Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
5 viewsChemical Equation
Chemical Equation
Uploaded by
KOVLEEN SAHNIA chemical equation summarizes a chemical reaction using symbols for the reactants and products and their quantities. It shows the molecular level changes that occur during a chemical reaction. A balanced chemical equation maintains equality between the number and type of atoms in the reactants and products. It provides information about the products, reactants, and mole ratios involved in the reaction. However, a chemical equation alone does not convey information about reaction conditions like time, temperature, or whether the reaction proceeds to completion.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
You might also like
- Grade 11 .. Life Sciences TOPIC TEST: Gaseous Exchange: Learners NameDocument5 pagesGrade 11 .. Life Sciences TOPIC TEST: Gaseous Exchange: Learners Namedeveloping habit and lifestyle of praise and worshNo ratings yet
- 01 Atoms, Molecules and StoichiometryDocument7 pages01 Atoms, Molecules and StoichiometryMalak Sellat100% (1)
- End of Term Exam Dec 2019 p1Document11 pagesEnd of Term Exam Dec 2019 p1Mustafa100% (1)
- 000-Essentials of Medical Chemistry and Biochemistry (Textbook)Document211 pages000-Essentials of Medical Chemistry and Biochemistry (Textbook)millinagi95No ratings yet
- Ict Notes Grade 8Document9 pagesIct Notes Grade 8S.K.Y sharmaNo ratings yet
- Module Organic Chemistry 3v - Tto v1Document14 pagesModule Organic Chemistry 3v - Tto v1Winston de GrootNo ratings yet
- 4.4, 4.5 Exam QuestionsDocument21 pages4.4, 4.5 Exam QuestionsRana Hassan TariqNo ratings yet
- Physical Chemistry in Brief (Knjiga)Document466 pagesPhysical Chemistry in Brief (Knjiga)ivzupicNo ratings yet
- Test As OrganicDocument17 pagesTest As OrganicKashif KhanNo ratings yet
- BIO iAL AS Material Assessment Question PaperDocument11 pagesBIO iAL AS Material Assessment Question Paperdina al amasiNo ratings yet
- Topic 4.4 WorksheetttttDocument2 pagesTopic 4.4 WorksheetttttEmanuelah CuriNo ratings yet
- Unit 4.7 - Spectroscopy and Chromatography - TestDocument15 pagesUnit 4.7 - Spectroscopy and Chromatography - TestSan SiddzNo ratings yet
- ALHAIL PRIVATE SCHOOL 9 2nd WorksheetDocument15 pagesALHAIL PRIVATE SCHOOL 9 2nd WorksheetdodoNo ratings yet
- 1Document80 pages1Razie HanafiNo ratings yet
- Chapter 1Document5 pagesChapter 1Tajryan TanimNo ratings yet
- Chemistry QuestionsDocument40 pagesChemistry Questionskaotao1No ratings yet
- Grade 7 WorksheetDocument5 pagesGrade 7 Worksheetsunitak115No ratings yet
- IGCSE - Bio - Worksheet 1 - Q8-9 Rate of RespirationDocument4 pagesIGCSE - Bio - Worksheet 1 - Q8-9 Rate of Respiration7170No ratings yet
- 4.1 TestDocument8 pages4.1 TestShermerNo ratings yet
- Xaqw 12Document3 pagesXaqw 12sn2nc22No ratings yet
- Questions On Respiration: Water, Energy, Glucose, Food, Carbon Dioxide, Cells, OxygenDocument1 pageQuestions On Respiration: Water, Energy, Glucose, Food, Carbon Dioxide, Cells, Oxygennouran94No ratings yet
- Ramin Habibi Marand - Master Thesis of EpidemiologyDocument54 pagesRamin Habibi Marand - Master Thesis of EpidemiologyRamin Habibi MarandNo ratings yet
- 2.10 HWDocument5 pages2.10 HWChong BengNo ratings yet
- Biology - Ordinary Level Sample: Coimisiún Na Scrúduithe Stáit State Examinations CommissionDocument15 pagesBiology - Ordinary Level Sample: Coimisiún Na Scrúduithe Stáit State Examinations CommissionHaidery SalumuNo ratings yet
- 1.1 General ............................................................Document7 pages1.1 General ............................................................مصطفى العباديNo ratings yet
- Revision Quiz 3 - EnzymesDocument2 pagesRevision Quiz 3 - EnzymesSakura MuRongNo ratings yet
