Professional Documents
Culture Documents
Internal Respiration Atf
Internal Respiration Atf
Uploaded by
patriciaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Internal Respiration Atf
Internal Respiration Atf
Uploaded by
patriciaCopyright:
Available Formats
Last edited: 9/13/2021
1. INTERNAL RESPIRATION
Internal Respiration Medical Editor: Dr. Sofia Suhada M. Uzir
OUTLINE (3) Carbon dioxide distribution
Comes from the tissues into the blood to be transported
I) EXTERNAL RESPIRATION to the alveoli
II) INTERNAL RESPIRATION
o 2-10 % dissolved in the blood plasma
III) REVIEW QUESTIONS
o 20% bound to the amino acids of the globin chains
IV) REFERENCES
o 70% hidden in the form of bicarbonate
(i) Carbon dioxide in the form of bicarbonate
They are abundant in the plasma
o Control acid base balance
The bicarbonates move into RBC
I) EXTERNAL RESPIRATION o Chloride moves out (chloride shift)
RBCs go through the pulmonary capillaries where the This is to balance the shift of ion moving into the
alveoli are located → gas exchange occur cell
o Oxygen o Interact and combine with the protons on RBC
Moves from the alveoli into the blood Happens when hemoglobin affinity for CO2,
o Carbon dioxide (CO2) protons and 2,3 BPG decrease
Moves from the blood into the alveoli Bicarbonates that combine with proton will
produce carbonic acid (H2CO3) → dissociate into
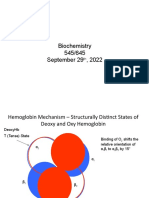
(A) DEOXYGENATED HEMOGLOBIN (T STATE) CO2 and water in the presence of carbonic
anhydrase enzyme
Low affinity for oxygen
(1) The structure of deoxygenated hemoglobin
Particularly HbA1, consist of
(i) 4 globin chains
2 Alpha chains
2 Beta chain
(ii) 4 iron containing heme groups
In the ferrous state, Fe 2+
It is bound to only 1 oxygen
(iii) Positive charged pocket Figure 1. The structures of hemoglobin in the T state vs in the
Made up of positively charged amino acids R state
o Histidine (B) POSITIVE COOPERATIVITY
o Arginine
o Lysine Happens when the oxygen moves from alveoli into the
blood to bind with the hemoglobin
(2) Molecules bound to the hemoglobin It is extremely hard for the first oxygen to bind to the
hemoglobin but it gets easier as more oxygen bind to it
(i) CO2
(C) OXYGENATED HEMOGLOBIN (R STATE)
Which is bound to amino acids at globin (protein) chains
o Account for 20% CO2 transported in the blood The structure of oxygenated hemoglobin changes from
circulation from the tissue to the alveoli→ carbamino the deoxygenated state
hemoglobin Affinity to oxygen is high
(ii) Protons (1) Partial pressures
Which are positively charged (i) Oxygen
o Bound to amino acids on the beta/alpha chains
In the alveoli is 104 mmHg
which are negatively charged
In the blood is 40 mmHg
The amino acids interact with the protons
→ protons bind to the hemoglobin → deoxy state (ii) Carbon dioxide
(T state) Figure 1
In the alveoli is 40 mmHg
(iii) 2,3 BPG In the blood is 45-46 mmHg
Even with the little difference, CO2 move easily from the
A negatively charged glycolytic intermediate
plasma to the alveoli because is it 20 times more
Bind to the positively charged pocket to stabilize the
soluble than the oxygen in the plasma and alveolar fluid
hemoglobin in the T state → affinity for oxygen is low
(2) Movement of gases
The gases move down the concentration gradient
o From high to low until equilibrium is reached
The alveoli = pulmonary arterial blood
INTERNAL RESPIRATION RESPIRATORY PHYSIOLOGY: Note #1. 1 of 4
The oxygen from the alveoli binds to iron containing the (B) MECHANISM OF GAS EXCHANGE
heme groups of hemoglobin in the blood
o A lot of oxygen bind causing → oxygen loading (1) Carbon dioxide (CO2)
CO2 unloaded → moves from the blood into the alveoli Is made through cellular respiration
o Bicarbonates converted to CO2 → unloaded In metabolizing oxygen, cellular respiration reaction takes
o CO2 dissolved in the plasma → unloaded place → giving off the CO2
o C6H12O6 (glucose) + 6 O2 (oxygen) → 6 CO2 + 6 H2O
The arterial blood leaving the lungs into the systemic + ATP + heat
circulation reaching the tissues will contain
