Professional Documents
Culture Documents
Intrinsic Risk Criteria Scoring For Drug Manufacturer
Intrinsic Risk Criteria Scoring For Drug Manufacturer
Uploaded by
autumn kimOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Intrinsic Risk Criteria Scoring For Drug Manufacturer
Intrinsic Risk Criteria Scoring For Drug Manufacturer
Uploaded by
autumn kimCopyright:
Available Formats
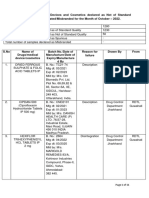
INTRINSIC RISK CRITERIA/SCORING FOR DRUG MANUFACTURER
Name of Established : ABCDE LABORATORIES, INC. LTO No.: LTO-3000001234567
Site Address : XYZ Industrial Park, Broadyway St., Manila
COMPLEXITY
SITE COMPLEXITY LOW = 1 MEDIUM =2 HIGH = 3 Score
1 Size 200 sqm and below 201 sqm to 500sqm 501sqm and above
2 No. of years of existence 1-5 years 6-10 years 11 years and above
3 Product line (liquid, solid, semi-solid & sterile, non- 1-3 line 4-6 lines 7 and above lines
sterile)
4 Dedication of equipment/campaign dedicated dedicated and campaign
campaign
5 No.of staff below 50 51-200 201 and above
6 Manufacture of own products/ no.of client manufacturer of own manufacturer of own manufacturer of own
product product and/or 1 to 3 product and/or 4 and
drug traders above drug traders
PROCESS COMPLEXITY
7 Product category HR,Medical Gas, Terminal sterilization Aseptic sterilization,
Non-sterile, TMHS,
Repacker
8 Parametric release without with
9 Critical steps involved - special environmental without Special with special
conditions (i.e heat sensitive, RH, light sensitive) requirements requirements
10 Potency non-potent modified-release potent
11 processing stages (i.e compounding, filling, packing) one stage process two to three four processes and
processes above
12 packer/repacker - primary and secondary includes primary and primary for non- primary packaging
secondary packaging sterile products for bulk sterile
product
13 biological processes without biological with biological
process process
PRODUCT COMPLEXITY
No.of components/pack - multiple active ingredients one two to three 4 and above
Special storage (ie radiopharma/ cold chain) room temp cool temp 8-15 C/ cold temp/ light
with RH requirement sensitive/ radioactive
barrier containment/
cryogenic storage
COMPLEXITY TOTAL 0
CRITICALITY
PRODUCT CRITICALITY LOW = 1 MEDIUM =2 HIGH = 3
1 essential product/ therapeutic category HR, TMHS, OTC Complementary List, Core list/ Anti
drugs, supplements/ dermatological Cancer, TB drugs,
vitamins products, others Antibiotics, Vaccine
& Biologicals, Blood
& Blood products,
Emergency Drugs,
2 availability of the product 21 and above 6 to 20 1-5
manufacturer manufacturers source/manufacturer
CRITICALITY TOTAL 0
COMPLEXITY SCORE = SUM TOTAL DIVIDE BY 15 0
*round off to the nearest whole number
CRITICALITY SCORE = SUM TOTAL DIVIDE BY 2 0
*round off to the nearest whole number
Prepared by:
Date:
You might also like
- Product ListDocument148 pagesProduct ListvivekNo ratings yet
- Module 5 PharmaceuticsDocument55 pagesModule 5 PharmaceuticsDenise Yanci Demiar100% (2)
- Strategic Management ProjectDocument84 pagesStrategic Management ProjectGaurav GopalNo ratings yet
- Safety Data Sheet: Trade Name: Töten Bakterien Page 1/2Document2 pagesSafety Data Sheet: Trade Name: Töten Bakterien Page 1/2Muhammad Ahmed Abd ElmonemNo ratings yet
- CC211 IA-2A Control V1.0 2020-05-18Document2 pagesCC211 IA-2A Control V1.0 2020-05-18Adams QuintanillaNo ratings yet
