Professional Documents
Culture Documents
Source V
Source V
Uploaded by
Armelle HoungangOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Source V
Source V
Uploaded by
Armelle HoungangCopyright:
Available Formats
m-PIMA HIV-1/2 VL ™
POINT-OF-CARE HIV
VIRAL LOAD IN LESS
THAN 70 MINUTES,
NOT MONTHS.
Scale-up of viral load testing is a global priority,
but countries face numerous challenges.
• Limited laboratory infrastructure
• Shortage of skilled clinical and
laboratory staff
• Weak specimen transport systems
• Inefficient systems for result feedback
• Lost samples, lost results and patients
lost to follow-up
m-PIMA HIV-1/2 VL ™
ACTIONABLE RESULTS IN LESS THAN 70 MINUTES WHILE THE PATIENT IS STILL PRESENT
• Instead of sending samples to a lab,
bring the test system to the patient
• Antiretroviral therapy (ART) failure can be detected early CURRENT ALTERNATIVES
• ART switching decisions can be made the same day WEEKS
• ART adherence problems can be addressed the same day
OR MONTHS
• Reduced sample transport costs
• Lost samples, lost results and patients lost to follow-up
potentially reduced
The World Health Organization (WHO) recommends that
all patients on ART receive a viral load test at 6 months and POINT OF CARE
12 months and annually thereafter if the patient is stable on
ART, 1 but very few patients receive that level of care.
LESS THAN
70 MINUTES
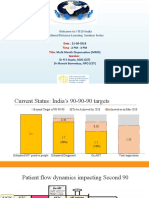
44% 90-90-90: 90% of patients on ART should be virally
suppressed by 2020, but only 44% actually are.2
50
Approximately 50% of the viral load testing need
% in low and middle-income countries is unmet.2
30
By 2021, low- to middle-income countries will
need an estimated 30 million viral load tests.3
million
VIRAL LOAD MONITORING HAS THE
POTENTIAL TO SUPPORT THE REALIZATION
OF THE 90-90-90 GOALS.
SPECIFICATIONS ORDER INFORMATION
• Limit of quantitation: 800 cp/mL for HIV-1 and HIV-2 RNA PART ORDER NUMBER QTY/BOX
(below the WHO cut-off value of < 1000 cp/mL) m-PIMA™ Analyser 27030R001 1
• m-PIMA™ HIV-1/2 VL, at a 95% confidence interval, in internal Accessory Pack (Energy Case, Printer w/Paper,
SB0189 1
and external data reported a limit of detection specific to HIV Connect Universal Gateway)
subtype: m-PIMA™ HIV-1/2 VL Cartridge (IVD) 27015-W50 50
ɝ HIV-1 group M: 342 cp/mL m-PIMA™ HIV-1/2 VL Cartridge (IVD-CE) 270150050 50
ɝ HIV-1 group O: 228 cp/mL Energy Case 5100643 1
ɝ HIV-2 group A: 364 cp/mL Connect Universal Gateway ACEU01 1
USB Printer 27040R007 1
• Method: reverse transcription and real-time PCR with
multiplexing capabilities Printer Paper 26040R009 1
• Time to results: less than 70 minutes Fingerstick Sample Collection Set (Capillary Blood
Collection Tubes, Safety Lancets, Plastic Bandage, 270400201 100
• Sample volume: 50 μL Adhesive Plasters, Nonwoven Swabs, Alcohol Pads)
• Sample type: fingerstick or venous EDTA plasma NOT CLEARED/APPROVED OR FOR SALE IN THE USA.
• Storage conditions: 4°C–30°C 1. WHO: What’s new in treatment monitoring: viral load and CD4 testing.
July 2017.
• Connectivity: enabled 2. Ending AIDS: progress towards the 90-90-90 targets 2017.
3. Lauren & Biyi, LMIC Forecast Demand for Diagnostic Tests 2016-2021,
Avenir Health.
