Professional Documents
Culture Documents
CAN 1 Ionic Bonding Mat 2
CAN 1 Ionic Bonding Mat 2
Uploaded by
Kev Watts0 ratings0% found this document useful (0 votes)
9 views1 pageThe document discusses the formation of ionic bonds and compounds. It involves 3 steps: 1) drawing the electronic configuration for each atom, 2) filling in the new electronic configuration for ions by gaining or losing electrons, and 3) labeling the charge on each ion. Ionic bonding results from electrostatic attraction between positive and negative ions in a giant ionic lattice structure. This ionic compound is stable and its properties differ from the original atoms.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document discusses the formation of ionic bonds and compounds. It involves 3 steps: 1) drawing the electronic configuration for each atom, 2) filling in the new electronic configuration for ions by gaining or losing electrons, and 3) labeling the charge on each ion. Ionic bonding results from electrostatic attraction between positive and negative ions in a giant ionic lattice structure. This ionic compound is stable and its properties differ from the original atoms.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
9 views1 pageCAN 1 Ionic Bonding Mat 2
CAN 1 Ionic Bonding Mat 2
Uploaded by
Kev WattsThe document discusses the formation of ionic bonds and compounds. It involves 3 steps: 1) drawing the electronic configuration for each atom, 2) filling in the new electronic configuration for ions by gaining or losing electrons, and 3) labeling the charge on each ion. Ionic bonding results from electrostatic attraction between positive and negative ions in a giant ionic lattice structure. This ionic compound is stable and its properties differ from the original atoms.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
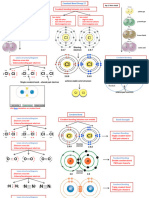
IONIC BONDING Formation of Ionic Compounds
1. draw in the electronic configuration for each atom.
2. Fill in the new electronic configuration for the ions.
3. Label the charge on the ion.
Formation of Ions
1. draw in the electronic configuration for each atom.
2. Fill in the new electronic configuration for the ion.
3. Label the charge on the ion.
IONIC BONDING Dot cross diagram 点叉图
Ionic 离子的
Electronic 电子排列
Bonding 键
configuration
Compound 化合物 Electrostatic attraction 静电吸引力
Positive charge 正电荷 force
Negative charge 负电荷 Giant ionic lattice 大型离子结构
Anion 阴离子
Stable 稳定的
Cation 阳离子
Outer shell 最外层
Metal 金属
Reactive 活泼的
Non-metal 非金属
Transfer 转移
You might also like
- Introduction To Plasma EtchingDocument58 pagesIntroduction To Plasma EtchingParashara Panduranga100% (1)
- Breakdown in GasesDocument28 pagesBreakdown in GasesGaurav kumarNo ratings yet
- Donate Electron Accept ElectronDocument2 pagesDonate Electron Accept ElectronPraveen Raj RajamaniNo ratings yet
- Core Bonding - Ionic BondingDocument21 pagesCore Bonding - Ionic BondingMarin PesicNo ratings yet
- AQA Combined Science Structure and BondingDocument2 pagesAQA Combined Science Structure and Bondingali.a.226No ratings yet
- Ionic Bonds Packet 1 WeeblyDocument4 pagesIonic Bonds Packet 1 WeeblyrajaijahNo ratings yet
- Electrocoagulation: Program Studi Teknik Pengolahan Limbah - PpnsDocument30 pagesElectrocoagulation: Program Studi Teknik Pengolahan Limbah - PpnsrizkaNo ratings yet