o 104 mmHg Oxygen CO2 moves from the tissue cells into the blood Figure 2
o 40 mmHg CO2
(i) 2-10% of CO2
(3) Molecules bound to the hemoglobin Dissolve in the blood plasma
(i) 2,3 BPG (ii) 20% of CO2
When the iron in the hemoglobin is bound the oxygen, the Bind to the amino acids of alpha/beta chains
positively charged moieties will not be exposed → 2,3 o In the form of carbamino hemoglobin
BPG cannot bind to it Figure 1
(iii) 70% of CO2
(ii) Carbon dioxide (CO2)
Taken into the RBC to bind with water → formation of
CO2 moves to the alveoli carbonic acid (H2CO3) in the presence of carbonic
anhydrase enzyme
(iii) Oxygen
Carbonic acid is very unstable → dissociate into proton
Oxygen binds to the hemoglobin molecule and bicarbonate
CO2 unloaded + oxygen loaded = Haldane effect o The protons bind to the negatively charged amino
acids on the globin chains → electrostatic
(D) HALDANE EFFECT
interactions
Blood coming to the lungs which had a low affinity o The bicarbonate goes to the plasma to act as a buffer
for oxygen becomes high system in the blood
This is due to the oxygen moving down its concentration Has pKa that closely match the pH of the blood
gradient and bind to iron containing heme groups As the bicarbonate goes out of the cell, chloride
o The hemoglobin affinity for CO2, protons and 2,3 BPG will enter in order to balance the shift of negative
decrease → unload ion leaving the cell (chloride shift)
o The hemoglobin affinity for oxygen increase →
positive cooperativity → oxygen loaded on the (2) Dissociation of oxygen
hemoglobin When protons and CO2 bind to amino acids of the globin
chain → allosterically regulating the hemoglobin
II) INTERNAL RESPIRATION o Changes the shape of the hemoglobin molecule
Oxygenated state of hemoglobin → travel to tissues o Weakens the bond between the iron and oxygen
Structure of oxygenated hemoglobin include → until eventually the bond breaks
o Oxygen bound to the iron o The heat produced as a biproduct from the metabolic
o Globin chains without CO2 process of the tissues also contribute to the weaking
o Hidden positively charged moiety and breaking of the iron and oxygen bond Figure 2
Not exposed to the 2,3 BPG → cannot bind The oxygen will dissociate and go to the tissues
When the oxygen dissociates, the positively charged
(1) Partial pressure during rest
pocket will be exposed for the binding of 2,3 BPG →
(i) Oxygen stabilize the hemoglobin in the T state → deoxy
hemoglobin
o In the tissue cells is 40 mmHg
o In the arterial blood is 104 mmHg (3) Oxygen
Oxygen leaves the blood and enters the tissue to be
(ii) Carbon dioxide (CO2)
utilized in metabolic processes
o In the tissue cells 45-46 mmHg o Produce ATP
o In the arterial blood 40 mmHg o Break down different types of molecules
Gases move from area of high pressure to low pressure o Produce heat
o Oxygen moves from the arterial blood into the tissue (4) Temperature/heat
cells
o CO2 moves from the tissue cells into the arterial Heat is the biproduct from the electron transport chain
blood that occurs in the tissues
It is used as the measurement of temperature /thermal
energy
o Delta H is the form of energy
Protein structures change when they’re exposed to
excessive heat and different types of pH
o Hemoglobin protein will undergo confirmational
change due to the heat → contributing to the breaking
of the iron and oxygen bond
2 of 4 RESPIRATORY PHYSIOLOGY: Note #1. INTERNAL RESPIRATION
(2) Arterial blood
The blood, which is now the arterial blood, leaving the
lungs will have the partial pressures of gases as follows
o Oxygen 104 mmHg
o CO2 40 mmHg
The gases will then again travel to the tissues and the
cycle continues
III) REVIEW QUESTIONS
1) Regarding oxygen, which is true
a) It travels from the blood to the alveoli
b) Binding of carbon dioxide to the hemoglobin helps in
the release of oxygen into the tissues
c) The partial pressure of oxygen in cell tissues is 104
mmHg
d) In internal respiration, it moves from the alveoli into
the blood
2) Which of the following is not a part of the circulatory
system?
