Professional Documents
Culture Documents
Enzyme Workbook
Enzyme Workbook
Uploaded by
LK chanellOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Enzyme Workbook
Enzyme Workbook
Uploaded by
LK chanellCopyright:
Available Formats
1
Enzymes
workbook
What are enzymes?
Enzymes are …………………………………that function as a biological
………………………………………………..……………………………………………………………………………………………………….
They are proteins in nature.
What is a catalyst?
……………………………………………………………………………………………………………………………………………….
……………………………………………………………………………………………………………………………………………….
5 Properties of enzymes
• Enzymes are made of protein
• Each enzyme catalyses one reaction at a time.
• They can be used again & again
• They are influenced by temperature
• They are influenced by pH
Mr Maurice IGCSE Biology
2
NAMING OF ENZYMES.
Enzymes can be named by the following ways.
1. The substrate the catalyse.
In this, you add suffix …..ase to the end of the substrate.
Eg.
Protein Protease
Lipids
Maltose
Sucrose
Carbohydrates
Amylose (Starch)
2. The process they catalyse.
In this, you add suffix …..ase to the end of the substrate.
e.g
Oxidation Oxidase
Reduction
Hydrolysis
3. Named after the person who discovered them.
e.g
Pepsin
Renin
Trypsin
Mr Maurice IGCSE Biology
3
Example.
Hydrogen peroxide (H2O2) is a substance that decomposes into Water (H2O) and
Oxygen (O2) if it is left in room temperature for a period of time.
This reaction could a long time, but it could be sped up if we add a catalyst.
Each catalyst can catalyse a specific substance and nothing but it. The catalyst for
Hydrogen peroxide is called Manganese (IV) oxide.
If it is added we will get water and oxygen gas in a very short time, and the
manganese (IV) oxide could be obtained again as it was, it remains unchanged.
Observe the class demonstration and write down the observation.
………………………………………………………………………………………………………………………………………………………..
…………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………
Mr Maurice IGCSE Biology
4
………………………………………………………………………………………………………………………………………......................
…………………………………………………………………………………………………………………………………………………………..
……………………………………………………………………………………………………………………………………………………………
Describe why enzymes are important in all living organisms
……………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………......................
…………………………………………………………………………………………………………………………………………………………..
……………………………………………………………………………………………………………………………………………………………
How Do Enzymes Work?
Enzymes work the same way as catalysts do, they can work with only one substrate
and they can be used more than once.
Enzymes have a structure that is called active site.
Only one substance can fit into the active site to be digested, and it is the only
substrate that this particular enzyme works with.
Mr Maurice IGCSE Biology
5
Task
Draw a well labelled diagram of an Enzyme. Include the substrate and products.
Mr Maurice IGCSE Biology
6
The figure above shows the function of enzymes:
• The substrate enters the active site of the enzyme.
• The reaction takes place.
• The substrate exits the enzyme as two simpler products.
You can also think of the way enzymes work as a key and a lock, the key is the
substrate and the lock is the enzyme.
The key should be exactly the right shape to fit in the lock, so does the
substrate to fit in the active site of the enzyme. The key could only open only one
lock, and the lock could be unlocked by only that key.
Enzymes - 'Lock and key' model
Enzymes are very specific, each kind of enzyme catalyse one kind of reaction only.
To catalyse a reaction, enzyme molecule and substrate molecule need to meet and
joint together by a temporary bond.
Each molecule has a special shape and
an active site into which its
substrate molecule fits exactly.
This enzyme is amylase, and its active
site is just the right size and shape
for a substrate molecule (starch in
this case).
Mr Maurice IGCSE Biology
7
The starch slots into the active site.
The starch is split into maltose
molecules
The enzyme is unaltered, and ready to
accept another part of the starch
molecule
Enzymes are two types,
❖ Builders and Breakers.
➢ Builder enzymes do the opposite of breaker enzymes.
➢ Breakers break large molecules into smaller simpler ones,
builders combine smaller ones to make large molecules.