- Topic 2.1Document1 pageTopic 2.1duneraoreedNo ratings yet
- Analysis of Concept Chemistry For Senior High School Grade 10 Subject Matter: Chemical BondingDocument4 pagesAnalysis of Concept Chemistry For Senior High School Grade 10 Subject Matter: Chemical BondingSiti Rahma Dani HarahapNo ratings yet
- Chemical BondingDocument131 pagesChemical BondingAnant VashishtNo ratings yet
- Ionic BondingDocument2 pagesIonic Bondingdigjhon6No ratings yet
- SARA PEREZ TRUJILLO - Molecules and Bonds 7BDocument4 pagesSARA PEREZ TRUJILLO - Molecules and Bonds 7BApuestas JulanoNo ratings yet
- Bonding Knowledge OrganiserDocument1 pageBonding Knowledge Organisermya thet htar sweNo ratings yet
- ChemistryDocument20 pagesChemistryFatma SharifNo ratings yet
- Chemical-Bonding Part 01Document1 pageChemical-Bonding Part 01Sampa SadhukhanNo ratings yet
- Mass SpectrosDocument20 pagesMass SpectrosAvinashNo ratings yet
- 06 CB Notes 2022Document6 pages06 CB Notes 2022Fitri armaya Jeffri (Greendaless)No ratings yet
- ElectricityDocument20 pagesElectricityJürgen GeermanNo ratings yet
- F4 C5 Chemical BondingDocument23 pagesF4 C5 Chemical BondingkaiqianNo ratings yet
- Bonding in Solids SummaryDocument2 pagesBonding in Solids SummaryarachnidkatNo ratings yet
- EVOs and Hutchison Effect PDFDocument26 pagesEVOs and Hutchison Effect PDFChiodo72No ratings yet
- Eicbornhabercycleposter 733782Document1 pageEicbornhabercycleposter 733782Joko SusiloNo ratings yet
- Dokumen - Tips Ib Chemistry On VseprDocument14 pagesDokumen - Tips Ib Chemistry On VseprIwona Agata GórnickaNo ratings yet
- 5.2 Ionic BondsDocument6 pages5.2 Ionic BondspapajahatNo ratings yet
- Types of SolidsDocument5 pagesTypes of SolidsBianca RolstonNo ratings yet
- Lesson 1-2Document11 pagesLesson 1-2joelikestwich7070No ratings yet
- Chapter 9 Periodic Table - PeriodicityDocument5 pagesChapter 9 Periodic Table - Periodicitysitinur qahirahNo ratings yet
- Chemical Bond Part 1.Document11 pagesChemical Bond Part 1.ali.safwan022007.28No ratings yet
- Year 10 Science 2024 Semester Exam Revision Sheet ANSWER KEYDocument25 pagesYear 10 Science 2024 Semester Exam Revision Sheet ANSWER KEYsports.kingdomNo ratings yet
- Chemical Bonding - BrahmastraDocument29 pagesChemical Bonding - BrahmastraTanvi MalikNo ratings yet
- Chemistry Test 2: Date: 24/09/2020 Day: Thursday Topics/Chapters InvolvedDocument2 pagesChemistry Test 2: Date: 24/09/2020 Day: Thursday Topics/Chapters InvolvedRXNOFCHMNo ratings yet
- S3 From Ion Formation 22-23 (Ans)Document4 pagesS3 From Ion Formation 22-23 (Ans)chun yau lamNo ratings yet
- Chapter 7 Ionic and Metallic BondingDocument56 pagesChapter 7 Ionic and Metallic BondingCharles GibbsNo ratings yet
- 1.3 Revision Guide Bonding AqaDocument3 pages1.3 Revision Guide Bonding AqaPragna AnanthNo ratings yet
- CHAPTER 5 - Chemical Bond (5.1 - 5.3) 8 JulyDocument25 pagesCHAPTER 5 - Chemical Bond (5.1 - 5.3) 8 JulyNur100% (1)
- Gen Chem Module 6 Answer PDFDocument5 pagesGen Chem Module 6 Answer PDFjhon paul espinarNo ratings yet
- BS Chemistry MCQsDocument6 pagesBS Chemistry MCQsRamsha saeedNo ratings yet
- Structure and Bonding. JobsjkkDocument77 pagesStructure and Bonding. JobsjkkkokodejoelNo ratings yet
