Professional Documents
Culture Documents
Amines All Sheet
Amines All Sheet
Uploaded by
Mahendra ShahOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Amines All Sheet
Amines All Sheet
Uploaded by
Mahendra ShahCopyright:
Available Formats
TG: @Chalnaayaaar
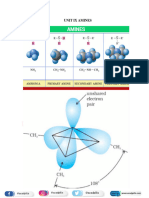
Amines
Part-01
Amines can be considered as derivatives of ammonia obtained by replacement of one, two or all the three
hydrogen atoms by alkyl/aryl groups.
Classification of Amines
Amines are classified according to the number of carbon atoms bonded directly to the nitrogen atom.
Methods of Preparation of amines
1. Reduction of nitro compounds
Nitro compounds are reduced to amines by passing hydrogen gas in the presence of finely divided nickel,
palladium or platinum and also by reduction with metals in acidic medium.
2. Reduction of nitriles(cyanides) and isonitriles (isocyanides)
Nitriles on reduction with lithium aluminium hydride (LiAlH4) or catalytic hydrogenation produce primary amines
but isonitriles produce secondary amines.
Digital Pvt. Ltd. [1]
TG: @Chalnaayaaar
Amines Part-01
3. Reduction of amides
Amides on reduction with lithium aluminium hydride (LiAlH4) produce amines.
4. Ammonolysis of alkyl halides
Alkyl halide on reaction with ethanolic solution of ammonia undergoes nucleophilic substitution in which the
halogen atom is replaced by amino (-NH2) group. The process of cleavage of C-X bond by ammonia is known
as Ammonolysis.
NH3 + R—X ⎯→ R—NH2
Ammonolysis reaction yields a mixture of primary, secondary, tertiary amines and also quaternary ammonium
salt. However primary amine can be obtained as major product taking large excess of ammonia and quaternary
ammonium salt will be major product if alkyl halide is taken in excess.
5. Gabriel phthalimide synthesis
Gabriel synthesis is used for the preparation of 1° aliphatic amines.
Phthalimide on treatment with ethanolic potassium hydroxide forms potassium salt of phthalimide which on
reaction with alkyl halide followed by alkaline or acidic hydrolysis produces the corresponding primary amine.
Mechanism
Digital Pvt. Ltd. [2]
TG: @Chalnaayaaar
Amines
Part-02
Methods of Preparation of amines
6. Hoffmann bromamide degradation reaction
Reagents that may be used for this reaction
1. X2 + alkaline medium
Br2 + NaOH
Br2 + KOH
2. NaOX (NaOCl, NaOBr etc.)
KOX (KOCl, KOBr etc.)
Physical Properties of Amines
Solubility
1. Lower aliphatic amines are soluble in water because they can form hydrogen bonds with water molecules.
2. Solubility decreases with increase in molar mass of amines due to increase in size of the hydrophobic alkyl part.
3. Higher amines are essentially insoluble in water.
Boiling point
Primary and secondary amines are engaged in intermolecular association due to hydrogen bonding between
nitrogen of one and hydrogen of another molecule.
This intermolecular association is more in primary amines than in secondary amines as there are two hydrogen
atoms available for hydrogen bond formation in it.
Tertiary amines do not have intermolecular association due to the absence of hydrogen atom required for
hydrogen bond formation.
The order of boiling points of isomeric amines is as follows:
Primary > Secondary > Tertiary
Digital Pvt. Ltd. [1]
TG: @Chalnaayaaar
Amines Part-02
Chemical reactions of Amines
(1) Reaction showing basic nature
Amines being basic in nature react with acids to form salts.
Amine salts on treatment with a base like NaOH, regenerate the parent amine.
(2) Acylation reaction
Aliphatic and aromatic primary and secondary amines react with acid chlorides, anhydrides and esters by
nucleophilic substitution reaction.
(3) Hoffmann's carbylamine reaction or isocyanide test
Aliphatic and aromatic primary amines on heating with chloroform and ethanolic potassium hydroxide form
isocyanides or carbylamines which are foul smelling substances.
Note: Dichlorocarbene is formed in this reaction.
Digital Pvt. Ltd. [2]
TG: @Chalnaayaaar
Amines
Part-03
Chemical reactions of Amines
(4) Reaction with Benzenesulphonyl chloride (Hinsberg’s reagent)
Note:
Hinsberg test is used to distinguish between primary, secondary and tertiary amines.