Mr Maurice IGCSE Biology
8
Using the knowledge of previous topic discussed (Biological molecules), give an
example for a builder enzyme and a breaker enzyme in action.
………………………………………………………………………………………………………………………………………………….
…………………………………………………………………………………………………………………………………………………
Breaker enzymes are used in the digestive system to break down large insoluble
molecules into simpler soluble ones to be used by the body.
They are also present in cells that respire to break down sugars and oxygen into
carbon dioxide, water and energy.
Builder enzymes are present in plants to be used in photosynthesis, the opposite of
respiration, in photosynthesis, oxygen and water are combined together to form
carbon dioxide and sugars.
Mr Maurice IGCSE Biology
9
TASK
Sp.p03
Mr Maurice IGCSE Biology
10
S12p21
Explain the specificity of enzymes
………………………………………………………………………………………………………………………………………………………..
………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………..................
………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………
Because enzymes are proteins, they are denatured by heat or some chemicals
What does Denaturing mean?
………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………
Mr Maurice IGCSE Biology
11
Effect of temperature on the enzyme’s activity:
Each enzyme has an optimum temperature, this is the temperature at which the
enzyme is most active, below this temperature the activity of the enzyme
decreases until it becomes inactive at low temperatures, above this optimum
temperature the enzyme becomes denatured and can no longer work.
At low temperatures the enzyme and the substrate are moving very slowly and
collide weakly, the enzyme is said to be inactive and doesn’t work.
As the temperature increases, the enzyme and substrate gain more kinetic energy
and move faster colliding more, the enzyme becomes more active and the reaction
takes place.
When the enzyme reaches it’s optimum temperature, it is in its most active mood,
if the temperature crosses the optimum the enzyme begins to get destroyed and
become denatured.
The enzymes become denatured when the shape of their active site changes as a
result of high temperature, thus the substrate cannot fit into the active site and
the enzyme is useless.
Each enzyme has its own optimum temperature; enzymes in humans have optimum
temperatures of around 40 degrees. Plants have enzymes with optimum
temperature of about 25 degrees.
Mr Maurice IGCSE Biology
12
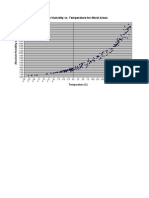
Effect of temperature on enzymes
The optimum (best) temperature for enzyme-controlled reactions is 370C (body
temperature).
As the temperature increases, the rate of reaction increases. But very high
temperatures denature enzymes.
The graph shows the typical change in an enzyme's activity with increasing
temperature.
The enzyme activity gradually increases with temperature up to around 37ºC, or
body temperature. Then, as the temperature continues to rise, the rate of
reaction falls rapidly as heat energy denatures the enzyme. Most enzymes are
denatured above 500C.
Mr Maurice IGCSE Biology
13
Describe the Graph.
………………………………………………………………………………………………………………………………………………………..
………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………..................
………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………
Explain the Graph
………………………………………………………………………………………………………………………………………………………..
………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………..................
………………………………………………………………………………………………………………………………………………………
Mr Maurice IGCSE Biology
14
The Effect of pH on the enzyme’s activity:
As in temperature, enzymes have an optimum pH. The pH is a scale measuring the
acidity or alkalinity of a substance or solution. The scale runs from 1 to 14. pH 7 is
neutral, below that it is acidic and above that it is alkaline.
Each enzyme has an optimum pH, if this pH changes, the shape of the active site of
the enzyme is changed, thus the substrate will not be able to fit in it, and the
enzyme becomes useless.
Effect of pH on enzymes
❖ The pH of a solution is how acidic or alkaline it is.
❖ Different enzymes work best at different pH values.
❖ The optimum pH for an enzyme depends on where it normally works.
❖ It is around neutral (pH= 7) for most enzymes but there are some
exceptions.
Changes in pH also alter an enzyme’s shape and slow down its activity, but this
can usually be reversed if the optimum pH is restored.
An extreme pH can denature enzymes – the active site is deformed
permanently.
Mr Maurice IGCSE Biology
15
Mr Maurice IGCSE Biology
16
Mr Maurice IGCSE Biology
17
Describe the Graph.
