Professional Documents
Culture Documents
Org Chem Rxns
Org Chem Rxns
Uploaded by
Akshit agarwalOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Org Chem Rxns
Org Chem Rxns
Uploaded by
Akshit agarwalCopyright:
Available Formats
Handout:
Organic Chemistry Reactions
Reactions Organized by Compound Families
Alkanes
1. Combustion
2. Halogenation
Alkenes and Alkynes
1. Additions: hydrogenation, halogenation,
hydrohalogenation, hydration
2. Polymerization
Aromatic Compounds
Substitutions: nitration, halogenation, sulfonation
Alcohols
1. Elimination: dehydration
2. Oxidations
Thiols
Oxidation
Amines
Acid-Base reactions
Aldehydes and Ketones
1. Addition: Acetal/hemiacetal formation by alcohol
addition
(Reverse rxn: Acetal hydrolysis with acid)
2. Oxidation and Reduction for aldehydes
(*Ketones go through reduction only)
Carboxylic Acids
1. Substitutions: esterification, amidation
(Reverse rxns: ester hydrolysis with acid or base, amide hydrolysis with acid or base)
2. Acid-base reactions
Phosphoric acid and Phosphates
Phosphoric acid: Esterification with alcohol
Phosphates: Phosphorylation
H. Kim for Chem 30B 1
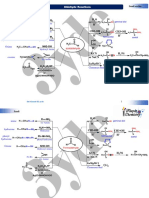
Reactions Organized by Reaction Type
Family Reaction Example
1. ADDITION
Alkenes and Hydrogenation H H Pd catalyst H H
C C + H2
alkynes H H
H C C H
H H
H H
Pd catalyst
H C C H + 2 H2 H C C H
H H
Halogenation H H Cl Cl
C C + Cl2 (or Br2) H C C H
H H
H H
Hydro- H H
H Cl Markonikov’s Rule
halogenation C C + HCl (or HBr) H C C H applies: H adds to C
H CH 3
H CH 3 with more Hs already.
Hydration H H H+ catalyst
H OH
C C + H2O H C C H Markonikov’s Rule
H CH 3
H CH 3 applies.
Aldehydes Addition of O OH
H + catalyst
and ketones alcohol to get H 3C C H(R) + CH 3CH2OH H 3C C H(R) + CH 3CH2OH
hemiacetals or O CH2CH 3
acetals hemiacetal
O CH2CH 3
H + catalyst
H 3C C H(R) + H 2O
O CH2CH 3
acetal
2. SUBSTITUTION
Aromatic Nitration H2SO 4 catalyst NO 2
compounds + HNO3
Halogenation FeCl3 (or FeBr3) catalyst Cl
+ Cl2 (or Br2)
Sulfonation H2SO 4 catalyst SO 3H
+ SO3
Carboxylic Esterification O H+ catalyst O
acids with alcohol H 3C C OH + CH 3CH2OH H 3C C OCH2CH 3 + H 2O
Amidation with O
heat
O
amine H 3C C OH + CH 3NH 2 H 3C C NHCH 3 + H 2O
Phosphoric Phosphoric acid: O O
acid and Esterification HO P OH + CH 3OH HO P OCH3 + H 2O
Phosphates with alcohol OH OH
Phosphates: O O O O O O
O P O P O- + ROH Adenosine O P O P O- +
Phosphorylation Adenosine O P RO P O- + Energy
O- O- O- O- O- O-
H. Kim for Chem 30B 2
3. ELIMINATION
Alcohols Dehydration H OH H2SO 4 catalyst H CH 3
H
H 3C C C CH 3 C C + H 3C C C CH2 + H 2O
H 3C H
H H H H
major minor
Zaitsev’s Rule applies: More highly substituted alkene is major
product.
