Professional Documents
Culture Documents
DWSS MODULE 5 Water Supply Systems Module
DWSS MODULE 5 Water Supply Systems Module
Uploaded by
GilbertOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
DWSS MODULE 5 Water Supply Systems Module
DWSS MODULE 5 Water Supply Systems Module
Uploaded by
GilbertCopyright:
Available Formats
Module
Postgraduate Diploma in Water Supplies & Sanitation 4
WATER SUPPLY
SYSTEMS
Institute Of Water And Sanitation Development
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
POST GRADUATE DISTANCE DIPLOMA IN WATER
SUPPLIES AND SANITATION
MODULE
WATER SUPPLY SYSTEMS
Copyright 2001 by: Institute of Water and Sanitation Development
7 Maasdorp Avenue
Alexandra Park
Harare
Zimbabwe
All rights reserved. No part of this module may be reproduced in any form or by any
means without permission in writing from IWSD.
Institute of Water and Sanitation Development ii 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
ABOUT THE COURSE
This course is aimed at participants working in the water and sanitation sector within the
Southern African Region. The course has been designed for engineers, environmental
health professionals, social scientists in management, planning and administration in the
related field.
The basic assumption is that participants attending the diploma course are busy
professionals who cannot be away from their work places for long periods of time. This
therefore means that there already exists some knowledge and experiences. Furthermore
given the ever-increasing costs of attending courses, long drawn out courses are not
affordable to most individuals. It is against these assumptions that this diploma is designed
as a distance course aiming at both quality and quantity in a short space of time.
The course is divided into a number of modules with each module having specific objectives and
expected outputs. The modules are divided as follows:
• Water supply systems
• Community Participation and Management
• Monitoring and evaluation
• Planning and management
• Waste water Excreta Disposal Systems
• Health and epidemiology
• Project
Institute of Water and Sanitation Development iii 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Instruction to the reader
How does this distance course work?
The training course allows you to progress at your own pace and to study the material when and
where you prefer. The module is written in self-instructional style. It is designed to have you teach
yourself, with a minimum time and effort on your part.
As you work through this module, it is important that you follow instructions. After going through
some paragraphs, you will often find questions relating to the information you will have read. Be
sure to answer these questions, as they are designed to ensure that you understand the topic.
How long does it take to complete the module?
The time that is required to complete the module varies with individual and course. You set your
own pace. We advise that you establish a regular schedule – one or two evenings a week or one
morning on the weekend. We would be very interested to know your time demand for the self-
studying.
What should I do when points are unclear?
Find a buddy for learning! It is easier and more efficient for proceed learning in a group. First read
the module alone. Afterwards sit together with you buddy and discuss the session, which you
would have worked through. Learning in a group also is more fun than learning in isolation!
If you are not able to solve questions in the learning group you are free to contact your facilitator for
assistance.
If you make an appointment to see your facilitator, prepare your questions in advance. It would be
convenient for both you and facilitator. If you could send your questions in advance the facilitator
might identify other participants with the same problems as you and set aside a day when more
people can benefit. This will give you better support and can also open up networking chances with
other participants.
Institute of Water and Sanitation Development iv 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
TABLE OF CONTENTS
1. SECTION I - WATER RESOURCES .....................................................................................1
1.1. THE HYDROLOGICAL CYCLE..................................................................................................................... 1
1.2. WATER SOURCES ........................................................................................................................................... 4
1.2.1. Rainwater ........................................................................................................................................................ 4
1.2.2. Surface water .................................................................................................................................................. 4
1.2.3. Groundwater ................................................................................................................................................... 5
1.2.4. Water Source Selection Considerations.......................................................................................................... 5
1.3. INTAKES............................................................................................................................................................ 9
1.3.1. Methods of getting water to the plant ............................................................................................................. 9
2. SECTION II - WATER QUALITY .........................................................................................15
2.1. WATER QUALITY CONSIDERATIONS ..................................................................................................... 15
2.2. CHARACTERISTICS OF WATER................................................................................................................. 16
2.2.1. Physical Characteristics ................................................................................................................................ 16
2.2.2. Chemical Characteristics Of Water .............................................................................................................. 18
2.2.3. Biological Characteristics ............................................................................................................................. 19
2.2.4. Microbiological Characteristics.................................................................................................................... 19
2.3. WATER SAMPLING....................................................................................................................................... 20
3. SECTION III - PLANNING WATER SUPPLY SYSTEM .....................................................22
3.1. PLANNING GUIDELINES ............................................................................................................................. 22
3.2. WATER DEMAND.......................................................................................................................................... 23
4. SECTION IV - WATER TREATMENT .................................................................................27
4.1. THE NEED TO TREAT WATER.................................................................................................................... 27
4.2. PRINCIPLES AND PROCESSES ................................................................................................................... 27
4.3. PRE-SEDIMENTATION ................................................................................................................................. 28
4.4. COAGULATION AND FLOCCULATION.................................................................................................... 28
4.5. INTRODUCTION............................................................................................................................................. 29
4.5.1. Purpose Of Coagulation And Flocculation................................................................................................... 29
4.5.2. Nature Of Particles In Water......................................................................................................................... 30
4.5.3. The Chemistry Of Coagulation..................................................................................................................... 32
4.5.4. Primary Coagulants....................................................................................................................................... 34
4.5.5. Factors Affecting The Coagulation Process ................................................................................................. 40
4.5.6. FLOCCULATION ....................................................................................................................................... 41
4.6. CLARIFIERS / SEDIMENTATION BASINS. ............................................................................................... 44
4.7. FILTRATION ................................................................................................................................................... 45
4.7.1. Slow Sand Filters: ......................................................................................................................................... 45
4.7.2. Rapid Gravity Sand Filters: .......................................................................................................................... 46
4.7.3. Pressure Filters:............................................................................................................................................. 48
4.8. DISINFECTION OR STERILISATION.......................................................................................................... 49
4.8.1. Chlorination .................................................................................................................................................. 50
4.9. OTHER TREATMENT PROCESSES............................................................................................................. 53
4.10. OPERATION AND MAINTENANCE FOR TREATMENT PLANT........................................................... 54
4.10.1. Maintenance.................................................................................................................................................. 54
5. SECTION V - WATER SUPPLY SERVICES.......................................................................58
5.1.1. STORAGE RESERVOIRS .......................................................................................................................... 58
5.1.2. Pumps - types, choice and maintenance ....................................................................................................... 60
6. ANSWERS SESSION ...........................................................................................................63
Institute of Water and Sanitation Development v 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
SECTION I - WATER RESOURCES
Objectives
At the end of the session you are expected to be able to;
• Define and describe the term ‘hydrological cycle’
• Discuss advantages and disadvantages or different sources of water
• Define the term ‘aquifer’ and describe at least four different aquifers
• State the purpose of an intake
• Describe the different intakes for different sources of water
1.1. THE HYDROLOGICAL CYCLE
To understand water resources you need to know about hydrology and its links to sources of water.
Hydrology is a science that deals with the occurrence and distribution of water in both space and
time, taking into account its chemical and physical characteristics and also its interaction with the
environment.
The term hydrological cycle refers to the continuous movement of water from the earth’s surface to
the atmosphere and back again through natural processes. Note that the definition has two
important terms i.e. ‘continuous’ and ‘natural processes’
The various hydrological processes can be summarised as follows:
Precipitation -a product of water vapour condensation in the atmosphere delivered to earth as
solid (snow or hail) or liquid (rain)
Interception -precipitation falling on or retained by vegetation
Infiltration -the movement of water into the soil
Percolation -vertical movement of water through soil in the aeration zone
Seepage -movement of water from one medium into another (from soil into a dam)
Surface runoff-rainfall water which travels over earth’s surface which discharges into streams rivers
and lakes.