These days benzenesulphonyl choride is replaced by p-toluenesulphonyl chloride (TsCl)
(5) Reaction with nitrous acid (HNO2)
Digital Pvt. Ltd. [1]
TG: @Chalnaayaaar
Amines Part-03
(a) 1° Aliphatic amine
Mechanism
(b) 1° Aromatic amine
If the temperature of the diazonium salt solution is allowed to rise upto 283K, the salt gets hydrolysed to phenol.
(c) 2° Aromatic amine
(d) 3° Aromatic amine
Digital Pvt. Ltd. [2]
TG: @Chalnaayaaar
Amines Part-03
Chemical reaction of Diazonium salts
A. Reactions involving displacement of nitrogen
B. Reactions involving retention of diazo group
Coupling reaction of benzene diazonium chloride (ESR)
Benzene diazonium chloride reacts with aromatic compounds containing strongly activating groups like
–OH, –NH2, –NHR, –NR2
Digital Pvt. Ltd. [3]
TG: @Chalnaayaaar
Amines
Part-04
Chemical reactions of Amines
(6) Electrophilic Substitution Reaction (ESR)
Halogenation of Aniline
Nitration of Aniline
Illustration
Sulphonation of Aniline
Note:
Aniline does not show Friedel Crafts reaction (alkylation and acylation)
Digital Pvt. Ltd. [1]
TG: @Chalnaayaaar
Amines Part-04
Reduction of Nitrobenzene
Digital Pvt. Ltd. [2]
You might also like
- Chemistry Paper 2 SLDocument16 pagesChemistry Paper 2 SLSonia InezaNo ratings yet
- 04 Reaction Mechanisms Full Reddy SirDocument114 pages04 Reaction Mechanisms Full Reddy SirMihica KhareNo ratings yet
- 15.coordination Compounds PDFDocument40 pages15.coordination Compounds PDFP. E. I. AcademicsNo ratings yet
- 12 Chemistry Notes ch13 AminesDocument11 pages12 Chemistry Notes ch13 AminesAkNo ratings yet
- Revision Notes For Class 12 CBSE Chemistry, Amines - TopperlearningDocument11 pagesRevision Notes For Class 12 CBSE Chemistry, Amines - TopperlearningRishabh BhandariNo ratings yet
- NamereactionorganicDocument13 pagesNamereactionorganicdeykrishna654100% (1)
- DPP Percentage - 2 (1) 5321633Document2 pagesDPP Percentage - 2 (1) 5321633netra7222No ratings yet
- Hydrocarbons NEET PYQ SOLUTIONDocument22 pagesHydrocarbons NEET PYQ SOLUTIONsomeone nooneNo ratings yet
- CBSE Salt AnalysisDocument4 pagesCBSE Salt Analysiskarmanya67% (6)
- Important Questions For CBSE Class 12 Chemistry The P-Block ElementsDocument41 pagesImportant Questions For CBSE Class 12 Chemistry The P-Block ElementsyndtfndtgndNo ratings yet
- Neet Mock Test - 1Document20 pagesNeet Mock Test - 1won wonNo ratings yet
- Acid Bases and Salts - Shobhit NirwanDocument21 pagesAcid Bases and Salts - Shobhit NirwanBhaskar 8287100% (1)
- Class 12 Chemistry MCQDocument19 pagesClass 12 Chemistry MCQTayseer SaudiaNo ratings yet
- DPP GoccccDocument10 pagesDPP GoccccMayur Khichi0% (1)
- Mole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations atDocument1 pageMole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations atmyiitchemistry50% (2)
- CLS Aipmt 19 20 XII Che Study Package 4 Level 1 Chapter 10Document36 pagesCLS Aipmt 19 20 XII Che Study Package 4 Level 1 Chapter 10Utkarsh KumarNo ratings yet
- CLS Aipmt-18-19 XI Che Study-Package-2 SET-2 Chapter-6 PDFDocument50 pagesCLS Aipmt-18-19 XI Che Study-Package-2 SET-2 Chapter-6 PDFSarang2426No ratings yet
- DPP Quantum Number JH Sir-3574Document5 pagesDPP Quantum Number JH Sir-3574shiva royNo ratings yet
- S - Block ElementsDocument34 pagesS - Block ElementssubesinghNo ratings yet
- Part - A Section - I: CLASS X (2020-21) Mathematics Standard (041) Sample Paper-07Document11 pagesPart - A Section - I: CLASS X (2020-21) Mathematics Standard (041) Sample Paper-07Anand Balakrishnan100% (1)