4. OXIDATION/REDUCTION
Alcohols Oxidation OH
[O] O [O] O
1º R C H R C H R C OH
H aldehyde carboxylic acid
OH O
[O]
2º R C H R C R
R Ketone
OH
[O]
3º R C H No Rxn
R
Thiols Oxidation [O]
[H]
Reverse rxn: R-S-S-R 2 RSH
2 RSH R-S-S-R
Aldehydes Oxidation O [O] O
R C H R C OH
aldehyde carboxylic acid
Reduction O [H] OH
(also addition) R C H R C H
H
Ketones Reduction O
[H] OH
(also addition) R C CH 3 R C CH 3
H
5. ACID-BASE
Amines Rxn with water CH 3CH2NH 2 + H 2O CH 3CH2NH 3+ + OH –
(Basic)
Neutralization CH 3CH2NH 2 + H 3O+ CH 3CH2NH 3+ + H 2O
with strong acid CH 3CH2NH 2 + HCl CH 3CH2NH 3+ Cl –
to get ammonium Reverse rxn: ammonium
CH 3CH2NH 3+ Cl – + NaOH CH 3CH2NH 2 + NaCl + H 2O
ion or salt ion or salt + base
Carboxylic Rxn with water O O
acids H 3C C OH + H 2O
H 3C C O– + H 3O+
(Acidic) Neutralization O
O
with strong base H 3C C OH (aq) + NaOH (aq)
H 3C C O – Na +(aq) + H 2O
to get carboxylic
acid salt
Phenols Neutralization OH O –Na +
(very weakly with strong base + NaOH + H 2O
acidic) to get salt
H. Kim for Chem 30B 3
Other Reaction Types
Family Reaction Example
RADICAL REACTIONS
Alkanes Combustion
(Also heat
oxidation/reduction CH4(g) + 2O2(g) CO2 + 2H2O + Heat
reaction)
Halogenation light or heat
Cl
CH 3CH2CH 3 + Cl2 HCl + CH 3CH2CH2Cl + CH 3CHCH 3
Cl Cl
Cl Cl
Cl Cl
+ CH CH CH + + CH 3CCH 3 + CHCH2CH 3
2 2 2 CH2CHCH 3
Cl Cl
Mixed products
Alkenes Polymerization initiator CH 3 CH 3
CH 3CH CH2 H2 H2
( C C C C )
H H
HYDROLYSIS
Esters Acid hydrolysis to get O
H+ catalyst
O
(Reverse rxns of carboxylic acid and R C O CH 3 + H 2O R C OH + CH 3OH
carboxylic acid alcohol
esterification) Saponification: Base O
O
H2O
hydrolysis to get R C O CH 3 + NaOH (aq)
C O – Na + + CH 3OH
carboxylic acid salt and
R
alcohol
Amides Acid hydrolysis to get O
H H + catalyst
O
(Reverse rxns of amine H 3C C N CH 3 + H 2O H 3C C OH + CH 3NH 2
carboxylic acid Base hydrolysis to get O O
H2O
amidation) amine H 3C C
H
N CH 3 + NaOH (aq) H 3C C OH + CH 3NH 2
Acetals Acid hydrolysis to get H H + catalyst
O
H 3C C O CH2CH 3 + H 2O
(Reverse reaction aldehydes of ketones H 3C C OH + 2 CH 3CH2OH
of acetal formation (reverse of acetal O CH2CH 3
from formation)
aldehydes/ketones)
*Note that organic chemistry reactions can be classified in several different ways.