- Carbon Based Compounds Practice QuestionDocument1 pageCarbon Based Compounds Practice QuestionTijana TosicNo ratings yet
- Primary Science OlympiadsDocument8 pagesPrimary Science Olympiadschinyembakelvin947No ratings yet
- Peadiatric History Taking: Patient IdentificationDocument7 pagesPeadiatric History Taking: Patient Identificationpriyanjali sainiNo ratings yet
- Wu3c7 by Adel KhamisDocument7 pagesWu3c7 by Adel KhamisAdel KhamisNo ratings yet
- Worksheet ThermochemistryDocument44 pagesWorksheet ThermochemistryHARIYANTONo ratings yet
- Dommelen Solution ManualDocument197 pagesDommelen Solution Manualclhs8860No ratings yet
- 4.6, 4.7 TestDocument7 pages4.6, 4.7 Testriditha0% (1)
- TEOS 10 ManualDocument218 pagesTEOS 10 ManualJose EscanezNo ratings yet
- Uganda Lower Secondary Certificate of Education. Chemistry Senior Two End of Term One 2023Document12 pagesUganda Lower Secondary Certificate of Education. Chemistry Senior Two End of Term One 2023Owani Jimmy100% (1)
- Total Synthesis of Natural ProductsDocument291 pagesTotal Synthesis of Natural ProductsSDNo ratings yet
- TEOS-10 Manual PDFDocument220 pagesTEOS-10 Manual PDFBYRON TERANNo ratings yet
- Harmacology: Year: Fourth Year Semester: First Academic Year: 2021 - 2022Document3 pagesHarmacology: Year: Fourth Year Semester: First Academic Year: 2021 - 2022MOHAMED AZIZNo ratings yet
- Practica Dec. 2022Document4 pagesPractica Dec. 2022JOSÉ ALEXANDER TRUJILLO ARANDANo ratings yet
- Physical ChemistryDocument466 pagesPhysical Chemistryashutoshp600No ratings yet
- Alternative Anode Materials For Methane Oxidation in Solid Oxide Fuel CellsDocument266 pagesAlternative Anode Materials For Methane Oxidation in Solid Oxide Fuel CellsKarthi KeyanNo ratings yet
- Physical Chemistry Course OutlineDocument6 pagesPhysical Chemistry Course OutlineAmanda MhlopeNo ratings yet
- 1.3 TestDocument7 pages1.3 TestAlvin HanNo ratings yet
- CRE-CSTR NotesDocument89 pagesCRE-CSTR Notesdeepshikhasingh0% (1)
- S.5 Organic Chemistry Test One by Doctor Bamutalira 1Document6 pagesS.5 Organic Chemistry Test One by Doctor Bamutalira 1annetbulyaba0No ratings yet
- Synthesis of Bromochloromethane Using Phase Transfer CatalysisDocument101 pagesSynthesis of Bromochloromethane Using Phase Transfer CatalysisrgNo ratings yet
- Design and Synthesis of Novel Chloroquine-Based AntimalarialsDocument77 pagesDesign and Synthesis of Novel Chloroquine-Based AntimalarialsHHWegenerNo ratings yet
- Thesis ManuscriptDocument107 pagesThesis ManuscriptEunice SaavedraNo ratings yet
- LC025ALPO00EVDocument12 pagesLC025ALPO00EVrianrocheNo ratings yet
- The Experimental Determination of SolubilitiesFrom EverandThe Experimental Determination of SolubilitiesG. T. HefterRating: 2.5 out of 5 stars2.5/5 (1)
- Chirality in Transition Metal Chemistry: Molecules, Supramolecular Assemblies and MaterialsFrom EverandChirality in Transition Metal Chemistry: Molecules, Supramolecular Assemblies and MaterialsNo ratings yet
- Statistical Tolerance Regions: Theory, Applications, and ComputationFrom EverandStatistical Tolerance Regions: Theory, Applications, and ComputationNo ratings yet
Chemical Equation
Chemical Equation
Uploaded by
KOVLEEN SAHNI0 ratings0% found this document useful (0 votes)
5 views2 pagesA chemical equation summarizes a chemical reaction using symbols for the reactants and products and their quantities. It shows the molecular level changes that occur during a chemical reaction. A balanced chemical equation maintains equality between the number and type of atoms in the reactants and products. It provides information about the products, reactants, and mole ratios involved in the reaction. However, a chemical equation alone does not convey information about reaction conditions like time, temperature, or whether the reaction proceeds to completion.