Evaporation -loss of water from the earth’s surface
Transpiration-loss of water from plants
Institute of Water and Sanitation Development 1 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Let us discuss the hydrologic cycle in more detail. The earth's water is constantly circulating from
atmosphere to ground to atmosphere again by a process called the hydrologic cycle. This chart
shows what happens in the hydrologic cycle.
Institute of Water and Sanitation Development 2 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Whenever water is exposed to the atmosphere, the
sun causes it to evaporate and rise as a water vapour.
Moisture is also given off to the air by plants. This is
called transpiration. For example on a hot day a large
fig tree may transpire or give off as much as a four
litres of water a minute.
The moisture in the air forms clouds. When the clouds
cool, the water vapour condenses or changes into
water droplets or snow. When heavy enough, the rain
or snow precipitates or falls to the ground. The largest
part of this water returns to the air through evaporation
and transpiration.
Although it will depend upon soil conditions, generally about 10% of the water that falls to the
ground will soak into the ground. The rest is taken up by oceans, lakes and rivers.
Basically three things can happen to water that
infiltrates or soaks into the soil.
1. The water may be pulled back to the surface by
capillary force and be evaporated into the atmosphere.
2. The water can be absorbed by plant roots growing
in the soil and re-enter the atmosphere by
transpiration.
3. Water that has infiltrated the soil deeply enough
may be pulled on downward by gravity, until it reaches
what is called the zone of saturation. This is the ground
water reservoir that supplies water to wells. (In this
zone of saturation, the spaces or openings between
the particles in the soil are completely filled with water.)
Ground water can flow in the saturated zone until it is
exposed to the ground surface and evaporate into the
atmosphere. The hydrological cycle can start again.
Question 1
Describe the hydrological cycle in full.
(Answer on next page)
Institute of Water and Sanitation Development 3 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
1.2. WATER SOURCES
Water sources relate to the basic phases of the hydrological cycle of rain, surface and
groundwater. These can be further categorized as follows:
Rainwater: roof, ground and rock catchments
Groundwater: wells, boreholes, springs, sand abstraction from riverbeds
Surface water: streams, rivers and lakes
1.2.1. Rainwater
Rain water from roofs has the advantage that it is available almost everywhere (close to users),
requires neither pumping nor abstraction works and is relatively pure. However the following
observations are worth noting:
many homes are roofed with thatch, a material which contributes to the deterioration of the
rainwater quality
rainfall patterns may require large and expensive storage tanks for guaranteed year round supply.
Hence this source option may be suitable only with reliable rainfall all year round or where other
sources are not available.
air pollution may lead to unacceptable quality.
Answer for 1
Your answer is correct if you;
you started and ended with the same process. (If you started from evaporation you should
have ended with evaporation.)
described all processes that take the water up
described all the processes that bring down water (precipitation)
stated what happens when water reaches the ground
described what happens to the water that has infiltrated the soil tracing it until it is released to
the atmosphere.
1.2.2. Surface water
Surface water has the following characteristics:
• it is readily available
• availability is easy to determine without sophisticated surveys
• is easy to abstract
• is easy to measure
• can be very polluted (faecal pollution – in streams, lakes and ponds), therefore extensive
treatment is normally required, since its use may be unavoidable. (Sea water contains
3500mg/l of chlorides. - Desalination is expensive)
• normally requires extensive pumping
• it is prone to large evaporation
• It compete for land use
Institute of Water and Sanitation Development 4 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
1.2.3. Groundwater
Groundwater exhibits excellent parameters for utilization the major ones being:
• it generally has good quality, and is bacteriologically safe
• normally available irrespective of season
• no evaporation losses
does not compete for land
• can be close to consumers normally
• preferable if it can be used for domestic supply with reasonable ease since it is purified by
the filtering action of soil
• predominant source of water in rural areas
Its disadvantages include the following:
• might contain high levels of iron, manganese, salt, fluoride, and other undesirable
parameters which can make its utilization undesirable or unpleasant.
• it is difficult to determine available quantities
• sophisticated equipment might be required to determine its location
• pumping is sometimes required
• moderate to high construction costs are normally involved
A well next to a surface water source may give fresh water and is to be preferred. For a village,
groundwater quantities from a well or a spring may be sufficient.
1.2.4. Water Source Selection Considerations
Priority System
1st No treatment and no pumping
2nd No treatment but pumping
3rd Treatment but no pumping
Generally the selection of a water source should
take into account the demand, quality, sanitary,
development, operation and maintenance
considerations. The least cost opinion should
require neither pumping nor treatment. Springs
should be the first option if these are available and
reliable since there is no requirement for pumping
or treatment. Some form of protection is essential
to prevent pollution by surface water and harmful
bacteria. After natural springs groundwater would
be the most appropriate, particularly for low cost
solutions like shallow or medium depth wells.
In cases where a village has no reliable year-round
source, a more distant source may be supplemented by a wet season well. This would have a
major impact as it would be during the period of greatest disease incidence and peak hour
demand.
Institute of Water and Sanitation Development 5 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Since treatment plants are generally unreliable under rural conditions, the best sources are those
which do not require any treatment.
Examine the diagram below. In this diagram we have combined information presented in the past
few pages.
This is an example of a ground water reservoir in a porous overburden material. Note that the
consolidated rock acts as a confining layer beneath this natural reservoir. The aquifer is a reservoir
because it is porous and will yield water to a well or spring.
This type of aquifer is called a water table aquifer. A water table aquifer does not have an upper
confining layer, and the water in the well will rise only to the water table - which coincides with the
top of the aquifer.
There are other types of aquifers, which can be used as a source of water. The diagram below
illustrates an artesian aquifer, and wells tapping this source of water are called artesian wells.
These can be flowing or non-flowing wells, depending on the relationship of the well top and the
aquifer water pressure. In artesian aquifers, the water is under pressure and will rise above the top
of the aquifer.
Institute of Water and Sanitation Development 6 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Another type of aquifer is the perched aquifer. These aquifers are usually very small and represent
infiltrating water trapped above the true zone of saturation.
We have been talking about aquifers and conventional vertical wells as sources of water.
At this point, since we are talking about sources of water, we should also mention infiltration
galleries.
An infiltration gallery is essentially a well that collects water through horizontal pipes. Such
galleries are laid in porous material near a body of surface water or even beneath a lake or river.
On the next page is a diagram to show what is meant by ‘infiltration gallery’.
An aquifer has two important functions:
-it stores water, and
-it conducts water, like a pipeline.
Both of these functions result from the use of openings or pores in the water bearing formation.
That is, the capacity of the reservoir has to be considered in terms of the space available between
particles of the aquifer formation that can be filled with water.
Institute of Water and Sanitation Development 7 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
QUESTION: Would you expect a higher yield of water from a clay, or a gravel aquifer?
ANSWER: The openings between the gravel particles are larger, so you would expect it to be
the higher yielding aquifer. More water can be stored, and water can easily move through the
larger openings.
Generally, with larger openings between the particles in the aquifer formation, more water can be
stored, and water can move more easily through the openings. (What actually happens also
depends on other factors, like grain or particle size, and grading or uniformity of these particles.)
There are special names given to these two functions of the aquifer.
Porosity is the term used to refer to the amount of water an aquifer will hold. (This is not
necessarily the amount of water that you can obtain from an aquifer.)