H. Kim for Chem 30B 4
You might also like
- Chemsheets AS 1077 Petroleum and AlkanesDocument4 pagesChemsheets AS 1077 Petroleum and Alkanescharlesma123No ratings yet
- Chemistry - Organic Chemistry Reaction SchemeDocument19 pagesChemistry - Organic Chemistry Reaction Schemehelixate94% (16)
- Mechanism Summary For A-Level AQA Chemistry: BR BRDocument5 pagesMechanism Summary For A-Level AQA Chemistry: BR BRamrhkmhNo ratings yet
- 6 - Organic ChemistryDocument27 pages6 - Organic ChemistryAlvaro CatalaNo ratings yet
- New CHY3201 Chapter 9 Addition ReactionDocument31 pagesNew CHY3201 Chapter 9 Addition Reaction222418No ratings yet
- Che 91165 FlashcardsDocument5 pagesChe 91165 FlashcardsLê Minh DuyNo ratings yet
- Reductions PPT 29-08-2020Document12 pagesReductions PPT 29-08-2020jkc collegeNo ratings yet
- 102 Lecture Ch13Document36 pages102 Lecture Ch13macybnzNo ratings yet
- 6 Organic Chemistry IDocument27 pages6 Organic Chemistry IAwil AhmedNo ratings yet
- Reaction Mechanism: C C H H H O Hgso H SO CHO H CDocument6 pagesReaction Mechanism: C C H H H O Hgso H SO CHO H CFATHIMA THANHA T NNo ratings yet
- Basic Concepts and Hydrocarbons PDFDocument6 pagesBasic Concepts and Hydrocarbons PDFDr.CharinNo ratings yet
- Organic Chemistry Slides 2Document34 pagesOrganic Chemistry Slides 2johnnfjayNo ratings yet
- Organic Chemistry I EdexcelDocument28 pagesOrganic Chemistry I EdexcelAinara Román MacíasNo ratings yet
- Cyclohexane Cyclohexene Lab AnswersDocument6 pagesCyclohexane Cyclohexene Lab AnswersTingYuan HoiNo ratings yet
- Aqa Mechanisms A Level SummaryDocument5 pagesAqa Mechanisms A Level SummaryRS JNo ratings yet
- Alkynes Medorg2Document6 pagesAlkynes Medorg2AR LazagaNo ratings yet
- Organic Chemistry I EdexcelDocument28 pagesOrganic Chemistry I Edexcelvyb8qpq57yNo ratings yet
- Functional Group Naming TableDocument1 pageFunctional Group Naming Tableinfo.acc231No ratings yet
- Aqa Mechanisms Summary AsDocument3 pagesAqa Mechanisms Summary AsRS JNo ratings yet
- 3.1 Revision Guide Introduction Organic AqaDocument9 pages3.1 Revision Guide Introduction Organic AqaIman KhanNo ratings yet
- 01 Aldket ReducDocument1 page01 Aldket ReducSarvik The Pokémon Master RaiNo ratings yet
- CH2211 Mechanisms: NH + OHDocument5 pagesCH2211 Mechanisms: NH + OHneemNo ratings yet
- 3.1 Revision Guide Introduction Organic AqaDocument9 pages3.1 Revision Guide Introduction Organic Aqashafiqur rahmanNo ratings yet
- 3.5 Revision Guide Alcohols AqaDocument7 pages3.5 Revision Guide Alcohols Aqashafiqur rahmanNo ratings yet
- MEQ L (Introductory Organic Chemistry)Document62 pagesMEQ L (Introductory Organic Chemistry)Furious SKNo ratings yet
- Coursebook Answers Chapter 18 Asal ChemistryDocument4 pagesCoursebook Answers Chapter 18 Asal ChemistryMarin PesicNo ratings yet
- Aqa Mechanisms A21Document4 pagesAqa Mechanisms A21Sarah INo ratings yet
- Mechanism Summary For AS AQA Chemistry: HO: NCDocument4 pagesMechanism Summary For AS AQA Chemistry: HO: NCjohn mNo ratings yet
- Alcohols PDFDocument8 pagesAlcohols PDFVon Valentine MhuteNo ratings yet