© 2020 Abbott. All rights reserved. All trademarks referenced are trademarks
of either the Abbott group of companies or their respective owners. Any photos
displayed are for illustrative purposes only. Any person depicted in such photos
is a model. 120001793-02 10/20
You might also like
- HCV Package Insert GeneXpertDocument26 pagesHCV Package Insert GeneXpertHassan Gill100% (1)
- Hkdse English Reading全方位實戰神技精讀 主筆記 Sample 1643026918Document23 pagesHkdse English Reading全方位實戰神技精讀 主筆記 Sample 1643026918SupercellNo ratings yet
- Thermador DWHD64EPDocument1 pageThermador DWHD64EPPurcellMurrayNo ratings yet
- Test Code 7300: CDC Priority 3 Testing For SARS-COVID-2Document2 pagesTest Code 7300: CDC Priority 3 Testing For SARS-COVID-2Cecil James BuguisNo ratings yet
- Day 2 TB Diagnostics - MacalaladDocument45 pagesDay 2 TB Diagnostics - MacalaladRye CalderonNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareSyed's Way PoolNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareParmesh PatelNo ratings yet
- Teste Diferenciação de HIVDocument21 pagesTeste Diferenciação de HIVRitaPinto37No ratings yet
- Medical Laboratory Report: Specimen Nasopharyngeal / Oropharyngeal Swab Covid-19 Qualitative PCRDocument1 pageMedical Laboratory Report: Specimen Nasopharyngeal / Oropharyngeal Swab Covid-19 Qualitative PCRSuhas KandNo ratings yet
- Xpert HCV Viral Load ENGLISH Package Insert 301-3019 Rev BDocument24 pagesXpert HCV Viral Load ENGLISH Package Insert 301-3019 Rev BAye MaNo ratings yet
- Package Insert Hiv-1 VLDocument26 pagesPackage Insert Hiv-1 VLSagir AlvaNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Name Ref. by Test Asked::: Patientid: Home CollectionDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Name Ref. by Test Asked::: Patientid: Home Collectionrohit singhNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareKAUSHAL KUMAR SHUKLANo ratings yet
- Diagnosing Patients at Point of Care: Busisiwe Vilakazi, Pieter Roux, Kevin LandDocument19 pagesDiagnosing Patients at Point of Care: Busisiwe Vilakazi, Pieter Roux, Kevin LandextrakaamkliyeNo ratings yet
- Evidence-Based Medicine,: Tuntutan Baru Dalam Profesionalisme Pengobatan Seorang PenderitaDocument40 pagesEvidence-Based Medicine,: Tuntutan Baru Dalam Profesionalisme Pengobatan Seorang PenderitaWinda A. PanjaitanNo ratings yet
- Evidence-Based Medicine,: Iwan Darmansjah Pusat Uji Klinik Obat, FkuiDocument40 pagesEvidence-Based Medicine,: Iwan Darmansjah Pusat Uji Klinik Obat, FkuiIsmail Andi BasoNo ratings yet
- Webinar 10 12 17 PDFDocument73 pagesWebinar 10 12 17 PDFDanielaGarciaNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Thyrocare077 - Ubaid dhansayNo ratings yet
- SD Who BioloineDocument14 pagesSD Who Bioloinesandhyakul9736No ratings yet
- LSERO 0001 - Screening Test For HIV Using KHB FINALDocument6 pagesLSERO 0001 - Screening Test For HIV Using KHB FINALjohnmutiso245No ratings yet
- Pointcare Now Brochure EngDocument10 pagesPointcare Now Brochure EngAnnisa ChaeraniNo ratings yet
- NDLS - Dr. RS Gupta - Dr. Manish - MMD - 21-08-2018Document32 pagesNDLS - Dr. RS Gupta - Dr. Manish - MMD - 21-08-2018fanwindowNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareSuraj IngaleNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareVinay DubeyNo ratings yet
- Test Result Report: Interpretation GuidelinesDocument2 pagesTest Result Report: Interpretation GuidelinesMohammed Shafi CpNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareVinay DubeyNo ratings yet
- Test Result Report: Interpretation GuidelinesDocument2 pagesTest Result Report: Interpretation GuidelinesMohammed Shafi CpNo ratings yet