Permeability of a water-bearing formation refers to the ease with which water can move through a
geological formation to a collection point.
Wells, boreholes and aquifer water levels
When a well is pumped, the water level in the well falls below the water level normal to the rest of
the aquifer. Water immediately begins to flow from all directions toward the well. As a result, the
water surface in the aquifer takes the shape of an inverted cone or curved funnel. This cone is
called the cone of depression. This diagram shows what we mean:
If the material of the aquifer transmits water easily, the cone of depression is flat and wide-spread.
If it transmits poorly, the cone will be steep. Also, this cone does not have a fixed shape. Within
minutes, it becomes deeper and flatter as the well is pumped. Ongoing measurement of this cone
is an important aspect of proper well operation.
Institute of Water and Sanitation Development 8 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
1.3. INTAKES
The purpose of an intake is to draw the highest quality water from a source. As will be discussed
later there are so many different types of intakes.
Structural considerations
gravity pipelines should have self cleansing velocities, i.e. at least 0.7m/s
the suction pipe should be designed for very low velocities to reduce friction loss and indraw of
sediments. Regular cleaning by backwashing is essential
a non-return valve, installed at strainer is essential
since the pipeline may contain air an anchor or come amount of preloading essential
A strainer mounted on the inlet should be installed to prevent debris, leaves, etc from entering.
This should preferably be located at least 1m below water level and at least 1m above the bottom.
In cases of turbulent flow a removable screen is required to protect the strainer. This should be
raked regularly.
Lake and dam intakes should preferably be at least 100m from the shore in lakes with bilharzia.
Underwater pipes should be flexible through the pipe material or joints.
Spring intakes need protection against contamination from surface water and animals. A drainage
ditch should be erected to divert any surface water above the spring site. Other essential
components are air vents, overflows pipes, access cover and scour pipes.
The intake is the first element of a water supply system wherever the source of supply is surface
water. The source of supply is usually a lake or river, and an intake structure is placed in the
surface water source to withdraw water.
The two basic things that will have been considered when the point of the water intake and the
source of the water supply were decided upon are;
1. quantity of water
2. quality of water
Obviously, the system must tap a source of water that will be continuously adequate in quantity to
satisfy present and reasonable future demands. Many rivers and lakes are used in this way.
1.3.1. Methods of getting water to the plant
There are cases where the elevation of the water source will allow movement of the water to the
plant by gravity.
More often, though, a pumping station will be required, as shown in this diagram:
Institute of Water and Sanitation Development 9 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Besides quantity of water, we are also concerned about the quality of water obtained from the
water source. To a large extent, this will be controlled by where the intake is positioned and by the
intake structure itself.
The intake will be positioned to avoid sources of pollution, like the community’s wastewater and
industrial wastes. This usually means that the intake will be placed as far from shore as
economically feasible, to take advantage of deep water.
The intake structure will provide necessary protection against debris, ice, prevailing winds, and
surface and subsurface currents. A buoy is often used to warn water traffic.
The position and design of the intake also has to take into account the turbidity of the water
collected. This refers to the amount of sand, silt, and other sediment that comes in along with the
water.
As you can see, the positioning and design of the intake can be complicated. Often lengthy studies
are required to determine the best location and design of the intake.
Let's look at the common types of intake structures.
Here is a diagram of a typical intake structure for a lake or other source of 'still' water.
Institute of Water and Sanitation Development 10 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Notice that the cover and the sidebars help prevent larger objects from entering the intake. The
typical intake structure used for moving waters like rivers, is somewhat different.
Institute of Water and Sanitation Development 11 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
This diagram shows a typical river intake.
Notice the ‘bell shaped’ intake. The lip of the intake is usually curved out to prevent debris and
sediments from entering the intake. A number of these structures could be used to provide water to
the intake pipe.
Notice too that the opening to the intake faces down-stream. Why?
________________________________________________________________________
________________________________________________________________________
________________________________________________________________________
________________________________________________________________________
______________________
Other structures that can be used are as follows.
Your intake structure may be as simple as a surface diversion, a small concrete structure
containing a slide gate as shown here
Institute of Water and Sanitation Development 12 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
When the slide gate is removed or raised, water spills into a canal, ditch or pipeline, which then
carries the water to the treatment plant.
This type of structure is sometimes used to divert water from small lakes and streams. Another
type of intake structure that could be used, specially for larger installations, is called an intake
tower.
A diagram of a intake tower is as shown here:
Institute of Water and Sanitation Development 13 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
An intake tower is usually provided with multiple inlets. This allows the operator to draw water from
different depths to compensate for changing water surface elevation to select the depth from which
to withdraw water of the most favourable temperature and quality.
Temperature Stratification
Temperature stratification is more significant in summer in dams that are more than 8 m deep.
The upper layer is warmer that the lower level layers. The water in this layer is continually
circulated through the action of wind and temperature. Dissolved oxygen content is often close to
saturation. Water from this layer is usually the easiest to treat.
The next layer is the middle layer, which is one to two metres thick. The temperature in this zone
decreases with depth until the lower layer. Oxygen level in the middle layer is lower than in the
upper layer but the quality of the water might be better. It is always advisable to seek a chemist’s
advice to determine the depth at which one should extract water.
The organic matter that settles out of the first two layers accumulates in the lower layer. The
substances are decomposed by bacterial action thereby using up oxygen in this layer, causing
anaerobic conditions to develop. The remaining organic matter is decomposed anaerobically
producing soluble organic matter and insoluble forms, which are difficult to remove in a treatment
works. It is never advisable to extract water from this layer.
When the water level in the reservoir increases or decreases the level of the take off point should
be changed accordingly. A chemist will be of help to determine the take-off point to be used.
During winter “turn over” might occur. This happens when the upper layer is cooled suddenly to
temperatures below the lower layer. The upper layer becomes the denser layer and sinks to the
bottom causing the turnover. This results in deoxygenated water at the surface and this can cause
large fish kills.
Final exercise for Session 1
1. State the three things that happen to water that has infiltrated the soil.
2. State any advantages of ground water
3. State the two factors that affect siting of an intake.
Institute of Water and Sanitation Development 14 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
SECTION II - WATER QUALITY
Objectives
At the end of the session you should be able to;
• State the characteristics of water
• Explain why coliforms can be used as indicators of pollution
• State factors that affect sampling frequency
1.4. WATER QUALITY CONSIDERATIONS
Water is essential for human life and many other uses. Each use has its own quality requirements
as shown below.
Fishing limited organic matter, suspended solids and turbidity, toxic materials, oil and
grease plus nutrients
Navigation free from floating masses
Power generation as for navigation plus limit on suspended solids
Amenity limit on organic content (biochemical oxygen demand), oil, grease, etc (appearance
only)
Recreation as for Amenity
Irrigation limit on dissolved salts, boron, heavy metals (cadmium, mercury, lead, nickel, iron,
manganese, chromium, copper)
Industrial varies from treated to raw, but generally use drinking quality
Water for human consumption should be;
- potable – fit and safe for human consumption
- palatable – aesthetically attractive
The water should also be free from
-visible suspended matter
-excessive colour
-taste and odour
-objectionable dissolved matter
-aggressive constituents (acidic or corrosive)
-bacteria indicative of pollution
Water from any natural source is likely to contain impurities from one or more of the following:
- suspended inorganic matter e.g. dirt or grit
- dissolved inorganic matter – chemical elements & compounds
- micro-organisms – bacteriological contamination
Surface Water
Surface water constitutes most of the water used in Southern Africa region for urban supplies. It is
likely to contain suspended matter especially during periods of rainfal flow. It is more prone to
pollution from
Institute of Water and Sanitation Development 15 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
- animal and human excreta
- industrial effluents
- agricultural pollution
Groundwater
- there is a possible pollution from seepage from septic tanks, toilets, dumping sites,
etc if these are too close
- very hard water aquifer is situated in limestone (or other calcareous rock)
- may have high saline content
Rainwater
-air pollution from chimneys, exhausts, acid rain, etc is possible
1.5. CHARACTERISTICS OF WATER
Clean surface water or potable water is increasingly becoming scarce, mainly due to more
intensive industrialisation, urbanisation and commercial agriculture. It is becoming clearer to more
people that clean water is a limited resource which should be continuously recycled if development
is to continue without being limited by the quality of available water. The quality of water can be
described by its chemical, physical, bacteriological and biological characteristics.