Original Description:
Worksheet
Copyright
© © All Rights Reserved
Available Formats
DOC, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentA chemical equation summarizes a chemical reaction using symbols for the reactants and products and their quantities. It shows the molecular level changes that occur during a chemical reaction. A balanced chemical equation maintains equality between the number and type of atoms in the reactants and products. It provides information about the products, reactants, and mole ratios involved in the reaction. However, a chemical equation alone does not convey information about reaction conditions like time, temperature, or whether the reaction proceeds to completion.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
0 ratings0% found this document useful (0 votes)
5 views2 pagesChemical Equation
Chemical Equation
Uploaded by
KOVLEEN SAHNIA chemical equation summarizes a chemical reaction using symbols for the reactants and products and their quantities. It shows the molecular level changes that occur during a chemical reaction. A balanced chemical equation maintains equality between the number and type of atoms in the reactants and products. It provides information about the products, reactants, and mole ratios involved in the reaction. However, a chemical equation alone does not convey information about reaction conditions like time, temperature, or whether the reaction proceeds to completion.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
You are on page 1of 2
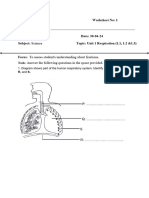
Chemical Equations
Q1. Define chemical equation.
A chemical equation is a …………………….representation of the……………………..reaction using……………………..and………………of
the………………………………..involved in the………………………………..
Q2. What is a balanced chemical equation?
It is an…………………in which the…………………..number of……………..of each…………………………in the…………………………on the
…………side of the……………………….is same as the number of atoms in………………………………formed on
the……………………….side of the …………………………….
Q3. What information is conveyed by a balanced chemical equation?
2NaOH + H2SO4 Na2SO4 + 2H2O
1. It tells about the …………………….result of a chemical change.
2. About the……………………..involved and the products…………………………….as a result of the reaction.
3. It tells about the number of ………………………………of each……………………………taking part and ………………….in the reaction.
4. About the chemical…………………………………of the respective…………………………………
5. It gives information about………………………….eg mass of H2SO4 is………………….
6. It also proves the ………………..of ……………………………………..of ………………….
Q4. State the limitations of a chemical equation.
A chemical equation does not tell:
1. ……………………..taken for ………………………………of reaction.
2. Respective………………………………..of …………………………..and ………………………………………….
3. The ………………………..at which the………………………proceeds.
4. Whether the ………………………is ………………………..or not completed.
Q5. Define relative atomic mass.
It is also called………………………It is the number of times………….atom of the…………………is ……………….than……..times
the……………….of an atom of………………….
Hence relative atomic mass =
Q6. Atomic mass unit id defined as …………….the mass of …………….
Q7. What is the value of 1amu in grams?
Q8. Define relative molecular mass.The…………………….molecular…………………of an……………………..or a ……………………….is
the………………..that represents how many times one…………………………..of the…………………………..is heavier than ………………
the mass of ……………………………….
Q9. Molecular mass of a substance is ………………..of ………………….masses of the……………………….atoms present in
one………………………of that……………………..
eg molecular mass of H2SO4 is :
Q10. Define percentage composition of a compound.