- Alcohol and Carboxylic AcidDocument6 pagesAlcohol and Carboxylic AcidPutri Nur SyafieqahNo ratings yet
- Contoh: + Butan-2-On Metanol H SODocument2 pagesContoh: + Butan-2-On Metanol H SOElis TianiNo ratings yet
- Contoh: + Butan-2-On Metanol H SODocument2 pagesContoh: + Butan-2-On Metanol H SOElis TianiNo ratings yet
- Corso Dottorato 2 CFU Lecture 3Document32 pagesCorso Dottorato 2 CFU Lecture 3Serena DamianNo ratings yet
- Problem Set 1 PDFDocument4 pagesProblem Set 1 PDFRay BinasNo ratings yet
- HalogenalkaneDocument4 pagesHalogenalkanePutri Nur SyafieqahNo ratings yet
- 6: Organic Chemistry I: 6A. Introduction To Organic Chemistry Basic Definitions To KnowDocument27 pages6: Organic Chemistry I: 6A. Introduction To Organic Chemistry Basic Definitions To KnowJam GeejeeNo ratings yet
- Loudon Chapter 4 & 5 Review: Reactions of Alkenes: CHEM 3311, Jacquie Richardson, Spring 2010 - Page 1Document3 pagesLoudon Chapter 4 & 5 Review: Reactions of Alkenes: CHEM 3311, Jacquie Richardson, Spring 2010 - Page 1xryceuNo ratings yet
- Organic ChemistryDocument36 pagesOrganic Chemistryj.obriain94No ratings yet
- Chemistry Heinemann U4 Workbook Solutions, 2 (3rd Edition)Document20 pagesChemistry Heinemann U4 Workbook Solutions, 2 (3rd Edition)Tiana Pedulla100% (1)
- BSC 2nd Year Trihydric Alcohol Important ReactionsDocument1 pageBSC 2nd Year Trihydric Alcohol Important ReactionsAbhishek AgarwalNo ratings yet
- 4 Introductory Organic Chemistry and AlkanesDocument12 pages4 Introductory Organic Chemistry and AlkanesChristina HerculesNo ratings yet
- Carbonyl Compounds A-Level NotesDocument4 pagesCarbonyl Compounds A-Level Notesbumblebee9323No ratings yet
- Organic Reactions Summary SheetDocument2 pagesOrganic Reactions Summary Sheetknprop134No ratings yet
- Aldehyde Ketone and AcidDocument15 pagesAldehyde Ketone and AcidSsNo ratings yet
- Alkenes Reactions NotesDocument14 pagesAlkenes Reactions NotesMartin AlvinNo ratings yet
- CY2102Document2 pagesCY2102Prarabdha SharmaNo ratings yet
- 맥머리의 유기화학 9판 (답)Document29 pages맥머리의 유기화학 9판 (답)빅옹일No ratings yet
- Exp 7 Preparation of AlkenesDocument14 pagesExp 7 Preparation of AlkenesGeorge PiliposyanNo ratings yet
- Aldehyde & Ketone RXDocument5 pagesAldehyde & Ketone RXhaNo ratings yet
- MechanismsDocument5 pagesMechanismsnajifaahmed223No ratings yet
- C H H H: 11.1 Fuels and Crude OilDocument7 pagesC H H H: 11.1 Fuels and Crude OilDavid SutantoNo ratings yet
- Topic 16 Aldehydes, Ketones and Optical Isomerism Reactions of Aldehydes and Ketones Optical IsomerismDocument15 pagesTopic 16 Aldehydes, Ketones and Optical Isomerism Reactions of Aldehydes and Ketones Optical Isomerismclip215No ratings yet
- Chapter 8-Reactions of AlkenesDocument35 pagesChapter 8-Reactions of Alkenes張湧浩No ratings yet
- Dia orDocument8 pagesDia orNaman MahawarNo ratings yet
- Alcohol Phenol Ether (1) 4Document9 pagesAlcohol Phenol Ether (1) 4subhashitamurapakaNo ratings yet
- Alcohol Phenol Ether (1) 6Document9 pagesAlcohol Phenol Ether (1) 6sdnishacNo ratings yet