1.5.1. Physical Characteristics
Temperature, tastes and odours, colour, turbidity, solids, electrical conductivity, density and
viscosity are some of the physical characteristics.
Temperature
The importance of temperature basically arises from its effect on other properties of water e.g.
chemical and biological reactions are speeded up by higher temperatures. Higher temperatures
accentuate tastes and odours. The solubility of gases for example of oxygen is reduced. Hence
higher temperatures result in lower oxygen content. Ideally water temperature should be a few
degrees below ambient temperature.
Tastes and Odours
These are due to dissolved impurities, normally organic in nature e.g. phenols from industrial
effluents. Other causes are excessive chlorination at the treatment plant and the presence of
algae. Water is classified as objectionable or unobjectionable. “Taste” problems in water supplies
constitute the major consumer complaint. Changes in the normal taste of a public water supply
may be due to changes in the quality of the raw water source or deficiencies in the treatments
process. Tastes and odours are subjective properties and are thus difficult to measure. The
guideline criterion is “not offensive to most of the consumers”.
Colour
Most natural waters have a colour, especially water from upland areas. Even pure water has a
pale green-blue tint in large volumes. The colour may be harmless but consumers drawing water
Institute of Water and Sanitation Development 16 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
from a piped water scheme may find it objectionable on purely aesthetic grounds. Colour can be
an indication of feacal pollution. Coloured water may be unacceptable for certain industrial
processes like high-quality paper production or clothing industries. There is need to distinguish
between true / natural colour and apparent colour. True colour is due to absorption of light arising
from the presence of products of organic decomposition particularly in streams rising upland
swamp catchments. Apparent is due to substances in solution or suspended matter.
Colour is measured by colour discs (visual comparison by colour comparator) or spectrometer. .
Turbidity
Turbidity is the cloudy appearance of water due to presence of colloidal particles which are in
suspension. This arises from the effect of light scattering caused by suspended solids. This is
aesthetically unattractive & may be harmful.
Turbidity in water can be due to clay and silt particles; discharges of sewage and industrial waste;
or the presence of large numbers of micro-organisms. The significance is not only aesthetic but is
important also in purification by disinfection and slow sand filtration, since high levels of turbidity
can protect micro-organisms from the effects of disinfection, stimulate bacterial growth and exert a
significant chlorine demand. So where disinfection in practised turbidity must always be low,
preferably below 1NTU.
Turbidity is measured by the Tube turbidimeter which is useful for field measurements, the
spectrophotometer or the turbidimeter which is useful for raw water. The WHO guideline is 5
neophelometric turbidity units (NTU) or 5 Jackson turbidity units (JTU). Turbidities above 5 NTU (5
JTU) may be objectionable to consumers. For higher turbidities coagulation, sedimentation &
filtration are essential.
Solids
These may be present in suspension and / or in solution and are either organic or inorganic matter.
Total dissolved solids, TDS, are due to soluble matter. Suspended solids, SS, are discrete
particles which can be measured by filtering a sample through a fine paper. These are removable
by filtration through such a fine paper. Settleable solids are those that settle out readily in a
graduated cone after standing for a fixed period, usually 2 hours. This standard settling procedure
is useful in measuring the performance of sedimentation units.
Electrical Conductivity
The electrical conductivity (EC) of water depends on the quantity of dissolved salts present. For
dilute solutions the conductivity if roughly proportional to the TDS content. Hence the conductivity
of a solution is a quick indicator of TDS content. It is also useful in measuring salinity (salt content).
Measurement is by using a conductivity meter.
Density
This is the mass per unit volume of a substance. For fresh water this is 1000 kg/m3. For sea water
this is 1025 kg/m3.
Viscosity
Institute of Water and Sanitation Development 17 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
This is the ability to resist change in the arrangement of molecules
1.5.2. Chemical Characteristics Of Water
pH
This is a measure of hydrogen ions present [H+] and is important in that it controls several chemical
and biological reactions including chlorination.
pH = -log10[H+]
It is a measure of the degree of acidity or alkalinity of a sample. The measurement is achieved
using a scale from 0 to 14 categorized as follows:
pH of 7 is neutral
pH below 7 is acidic and water is bitter
pH above 7 is alkaline
Highly acidic or alkaline waters are undesirable because of corrosion hazards and difficulties in
treatment.
Alkalinity
This refers to the buffering capacity to resisting change in pH. This is due to the presence of
carbonates, bi-carbonates or hydroxides of e.g. Ca, Mg. A little bit of alkalinity preferred. Alkalinity
is measured by titration with acid using methyl orange or phenolphthalein
Acidity
Acidic water is corrosive and reduces the life of pipes and pumps and results in a bitter taste.
There are two forms;- carbon dioxide acidity and mineral acidity:
CO2 acidity due to natural CO2 or acid rain, or the decomposition of organic matter.
Mineral acidity indicates the presence of industrial wastes in water. Acidity is expressed in terms of
mg/l (of CaCO3)
Hardness
This is the property of water that prevents lather formulation with soap and produces scale in hot
water systems. There is no health hazard but is undesirable (heart disease with very soft water).
Hardness is due mainly to the presence of metallic ions: Ca2+, Mg2+; but Fe2+ and Sr2+ are also
responsible. The economic disadvantages of a hard water include:
increased soap consumption
higher fuel costs
There are two forms of hardness: carbonate and non-carbonate
Institute of Water and Sanitation Development 18 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Dissolved Oxygen (DO)
This is the most important element in water quality control and is measured in mg/l. The presence
of dissolved oxygen is necessary to maintain the higher forms of biological life. The effect of a
waste discharge on a river is largely determined by the oxygen balance of the river system. Any
pollution depletes the level of DO. Clean surface water usually is saturated with DO. Saturated
water has a pleasant taste. Low DO leads to an insipid taste as is the case with groundwater
which has recently come up.
Chloride
Chloride is responsible for brackish (salty) water and is an indicator of sewage pollution because of
the chloride content of urine. chlorides are expressed in mg/l.
Nitrogen
Nitrogen is important because biological reactions can only proceed in the presence of sufficient
nitrogen . It occurs in four main forms:-
organic nitrogen – usually from domestic wastes e.g. proteins and urea.
ammonia nitrogen – as ammonium salts of free ammonia.
nitrite nitrogen – an intermediate oxidation state.
nitrate nitrogen – final oxidation product of nitrogen.
The relative concentration of each form gives an indication of the nature and strength of a sample.
Nitrogen content can be used to assess the quality of water prior to the availability of
bacteriological analysis.