Percentage ………………….of a ………………………is the…………………………by ………………………..of each………………………present in a
………………………………
Formula :
You might also like
- Grade 11 .. Life Sciences TOPIC TEST: Gaseous Exchange: Learners NameDocument5 pagesGrade 11 .. Life Sciences TOPIC TEST: Gaseous Exchange: Learners Namedeveloping habit and lifestyle of praise and worshNo ratings yet
- 01 Atoms, Molecules and StoichiometryDocument7 pages01 Atoms, Molecules and StoichiometryMalak Sellat100% (1)
- End of Term Exam Dec 2019 p1Document11 pagesEnd of Term Exam Dec 2019 p1Mustafa100% (1)
- 000-Essentials of Medical Chemistry and Biochemistry (Textbook)Document211 pages000-Essentials of Medical Chemistry and Biochemistry (Textbook)millinagi95No ratings yet
- Ict Notes Grade 8Document9 pagesIct Notes Grade 8S.K.Y sharmaNo ratings yet
- Module Organic Chemistry 3v - Tto v1Document14 pagesModule Organic Chemistry 3v - Tto v1Winston de GrootNo ratings yet
- 4.4, 4.5 Exam QuestionsDocument21 pages4.4, 4.5 Exam QuestionsRana Hassan TariqNo ratings yet
- Physical Chemistry in Brief (Knjiga)Document466 pagesPhysical Chemistry in Brief (Knjiga)ivzupicNo ratings yet
- Test As OrganicDocument17 pagesTest As OrganicKashif KhanNo ratings yet
- BIO iAL AS Material Assessment Question PaperDocument11 pagesBIO iAL AS Material Assessment Question Paperdina al amasiNo ratings yet
- Topic 4.4 WorksheetttttDocument2 pagesTopic 4.4 WorksheetttttEmanuelah CuriNo ratings yet
- Unit 4.7 - Spectroscopy and Chromatography - TestDocument15 pagesUnit 4.7 - Spectroscopy and Chromatography - TestSan SiddzNo ratings yet
- ALHAIL PRIVATE SCHOOL 9 2nd WorksheetDocument15 pagesALHAIL PRIVATE SCHOOL 9 2nd WorksheetdodoNo ratings yet
- 1Document80 pages1Razie HanafiNo ratings yet
- Chapter 1Document5 pagesChapter 1Tajryan TanimNo ratings yet
- Chemistry QuestionsDocument40 pagesChemistry Questionskaotao1No ratings yet
- Grade 7 WorksheetDocument5 pagesGrade 7 Worksheetsunitak115No ratings yet
- IGCSE - Bio - Worksheet 1 - Q8-9 Rate of RespirationDocument4 pagesIGCSE - Bio - Worksheet 1 - Q8-9 Rate of Respiration7170No ratings yet
- 4.1 TestDocument8 pages4.1 TestShermerNo ratings yet
- Xaqw 12Document3 pagesXaqw 12sn2nc22No ratings yet
- Questions On Respiration: Water, Energy, Glucose, Food, Carbon Dioxide, Cells, OxygenDocument1 pageQuestions On Respiration: Water, Energy, Glucose, Food, Carbon Dioxide, Cells, Oxygennouran94No ratings yet
- Ramin Habibi Marand - Master Thesis of EpidemiologyDocument54 pagesRamin Habibi Marand - Master Thesis of EpidemiologyRamin Habibi MarandNo ratings yet
- 2.10 HWDocument5 pages2.10 HWChong BengNo ratings yet
- Biology - Ordinary Level Sample: Coimisiún Na Scrúduithe Stáit State Examinations CommissionDocument15 pagesBiology - Ordinary Level Sample: Coimisiún Na Scrúduithe Stáit State Examinations CommissionHaidery SalumuNo ratings yet
- 1.1 General ............................................................Document7 pages1.1 General ............................................................مصطفى العباديNo ratings yet
- Revision Quiz 3 - EnzymesDocument2 pagesRevision Quiz 3 - EnzymesSakura MuRongNo ratings yet