a) Pulmonary ventilation
b) External respiration
Figure 2. The overall mechanism of internal respiration c) Transport
d) Internal respiration
(C) BOHR EFFECT
3) The most important factor that determines how much
Hemoglobin affinity for oxygen that was initially high
oxygen combines with hemoglobin is
becomes low
a) Oxygen
This is due to the CO2, protons and 2,3 BPG which bind
b) Temperature
to the hemoglobin with the contribution of heat from the
c) CO2
tissues → change overall structure of hemoglobin
d) 2,3 DPG
o This change eventually breaks iron and oxygen bond
→ oxygen dissociate →decrease in hemoglobin 4) Hemoglobin has a tendency to release oxygen where
affinity for oxygen → unload the oxygen a) temperature is lower
b) pH is more acidic
Hemoglobin affinity for CO2, proton, 2,3 BPG increase c) partial pressures of carbon dioxide are lower
o Loaded on the hemoglobin d) pH is more alkaline
(D) PRESSURES OF GASES 5) Most of the carbon dioxide transported by the blood
The partial pressures of gases when the blood drains is
from the capillary bed going to the vein a) converted to bicarbonate ions and transported in
plasma
(i) Oxygen b) dissolved in plasma
c) bound to the same protein as carbon dioxide
40 mmHg in the cells
d) bound to hemoglobin
104 mmHg in the capillary bed
Hence the oxygen will move from the capillary bed into 6) Approximately 20% of carbon dioxide is transported
the cells until it reaches the equilibrium of 40 mmHg in the blood as
a) bicarbonate ions
(ii) CO2 b) carbaminohemoglobin
45-46 mmHg in the cells c) hydrogen ions
40 mmHg in the capillary bed d) dissolved gas in the plasma
Hence, the CO2 will move from the cells into the capillary 7) In the alveoli, the partial pressure of oxygen is
beds until it reaches the equilibrium of 45 mmHg a) lower than the PO2 of venous blood
b) much higher than the PO2 of arterial blood
Remember c) the same as the PO2 of venous blood.
Gas exchange occurs until the concentration of gases in the d) about 104 mm Hg
blood is equal to the tissues
8) In the internal respiration
(1) Veinous blood a) Oxygen moves from the tissues into the alveoli
b) Oxygen moves from the blood to the alveoli
When the blood leaves the capillary bed, and enters the c) Oxygen moves from the blood into the tissues
vein, the partial pressure will be as follows d) Oxygen moves from the tissues into the blood
o CO2 45 mmHg
o Oxygen 40 mmHg 9) Regarding the Bohr effect
a) The hemoglobin affinity to oxygen decreases
These gases will go to the lungs b) The hemoglobin affinity to carbon dioxide decreases
In the lungs, once again the gas exchange occurs down c) The carbon dioxide leaves the hemoglobin
the concentration gradient d) Oxygen will bind to the hemoglobin
o The oxygen from the alveoli will enter the blood
From 104 mmHg to 40 mmHg until the equilibrium 10) Breaking of the iron and oxygen bond is caused by
of 104 mmHg is reached a) Change in the red blood cell membrane potential
o The CO2 will move from the blood to the alveoli b) Release of carbon dioxide into the plasma
From 45-46 mmHg to 40 mmHg until the c) Binding of carbon dioxide to the hemoglobin
equilibrium of 40 mmHg is reached d) Decrease in the 2,3 BPG levels in the blood
CHECK YOUR ANSWERS
INTERNAL RESPIRATION RESPIRATORY PHYSIOLOGY: Note #1. 3 of 4
IV) REFERENCES
● Sabatine MS. Pocket Medicine: the Massachusetts General
Hospital Handbook of Internal Medicine. Philadelphia: Wolters
Kluwer; 2020.