- Class 10th Chemistry - Maha Marathon (Prashant Kirad)Document114 pagesClass 10th Chemistry - Maha Marathon (Prashant Kirad)LMRP2 LMRP2100% (1)
- Aakash Chemistry Study Package 6 SolutionsDocument103 pagesAakash Chemistry Study Package 6 SolutionsSamuel Moris Mandanakka100% (1)
- Course-Planner Full DetailsDocument8 pagesCourse-Planner Full Detailsmanish365No ratings yet
- CLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFDocument40 pagesCLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFThavasimariselvam N100% (1)
- Organic Named Reactions PDFDocument8 pagesOrganic Named Reactions PDFAshis BisoyiNo ratings yet
- Name ReactionsDocument10 pagesName ReactionsParam SoniNo ratings yet
- Ms Chauhan Organic Chemistry PDFDocument2 pagesMs Chauhan Organic Chemistry PDFLovely Chetan0% (1)
- CBSE G+10 Chemical+Reactions+and+Equations NotesDocument9 pagesCBSE G+10 Chemical+Reactions+and+Equations Notesrd199159No ratings yet
- Amines PDFDocument34 pagesAmines PDFRam KhannaNo ratings yet
- Chemistry Kcet 2021Document9 pagesChemistry Kcet 2021Kirti Vinodkumar JainNo ratings yet
- Paper A93Document25 pagesPaper A93MP12No ratings yet
- SN DPPDocument34 pagesSN DPPtyagiabhishek145No ratings yet
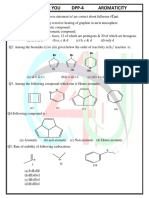
- Aromaticity DPP 4Document4 pagesAromaticity DPP 4SubhadeepNo ratings yet
- Nomenclature SheetDocument24 pagesNomenclature SheetEkta MishraNo ratings yet
- 01 Some Basic Concept of Chemistry Formula Sheets Quizrr PDFDocument7 pages01 Some Basic Concept of Chemistry Formula Sheets Quizrr PDFKalyana ChakravarthiNo ratings yet
- Grignard ReagentDocument7 pagesGrignard Reagentd anjilappa100% (1)
- IIT JEE Main Advnaced Physical Chemistry 12th ElectrochemistryDocument56 pagesIIT JEE Main Advnaced Physical Chemistry 12th ElectrochemistrySesha Sai KumarNo ratings yet
- ELECTROCHEMISTRYDocument4 pagesELECTROCHEMISTRYSAIRAJ PRABHAKAR PAWARNo ratings yet
- JA DPP No.A1 To A10 Answer KeyDocument2 pagesJA DPP No.A1 To A10 Answer KeyAkkaldevi Saivinayak CR100% (2)
- Aromatic CompoundsDocument107 pagesAromatic CompoundsNishantNo ratings yet
- Chemical Kinetic PDFDocument44 pagesChemical Kinetic PDFAmogh VaishnavNo ratings yet
- 33stoichiometry PDFDocument11 pages33stoichiometry PDFJessica ShamoonNo ratings yet
- 1001-Class XI - C-232.Mole Concept Assignment - 1Document5 pages1001-Class XI - C-232.Mole Concept Assignment - 1The GentlemanNo ratings yet
- Goc and Isomerism Notes - PMD - 1 PDFDocument46 pagesGoc and Isomerism Notes - PMD - 1 PDFrutvik bhoraniyaNo ratings yet
- VMC Salt AnalysisDocument53 pagesVMC Salt AnalysisAkash Mukherjee100% (3)
- CH13 Hydrocarbons Shobhit NirwanDocument58 pagesCH13 Hydrocarbons Shobhit NirwanpujaNo ratings yet
- Ncert Solutions For Class 11 Chemistry Jan11 Chapter 12 Organic Chemistry Some Basic Principles and TechniquesDocument32 pagesNcert Solutions For Class 11 Chemistry Jan11 Chapter 12 Organic Chemistry Some Basic Principles and TechniquesSachit GuptaNo ratings yet
- Last Leap 2Document395 pagesLast Leap 2mahendrasinghsumankumarNo ratings yet
- 28 Salt Analysis Revision Notes QuizrrDocument46 pages28 Salt Analysis Revision Notes Quizrrprince thakur100% (2)
- Chapter 10 Haloalkanes and HaloarenesDocument24 pagesChapter 10 Haloalkanes and HaloarenesSuhas GowdaNo ratings yet
- Qualitative Analysis - DPP'sDocument15 pagesQualitative Analysis - DPP'sVanshaj GuptaNo ratings yet