- Carbon Based Compounds Practice QuestionDocument1 pageCarbon Based Compounds Practice QuestionTijana TosicNo ratings yet
- Primary Science OlympiadsDocument8 pagesPrimary Science Olympiadschinyembakelvin947No ratings yet
- Peadiatric History Taking: Patient IdentificationDocument7 pagesPeadiatric History Taking: Patient Identificationpriyanjali sainiNo ratings yet
- Wu3c7 by Adel KhamisDocument7 pagesWu3c7 by Adel KhamisAdel KhamisNo ratings yet
- Worksheet ThermochemistryDocument44 pagesWorksheet ThermochemistryHARIYANTONo ratings yet
- Dommelen Solution ManualDocument197 pagesDommelen Solution Manualclhs8860No ratings yet
- 4.6, 4.7 TestDocument7 pages4.6, 4.7 Testriditha0% (1)
- TEOS 10 ManualDocument218 pagesTEOS 10 ManualJose EscanezNo ratings yet
- Uganda Lower Secondary Certificate of Education. Chemistry Senior Two End of Term One 2023Document12 pagesUganda Lower Secondary Certificate of Education. Chemistry Senior Two End of Term One 2023Owani Jimmy100% (1)
- Total Synthesis of Natural ProductsDocument291 pagesTotal Synthesis of Natural ProductsSDNo ratings yet
- TEOS-10 Manual PDFDocument220 pagesTEOS-10 Manual PDFBYRON TERANNo ratings yet
- Harmacology: Year: Fourth Year Semester: First Academic Year: 2021 - 2022Document3 pagesHarmacology: Year: Fourth Year Semester: First Academic Year: 2021 - 2022MOHAMED AZIZNo ratings yet
- Practica Dec. 2022Document4 pagesPractica Dec. 2022JOSÉ ALEXANDER TRUJILLO ARANDANo ratings yet
- Physical ChemistryDocument466 pagesPhysical Chemistryashutoshp600No ratings yet
- Alternative Anode Materials For Methane Oxidation in Solid Oxide Fuel CellsDocument266 pagesAlternative Anode Materials For Methane Oxidation in Solid Oxide Fuel CellsKarthi KeyanNo ratings yet
- Physical Chemistry Course OutlineDocument6 pagesPhysical Chemistry Course OutlineAmanda MhlopeNo ratings yet
- 1.3 TestDocument7 pages1.3 TestAlvin HanNo ratings yet
- CRE-CSTR NotesDocument89 pagesCRE-CSTR Notesdeepshikhasingh0% (1)
- S.5 Organic Chemistry Test One by Doctor Bamutalira 1Document6 pagesS.5 Organic Chemistry Test One by Doctor Bamutalira 1annetbulyaba0No ratings yet
- Synthesis of Bromochloromethane Using Phase Transfer CatalysisDocument101 pagesSynthesis of Bromochloromethane Using Phase Transfer CatalysisrgNo ratings yet
- Design and Synthesis of Novel Chloroquine-Based AntimalarialsDocument77 pagesDesign and Synthesis of Novel Chloroquine-Based AntimalarialsHHWegenerNo ratings yet
- Thesis ManuscriptDocument107 pagesThesis ManuscriptEunice SaavedraNo ratings yet
- LC025ALPO00EVDocument12 pagesLC025ALPO00EVrianrocheNo ratings yet
- The Experimental Determination of SolubilitiesFrom EverandThe Experimental Determination of SolubilitiesG. T. HefterRating: 2.5 out of 5 stars2.5/5 (1)
- Chirality in Transition Metal Chemistry: Molecules, Supramolecular Assemblies and MaterialsFrom EverandChirality in Transition Metal Chemistry: Molecules, Supramolecular Assemblies and MaterialsNo ratings yet
- Statistical Tolerance Regions: Theory, Applications, and ComputationFrom EverandStatistical Tolerance Regions: Theory, Applications, and ComputationNo ratings yet