1.5.3. Biological Characteristics
It is difficult to give guidelines on biological hazards that are generally applicable since many
parasites have a complex geographical distribution and it may be unnecessary to take precautions
against those not occurring locally. The majority of waterborne parasites are also transmitted by
other routes, e.g. food and direct faecal-oral spread, and these should also be considered.
Therefore where possible use drinking water sources not subject to contamination.
1.5.4. Microbiological Characteristics
Drinking water should not contain any pathogens (disease causing) micro-organisms. It should
also be free from bacteria indicative of excremental pollution. Therefore regularly examine
samples for indicators of faecal pollution. The main bacterial indicator recommended for this
purpose is the coliform group, in particular Escherichia coli. (E-coli).
Coliform bacteria are present in human intestines in large numbers. There are two groups:-
• E-coli which inhabits the intestinal tracts of man and mammals – warm blooded and whose
presence indicates faecal pollution
• The other group is found in many environments. Their presence could be due to coliform
from soil or humus and therefore does not necessarily imply faecal pollution.
Institute of Water and Sanitation Development 19 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
This is made use of because pathogens are difficult to look for. The indicator organisms have the
advantage that they are:-
-non-pathogenic
-restricted to faeces
-present in large numbers, therefore easy to enumerate.
-are as resistant as pathogens to light and chemicals
-easy to detect by simple procedures, (answers in a day compared to at least a week for
the pathogenic organisms).
Chlorination can produce virus-free water from faecally polluted source of waters when the
concentration of free residual chlorine is at least 0,5mg/l for a minimum contact period of 30
minutes at a pH below 8,0 and a turbidity of 1 NTU or less.
It is also desirable to maintain a free residual chlorine level of 0,2 – 0,5 mg/I in the distribution
system to reduce risk of microbial regrowth. Detection of chloride in this range indicates the
absence of post-treatment contamination.
1.6. WATER SAMPLING
The bacteriological testing of water requires sampling to collect and deliver a water sample for
analysis. The sample should be taken so that it is representative of the bulk of the water being
examined. The sampling of the water is therefore as important as the analysis. Precautions
should be taken to avoid any accidental contamination of the sample during sampling and
transportation.
Utilization of the results of bacteriological analysis:
• to check the quality of treated drinking water in the distribution system (at taps and other
outlets)
• to evaluate the contamination of water sources like boreholes, lakes, streams, springs, etc.
Guidelines for water quality tests
• avoid contaminating sampling point
• avoid use of dirty bottles
• use sterilized bottles
• resample in cases where contamination is suspected
• keep samples in covered and cool container and deliver within 24 hours
• properly label the bottles with sample details – location, date, time, etc
• assess chlorine residual on site
• take measures to avoid the risk of infection e.g. bilharzia
Sampling frequency
(WHO Guidelines for drinking water quality)
Sampling should be undertaken as often as possible and to cover as many network points as
possible.
The factors that affect sampling frequency are as follows;
Institute of Water and Sanitation Development 20 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
• Type of water source (Some sources are more prone to pollution than others)
• Use of water (Drinking water requires frequent tests)
• disease prevalence (In case of disease outbreak monitoring is more often)
• Type of water sample
• Season (In the rainy season there are more chances of pollution)
• Risk of contamination (When there is a settlement upstream of the source)
• Resources available to carry out the sampling and tests
Bacteriological Water Testing of water
The purpose of bacteriological testing of water is to detect and determine the concentrations of
faecal bacteria in water supplies since these should be free from pathogens and be safe for human
consumption.
There are two major methods of detecting and measuring indicator bacteria in water – the
membrane filtration method and the most probable number (MPN) multiple tube method.
Procedure for the membrane filtration method
• filter the water sample
• provide bacteria with nutrients
• incubate at 440C in an incubator for 24 hours for growth
• count the individual colonies
• calculate the number of colonies per 100 ml assuming each colony arose from a single
bacterium
Procedure for the most probable number (MPN) multiple tube method.
• add measured volumes of sample to sets of sterile tubes or bottles with suitable liquid
medium
• incubate at 440C; the faecal coliforms produce acid and gas
• detect any evidence of gas production with an inverted glass test tube
• detect any presence of acid using the various pH indicators
• record the number of tubes showing positive reactions
• estimate the most probable number (MPN) of organisms present in the original sample
from statistical tables.
It is essential to use different dilutions to get both positive and negative reactions
Final Question for Session II
1. Name two types of hardness in water?
2. Why can coliform bacteria be used as indicators of quality for drinking water?
3 State and five factors that affect sampling frequency.
Institute of Water and Sanitation Development 21 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
SECTION III - PLANNING WATER SUPPLY SYSTEM
Objectives
At the end of the session you should be able to;
• Explain the planning aspects considered in water supply systems
• Define the term ‘design period’
• State the factors that affect demand
• Define water consumption and water demand
• State the aspects considered in pipe selection
1.7. PLANNING GUIDELINES
Legal aspects include:-
• procedures in getting water rights
• rights to public water
• water servitudes
• water pollution control
• safety of dams
In addition to the above there is the relevant legislation. This gives the relevant requirements,
regulations, responsibilities, etc. regarding – impoundment, abstraction, use, pollution control and
safety of ancilliary structures.
There are various constraints to the development of water supplies. These include:
Physical
-quantity may not be enough in the catchment
-quality – some sources may be ruled out because of poor quality
Financial
Administrative
-may want to protect good areas from being flooded by dams
-best source may lie in another country
Legal
-there are legal restrictions on what one can do (water rights or permits)
A water supply project is normally either a new system or an augmentation, extension or
rehabilitation. The preliminary considerations for a proposed water supply scheme are:
• approximate estimate of cost
• assessments of political, economic and legal feasibility of alternative proposals
• interdepartmental collaboration of government departments for multi-purpose schemes
• inter-government agreement for boundary rivers
• local community considerations
Institute of Water and Sanitation Development 22 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Design period
The design period is the horizon during which a project is expected to meet a community’s
requirements. This depends on the uncertainty of demand and the time it takes to provide the new
source and other ancillliary works. The total water demand is initially calculated for design period.
Design period and Design flows
The design of a water supply system should seek to cater for the expected total population at the
end of the design period. Various aspects need to be considered in estimating this future
population which is determined by the birth / death rate ratio, rural –urban migration and a general
increase in longevity as health services improve.
The design flow should also allow for the higher future population and in particular allow for:
• an increase in per capita consumption during economic life of a system due to improved
living standards and expansion of industry
• cases of unsewered towns where a sudden increase in water requirements often occurs
when a sewerage system comes into service.
However it should be noted that the per capita demand can go down due to greater reuse by
industry, increases in water charges and the installation of individual meters in areas not previously
metered.
1.8. WATER DEMAND
Demand can be defined as the amount of water used when it is freely available, and this includes
losses and unaccounted for water. Supply is the water entering the distribution system.
Consumption rate is the water actually used by a consumer.
Factors affecting demand in a water supply schemes
1. The climate, season, and weather. Usually, water use will be highest in hot, dry
climates, particularly during summer when people are watering lawns and gardens.
2. The type of community. Water use will depend on whether it is used only for
domestic purposes, or for other purposes as well (such as irrigation or industry).
3. The standard of living in the community. Usually, water use per person will be higher
in communities with relatively high standards of living. This is because the water is used
for "luxury" purposes (such as filling swimming pools; operating garbage disposals, air
conditioners, and dishwashers; and the use of more than one bathroom).
4. The day of the week. The day-to-day variation in water use varies depending on the
habits of the community, but within a season the pattern is usually the same,
week after week with only minor variations.
