Professional Documents
Culture Documents
Distillation ppt3
Distillation ppt3
Uploaded by
Limon C. Uriel0 ratings0% found this document useful (0 votes)
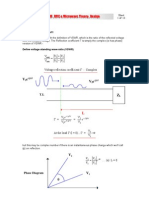
7 views1 pageThis document describes a single-stage equilibrium distillation process for separating two components, A and B. The process involves a constant molar overflow where the liquid and vapor flows into and out of the stage are equal. Mass balances are written for the total material and specifically for component A. These equations are solved graphically by finding the intersection between the equilibrium line and the overall material balance line to determine the unknown mole fractions of A in the liquid and vapor phases.
Original Description:
apuntes
Original Title
433871883-Distillation-ppt3
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document describes a single-stage equilibrium distillation process for separating two components, A and B. The process involves a constant molar overflow where the liquid and vapor flows into and out of the stage are equal. Mass balances are written for the total material and specifically for component A. These equations are solved graphically by finding the intersection between the equilibrium line and the overall material balance line to determine the unknown mole fractions of A in the liquid and vapor phases.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
7 views1 pageDistillation ppt3
Distillation ppt3
Uploaded by
Limon C. UrielThis document describes a single-stage equilibrium distillation process for separating two components, A and B. The process involves a constant molar overflow where the liquid and vapor flows into and out of the stage are equal. Mass balances are written for the total material and specifically for component A. These equations are solved graphically by finding the intersection between the equilibrium line and the overall material balance line to determine the unknown mole fractions of A in the liquid and vapor phases.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
3.
Equilibrium Stages - Single-stage
equilibrium
contactor
Binary distillation - components A & B
yA1 yA2
xA0 xA1
Constant molal overflow: V1 = V 2
L0 = L1
Total material balance: L0 + V2 = L1 + V1
Balance on A: L0xA0 + V2yA2 = L1xA1 + V1yA1
Unknown : x1 & y1 – solve simultaneously (graphically) between equilibrium line
& overall material balance
13
You might also like
- Chem3002-3-Stage and Continuous Gas Liquid Separation ProcessesDocument116 pagesChem3002-3-Stage and Continuous Gas Liquid Separation ProcessesJezreel CardinalesNo ratings yet
- Solid-Liquid Extraction (Leaching)Document4 pagesSolid-Liquid Extraction (Leaching)Dozdi83% (6)
- CHE3165 Formula SheetDocument7 pagesCHE3165 Formula SheetAbdurrakhman MukhyiddinNo ratings yet
- 單元操作PPT- chapter 20 PDFDocument52 pages單元操作PPT- chapter 20 PDFFucKerWengieNo ratings yet
- Introduction To Extraction ProcessesDocument7 pagesIntroduction To Extraction ProcessesBangkit GotamaNo ratings yet
- Liquid Liquid ExtractionDocument7 pagesLiquid Liquid ExtractionDozdi71% (7)
- Liquid-Liquid ExtractionDocument39 pagesLiquid-Liquid ExtractionHassanNo ratings yet