● Le T. First Aid for the USMLE Step 1 2020. 30th anniversary
edition: McGraw Hill; 2020.
● Jameson JL, Fauci AS, Kasper DL, Hauser SL, Longo DL,
Loscalzo J. Harrison's Principles of Internal Medicine, Twentieth
Edition (Vol.1 & Vol.2). McGraw-Hill Education / Medical; 2018
● Marieb EN, Hoehn K. Anatomy & Physiology. Hoboken, NJ:
Pearson; 2020.
● Boron WF, Boulpaep EL. Medical Physiology.; 2017.
● Guyton and Hall Textbook of Medical Physiology. Philadelphia,
PA: Elsevier; 2021.
4 of 4 RESPIRATORY PHYSIOLOGY: Note #1. INTERNAL RESPIRATION
You might also like
- Plant Design Project Report-Group 6BDocument275 pagesPlant Design Project Report-Group 6BNana kwadwo100% (2)
- Schaum's Easy Outline of Organic Chemistry, Second EditionFrom EverandSchaum's Easy Outline of Organic Chemistry, Second EditionRating: 3.5 out of 5 stars3.5/5 (2)
- PAHS Curriculum Final Version NepalDocument249 pagesPAHS Curriculum Final Version Nepalpreeti A100% (2)
- 17 CRISP PhysiologyDocument5 pages17 CRISP PhysiologySyed Aijaz44% (9)
- Proposal For Chemistry Project STPMDocument2 pagesProposal For Chemistry Project STPMChan Choon Yee100% (2)
- Measuring PH of Soil For Use in Corrosion Testing: Standard Test Method ForDocument3 pagesMeasuring PH of Soil For Use in Corrosion Testing: Standard Test Method ForHadi HowaidaNo ratings yet
- 232 Respiratory Physiology External Respiration Partial PressuresDocument3 pages232 Respiratory Physiology External Respiration Partial PressuresAhmed JabbarNo ratings yet
- External Respiration - Partial Pressures & Solubilities (2) AtfDocument3 pagesExternal Respiration - Partial Pressures & Solubilities (2) AtfiimjokerssNo ratings yet
- Transport of Oxygen and CarbondioxideDocument27 pagesTransport of Oxygen and CarbondioxideDhanush SatheeshNo ratings yet
- Gas Exchange - Respirasi Blok 2.1.maret.15Document29 pagesGas Exchange - Respirasi Blok 2.1.maret.15Fatiha TamadNo ratings yet
- Lecture 6 Gas TransportDocument37 pagesLecture 6 Gas Transport7mb767gqyvNo ratings yet
- Transportation of OxygenDocument37 pagesTransportation of OxygenDrAbhilasha SharmaNo ratings yet
- Breathing BookletDocument6 pagesBreathing BookletANo ratings yet
- UAS Kritis 1 (T4) - Monitoring Fungsi Pernapasan - Bu HY (A11)Document108 pagesUAS Kritis 1 (T4) - Monitoring Fungsi Pernapasan - Bu HY (A11)rifqifuadiNo ratings yet
- Breathing Exchange and Gases 5Document4 pagesBreathing Exchange and Gases 5Dr. D. kumar Harshit -apexNo ratings yet
- Oxygen & Carbon Dioxide Transport by Blood: Page 58 Respiratory PhysiologyDocument15 pagesOxygen & Carbon Dioxide Transport by Blood: Page 58 Respiratory PhysiologygerginNo ratings yet
- 08-The Mamalian Transport SystemDocument4 pages08-The Mamalian Transport SystemTia wkwkNo ratings yet
- Biochem PresentationDocument16 pagesBiochem PresentationRushana SadaqatNo ratings yet