- Introductory Chemistry: Chemical BondDocument42 pagesIntroductory Chemistry: Chemical BondmayNo ratings yet
- Acis Bonding SLDocument69 pagesAcis Bonding SLJeenal AgrawalNo ratings yet
- Chemical Bonding SummaryDocument1 pageChemical Bonding Summarychong56No ratings yet
- Spec Sony Us18650Document8 pagesSpec Sony Us18650SpyNo ratings yet
- Ionic Bonding - FactRecallDocument1 pageIonic Bonding - FactRecallgabby fosterNo ratings yet
- Ian Appelbaum: Electrical and Computer Engineering University of DelawareDocument39 pagesIan Appelbaum: Electrical and Computer Engineering University of DelawareLIAKMANNo ratings yet
- 2nd Quarter Reviewer - CHEMISTRYDocument2 pages2nd Quarter Reviewer - CHEMISTRYKate Ashley CailingNo ratings yet
- Topics 1-4 Concept ListDocument2 pagesTopics 1-4 Concept ListSteve JacobsNo ratings yet
- Electricity & MagnetismDocument34 pagesElectricity & MagnetismMark Niño JavierNo ratings yet
- Structural Analysis of NanomaterialsDocument27 pagesStructural Analysis of NanomaterialswinnieNo ratings yet
- W3W4 BTD1123 Chapter 2 Mat Structure N BondingDocument62 pagesW3W4 BTD1123 Chapter 2 Mat Structure N BondingHakim ShahmiNo ratings yet
- MECH 3830 1: Chapter 1-TextbookDocument8 pagesMECH 3830 1: Chapter 1-TextbookharnoorNo ratings yet
- Why Are Metals Good Conductors?Document1 pageWhy Are Metals Good Conductors?Paul BurgessNo ratings yet
- Electronegativity Bond Polarity: Electronegativity of An ElementDocument12 pagesElectronegativity Bond Polarity: Electronegativity of An ElementSamNo ratings yet
- TA B L E 2.1. Overview of The Ion Generation Methods Described in This ChapterDocument18 pagesTA B L E 2.1. Overview of The Ion Generation Methods Described in This ChapterLaura Daniela Rodríguez CastroNo ratings yet
- Science9.Chapter5.Lesson1 - Ionic BondingDocument51 pagesScience9.Chapter5.Lesson1 - Ionic BondingടHՕՕꝄedкƲȠNo ratings yet
- CH 2 Chemical Bonding PDFDocument26 pagesCH 2 Chemical Bonding PDFTonald DrumpNo ratings yet
- Chemical Bonding One Day One Chapter Nitesh DevnaniDocument41 pagesChemical Bonding One Day One Chapter Nitesh Devnanivrinda11xxNo ratings yet
- Chemistry: Writing Ionic Formulas For CompoundsDocument5 pagesChemistry: Writing Ionic Formulas For CompoundsTiffany GallinaNo ratings yet
- Corrosion Resistance of Aluminum and Magnesium Alloys: Understanding, Performance, and TestingFrom EverandCorrosion Resistance of Aluminum and Magnesium Alloys: Understanding, Performance, and TestingNo ratings yet
- Unit 8 When IsDocument47 pagesUnit 8 When IsKev WattsNo ratings yet
- The Universe v2.0Document38 pagesThe Universe v2.0Kev WattsNo ratings yet
- Bonding MatDocument1 pageBonding MatKev WattsNo ratings yet
- All About Chemical Bonding - IonicDocument7 pagesAll About Chemical Bonding - IonicKev WattsNo ratings yet
- AstrophysicsDocument39 pagesAstrophysicsKev Watts100% (1)
- New Spec Revision NotesDocument43 pagesNew Spec Revision NotesKev WattsNo ratings yet
- How Different Conditions Affect The Yield of AmmoniaDocument1 pageHow Different Conditions Affect The Yield of AmmoniaKev WattsNo ratings yet
- How To Make A Concept - MapDocument4 pagesHow To Make A Concept - MapKev WattsNo ratings yet
- List of What Pupils Need To knowGCSE Biology Learnt in KS3Document1 pageList of What Pupils Need To knowGCSE Biology Learnt in KS3Kev WattsNo ratings yet