- CHE3165 Formula Sheet - TN3 PDFDocument7 pagesCHE3165 Formula Sheet - TN3 PDFnathish karanNo ratings yet
- Announcements: Read 12.8 and 12.9 After Monday Lecture, Skip 12.10 Homework 7 Due Friday, November 19Document23 pagesAnnouncements: Read 12.8 and 12.9 After Monday Lecture, Skip 12.10 Homework 7 Due Friday, November 19Kiran PatilNo ratings yet
- L4 Liquid-Liquid ExtractionDocument17 pagesL4 Liquid-Liquid ExtractionrickNo ratings yet
- Extraction CHAPTER 7 UpdatesDocument22 pagesExtraction CHAPTER 7 UpdatesSyakirin SpearsNo ratings yet
- Distillation by McCabe Thiele Method 1Document17 pagesDistillation by McCabe Thiele Method 1HassanNo ratings yet
- EE 740 Professor Ali Keyhani Lecture #13: Symmetrical ComponentsDocument11 pagesEE 740 Professor Ali Keyhani Lecture #13: Symmetrical ComponentsMohamed A. Hussein0% (1)
- EE204 Lecture Notes Lecture 05Document12 pagesEE204 Lecture Notes Lecture 05Quantum SaudiNo ratings yet
- Separation - AbsorptionDocument51 pagesSeparation - AbsorptionJason Yap Han YuenNo ratings yet
- Single Phase and Three Phase Rectifiers NumericalsDocument71 pagesSingle Phase and Three Phase Rectifiers Numericalsaryan singhNo ratings yet
- Ekstraksi Cair-Cair Pertemuan 3Document21 pagesEkstraksi Cair-Cair Pertemuan 3teguh aditya NNo ratings yet
- Chapter 3.2 Liquid-Liquid ExtractionDocument9 pagesChapter 3.2 Liquid-Liquid ExtractionJivashiniBalasupurManiamNo ratings yet
- EE 740 Professor Ali Keyhani Lecture #5: Three Phase Transformer Connections and Phase ShiftDocument10 pagesEE 740 Professor Ali Keyhani Lecture #5: Three Phase Transformer Connections and Phase ShiftMohamed A. HusseinNo ratings yet
- 13 Operational AmplifiersDocument51 pages13 Operational Amplifiers陳浚維No ratings yet
- Practice Exam 1 Fall 2017Document2 pagesPractice Exam 1 Fall 2017Allie JuneNo ratings yet
- Thevenins TheoremDocument18 pagesThevenins TheoremVitaliana VillaverdeNo ratings yet
- Toaz - Info Thevenins Theorem Lab Report PRDocument9 pagesToaz - Info Thevenins Theorem Lab Report PRhsejmal12345No ratings yet
- W-1-Day-4-D - Symmetrical ComponentsDocument18 pagesW-1-Day-4-D - Symmetrical ComponentsKASHIF100% (1)
- Stage & Continuous Gas-Liquid Separation Processes: Chapter ThreeDocument116 pagesStage & Continuous Gas-Liquid Separation Processes: Chapter ThreeDoni RopawandiNo ratings yet
- Chem3002-3-Stage and Continuous Gas Liquid Separation ProcessesDocument116 pagesChem3002-3-Stage and Continuous Gas Liquid Separation Processesthvindren anandarajahNo ratings yet
- 04-MB-4 Version Anglaise - Mai 2014Document4 pages04-MB-4 Version Anglaise - Mai 2014LuisAranaNo ratings yet
- Extraction 3Document19 pagesExtraction 3Kiran PatilNo ratings yet
- Announcements: //WWW - Engin.umich - Edu/class/che343Document19 pagesAnnouncements: //WWW - Engin.umich - Edu/class/che343Kiran PatilNo ratings yet
- Smith Chart Part 1Document13 pagesSmith Chart Part 1api-3823128No ratings yet
- Separation Process I: Dr. Lam Man Kee Department of Chemical EngineeringDocument25 pagesSeparation Process I: Dr. Lam Man Kee Department of Chemical EngineeringSENG LEE LIMNo ratings yet
- Circuit Analysis Using KCL (Node Voltage) MethodDocument3 pagesCircuit Analysis Using KCL (Node Voltage) MethodOshane So-confidential Sawyers100% (1)