- Gas Transport 2Document29 pagesGas Transport 2Charmaine LucNo ratings yet
- 8-3 HB, Dissociation Curves (2022 Syllabus)Document40 pages8-3 HB, Dissociation Curves (2022 Syllabus)xbb2b9pmpwNo ratings yet
- Hemoglobin Structure & FunctionDocument28 pagesHemoglobin Structure & Functiondeepak3027315No ratings yet
- Transport of GasesDocument39 pagesTransport of GasesJayballabh KumarNo ratings yet
- Renal Physiology The Collecting DuctDocument3 pagesRenal Physiology The Collecting DuctSahnusha SryenNo ratings yet
- O2 Transport CostanzoDocument10 pagesO2 Transport CostanzoStudent1010No ratings yet
- Acid-Base BalanceDocument11 pagesAcid-Base Balancemostafadiab074No ratings yet
- Biochemistry of HB Oxydation-ReductionDocument5 pagesBiochemistry of HB Oxydation-ReductionYasmine AzzackiyahNo ratings yet
- Gas Transport & PH Regulation UDocument56 pagesGas Transport & PH Regulation UAhmad MirzaNo ratings yet
- Co2 PDFDocument4 pagesCo2 PDFSITI SARAH BTE NOOR MEAHNo ratings yet
- Transport of Carbon DioxideDocument17 pagesTransport of Carbon Dioxideyasir ishaqNo ratings yet
- Heme Synthesis Breakdown HBDocument18 pagesHeme Synthesis Breakdown HBDr.P.NatarajanNo ratings yet
- Carbon Dioxide TransportDocument3 pagesCarbon Dioxide TransportAurelia AlexandraNo ratings yet
- Bioinorganic Chemistry PDFDocument46 pagesBioinorganic Chemistry PDFabhay kesharwaniNo ratings yet
- Gas Transport in Blood PPT 1Document34 pagesGas Transport in Blood PPT 1CLEMENTNo ratings yet
- Zoology Restart - Daily Practice Paper: Chapter - Breathing and Exchange of Gases Topic - Transport of Gases Part 2Document2 pagesZoology Restart - Daily Practice Paper: Chapter - Breathing and Exchange of Gases Topic - Transport of Gases Part 2www.ksarma55No ratings yet
- Trans. of OxygenDocument32 pagesTrans. of OxygenSuraiya IslamNo ratings yet
- Myoglobin & HemoglobinDocument53 pagesMyoglobin & HemoglobinAgnetasia PriscilliaNo ratings yet
- Gas TransportDocument10 pagesGas TransportDaveNo ratings yet
- 1-The Bicarbonate Buffer System-2022Document41 pages1-The Bicarbonate Buffer System-2022Keerthi Chowdary AmaraneniNo ratings yet
- Biochem Ratio 7 FinalDocument31 pagesBiochem Ratio 7 FinalJimell ObradorNo ratings yet
- 2022.01.07 Live Session Article EquilibriumDocument4 pages2022.01.07 Live Session Article EquilibriumInês AlmeidaNo ratings yet
- Dissolved Carbon Dioxide 2. Bound To Haemoglobin & Plasma ProteinDocument2 pagesDissolved Carbon Dioxide 2. Bound To Haemoglobin & Plasma Proteinarun139No ratings yet
- Ransport Of: Dr. Roopa Kotha Dr. Shaji MathewDocument36 pagesRansport Of: Dr. Roopa Kotha Dr. Shaji MathewRenganathan SockalingamNo ratings yet
- Gas Transportation: Dr. Abdalla Adam AbdallaDocument12 pagesGas Transportation: Dr. Abdalla Adam AbdallaR00r0 M0h4mm3dNo ratings yet
