Professional Documents
Culture Documents
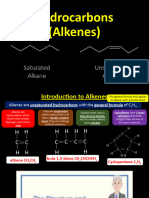
A Electrophilic Addition
A Electrophilic Addition
Uploaded by
赵倞Original Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
A Electrophilic Addition
A Electrophilic Addition
Uploaded by
赵倞Copyright:
Available Formats
HL Answers to Electrophilic addition questions
1. i. Bromine is non-polar as the bonding electron pair is shared equally between the two bromine
atoms. As the bromine molecule approaches the electron rich C=C double bond the electrons in the
double bond repel the bromine electrons on the nearest bromine atom inducing a dipole in the
molecule so that one of the Br atoms becomes δ+ (and can act as an electrophile) and the other
becomes δ−.
ii. 1,2-dibromoethane
iii. The intermediate carbocation formed is CH2Br-CH2+. In the presence of water the OH- from the
water adds to the carbocation in place of Br− as it is a better nucleophile. (Alternatively water adds
to give CH2Br-CH2OH2+ and then the Br− removes H+ to for the organic product and HBr.)
2.
The IB will usually award four marks for the answers involving this type of mechanism.
One mark each for:
curly arrow going from C=C to H of HBr and curly arrow showing Br– leaving;
correct representation of carbocation;
curly arrow going from lone pair/negative charge on :Br– to C+;
formation of 2-bromopropane, CH3CHBr(CH3);
(Note that in the past they have not awarded a mark for showing the δ+ and the δ- on the H―Br but I
think that it is good practice to include this and it is now specifically mentioned on the syllabus under
'Guidance').
3. H2IC―CHCl―CH2―CH3 2-chloro-1-iodobutane (1-iodo-2-chlorbutane would also be accepted).
4.
© Dr Geoffrey Neuss, InThinking
http://www.thinkib.net/chemistry 1
You might also like
- Organic Nitrogen Compounds Tutorial SolutionsDocument8 pagesOrganic Nitrogen Compounds Tutorial SolutionsDomNo ratings yet
- Pages From Organic Chemistry Handbbok For Exams Current 1, eDocument10 pagesPages From Organic Chemistry Handbbok For Exams Current 1, eTawanda ChimbwinyaNo ratings yet
- (123doc) - Tieu-Luan-Phan-Ung-Cong-AeDocument60 pages(123doc) - Tieu-Luan-Phan-Ung-Cong-Ae58.Nguyễn Thị Diệu LinhNo ratings yet
- Tutorial Letter 203/2/2017: General Chemistry 1BDocument18 pagesTutorial Letter 203/2/2017: General Chemistry 1BLeigh MakanNo ratings yet
- Radical Additions Anti-Markovnikov Product FormationDocument2 pagesRadical Additions Anti-Markovnikov Product Formationmegamon asabeNo ratings yet
- Tutorial Letter 203/1/2016: General Chemistry 1BDocument22 pagesTutorial Letter 203/1/2016: General Chemistry 1BLeigh MakanNo ratings yet
- Hydrocarbon Answer KeyDocument7 pagesHydrocarbon Answer KeyVK CREATIONNo ratings yet
- 187 Avoidable Errors in Reaction Mechanism QuestionsDocument4 pages187 Avoidable Errors in Reaction Mechanism QuestionsM DiNo ratings yet
- A Halogenoalkanes - BenzeneDocument1 pageA Halogenoalkanes - Benzene赵倞No ratings yet
- AQA - A Level - Chem - 1 - Answers Ch13.inddDocument4 pagesAQA - A Level - Chem - 1 - Answers Ch13.inddMahebul MazidNo ratings yet
- El Ctrophilic: SolutionDocument41 pagesEl Ctrophilic: SolutionManaswini PNo ratings yet
- Adisi Elektrofilik Kimia OrganikDocument25 pagesAdisi Elektrofilik Kimia OrganikAcikaNo ratings yet
- Chap 2Document47 pagesChap 2h2300147No ratings yet
- Adisi AlkeneDocument15 pagesAdisi Alkenekurniatriwijaya.2410No ratings yet
- Chemistry Set-1 BSSC Term IDocument10 pagesChemistry Set-1 BSSC Term IKavin SatyaNo ratings yet
- Reactivity and Mechanism: Presented By: Dr. Faisal Almalki (Male)Document30 pagesReactivity and Mechanism: Presented By: Dr. Faisal Almalki (Male)battal eduNo ratings yet
- AS Level - AlkenesDocument17 pagesAS Level - AlkenesRegine BalagtasNo ratings yet
- Chapter 7 (Org) - Part 2Document22 pagesChapter 7 (Org) - Part 2Wacka FlockaNo ratings yet
- 2014 H2 Alkanes Tut (Teachers)Document14 pages2014 H2 Alkanes Tut (Teachers)Chen ZhihaoNo ratings yet
- Alkenes and Alkynes CHM456Document93 pagesAlkenes and Alkynes CHM456nanaNo ratings yet
- L1 AlkenesDocument21 pagesL1 AlkenesCheng FuNo ratings yet
- Solution 927200Document3 pagesSolution 927200niveditasingh2472No ratings yet
- Hcno C H H H C H H H C H H H: Remains +1 ThroughoutDocument4 pagesHcno C H H H C H H H C H H H: Remains +1 ThroughoutpNo ratings yet
- GOC TheoryDocument11 pagesGOC Theorygargreyansh830No ratings yet