…………………………………………………………………………………………………………………………………………………
…………………………………………………………………………………………………………………………………………………
…………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………............
………………………………………………………………………………………………………………………………………………….
………………………………………………………………………………………………………………………………………………
Explain the Graph
…………………………………………………………………………………………………………………………………………………
…………………………………………………………………………………………………………………………………………………
…………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………............
……………………………………………………………………………………………………………………………………………….
…………………………………………………………………………………………………………………………………………………
Mr Maurice IGCSE Biology
You might also like
- TDS-11SA Service Manual PDFDocument236 pagesTDS-11SA Service Manual PDFBoris Zaslichko88% (34)
- Iso 18295 1 2017Document11 pagesIso 18295 1 2017Duilio HomarNo ratings yet
- Enzyme NotesDocument4 pagesEnzyme NotesMaulana Makhmud100% (2)
- GCSE EnzymesDocument32 pagesGCSE EnzymesFrankie BarnesNo ratings yet
- The First Computers Were PeopleDocument39 pagesThe First Computers Were Peopleskywriter36No ratings yet
- Unit 3 - EnzymesDocument5 pagesUnit 3 - EnzymesWaseem AhmedNo ratings yet
- Enzymes - Theory (SC)Document10 pagesEnzymes - Theory (SC)Nithika De SilvaNo ratings yet
- MIQ B1 - Spec Point Q & ADocument9 pagesMIQ B1 - Spec Point Q & ADaniyal MehmoodNo ratings yet
- Bio-Ch 5-EnzymesDocument6 pagesBio-Ch 5-EnzymesHassan RiazNo ratings yet
- Enzyme wks-1 O-Level PDFDocument3 pagesEnzyme wks-1 O-Level PDFMasthankhan Patan100% (1)
- B2.2 - Digestive System Gap Fill 2018 (Combined & Bio)Document4 pagesB2.2 - Digestive System Gap Fill 2018 (Combined & Bio)roger busbyNo ratings yet
- Chapter5 Bio f5Document8 pagesChapter5 Bio f5JiayingNo ratings yet
- Enzyme PracticeDocument2 pagesEnzyme Practiceziming linNo ratings yet
- Chapter 5 Metabolism and EnzymesDocument8 pagesChapter 5 Metabolism and Enzymes绿谷出久No ratings yet
- General-Biology 1Document21 pagesGeneral-Biology 1Edreyan Adong Cortez LimbagaNo ratings yet
- Chapter 5 Biological CatalystsDocument51 pagesChapter 5 Biological Catalystsksmohammed2008No ratings yet
- Enzyme NoteDocument4 pagesEnzyme NoteYolanda JesslinaNo ratings yet
- Catalyst: Enzyme-Substrate Complex Specific Complementary StructureDocument2 pagesCatalyst: Enzyme-Substrate Complex Specific Complementary Structureapi-296833859No ratings yet
- Chapter 1Document9 pagesChapter 1Abubakar Siddiq RaminNo ratings yet
- Year 10 Revision BookletDocument72 pagesYear 10 Revision BookletwolfpackoftrioNo ratings yet
- Energy Relationships in Metabolic Processes and EnzymesDocument16 pagesEnergy Relationships in Metabolic Processes and EnzymesMighty Warrior GSRNo ratings yet
- Enzymes Form 3Document4 pagesEnzymes Form 3Ralph MuchingamiNo ratings yet
- EnzymesDocument3 pagesEnzymescpagente01No ratings yet
- Enzymes As Biological CatalystDocument19 pagesEnzymes As Biological CatalystJuliet Ileto Villaruel - AlmonacidNo ratings yet
- Reminder B5Document10 pagesReminder B5Daniel thanh ducNo ratings yet
- EnzymesDocument40 pagesEnzymesVeronica HanyNo ratings yet