- Physeo - Lecture 3Document21 pagesPhyseo - Lecture 3Mahmoud hilmyNo ratings yet
- O2 and CO2 TransportDocument70 pagesO2 and CO2 TransportThomas HuxleyNo ratings yet
- Oxygen Transport JOHN W. BAYNES, MAREK H. DOMINICZAK - Medical Biochemistry-Elsevier Inc. (2019)Document15 pagesOxygen Transport JOHN W. BAYNES, MAREK H. DOMINICZAK - Medical Biochemistry-Elsevier Inc. (2019)Malika MohNo ratings yet
- Lecture 11 9-29-22Document20 pagesLecture 11 9-29-22Caleb HeNo ratings yet
- Functions of HemoglobinDocument63 pagesFunctions of HemoglobinMadeline UdarbeNo ratings yet
- 18 Diuretics (Notes) AtfDocument19 pages18 Diuretics (Notes) AtfSeerat ShabirNo ratings yet
- DPN-Biochem I - Lecture 8 - Myoglobin and HemoglobinDocument41 pagesDPN-Biochem I - Lecture 8 - Myoglobin and Hemoglobinchienyu2002No ratings yet
- BIOLOGY, CHEMISTRY (A Tiny Bit of Physics) MinutesDocument11 pagesBIOLOGY, CHEMISTRY (A Tiny Bit of Physics) MinutesCarlos OrtizNo ratings yet
- a40570bb-d279-423e-9cdb-8703c47f18cf (1)Document2 pagesa40570bb-d279-423e-9cdb-8703c47f18cf (1)Ashish SharmaNo ratings yet
- Transport of O2 and Co2Document22 pagesTransport of O2 and Co2JaclinNo ratings yet
- Unit 6 Notes - Gas Exchange and TransportDocument36 pagesUnit 6 Notes - Gas Exchange and Transportshrilpatel2001No ratings yet
- Subject ChemistryDocument13 pagesSubject ChemistrykottooranjohnbNo ratings yet
- CC2 - Lec9 1Document5 pagesCC2 - Lec9 1Glecy PaciaNo ratings yet
- Chapter 9 The Respiratory SystemDocument56 pagesChapter 9 The Respiratory SystemNurarief AffendyNo ratings yet
- 09 Oxygen CurvesDocument3 pages09 Oxygen Curvesramloghun veerNo ratings yet
- Biochemistry Chapter 7-HemoglobinDocument7 pagesBiochemistry Chapter 7-HemoglobinKyle MinorNo ratings yet
- Uas Fishew AminDocument31 pagesUas Fishew Aminwahyu amin pratiwiNo ratings yet
- Transport of Oxygen and Carbon DioxideDocument22 pagesTransport of Oxygen and Carbon DioxideSajid Nadaf100% (1)
- Lab Series Blood Gas Analysis and Fundamentals of Acid Base BalanceDocument4 pagesLab Series Blood Gas Analysis and Fundamentals of Acid Base Balanceyuta titeNo ratings yet
- ABG Analysis in Clinical Setting: Rajesh Mahajan, Suman SethiDocument5 pagesABG Analysis in Clinical Setting: Rajesh Mahajan, Suman SethiLord GrimmNo ratings yet
- HB Function and SynthesisDocument4 pagesHB Function and SynthesisSubhasish BarikNo ratings yet
- Refractometry: Theory & Principle Instrumentation Applications DisadvantagesDocument16 pagesRefractometry: Theory & Principle Instrumentation Applications DisadvantagesVanessa Lim100% (1)
- Lecture 3 Notes - EnzymologyDocument9 pagesLecture 3 Notes - EnzymologyYi Gong100% (3)
- USP Chapter 645 ConductivityDocument4 pagesUSP Chapter 645 ConductivitySulistiaNo ratings yet