- Term Paper ON: Correlation Between Rapid Test and Automated Methods in Diagnosing Infectious Disease Like TyphoidDocument22 pagesTerm Paper ON: Correlation Between Rapid Test and Automated Methods in Diagnosing Infectious Disease Like TyphoidSoniya DhyaniNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Thyrocarenarendra99No ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareGurushantayya SwamiNo ratings yet
- Bosch Vivalytic Sti Cartiladge and QC IFU Update 17-3 Last UpdateDocument7 pagesBosch Vivalytic Sti Cartiladge and QC IFU Update 17-3 Last UpdateMohammed AlshnqityNo ratings yet
- PreciControl Anti-HCV - Ms 03290379190.V9.EnDocument2 pagesPreciControl Anti-HCV - Ms 03290379190.V9.EnARIF AHAMMED PNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareAravinth raj kumar. MNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Thyrocareram kumar goelNo ratings yet
- HBC IgM Outside US - ADVIA Centaur, XP, and XPT - Rev N DXDCM 09008b838072868f-1412760601006Document16 pagesHBC IgM Outside US - ADVIA Centaur, XP, and XPT - Rev N DXDCM 09008b838072868f-1412760601006Sandy SousaNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareAryanNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Thyrocaresaqlain mhaldarNo ratings yet
- L'Helgouach-2020-EasyCOV - LAMP Based Rapid deDocument14 pagesL'Helgouach-2020-EasyCOV - LAMP Based Rapid deGerman MadrigalNo ratings yet
- CCO Cytokine TX in MM ExpressPointsDocument18 pagesCCO Cytokine TX in MM ExpressPointsveaceslav coscodanNo ratings yet
- Gueudin2016 PDFDocument21 pagesGueudin2016 PDFibnuNo ratings yet
- Pi M Pima 01 en v12 M Pima Hiv 12 Detect Cartridge Guide - enDocument40 pagesPi M Pima 01 en v12 M Pima Hiv 12 Detect Cartridge Guide - enismail kayongoNo ratings yet
- 25.signal HIV Immuno DotDocument5 pages25.signal HIV Immuno DotprastacharNo ratings yet
- Jose Gomez-Marquez: MIT D-Lab D-Lab - Mit.edu Twitter: JFGM Jfgm@mit - EduDocument76 pagesJose Gomez-Marquez: MIT D-Lab D-Lab - Mit.edu Twitter: JFGM Jfgm@mit - EduInnovations in International HealthNo ratings yet
- PreciControl Anti CCP.05115680001.V3.EnDocument2 pagesPreciControl Anti CCP.05115680001.V3.EnARIF AHAMMED PNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareVinay DubeyNo ratings yet
- Product Datasheet: Zika Virus (SPH2015) NS1 ELISA Kit (Colorimetric) NBP2-80364Document3 pagesProduct Datasheet: Zika Virus (SPH2015) NS1 ELISA Kit (Colorimetric) NBP2-80364rameshmrsNo ratings yet
- Implementation of MALDI-TOF MS in The Routine Clinical LaboratoryDocument69 pagesImplementation of MALDI-TOF MS in The Routine Clinical LaboratorycavabNo ratings yet
- MR Praveen Borkar 002Document1 pageMR Praveen Borkar 002vansh taraNo ratings yet
- Anti HBs Total II 30318Document8 pagesAnti HBs Total II 30318armada thamNo ratings yet
- WHO Use Malaria Rapid - Diagnostic Test 2004Document20 pagesWHO Use Malaria Rapid - Diagnostic Test 2004Ahmed AdemNo ratings yet
- BP160050.0. Elecsys® HIV Combi PT Immunoassay. FINAL PIDocument21 pagesBP160050.0. Elecsys® HIV Combi PT Immunoassay. FINAL PIDaniel Gutierrez GutierrezNo ratings yet
- en-USDocument3 pagesen-USpsychejaneNo ratings yet
- Post-Exposure Prophylaxis and Needle Stick InjuryDocument29 pagesPost-Exposure Prophylaxis and Needle Stick InjuryNabila Qatrah NadaNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument3 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareVinay DubeyNo ratings yet
- Covid PanelDocument66 pagesCovid PanelJoshua RuizNo ratings yet
- CCO 2020 Contemporary Management of HIV Emerging Approaches DownloadableDocument52 pagesCCO 2020 Contemporary Management of HIV Emerging Approaches Downloadableluis hernandezNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: ThyrocareKAUSHAL KUMAR SHUKLANo ratings yet
- HbsAg TechincalDocument21 pagesHbsAg Techincalspike9mNo ratings yet