- The D and F Block ElementsDocument48 pagesThe D and F Block ElementsRocking vevo100% (1)
- Name Reactions of Organic ChemistryDocument7 pagesName Reactions of Organic ChemistryNaynam SharmaNo ratings yet
- Allen ChemistryDocument95 pagesAllen ChemistryAtharv SoniNo ratings yet
- SHM Formula Sheet @cbseinfiniteDocument6 pagesSHM Formula Sheet @cbseinfiniteRekha JainNo ratings yet
- 5.surface Chemistry Final 4-3-2014 PDFDocument16 pages5.surface Chemistry Final 4-3-2014 PDFArinjayNo ratings yet
- AminesDocument13 pagesAminesSohamNo ratings yet
- Amines Class 12 Notes NEET Chemistry (PDF)Document19 pagesAmines Class 12 Notes NEET Chemistry (PDF)Scar LNo ratings yet
- 12 Notes Ammines and AcidsDocument11 pages12 Notes Ammines and AcidsXyzdevuNo ratings yet
- CBSE Class 12 Chemistry - Amines Chapter NotesDocument12 pagesCBSE Class 12 Chemistry - Amines Chapter NotesGayathiriNo ratings yet
- AminesDocument12 pagesAminesEmelda BanumathyNo ratings yet
- SheetDocument30 pagesSheetMahendra ShahNo ratings yet
- EXERCISEDocument24 pagesEXERCISEMahendra ShahNo ratings yet
- Board Question Paper: July 2019: PhysicsDocument3 pagesBoard Question Paper: July 2019: PhysicsMahendra ShahNo ratings yet
- Biomolecules SheetDocument21 pagesBiomolecules SheetMahendra Shah100% (1)
- Atoms StructureDocument47 pagesAtoms StructureMahendra ShahNo ratings yet
- Cbse 11 Chap 1,2,3 MathstDocument2 pagesCbse 11 Chap 1,2,3 MathstMahendra ShahNo ratings yet
- Maths 11 SecondunitDocument1 pageMaths 11 SecondunitMahendra ShahNo ratings yet
- Chapter 11 (Dual Nature of Radiation and Matter) Unsolved-1Document4 pagesChapter 11 (Dual Nature of Radiation and Matter) Unsolved-1Mahendra ShahNo ratings yet
- Kendriya Vidyalaya Gachibowli, Hyderabad: Sample Paper 03: Periodic Test - 1 (2019 - 20) Class - Xi MathematicsDocument2 pagesKendriya Vidyalaya Gachibowli, Hyderabad: Sample Paper 03: Periodic Test - 1 (2019 - 20) Class - Xi MathematicsMahendra ShahNo ratings yet
- Atoms NuclearDocument39 pagesAtoms NuclearMahendra ShahNo ratings yet
- Hotel Booking Ref-2409210054733Document3 pagesHotel Booking Ref-2409210054733Mahendra ShahNo ratings yet
- Maths 2 NdunitDocument1 pageMaths 2 NdunitMahendra ShahNo ratings yet
- Toaz - Info CH 11 Three Dimentional Geometry Multiple Choice Questions With Answers PRDocument5 pagesToaz - Info CH 11 Three Dimentional Geometry Multiple Choice Questions With Answers PRMahendra ShahNo ratings yet
- Chapter 2 (Electrostatic Potential and Capacitance) UnsolvedDocument8 pagesChapter 2 (Electrostatic Potential and Capacitance) UnsolvedMahendra ShahNo ratings yet
- Chapter 11 (Dual Nature of Radiation and Matter) Unsolved-1Document4 pagesChapter 11 (Dual Nature of Radiation and Matter) Unsolved-1Mahendra ShahNo ratings yet
- Chapter 12 (Atoms) UnsolvedDocument4 pagesChapter 12 (Atoms) UnsolvedMahendra ShahNo ratings yet
- Board Question Paper: July 2018: PhysicsDocument3 pagesBoard Question Paper: July 2018: PhysicsMahendra ShahNo ratings yet
- Complete GOCand Mechanismwithkeysand SolutionsDocument139 pagesComplete GOCand Mechanismwithkeysand Solutionsarryan keshanNo ratings yet
- Chemistry Microproject 1Document12 pagesChemistry Microproject 1Aachal GulhaneNo ratings yet
- Elements Compounds Mixtures KuizDocument1 pageElements Compounds Mixtures KuizMOHD SAIFUNIZAM BIN ZAKARIA MoeNo ratings yet
- Lecture 2 - Atomic Structure and BondingDocument29 pagesLecture 2 - Atomic Structure and BondingMercylin MakambureNo ratings yet