5. The time of day. In most residential communities, water use is lowest during the early
morning hours, rises steadily until noon, stabilises during the afternoon, and peaks during the early
evening.
6. The available water pressure. As water pressure increases, the amount used also
Institute of Water and Sanitation Development 23 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Increases. A pressure of 175 to 350 kPa is considered normal. By increasing a 175 kPa
service to 350 kPa, water use can be expected to increase by as much as 35 percent.
7. Metering. Communities having flat-rate water services often use about 25 percent more
water per person than they would if the services were metered.
8. Sewers. The availability of municipal sewer systems usually increases water use by 50
to 100 percent. This is because people need not be concerned with the capacity of
private disposal systems such as a septic tank.
9. Unusual events (such as a fire or water-main break). Although the total amount of
water used during events such as these is small, the instantaneous flow can be very
large. This can result in a drastic drop in system pressure and rapid depletion of reservoir
storage.
10. Condition of the distribution system. All distribution systems leak, some much
more than others. If the system is in poor condition, the amount of water lost by leakage
can equal or exceed the amount used by consumers.
11. Reservoir level control. All reservoirs are provided with overflows. Water level in
the reservoir can be controlled in one of several ways, such as by means of an altitude
(level control) valve on the inlet line. If the level control system is improperly adjusted, water
may be lost by overflowing. This is sometimes difficult
to detect and may continue for a long time.
12. Water service Level. Less water is used when the sources is a standpipe as compared to
a house connection with multiple taps.
Because of all these variables, it is difficult or impossible to predict what the water consumption of
a particular community will be. As a very general guide, a "typical" Zimbabwean urban or suburban
community (if such a thing exists) will use an average of about 100 litres of water per person per
day.
Demand Forecasting
Extrapolation of historical trends. This requires a good length of record, at least 20 years. It is
accurate but very uncertain, very risky and prone to large errors. It is therefore suitable for the
short term.
For medium to long term, examine per capita consumption since city boundaries can change.
Divide demand into components
-power (cooling)
-institutional
-residential
-commercial
-industrial
-agricultural
-rural (domestic)
Institute of Water and Sanitation Development 24 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Variations of demand
Water consumption varies from hour to hour during the day, from day to day during the week, and
from week to week during the year. All consumption rates are expressed in annual average daily
rates. These various rates are used to fix capacities of the various components of a water supply
system like raw water reservoirs, treatment plant, transmission mains, reservoirs, pumps, etc.
Maximum rates are higher in small community and in predominantly residential than industrial or
rural areas.
Peak factors are used to describe the highest established demands over a given period. As an
example the peak daily demand (factor) is the established daily demand in a year. This is normally
taken as 1,5 times the annual daily average demand. This factor is 1,47 for Zimbabwean cities and
1.61 for Zimbabwean towns.
The peak hourly factor gives the highest hourly demand in any one day. This is 2.0 times the daily
demand. The peak week factor describes the ration of the highest weekly demand in any year and
us 1,3 for Zimbabwean cities and 1,37 for Zimbabwean towns.
In order to allow for the hourly peak during a peak day demand the peak hourly factor becomes the
product of these two, i.e. peak hourly factor = 2,0 x 1,5 x Average Daily
Demand, or 3,0 x average daily demand.
The peak hourly factor allows the reticulation to have the residual head at any point.
Exercise:
Calculate the peak hourly and peak daily demand for a town with the following water requirements
(average daily demand):
Demand M3/day
_________________________________________
Residential 100
Institutional 40
Commercial and industrial 30
Livestock 20
Garden and watering 10
Exercise
Calculate the average daily and peak daily demands given a town of population 60 000 made up of
low, medium and high density residential areas each of which has a third of the population. Per
capita consumption for these areas is 300, 200 and 85 litres respectively.
Pipe Selection
Economics of pipe selection:
The following aspects should be considered:
-pressures
-quality of water to be conveyed
-delivery times to be conveyed
-volume of water to be conveyed
-nature of ground (aggressive, expansive soil)
Institute of Water and Sanitation Development 25 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
-transport and handling costs
-supervision in laying
-cost, availability of pipes and fittings for specific pressures
Standardisation
It is worthwhile standardizing all pipes in your area e.g. local authority
Pump choice
Pumping plant design depends on:
• Water demand, Q
Fluctuating demand on pump and power source is to be avoided
• Pumping head, H
H = sum of (static and friction heads) in system on pump’s pressure side.
Pump applications
-water from wells, boreholes and surface water intakes to treatment plant and reservoir (ground)
-water from source, treatment plant and reservoir to elevated tank or reservoir at a high level
-pumping water and pressure in distribution system (booster station)
Operation and maintenance
In instances where the demand is fast approaching the supply, it may be appropriate to take
measures which assure the consumers continuity of supply. These supply – demand balance
measures include:
Demand modification through price (if metered), education (urge consumers), technological
change (industrial or residential).
Supply changes either by reducing loses or increasing the frequency of shortages.
Alternatively install standpipes the in street and close to house taps.
Increasing sources through surface water, ground water, desalination of contaminated
groundwater, wastewater reuse, conjunctive use (use several source together – shallow
water and ground water), etc.
With groundwater there are far less controls and conditions than with surface water. Before costly
drilling / construction works there is no to engage the assistance of an experienced hydrogeolist to
investigate the area and make recommendations for potential well/borehole sites and their
expected yield.
Compare anticipated yields with consumer demand and the conveyance cost (to consumer).
Where possible, analyse water samples from nearby wells/boreholes for water quality and
treatability.
Institute of Water and Sanitation Development 26 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
SECTION IV - WATER TREATMENT
1.9. THE NEED TO TREAT WATER
The aim of treating water is to turn water from an existing source (raw water) into drinking water
through meeting the basic drinking water quality requirements. The resulting water should be free
from pathogens and toxic substances, have a pleasant appearance and a neutral smell and taste.
Therefore the process should be based on quality of existing water and the use to which the
treated water will be put.
1.10. PRINCIPLES AND PROCESSES
The main treatment consists of physical, biological and chemical processes, mainly:-
- aeration
- sedimentation
- coagulation and flocculation
- filtration and disinfection.
Aeration
Aeration is the addition of oxygen where the natural oxygen content is depleted due to large
amounts of organics. This aids subsequent biological treatment in slow and filtration. Aeration
accomplishes several objectives, among them:-
• removal of dissolved iron and manganese. These are oxidized to nearly insoluble
hydroxide sludges which are removed in a settling tank of by a coarse filter. Groundwater
might have iron and manganese
• Removal of excess carbon dioxide which causes corrosion of metal and concrete surfaces.
• Removal of hydrogen sulphide, methane and other volatile compounds which produce
objectionable taste and colour.
• Temperature reduction.
Aerators
There are various aerators:
• Multiple tray or trickling aerators which consist of inlet, tray, water dispenser with coarse
material, collection basin and outlet. Water dispersed into fine droplets using trays with
wire mesh bottoms. The coarse material increases efficiency.
• Cascade aerators which provide fast and simple aeration. The principle involves passing
water over a lateral series of basins made out of masonry or concrete with a total height of
between 1 to 6 metres. This can suffice for small quantities of iron and manganese. If the
aim is just the addition of oxygen it is sufficient to install a small weir above the downstream
clarifying tank with a perforated feed pipe.
• Diffusion aerators force water into the air through fixed nozzles, setting up large contacts
Institute of Water and Sanitation Development 27 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
surfaces for gas transfer above a settling tank or a filter. These are the most efficient but
they are expensive and complex.