- Hicc For Nursing Excellence-Smh Sept 2018Document25 pagesHicc For Nursing Excellence-Smh Sept 2018RakshaNo ratings yet
- Advances of Basic Sciences - Diagnostic & Prognostic ApplicationDocument38 pagesAdvances of Basic Sciences - Diagnostic & Prognostic ApplicationKamini WaliaNo ratings yet
- Amruta RTPCR 04 June 2021Document2 pagesAmruta RTPCR 04 June 2021sunnyNo ratings yet
- EH - UK - Employers - Basics of UK Payroll GuideDocument10 pagesEH - UK - Employers - Basics of UK Payroll GuidexiibzzxNo ratings yet
- Revolusi Industri 4.0: Impak Terhadap Perkembangan Pendidikan Tinggi Di MalaysiaDocument13 pagesRevolusi Industri 4.0: Impak Terhadap Perkembangan Pendidikan Tinggi Di Malaysiaeiman medinaNo ratings yet
- Polarization DensityDocument8 pagesPolarization DensityAshwin BalajiNo ratings yet
- Dikitanan Task1Document5 pagesDikitanan Task1Janna CrizetteNo ratings yet
- Commercial PoliciesDocument8 pagesCommercial Policiesapi-3706009No ratings yet
- The Spotify ModelDocument2 pagesThe Spotify ModelMuhammad SaadNo ratings yet
- GHMC taxGHMC TAXDocument12 pagesGHMC taxGHMC TAXpoornima_npNo ratings yet
- SCB N KingfiherDocument7 pagesSCB N KingfiherSergel Capsule100% (1)
- BangladeshDocument6 pagesBangladeshKhalid AhmedNo ratings yet
- November 2023 Timetable Zone 2Document12 pagesNovember 2023 Timetable Zone 2Horror666No ratings yet
- ASTM E8 E8M 2021 Tension Testing of Metallic MaterialsDocument30 pagesASTM E8 E8M 2021 Tension Testing of Metallic MaterialsSuvro ChakrabortyNo ratings yet
- Nut, Double Hexagon, Self Locking: Page 1/3Document3 pagesNut, Double Hexagon, Self Locking: Page 1/3Renato WatanabeNo ratings yet
- Indian School of Business (ISB) PGP (Intended Major: Finance) - GMAT: 740Document1 pageIndian School of Business (ISB) PGP (Intended Major: Finance) - GMAT: 740Suprateek BoseNo ratings yet
- Charging and TD Chart PlusDocument1 pageCharging and TD Chart PlusFaquruddin AliNo ratings yet
- Masidlak Eagles Form 2Document2 pagesMasidlak Eagles Form 2Rubie Anne SaducosNo ratings yet
- Instrumentation Group1Document16 pagesInstrumentation Group1Aruna KumbukageNo ratings yet
- MIUI 12 Latest Update Features 2020Document5 pagesMIUI 12 Latest Update Features 2020johnson veigasNo ratings yet
- 45 - 60055 - EE413 - 2015 - 4 - 2 - 1 - Sheet 06 Sheet-ADC & DACDocument2 pages45 - 60055 - EE413 - 2015 - 4 - 2 - 1 - Sheet 06 Sheet-ADC & DACZuhaili ZawawiNo ratings yet
- Step by Step Tutorial On BDC Session Method Program in Sap AbapDocument16 pagesStep by Step Tutorial On BDC Session Method Program in Sap AbapBipul KumarNo ratings yet
- (Vacancies May Increase (Or) Decrease at The Time of Recruitment) Eligibility: For Assistant Professors in Ap Medical Education ServicesDocument4 pages(Vacancies May Increase (Or) Decrease at The Time of Recruitment) Eligibility: For Assistant Professors in Ap Medical Education ServicesseravanakumarNo ratings yet
- Se 747Document16 pagesSe 747Roohian Ali100% (1)
- MENINGITIS Lesson PlanDocument12 pagesMENINGITIS Lesson PlannidhiNo ratings yet
- Apparel Internship Report 12Document118 pagesApparel Internship Report 12Ankita RanjanNo ratings yet
- Repayment Schedule - 164916682Document1 pageRepayment Schedule - 164916682Shivashakthi MaheshNo ratings yet
- GBS Project Service Center: Financial Process Monthly Reporting (IPPF)Document16 pagesGBS Project Service Center: Financial Process Monthly Reporting (IPPF)smgalloNo ratings yet
- CB-8466 ECF ClearFire Economizer PDFDocument16 pagesCB-8466 ECF ClearFire Economizer PDFAbdul RazzaqNo ratings yet
- Module 2 Solar EnergyDocument140 pagesModule 2 Solar EnergyMuhammad Ahad Rahman MiahNo ratings yet
- 1,023,695, Patented Apr. 16, 1912.: G. WaltherDocument3 pages1,023,695, Patented Apr. 16, 1912.: G. WaltherdfdfsdNo ratings yet