- Advanced Micro-And Nano - Manufacturing Technologies: Shrikrishna Nandkishor Joshi Pranjal Chandra EditorsDocument404 pagesAdvanced Micro-And Nano - Manufacturing Technologies: Shrikrishna Nandkishor Joshi Pranjal Chandra EditorsDaniel MăriuțaNo ratings yet
- Atomic StructureDocument20 pagesAtomic StructureshinNo ratings yet
- Physics Galaxy Advanced IllustrationsDocument2 pagesPhysics Galaxy Advanced Illustrationsshreyas shahNo ratings yet
- Nuclear Chemistry (Selected Topics)Document76 pagesNuclear Chemistry (Selected Topics)Mahamudul Hasan MunnaNo ratings yet
- 01-01-2021 GOC2 English ADocument39 pages01-01-2021 GOC2 English APrabhakar BandaruNo ratings yet
- Experiment 2 - Densities of A Solid and A LiquidDocument6 pagesExperiment 2 - Densities of A Solid and A LiquidGelina LuceñoNo ratings yet
- AtomsDocument29 pagesAtomsGunek ChaddhaNo ratings yet
- CHEM 201 Organic Chemistry 1: Polar Covalent Bonds Acids and BasesDocument95 pagesCHEM 201 Organic Chemistry 1: Polar Covalent Bonds Acids and BasesTRIXIA NICOLE GARCIANo ratings yet
- Physics Exam Quick Revision NotesDocument40 pagesPhysics Exam Quick Revision NotesPinky Ann DanielNo ratings yet
- Me 311: Heat Transfer Assignment 1: INSTRUCTION: Answer The Following QuestionsDocument2 pagesMe 311: Heat Transfer Assignment 1: INSTRUCTION: Answer The Following QuestionsMahusay Neil DominicNo ratings yet
- Torsion and Scalar Canceling Coil Basics For ZPEDocument8 pagesTorsion and Scalar Canceling Coil Basics For ZPEIchbinsNo ratings yet
- Emission Spectra Data and More ..Document15 pagesEmission Spectra Data and More ..Mikaelia NesbethNo ratings yet
- dpeulgjh, Qwhuqdwlrqdo$6 $/hyho: 0D/-XQH KrxuplqxwhvDocument20 pagesdpeulgjh, Qwhuqdwlrqdo$6 $/hyho: 0D/-XQH Krxuplqxwhvchaitanya khemaniNo ratings yet
- EMJ17104Document35 pagesEMJ17104s231060031-5No ratings yet
- CHM 101 - Hybridization and Shapes of MoleculesDocument12 pagesCHM 101 - Hybridization and Shapes of Moleculeslokpored NojporesNo ratings yet
- Synopsis & Assignments - EAS by Prof. Ajay Yashpal KapoorDocument78 pagesSynopsis & Assignments - EAS by Prof. Ajay Yashpal KapoorRudranarayan DuttaNo ratings yet
- PrialnikDocument338 pagesPrialnikLouis JohnsonNo ratings yet
- Assignment Chem 353.1 TU Attempt ReviewDocument16 pagesAssignment Chem 353.1 TU Attempt ReviewJason SummatNo ratings yet
- Physics Higher Secondary Second YearDocument237 pagesPhysics Higher Secondary Second YearDhanalakshmi DhanalakshmiNo ratings yet
- Types of Energy and Their Transformation - 065437Document12 pagesTypes of Energy and Their Transformation - 065437Daniel CamarilloNo ratings yet
- Pharmaceutical Instrumental Analysis 2Document90 pagesPharmaceutical Instrumental Analysis 2Ayat AborassNo ratings yet
- Chemical Energetics Chemistry AS/A LevelDocument4 pagesChemical Energetics Chemistry AS/A Levelyep okNo ratings yet
- Dwnload Full Earth An Introduction To Physical Geology Canadian 4th Edition Tarbuck Test Bank PDFDocument36 pagesDwnload Full Earth An Introduction To Physical Geology Canadian 4th Edition Tarbuck Test Bank PDFpropelomnifi.cw3100% (16)
- Dr. Santosh Kumar Yadav Ph.D. (IIT Bombay) M.Sc. (BHU)Document23 pagesDr. Santosh Kumar Yadav Ph.D. (IIT Bombay) M.Sc. (BHU)Aditya Kumar ChaubeyNo ratings yet
- Section - A: Holiday Homework For Grade XiDocument7 pagesSection - A: Holiday Homework For Grade XiGM Ali KawsarNo ratings yet