1.11. PRE-SEDIMENTATION
Sedimentation is a simple way of reducing suspended matter and partially, bacteria. This is a
natural lake and river purification process. Water is passed through settling basins or storage
tanks at low and uniform velocities.
Aims and processes in pre-sedimentation:
1. Turbidity reduction
Suspended matter settles out by gravity if its density is greater than that of water
The efficiency of the settling basin depends on the nature of the constituent particles –
shape, size and density. Sand and silt settle easily. Colloidal matter which is held in
suspension by electrostatic forces because of its density needs coagulents for removal.
Laboratory experiments should precede settling tank design to determine the contents of
settleable and non-settleable matter. Contamination by suspended can be prevented by
screening the tank inlets.
2. Reduction of pathogenic organisms
Simple sedimentation has an insignificant removal of pathogens. However two or four
weeks storage reduces bacteria population by 50 – 90% through biological processes.
Storage for more than one month can reduce the viral count.
The extent of purification depends on the severity of the population and on the variety
pollutants. Storage induced contamination (e.g. mosquito breeding due to algae growth)
can be reduced by covering the tank. Schistosoma larvae can not survive two days in
protected storage without snails.
3. Removal of colour
The removal of colour requires very long storage time if unaided by chemicals.
1.12. COAGULATION AND FLOCCULATION
Coagulants assist in the removal of finely dispersed and colloidal particles which cause turbidity
and colour but which cannot be removed sufficiently by ordinary sedimentation. These are
substances which react with water producing a bulky precipitate and are added downstream of any
pre-settling basin.
Institute of Water and Sanitation Development 28 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
1.13. INTRODUCTION
Coagulation and flocculation are two of several steps needed to purify raw water to acceptable
standards. Their purpose is to precondition the raw water so that the efficiency of the next process,
clarification, is greatly improved. In fact, the operation of these three processes coagulation,
flocculation, and clarification - are so interdependent that they are often considered as a single
process.
There are many different types of coagulation/flocculation/clarification systems, but they fall into
two broad categories.
The first, known as the conventional system, uses separate tanks for each of the three
processes. The second combines them in a single tank, in what is called the solids contact
process. Both are in common use world wide although in Zimbabwe we still use the conventional
system only.
1.13.1. Purpose Of Coagulation And Flocculation
All natural waters contain, to a greater or lesser degree, some turbidity and colour. The turbidity is
caused by very finely divided solid particles held in suspension, giving the water a cloudy
appearance. Colour is caused by dissolved and colloidal organic or inorganic material (from algae,
decayed vegetation, or bacteria) or by certain mineral substances. One of the purposes of water
treatment is the removal of as much turbidity and colour as possible.
Almost all surface water treatment plants use filtration (see Session 7) to remove suspended
particles from the water. However, filtration alone will not usually remove enough particles to
produce an acceptable product. This is because many of these particles are so small that they
pass through the pores of the filter medium. And because colour is often caused by dissolved
rather than suspended matter, it may not be removed by filtration.
For these reasons, the water must be pre-treated before it is filtered.
In the coagulation process, certain coagulant chemicals are added to the water. The chemicals,
the water, and the dissolved and suspended matter undergo several chemical and physical
reactions; these will be described later in this Session.
Put simply, the coagulant and the water react to form precipitates (insoluble reaction products)
which appear in the form of fairly large suspended particles. During the formation of the precipitates
(usually called floc) the very small particles of suspended matter in the original water are trapped
within the larger floc particles. The floc particles are relatively easy to separate from the water by
settling or filtration.
The coagulant chemical also reacts with dissolved colour-causing substances and converts them
into an insoluble form.
Coagulation is followed immediately by flocculation. In the flocculation process, the water
Institute of Water and Sanitation Development 29 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
is agitated very gently for a fairly long time (usually about 30 minutes. During this time,
the floc particles grow larger. As they are growing, they are brought into contact with (and
entrap) more particles of suspended matter.
The floc is removed in subsequent treatment stages, leaving a relatively clear and colourless
treated water.
Because coagulation and flocculation are so important to the proper treatment of water, and
because they are both complex processes, they will be discussed in some detail.
1.13.2. Nature Of Particles In Water
To understand the coagulation process, it is necessary to know something about the nature of the
particulate impurities in surface water.
Size Range
The particles in surface waters have a very broad size range. Large particles such as gravel, sand,
and heavy silt can be settled out by slowing the velocity of the water (for example, by passing it
through storage reservoirs or ponds). However, many of the smaller particles still remain. Figure
6.2 illustrates the approximate sizes of some of these.
It can be seen that the diameters of many particles (clays, colour, and viruses, for example) are
less than about 0.001 mm. These very small particles are considered to be non-settleable - that is,
for all practical purposes, they cannot be removed by most direct sedimentation or filtration
processes.
Forces On Particles
The particles suspended in the water are subject to several different forces acting on them
simultaneously. These forces are:
1. Brownian movement. The molecules in water (or any liquid) are always moving randomly.
A particle suspended in the water will be bombarded constantly by these moving molecules.
If the particle is relatively large, the bombardment will have little effect on it. However, if it is
very small, it will tend to be pushed back and forth by the collisions with the water molecules,
and will tend to move in random directions. Brownian movement is one of the reasons that
very small particles will never settle, even in completely still water.
2. Turbulence. All suspended particles are subject to movement caused by turbulence or
currents in the water. The degree to which the particles are affected depends on the velocity of
the turbulence or currents and the size of the particle.
3. Electrical repulsion. A basic rule of physics is that like (equivalent) electrical charges repel
each other. It so happens that almost all suspended particles in water have negative electrical
charges, so they will tend to repel each other and prevent particles from joining together to
form larger particles. One of the functions of the chemicals added during the coagulation
process is to neutralise (cancel) the negative electrical charges on particles.
Figure 3.3.1
Institute of Water and Sanitation Development 30 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Coagulation Process Description
The term coagulation describes the effect produced when certain chemicals are added to raw
water containing slow-settling or non-settling suspended particles. The reactions between the
Institute of Water and Sanitation Development 31 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
chemicals, the water, and the suspended particles result in the formation of relatively large particles
which can be removed from the water by sedimentation and filtration.
The mixing of the coagulant chemical and the raw water is commonly referred to as rapid mixing or
flash mixing. The main purpose of mixing is to distribute the coagulant chemical quickly and evenly
in the water. The actual coagulation reaction itself occurs in a very short time (a fraction of a
second) so good mixing and high turbulence is needed if all the water is to receive some of the
chemical.
Coagulant Chemicals
There are two types of chemicals used in the coagulation process.
Primary coagulants are the chemicals which are responsible for the main coagulation reactions -
formation of a floc and neutralisation of particle charges. Coagulant aids (sometimes called
flocculant aids) are used to increase the density of slow-settling floc particles, or to strengthen them
so that they do not break up easily during settling and filtration.
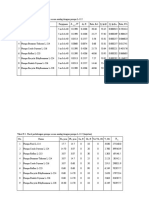
Table 3.5.1 lists the coagulants most commonly used in water treatment.