- Ketolaetal - Bioresources13432 49700 1 PBDocument25 pagesKetolaetal - Bioresources13432 49700 1 PBKaroline TavaresNo ratings yet
- Nitrosamines Science Based StrategyDocument15 pagesNitrosamines Science Based StrategyJaya AbrahamNo ratings yet
- Cellucap Licorice CombinedDocument3 pagesCellucap Licorice Combinedrafaeldelperu1982No ratings yet
- Corrsion AssessmentDocument27 pagesCorrsion Assessmentanilsmg09No ratings yet
- Kinetics & ThermodynamicsDocument36 pagesKinetics & ThermodynamicsHabibNo ratings yet
- Iso 4892-1Document20 pagesIso 4892-1ERDA TPI REPORTNo ratings yet
- Is Matter Around Us Pure? Separation TechniquesDocument15 pagesIs Matter Around Us Pure? Separation TechniquesAradhya RaiNo ratings yet
- Financial Statement ProjectDocument96 pagesFinancial Statement ProjectJhansi PasupulatiNo ratings yet
- Screenshot 2022-07-20 at 3.22.45 PMDocument22 pagesScreenshot 2022-07-20 at 3.22.45 PMSURYANSH SINGH 12-GNo ratings yet
- Ayesha JavedDocument39 pagesAyesha JavedArshad MadniNo ratings yet
- Astm F2328M 14Document5 pagesAstm F2328M 14Farhad MoravvejNo ratings yet
- Plastic CompoundDocument2 pagesPlastic Compoundstuart2333maleNo ratings yet
- Tech Doc Def Iso22241 Jul2022Document1 pageTech Doc Def Iso22241 Jul2022Wael MohammedNo ratings yet
- Activity On Chemical ReactionsDocument2 pagesActivity On Chemical Reactionsiyahh5.comNo ratings yet
- Granato 2018Document41 pagesGranato 2018An Bùi ĐứcNo ratings yet
- Tip Dan Trik Publish q1 q2Document65 pagesTip Dan Trik Publish q1 q2edward ngiiNo ratings yet
- Mud Solids AnalysisDocument1 pageMud Solids AnalysisstevebeardsleyNo ratings yet
- Form 2 8 Ionic BondingDocument64 pagesForm 2 8 Ionic BondingOsmany Madrigal100% (1)
- (Radiochimica Acta) Sonochemical Dissolution of Metallic Plutonium in A Mixture of Nitric and Formic Acid PDFDocument8 pages(Radiochimica Acta) Sonochemical Dissolution of Metallic Plutonium in A Mixture of Nitric and Formic Acid PDFSutanwi LahiriNo ratings yet
- Topic 6 Reversible Reactions MCQ Questions Copy Aoudi 2023Document9 pagesTopic 6 Reversible Reactions MCQ Questions Copy Aoudi 2023yamanfortnaimatNo ratings yet
- Astm D473-07 (2017)Document6 pagesAstm D473-07 (2017)Camilo Alfonso TorresNo ratings yet
- Determination of The Aromatic Content and Polynuclear Aromatic Content of Diesel Fuels and Aviation Turbine Fuels by Supercritical Fluid ChromatographyDocument6 pagesDetermination of The Aromatic Content and Polynuclear Aromatic Content of Diesel Fuels and Aviation Turbine Fuels by Supercritical Fluid ChromatographyahmedNo ratings yet
- Silica Sulfuric Acid An Efficient and Reusable Catalyst For The One-Pot Synthesis of 3,4-Dihydropyrimidin-2 (1H) - OnesDocument3 pagesSilica Sulfuric Acid An Efficient and Reusable Catalyst For The One-Pot Synthesis of 3,4-Dihydropyrimidin-2 (1H) - OnesDuy Phuc LeNo ratings yet