- IGCSE Revision Booklet Part 1 2018 2019 New Spec AnswersDocument87 pagesIGCSE Revision Booklet Part 1 2018 2019 New Spec Answersayten.sherifNo ratings yet
- CHM 152-Biochemistry EnzymesDocument2 pagesCHM 152-Biochemistry EnzymesfillereyesNo ratings yet
- Biology 7B Worksheet No.3Document6 pagesBiology 7B Worksheet No.3Ali AshrafNo ratings yet
- 3 EnzymesDocument12 pages3 Enzymesgj2c74k59xNo ratings yet
- Free Sample - Topic 2Document7 pagesFree Sample - Topic 2Amna AmerNo ratings yet
- What Are EnzymesDocument18 pagesWhat Are EnzymesJudy AbbottNo ratings yet
- KISS Maintaining A BalanceDocument32 pagesKISS Maintaining A BalanceL50% (2)
- Structure of Living OrganismsDocument28 pagesStructure of Living OrganismsDinangaNo ratings yet
- EnzymesDocument2 pagesEnzymesMichaelNo ratings yet
- EnzymeDocument15 pagesEnzymeAljon Lara ArticuloNo ratings yet
- Alwadi International School Biology Grade 9 5. Enzymes NotesDocument43 pagesAlwadi International School Biology Grade 9 5. Enzymes NotesMohammed HelmyNo ratings yet
- Enzymes (As Biology)Document42 pagesEnzymes (As Biology)Muhtasim FarhanNo ratings yet
- MICROORGANISMS BIOLOGY DOXDocument3 pagesMICROORGANISMS BIOLOGY DOXmaddi.ellistonNo ratings yet
- 5 - EnzymesDocument3 pages5 - EnzymesAvyay TopraniNo ratings yet
- Enzymes StructureDocument20 pagesEnzymes StructureBrandonNo ratings yet
- Enzymes and ReactionsDocument5 pagesEnzymes and Reactionstechninja256No ratings yet
- FREE SAMPLE - Topic HeDocument7 pagesFREE SAMPLE - Topic Hehbmffhnkw5No ratings yet
- Enzyme Activity ADocument3 pagesEnzyme Activity AKyla QuilantangNo ratings yet
- EnzymesDocument10 pagesEnzymeszena mohamedNo ratings yet
- Chapter 5 BioDocument23 pagesChapter 5 Biojoelmokwenxuan2No ratings yet
- Cape Biology - Unit 1 - EnzymesDocument7 pagesCape Biology - Unit 1 - EnzymesSteven IStudy Smith67% (3)
- Enzyme PowerpointDocument39 pagesEnzyme PowerpointlinzihaoleoNo ratings yet
- Enzyme WorksheetDocument3 pagesEnzyme Worksheetchinmayee chitturu100% (1)
- Gen Bio 1 2nd Quarter ModularDocument8 pagesGen Bio 1 2nd Quarter ModularAriane NicoleNo ratings yet
- EnzymesDocument17 pagesEnzymesapi-309893409No ratings yet
- Bio - Chapter 3 and 4Document13 pagesBio - Chapter 3 and 4Lim MonyratanakNo ratings yet
- Biology OlevelsDocument6 pagesBiology OlevelsMuhammad Usman AjmalNo ratings yet
- EnzymesDocument27 pagesEnzymesRussNo ratings yet
- Enzymes Material PDFDocument7 pagesEnzymes Material PDFSalma AhmedNo ratings yet
- Cell Bio Chapter 6Document32 pagesCell Bio Chapter 6GuteNo ratings yet
- Year 10 Revision Booklet AnswersDocument71 pagesYear 10 Revision Booklet AnswersArooj AbidNo ratings yet
- Enzymes Part 1Document23 pagesEnzymes Part 1judevolkovNo ratings yet
- Bio Investigatory (Study of Enzymes) - 3Document18 pagesBio Investigatory (Study of Enzymes) - 3Deepa Singh0% (1)
- Definitions:: Pakistan International School Jeddah (Es) Biology NotesDocument9 pagesDefinitions:: Pakistan International School Jeddah (Es) Biology NotesMaira. BalsaNo ratings yet