- Topic 10 Alkane TutorialDocument6 pagesTopic 10 Alkane TutorialTimNo ratings yet
- Styx Numbers and Rules of Boron HydridesDocument4 pagesStyx Numbers and Rules of Boron Hydridesrachzammit2003100% (1)
- Haloalkenes and HaloarenesDocument38 pagesHaloalkenes and Haloarenesshashwat201008No ratings yet
- Organic Unit Quiz Assignment 2020Document6 pagesOrganic Unit Quiz Assignment 2020ludihemicarNo ratings yet
- SAQ Ans 25Document1 pageSAQ Ans 25AbilashNo ratings yet
- Organic Chemistry: CollegeDocument34 pagesOrganic Chemistry: CollegeArwa AhmedNo ratings yet
- Organic Halogen Compounds UpdatedDocument20 pagesOrganic Halogen Compounds UpdatedDr. Dhondiba VishwanathNo ratings yet
- Cambridge University Pressorganic Chemistry PDFDocument47 pagesCambridge University Pressorganic Chemistry PDFluis peixoto100% (1)
- Practice Question & Worksheet For Chapter 10: Class-12 Haloalkanes & HaloarenesDocument3 pagesPractice Question & Worksheet For Chapter 10: Class-12 Haloalkanes & Haloarenesxajitib271No ratings yet
- PRACTICE MCQ HYDROCARBONS - 11ScADocument7 pagesPRACTICE MCQ HYDROCARBONS - 11ScAArda Rahmaini100% (1)
- Organic ChemistryDocument10 pagesOrganic ChemistrylovelykissNo ratings yet
- Premock A-Level Paper 1Document6 pagesPremock A-Level Paper 1Franck OmgbwaNo ratings yet
- BCHEM 2 - Lecture 2 - Organic Reaction MechanismDocument71 pagesBCHEM 2 - Lecture 2 - Organic Reaction Mechanismaduashongsolomon06No ratings yet
- Coursebook Answers: Self-Assessment QuestionsDocument2 pagesCoursebook Answers: Self-Assessment QuestionslizNo ratings yet
- Haloalkanes and HaloarenesDocument14 pagesHaloalkanes and Haloarenesshreyansh tanwarNo ratings yet
- CHM3201 Lab Report S2 2019-2020Document42 pagesCHM3201 Lab Report S2 2019-2020Halimatun MustafaNo ratings yet
- 10 Haloalkanes and Haloarenes 1 MK QuestionsDocument42 pages10 Haloalkanes and Haloarenes 1 MK QuestionsPriyanks RoutNo ratings yet
- Section A: 2018 Y6 H2 Chemistry Term 2 Common Test - Suggested SolutionsDocument18 pagesSection A: 2018 Y6 H2 Chemistry Term 2 Common Test - Suggested SolutionsImagreenbucklegirl SGNo ratings yet
- Hsslive-Xi-Chem-9. Hydrocarbons Q & ADocument14 pagesHsslive-Xi-Chem-9. Hydrocarbons Q & AshineNo ratings yet
- Haloalkanes and Arenes Q and AnsDocument66 pagesHaloalkanes and Arenes Q and AnsSandhya. SNo ratings yet
- CARBORANESDocument10 pagesCARBORANESDRISTIE KALITANo ratings yet
- STYX Rules and Structure of Higher BoranesDocument11 pagesSTYX Rules and Structure of Higher Boranesneharamkrishna75% (4)
- 241 QP PA-1 12-2023 - Answer KeysDocument3 pages241 QP PA-1 12-2023 - Answer KeyscaceyNo ratings yet
- Name of The Molecule Molecular Formula Hybridization Type Bond Angle GeometryDocument7 pagesName of The Molecule Molecular Formula Hybridization Type Bond Angle GeometryKerala MekuriyaNo ratings yet
- 2013 Alkane Tutorial (Solutions)Document7 pages2013 Alkane Tutorial (Solutions)Pinzhen ChenNo ratings yet
- Answers To Saqs: Cambridge International As Level ChemistryDocument2 pagesAnswers To Saqs: Cambridge International As Level ChemistryAlaNo ratings yet
- Dr. R. S. Shinde B.sc. I Sem-I Organic ChemistryDocument36 pagesDr. R. S. Shinde B.sc. I Sem-I Organic ChemistryHaroon Ur RashidNo ratings yet
- Mechanism of Organic Reactions 1Document23 pagesMechanism of Organic Reactions 1Suresh Vedpathak100% (2)
- SSGS 17-18 F.6 Final Exam 1 and 2 CHEMDocument37 pagesSSGS 17-18 F.6 Final Exam 1 and 2 CHEMKelvin ChowNo ratings yet
- CH1 C02-Structural and Molecular Organic Chemistry 2014Document22 pagesCH1 C02-Structural and Molecular Organic Chemistry 2014xapodi8776No ratings yet
- Metal Clusters:: Structure and Bonding in Higher BoranesDocument48 pagesMetal Clusters:: Structure and Bonding in Higher BoranesAnujNo ratings yet
- Alkenes Reactions NotesDocument14 pagesAlkenes Reactions NotesMartin AlvinNo ratings yet
- Organic Chem AnsDocument78 pagesOrganic Chem Ansrussell_mahmoodNo ratings yet
- Organometallic Chemistry: Plenary Lectures Presented at the Fourth International Conference on Organometallic ChemistryFrom EverandOrganometallic Chemistry: Plenary Lectures Presented at the Fourth International Conference on Organometallic ChemistryF. G. A. StoneNo ratings yet
- Plasma Chemistry: International Symposium on Plasma ChemistryFrom EverandPlasma Chemistry: International Symposium on Plasma ChemistryD. E. JensenNo ratings yet