- Haccp WorkDocument11 pagesHaccp WorkSude KaradağNo ratings yet
- Self-Contained Spore Ampoules.: Infection Control Division - Biological IndicatorsDocument4 pagesSelf-Contained Spore Ampoules.: Infection Control Division - Biological IndicatorsAngel CuervoNo ratings yet
- Haccp Forms 1-10Document21 pagesHaccp Forms 1-10hussam.workmailNo ratings yet
- TDS - Mergal K20 - 17-07-2014Document2 pagesTDS - Mergal K20 - 17-07-2014APEX SON100% (1)
- Product Description - BD125X-1 TRAZANTO Rev.26 PDFDocument6 pagesProduct Description - BD125X-1 TRAZANTO Rev.26 PDFEdwin Alexis Aguilar ContrerasNo ratings yet
- Mergal K14Document2 pagesMergal K14amila209No ratings yet
- Technical Information - BT20Document6 pagesTechnical Information - BT20David AlbertoNo ratings yet
- Ficha Tecnica Indicador Biologico BT20Document4 pagesFicha Tecnica Indicador Biologico BT20CATALINA FAJARDONo ratings yet
- Mergal X12NDocument2 pagesMergal X12NwtwilightwNo ratings yet
- Inteliq Homocysteine Control Levels 1, 2 and 3: - 2023-06 - 30013-00W-00 EnglishDocument2 pagesInteliq Homocysteine Control Levels 1, 2 and 3: - 2023-06 - 30013-00W-00 Englishjnyng222No ratings yet
- 564 Flowcrete Peran STCDocument8 pages564 Flowcrete Peran STCOscar PintoNo ratings yet
- Bakzid 10N - Product - Information - EN - PDSDocument2 pagesBakzid 10N - Product - Information - EN - PDSgaloisliu2020No ratings yet
- Liquichek D-Dimer Control Levels Low, 1, 2 and 3Document5 pagesLiquichek D-Dimer Control Levels Low, 1, 2 and 3Ade FeriyatnaNo ratings yet
- Tra-Bond Fda2t-EnDocument2 pagesTra-Bond Fda2t-En4445No ratings yet
- HACCP Plan With Flow Chart-1Document23 pagesHACCP Plan With Flow Chart-1Anonymous aZA07k8TXfNo ratings yet
- HPD Marmoleum Modular (Exp. 2024.10.08)Document7 pagesHPD Marmoleum Modular (Exp. 2024.10.08)Marco RaptusNo ratings yet
- Increase Productivity Rapid Micro Methods PharmaEnvironment Michael WasmannDocument31 pagesIncrease Productivity Rapid Micro Methods PharmaEnvironment Michael WasmannRonald SalasNo ratings yet
- At CRPDocument2 pagesAt CRPnacer remidiNo ratings yet
- HACCP Model For Traditional Swine SlaughterDocument17 pagesHACCP Model For Traditional Swine SlaughterMadhanNo ratings yet
- Module 5 - PharmaceuticsDocument55 pagesModule 5 - PharmaceuticsShaira Gayle T TechonNo ratings yet
- Ultima High Recycled Content HPDDocument9 pagesUltima High Recycled Content HPDJavier Enrique Diaz ChacinNo ratings yet
- Msds 3Document11 pagesMsds 3Amir masoud MoezziNo ratings yet
- HACCP Model NPIS Poultry SlaughterDocument23 pagesHACCP Model NPIS Poultry SlaughterjessycaduartesegoviaNo ratings yet
- Msds 2Document11 pagesMsds 2Amir masoud MoezziNo ratings yet
- 1084_TERRACLAD_Document6 pages1084_TERRACLAD_vishal shindeNo ratings yet
- Dipentaerythritol MSDSDocument5 pagesDipentaerythritol MSDSJaymit PatelNo ratings yet
- CC209-GADA Control-Instruction For UseDocument2 pagesCC209-GADA Control-Instruction For UseAdams QuintanillaNo ratings yet
- HPD Jaz Epoxy Sealer Clear UltraDocument5 pagesHPD Jaz Epoxy Sealer Clear UltraAhmad BougeisNo ratings yet
- Neo Guard: Life Guard Pharma Pvt. LTDDocument2 pagesNeo Guard: Life Guard Pharma Pvt. LTDRavi MadaanNo ratings yet
- Effisin® CHG 20 - AP - TDS ENDocument2 pagesEffisin® CHG 20 - AP - TDS ENKitra KiaraNo ratings yet