- 5 Enzymes: 0610 BiologyDocument9 pages5 Enzymes: 0610 BiologyAbdul HadiNo ratings yet
- Enzymes and Biotechnology: See Also Rates Notes at End of 2. Extra Organic ChemistryDocument4 pagesEnzymes and Biotechnology: See Also Rates Notes at End of 2. Extra Organic ChemistryYusra RasoolNo ratings yet
- SMOE Layout Man PDFDocument4 pagesSMOE Layout Man PDFCandrahasner SiregarNo ratings yet
- Arts 9 - Q4 - Mod4 - ChoreographyMovementAndGesturesFromWesternClassicalPlaysOperas - v4Document15 pagesArts 9 - Q4 - Mod4 - ChoreographyMovementAndGesturesFromWesternClassicalPlaysOperas - v4Nikko PatunganNo ratings yet
- USA Comparison6Document30 pagesUSA Comparison6api-27259648No ratings yet
- Baked Salmon SushiDocument2 pagesBaked Salmon Sushivetsnsr71No ratings yet
- Kaise KB1218 - Hoja de DatosDocument2 pagesKaise KB1218 - Hoja de DatosGermanYPNo ratings yet
- Reading Comprehension Power ToolsDocument6 pagesReading Comprehension Power ToolsCarlos Alberto Lopez MartinezNo ratings yet
- Bomba Peristáltica EspecificacionDocument1 pageBomba Peristáltica EspecificacionhugoNo ratings yet
- Lecture 5 - Aldehydes & KetonesDocument93 pagesLecture 5 - Aldehydes & KetonesQutaiba Ibrahim100% (1)
- DETAILED LESSON PLAN SubatomicparticlesDocument16 pagesDETAILED LESSON PLAN SubatomicparticlesMary Rose Ramos100% (3)
- Wem Pu 6089 A PDFDocument74 pagesWem Pu 6089 A PDFolalekanNo ratings yet
- PSYC 1504 Discussion Forum Unit 5Document2 pagesPSYC 1504 Discussion Forum Unit 5keshanirathnayake745No ratings yet
- Cheat Sheet PDFDocument1 pageCheat Sheet PDFGonçalo RaçõesNo ratings yet
- CPRG - V4 - Advanced Issues in Contract PricingDocument304 pagesCPRG - V4 - Advanced Issues in Contract Pricingjarod437No ratings yet
- B23 PDFDocument3 pagesB23 PDFHector SilvaNo ratings yet
- Common Law Pleading Regarding Name ChangesDocument54 pagesCommon Law Pleading Regarding Name ChangesgmsapiensNo ratings yet
- Lab 1a RASPlib Installation Instructions R5 2016b-2017aDocument10 pagesLab 1a RASPlib Installation Instructions R5 2016b-2017aaleNo ratings yet
- Circuit Diagram: Ecu A/7Document1 pageCircuit Diagram: Ecu A/7RubénNo ratings yet
- AKZ Asphaltmatters Europe1Document24 pagesAKZ Asphaltmatters Europe1jijimanNo ratings yet
- Propylene (Asia-Pacific) 31 Jan 2020Document5 pagesPropylene (Asia-Pacific) 31 Jan 2020Wira Nata S SinuhajiNo ratings yet
- Lecture 6 - RTL SynthesisDocument72 pagesLecture 6 - RTL SynthesisantoniocljNo ratings yet
- Complete PersepolisDocument7 pagesComplete Persepolisapi-301951533No ratings yet
- Halfen Cast-In Channels: ConcreteDocument92 pagesHalfen Cast-In Channels: ConcreteFernando Castillo HerreraNo ratings yet
- Gis Prac1Document126 pagesGis Prac1Brijesh JavakeNo ratings yet
- Chapter 1: Why Study Money, Banking, and Financial Markets?Document23 pagesChapter 1: Why Study Money, Banking, and Financial Markets?Nguyễn Thanh TrangNo ratings yet
- UG Bulletin2018FinalDocument39 pagesUG Bulletin2018FinalAman KumarNo ratings yet
- Techciti: Managed ServicesDocument6 pagesTechciti: Managed ServicesTechciti TechnologiesNo ratings yet