- ChemicalDocument5 pagesChemicalTika Nur Laili L HiperkesNo ratings yet
- Edexcel IAL Chemistry A-Level: Topic 20: Organic SynthesisDocument8 pagesEdexcel IAL Chemistry A-Level: Topic 20: Organic SynthesisCornflake 25No ratings yet
- Synthesis of Fenarimol From 2-Butene: An Organic Synthesis ProjectDocument20 pagesSynthesis of Fenarimol From 2-Butene: An Organic Synthesis ProjectRobinhood Jevons MartirezNo ratings yet
- Varian Catalog Gas ChromatographyDocument118 pagesVarian Catalog Gas ChromatographyReda HassanNo ratings yet
- BCH 303 - 1Document7 pagesBCH 303 - 1Isabel WilliamsNo ratings yet
- WEEK 3 Biochemistry As Chemical ScienceDocument4 pagesWEEK 3 Biochemistry As Chemical ScienceCosmic PhilippinesNo ratings yet
- Competency Exam in Organic ChemistryDocument3 pagesCompetency Exam in Organic ChemistryWinsletJoyDauagNo ratings yet
- 2016 Priority Health Choice FormularyDocument180 pages2016 Priority Health Choice FormularygicribarkNo ratings yet
- Phenol Handwritten Notes by LMDocument14 pagesPhenol Handwritten Notes by LMpanchaldalshukh46No ratings yet
- ZZZZZDocument36 pagesZZZZZwuriNo ratings yet
- 13 HydrocarbonsDocument2 pages13 HydrocarbonsPadhai tak : by Dr.Aditya guptaNo ratings yet
- Wade 18 PowerpointDocument55 pagesWade 18 PowerpointKosygin Leishangthem100% (2)
- PracticeTests Answers All Chem360Document109 pagesPracticeTests Answers All Chem360EthanNo ratings yet
- Alcohols Phenols and Ether by AarkumarDocument0 pagesAlcohols Phenols and Ether by AarkumarNikhil Surya MukhiNo ratings yet
- Iit Reductions PDFDocument71 pagesIit Reductions PDFAshish SinghNo ratings yet
- Solvent Physical PropertiesDocument1 pageSolvent Physical PropertiesJaime Alejandro Martinez AcostaNo ratings yet
- Microsoft Word - Buffer Solutions CompositionDocument1 pageMicrosoft Word - Buffer Solutions Compositiontushar84No ratings yet
- Cholinergic System: Parasympatholytics: ClassificationDocument3 pagesCholinergic System: Parasympatholytics: ClassificationAmit KochharNo ratings yet
- General Chemistry 1 Qt. 2 Week 5Document31 pagesGeneral Chemistry 1 Qt. 2 Week 5Nina Reca OmisolNo ratings yet
- 10april ITCM921E01Document10 pages10april ITCM921E01NanaiNo ratings yet
- Titration Questions Set 1Document8 pagesTitration Questions Set 1danielmahsaNo ratings yet
- Fats & Oils IntroductionDocument12 pagesFats & Oils IntroductionShibbu GangwarNo ratings yet
- Notes 6.1Document3 pagesNotes 6.1LEE LEE LAUNo ratings yet
- Viva Voice QuestionsDocument2 pagesViva Voice QuestionsVarun Sharma0% (1)
- Tugas Akhir KLP 4 MS Excel FIXDocument5 pagesTugas Akhir KLP 4 MS Excel FIXxhgxNo ratings yet
- Fatty Acids SynthesisDocument30 pagesFatty Acids SynthesisGhaidaa SadeqNo ratings yet
- Chapter3-pKa Table PDFDocument3 pagesChapter3-pKa Table PDFAnilesh BansalNo ratings yet
- PharmacologyDocument3 pagesPharmacologyWaseem RazaNo ratings yet
- ORGANIC REACTIONS GRADE 12 Summary 2018Document2 pagesORGANIC REACTIONS GRADE 12 Summary 2018Wanga MdallNo ratings yet
- COURSE OUTLINE IN CHEM 104-PyschDocument5 pagesCOURSE OUTLINE IN CHEM 104-PyschJesson BelenNo ratings yet