- Acusera Products RIT 2.10 (Online) Revision D Part 2 - Selling The Acusera BrandDocument93 pagesAcusera Products RIT 2.10 (Online) Revision D Part 2 - Selling The Acusera BrandAna Maria Tucno BarriosNo ratings yet
- Weekly ReportDocument7 pagesWeekly Reportpratistanadia_912023No ratings yet
- HPD Marmoleum Ohmex (Exp. 2022.01.09)Document10 pagesHPD Marmoleum Ohmex (Exp. 2022.01.09)Marco RaptusNo ratings yet
- Liquozyme SupraDocument2 pagesLiquozyme SupraHatem ZedanNo ratings yet
- Insert - Sample Cleaner 2.06529178001.V3.EnDocument1 pageInsert - Sample Cleaner 2.06529178001.V3.EnARIF AHAMMED PNo ratings yet
- Inteliq Cardiac Advance Control Levels 1, 2, 3 and 4Document2 pagesInteliq Cardiac Advance Control Levels 1, 2, 3 and 4jnyng222No ratings yet
- NordVal Certificate 033 Compact Dry TC Aerobic - Nissui - 24nov2021Document10 pagesNordVal Certificate 033 Compact Dry TC Aerobic - Nissui - 24nov2021RICHARD L. VAILATINo ratings yet
- Pro-Drill 568 MSDS: Performance Chemical CompanyDocument4 pagesPro-Drill 568 MSDS: Performance Chemical CompanyraedNo ratings yet
- MM TechDS 1601Document3 pagesMM TechDS 1601luisNo ratings yet
- 5a6f6 f680 4af2 9dc4 5f6b016cd633Document2 pages5a6f6 f680 4af2 9dc4 5f6b016cd633ISSL RSUDMWNo ratings yet
- g2 02 Clinker v2015 InglesDocument14 pagesg2 02 Clinker v2015 Inglesوائل شديوةNo ratings yet
- CC210-IAA Control-Instruction For UseDocument2 pagesCC210-IAA Control-Instruction For UseAdams QuintanillaNo ratings yet
- Quote: Pharm Canada PakistanDocument3 pagesQuote: Pharm Canada PakistanAli RazaNo ratings yet
- Microcare HDIDocument5 pagesMicrocare HDIdokterasadNo ratings yet
- TDS Bodoxin Ao, En, 2020Document2 pagesTDS Bodoxin Ao, En, 2020ckarantoniNo ratings yet
- HPD Marmoleum Sheet (Exp. 2024.09.12)Document8 pagesHPD Marmoleum Sheet (Exp. 2024.09.12)Marco RaptusNo ratings yet
- 054631Document5 pages054631sulajoNo ratings yet
- hpdc-91 DektonDocument9 pageshpdc-91 Dektonj.saiyathNo ratings yet
- MSDS Foodsafe Plus Greases 2, 1 & 00Document3 pagesMSDS Foodsafe Plus Greases 2, 1 & 00johanes kharismaNo ratings yet
- 251 Automatic Sliding Door Operator ES 200Document8 pages251 Automatic Sliding Door Operator ES 200celio BrandãoNo ratings yet
- Precipath HDL - LDL-C.11818171001.V10.en PDFDocument2 pagesPrecipath HDL - LDL-C.11818171001.V10.en PDFARIF AHAMMED PNo ratings yet
- Insert.C.f.a.s. Proteins.03554333001.V7.EnDocument2 pagesInsert.C.f.a.s. Proteins.03554333001.V7.EnonedarigirlNo ratings yet
- HACCP Model For Poultry SlaughterDocument19 pagesHACCP Model For Poultry Slaughterrimuru samaNo ratings yet
- Precinorm - Precipath Fructosamine.11934589001.V9.en PDFDocument2 pagesPrecinorm - Precipath Fructosamine.11934589001.V9.en PDFARIF AHAMMED PNo ratings yet
- 283MEDocument2 pages283MEqasim078No ratings yet
- MSDS Custodiol EnglischDocument4 pagesMSDS Custodiol EnglischHQ-Philippines CHAMPhmedNo ratings yet
- Revised March 2023Document1 pageRevised March 2023ghfjrcsk8vNo ratings yet
- Case Study: Beta-Lactam Decontamination and Cleaning Validation of A Pharmaceutical Manufacturing FacilityDocument5 pagesCase Study: Beta-Lactam Decontamination and Cleaning Validation of A Pharmaceutical Manufacturing FacilityМарина Рогоза100% (1)
- The Inventory Control Storage Facilities and Distr 230728 152955Document11 pagesThe Inventory Control Storage Facilities and Distr 230728 152955Yayan FadhlianoNo ratings yet