- Chapter 3: Symmetrical ComponentsDocument18 pagesChapter 3: Symmetrical ComponentsKvv BapirajuNo ratings yet
- Miller PDFDocument5 pagesMiller PDFSimone MolinaroNo ratings yet
- 2 - Power System Fault Analysis - Symmetrical Components and Unbalanced FaultsDocument39 pages2 - Power System Fault Analysis - Symmetrical Components and Unbalanced FaultsKristhian Q. HernandezNo ratings yet
- 05 ChE421 - Multistage OperationsDocument15 pages05 ChE421 - Multistage OperationsAllan PaoloNo ratings yet
- Tutorial 1 AnswersDocument2 pagesTutorial 1 AnswersAkash VermaNo ratings yet
- Absorption Part 1 Student - 0Document16 pagesAbsorption Part 1 Student - 0AmyNo ratings yet
- Beam Deflection Tables - MechaniCalcDocument8 pagesBeam Deflection Tables - MechaniCalcHAFIZNo ratings yet
- Symm PDFDocument19 pagesSymm PDFPrince JosephNo ratings yet
- Chapter 12 Liquid ExtractionDocument68 pagesChapter 12 Liquid ExtractionNurul AinNo ratings yet
- Integration II HL Math MSDocument17 pagesIntegration II HL Math MSRonald McdonaldNo ratings yet
- Simple Regression 1Document18 pagesSimple Regression 1AmirahHaziqahNo ratings yet
- EE 1352 Monograph UnitIVDocument17 pagesEE 1352 Monograph UnitIVafzalbaigsa100% (2)
- Pair of Linear Equations in Two VariablesDocument8 pagesPair of Linear Equations in Two VariablesshamNo ratings yet
- Problem Set 5 Physical Electronics IDocument3 pagesProblem Set 5 Physical Electronics IJuan-carlos IsipNo ratings yet
- Mod1 DC Circuit1Document19 pagesMod1 DC Circuit1sumanta.kundu318020No ratings yet
- Source-TransformationDocument16 pagesSource-Transformationraovinayakm2No ratings yet
- 5 Chopper - 2Q - 4Q-Controlled DC DrivesDocument17 pages5 Chopper - 2Q - 4Q-Controlled DC DrivesrajapeeeNo ratings yet
- Straight LinesDocument2 pagesStraight Linesnabhijain9No ratings yet
- 5ECA0 Lecture 2 v4Document114 pages5ECA0 Lecture 2 v4Jacob KwonNo ratings yet
- Lecture EEE 447 Chap 3 Diode Rectifiers v2Document43 pagesLecture EEE 447 Chap 3 Diode Rectifiers v2Md. Anisur RahmanNo ratings yet
- Chapter 09Document234 pagesChapter 09JUAN M100% (1)
- Module 1 Basic ConceptsDocument70 pagesModule 1 Basic ConceptsTanuja VNo ratings yet
- Basic Laws - Voltage and Current DivisionDocument12 pagesBasic Laws - Voltage and Current DivisionSTACEYLEEN CYENTHIA LADIUN BK22110177No ratings yet
- Introduction To Load FlowDocument5 pagesIntroduction To Load FlowFake NameNo ratings yet
- A_8599_ElectricDocument9 pagesA_8599_ElectricAbidNo ratings yet
- Workbook A2 (1) 55Document1 pageWorkbook A2 (1) 55Limon C. UrielNo ratings yet
- InglesDocument4 pagesInglesLimon C. UrielNo ratings yet
- Tanque 3Document1 pageTanque 3Limon C. UrielNo ratings yet
- Workbook A2 (156Document1 pageWorkbook A2 (156Limon C. UrielNo ratings yet
- Workbook A2 (1) 77Document1 pageWorkbook A2 (1) 77Limon C. UrielNo ratings yet
- Workbook A2 (3Document4 pagesWorkbook A2 (3Limon C. UrielNo ratings yet
- Workbook InglesDocument4 pagesWorkbook InglesLimon C. UrielNo ratings yet
- Present Progressive: Yes/No Questions / Short AnswersDocument1 pagePresent Progressive: Yes/No Questions / Short AnswersLimon C. UrielNo ratings yet
- Richard's Romance - Part 2: Unit 5Document3 pagesRichard's Romance - Part 2: Unit 5Limon C. UrielNo ratings yet