FailureTeddy MauriceNo ratings yet
- PMR 2022 0017 PDFDocument7 pagesPMR 2022 0017 PDFStory WA DokterNo ratings yet
- Hazard Identification Risk Assessment and Determine Control (HIRADC)Document28 pagesHazard Identification Risk Assessment and Determine Control (HIRADC)Shida ShidotNo ratings yet
- Benelam 2009 Nutrition - BulletinDocument48 pagesBenelam 2009 Nutrition - BulletinCarlos Julio Fonseca SilvaNo ratings yet
- CHN BawangDocument3 pagesCHN BawangNicole MapiliNo ratings yet