- Update Stok FMM - MaretDocument34 pagesUpdate Stok FMM - MaretErna RujuNo ratings yet
- Cbe 107 Sterile Products FinalDocument68 pagesCbe 107 Sterile Products Finalto van trungNo ratings yet
- Week 12 Short Answer QuestionsDocument2 pagesWeek 12 Short Answer Questionschristinaj98No ratings yet
- Development of A Topical Gel Containing A Dipeptidyl Peptidase-4 Inhibitor For Wound Healing ApplicationsDocument15 pagesDevelopment of A Topical Gel Containing A Dipeptidyl Peptidase-4 Inhibitor For Wound Healing ApplicationsGlobal Research and Development ServicesNo ratings yet
- Xtampza Vs Oxycontin - Main Differences and SimilaritiesDocument4 pagesXtampza Vs Oxycontin - Main Differences and SimilaritiesNicholas FeatherstonNo ratings yet
- Gastrointestinal Bioadhesive Drug Delivery Systems and Their ApplicationsDocument14 pagesGastrointestinal Bioadhesive Drug Delivery Systems and Their ApplicationsYIJINGNo ratings yet
- DP Ii ThesisDocument123 pagesDP Ii ThesisRohit PalNo ratings yet
- Administrative Medical Assisting 8th Edition French Test BankDocument13 pagesAdministrative Medical Assisting 8th Edition French Test Bankyearaastutecalml100% (36)
- Drug Alert List of Oct - 2022Document11 pagesDrug Alert List of Oct - 2022Gopal RaoNo ratings yet
- PDF Glee ProductsDocument1 pagePDF Glee ProductsSaurabh BishtNo ratings yet
- Low Endotoxin Recovery (LER) in Drug Products - Original - 31428Document2 pagesLow Endotoxin Recovery (LER) in Drug Products - Original - 31428Putri DozanNo ratings yet
- Contoh PembahasanDocument17 pagesContoh PembahasanNilam atika sariNo ratings yet
- KynaProductPriceList BFGHJJDocument11 pagesKynaProductPriceList BFGHJJAAradhya PharmaceuticalsNo ratings yet
- This Study Resource Was: NURS 6521N-26: Advanced PharmacologyDocument2 pagesThis Study Resource Was: NURS 6521N-26: Advanced Pharmacologywriter topNo ratings yet
- AI For Drug Discovery 2020Document130 pagesAI For Drug Discovery 2020Rajesh KrishnanNo ratings yet
- Common and Specific Antidotes: N-AcetylcysteineDocument2 pagesCommon and Specific Antidotes: N-AcetylcysteineMicolie GargalicanoNo ratings yet
- In Vitro, Sucralfate Adsorbs Bile SaltsDocument5 pagesIn Vitro, Sucralfate Adsorbs Bile SaltsZarbakht AliNo ratings yet
- Mono Rosemary - Oral EnglishDocument4 pagesMono Rosemary - Oral EnglishTom DelongeNo ratings yet
- Visiteur Pharmacie Grossiste Date: #DésignationDocument18 pagesVisiteur Pharmacie Grossiste Date: #DésignationLamine TABLI PHARMNo ratings yet
- BAMS IInd Year (06122017) PDFDocument22 pagesBAMS IInd Year (06122017) PDFDipesh MallickNo ratings yet
- Extractables and Leachables Characterization of Drug Products Packaging Manufacturing and Delivery Systems Medical Devices Dennis Jenke Full ChapterDocument68 pagesExtractables and Leachables Characterization of Drug Products Packaging Manufacturing and Delivery Systems Medical Devices Dennis Jenke Full Chapterbarbara.heinrich849100% (16)
- NDA Classification CodesDocument10 pagesNDA Classification CodesNathan RamosNo ratings yet
- Medicine ListDocument4 pagesMedicine ListAneela TabassumNo ratings yet
- Update Stock PT KMD 14Document42 pagesUpdate Stock PT KMD 14Trie Gusti linglingNo ratings yet
- 70 MG Once Weekly For Mrs. CordovaDocument2 pages70 MG Once Weekly For Mrs. CordovaSherrie Ann Delos SantosNo ratings yet