TABLE 3.5.1 COAGULANT CHEMICALS USED IN WATER TREATMENT
Chemical Chemical Primary Coagulant
Name Formula Coagulant Aid
Aluminium sulphate Al 2(S04).14H20 ¬
Ferrous sulphate FeSO4 7H20 ¬
Ferric sulphate Fe2(SO4)3.9H20 ¬
Ferric chloride FeCl3.6.H20 ¬
Cationic polymer Various ¬ <
Calcium hydroxide Ca(OH)2 ¬ <
Calcium oxide CaO ¬ <
Sodium aluminate Na2Al204 ¬ <
Bentonite Clay <
Sodium silicate Na2SiO3 <
Anionic polymer Various <
Nonionic polymer Various <
1.13.3. The Chemistry Of Coagulation
Because several different chemical substances contribute towards hardness in water, it is
convenient to convert them to a “common denominator” when discussing the chemistry of
coagulation . When this is done, these different substances can be compared to each other directly
and calculations are made much easier.
We are therefore going to consider the concept of equivalent weight.
By convention, hardness in water when discussing or calculating will be considered as calcium
carbonate equivalent.
Table 3.6.2 shows the molecular masses and calcium carbonate equivalent of various substances.
Institute of Water and Sanitation Development 32 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Table 3.6.2
SUBSTANCE FORMULA MOLECULAR EQUIVALENT
MASS MASS
Aluminium Sulphate Al2(SO 4 )314.3H2O 600 -
Ferrous Sulphate FeSO4 7H2O 278 -
Ferric chloride Fe Cl3 162 -
Aluminium Hydroxide Al(OH)3 78 -
Iron Hydroxide Fe (OH)3 107 -
Calcium Carbonate CaCo3 100 50
Calcium Hydrogen
Carbonate Ca (HCO3) 2 162 81
Calcium oxide CaO 56 28
Calcium Hydroxide Ca (OH)2 74 37
Sodium carbonate Na2CO3 106 53
_________________________________________________________________________
Example
Natural alkalinity in raw water is found to be 8,1mg/l of calcium hydrogen carbonate. Calculate the
calcium carbonate equivalent.
Answer
Calcium hydrogen carbonate equivalent mass = 81
Calcium carbonate equivalent mass = 50
Natural alkalinity as Ca(HCO3)2 = 8.1mg/l
Therefore Natural alkalinity as CaCO3 = 8.1 x 50
81
= 5 mg/l
Example
Institute of Water and Sanitation Development 33 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Calculate the calcium carbonate equivalent of a sample of water with 7mg/l of calcium oxide.
Answer
Calcium oxide equivalent mass = 28
Calcium carbonate equivalent mass = 50
Concentration of calcium oxide = 7mg/l
The calcium carbonate equivalent = 50 x 7 mg/l
28
= 12,5mg/l
1.13.4. Primary Coagulants
Of all the primary coagulants available aluminium sulphate (usually called alum) is by far the most
commonly used. It is used in well over 90% of the water treatment plants. For this reason
aluminium sulphate will be discussed in detail in this Session.
Aluminium Sulphate
Aluminium sulphate is the most commonly used coagulant in Zimbabwe. It reacts with the alkaline
in water to form a hydroxide. Optimum floc formation using alum occurs when the pH value of the
water is between 6.0 and 8.0. If insufficient alkalinity is present to react with alum, an alkali such as
lime must be added.
Reaction of alum with natural alkalinity
Al2(SO4)3 14.3H2O + 3Ca(HCO3)2 ⇒ 2Al(OH)3 + 3CaSO4 + 14.3H2O + 6CO2
Reaction of alum with added alkalinity, in this case lime Ca(OH)2
Al 2(SO4 ) 14.3H2 O + 3Ca(OH)2 ⇒ 2Al(OH)3 + 3CaSO4 + 14.3H2O
Advantages of Aluminium Sulphate
Alum is a relatively inexpensive coagulant if local production is possible. In some other
countries in Africa, however, alum must be imported at substantially increased costs.
Countries in West Africa, for example, import most of their alum from Europe, paying as
much as ten times as it would cost in the US. Accordingly, treatment plants in these
countries should be designed so that alum consumption is minimised.
Institute of Water and Sanitation Development 34 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
The dosage of alum may be reduced in some instances by
- pretreating excessively turbid river waters,
- direct filtration of low turbidity waters (< 50 NTU) waters,
- the use of coagulant aids, and
- optimum pH adjustments
The other advantages of using alum are that it may be the only chemical that needs to be added,
that it is not corrosive and forms good flocs.
Ferric Salts
Four types of ferric salts are used as coagulants: (1) ferrous sulphate (copperas), (2) chlorinated
copperas, (3) ferric sulphate, and (4) ferric chloride. The physical and chemical characteristics of
each are summarised in appendix A. In general, they give similar results when their doses are
compared in terms of iron content.
A number of practical differences between alum and ferric coagulants have been noted in standard
references (America Water Works Association, 1971; Cox, 1964).
1. Ferric hydroxide is insoluble over a wide range of pH values than aluminium hydroxide.
This is illustrated in the pH zone-coagulation relationship shown in figure 3.7.1 for
aluminium sulphate and ferric sulphate. The two curves indicate that for alum the pH zone
for optimal coagulation is relatively narrow (6.5 to 7.5), whereas for ferric sulphate it is much
broader, ranging from 5.5 to 9.0. Hence iron salts can be used over a wider range of pH
values.
2. Ferric hydroxide is formed at low pH values, so that coagulation is possible with ferric
sulphate at pH values as low as 4.0 and ferric chloride at pH values as low as 5.0.
3. The floc formed with ferric coagulants is heavier than alum floc. Therefore floc of ferric
coagulants settle faster than that of alum.
4 The ferric hydroxide floc does not redissolve at high pH values.
1. Ferric coagulants may be used in colour removal at the high pH values required for the
removal of iron and manganese and in the softening of water.
2. Iron salts, being good oxidising agents can remove hydrogen sulphide and its
corresponding tastes and odours from water.
3. Iron salts cause staining and promote growth of iron bacteria in the distribution system. This
results in red-water complaints from consumers.
4. The handling and storing of iron salts requires more skill and control, as they are corrosive
and deliquescent
Institute of Water and Sanitation Development 35 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Figure 3.7.1
Although ferric salts are not as widely available as alum (which may prevent their widespread
adoption as coagulants particularly in developing countries), the possibility of using ferric salts
should be investigated, especially when raw water pH is about 8.0 or higher. In Zimbabwe
Kwekwe municipality uses iron salts as coagulants.
a) Ferrous Sulphate
Ferrous sulphate prefers added alkalinity.
The reaction of Ferrous Sulphate with lime.
2Fe2 SO4.7H2O + 2Ca(OH)2 + ½O2 ⇒ 2Fe(OH)3 + 2CaSO4 + 13H2O
The disadvantage of ferrous sulphate is the need to add two chemicals which increases costs and
storage space.
Oxidation of ferrous sulphate
Ferrous sulphate oxidation can be achieved using chlorine. The reaction of the two yields ferric
sulphate and ferric chloride which can both be used as primary coagulants. See reaction below.
3FeSO4 .7H2O + 1.5Cl2 ⇒ Fe2(SO4 )3 + FeCl3 + 21H2O
(Ferrous Sulphate) +1.5(chlorine) = (Ferric sulphate) + (Ferric Chloride) + 21 (water molecules)
b) Ferric sulphate
This is a corrosive and staining expensive product, which is difficult to dissolve. It has advantages
in certain processes like decolouration in low pH waters and removal of manganese at high pH
waters.
Institute of Water and Sanitation Development 36 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Ferric sulphate reacts with natural or added alkalinity through the following reactions:
The reaction with natural alkalinity
Fe2 (SO4 )3 + 3Ca(HCO 3) 2 ⇒ 2Fe(OH)3 + 3CaSO4 + 6 CO2
The reaction with added alkalinity
Fe2(SO4)3 + 3Ca(OH)3 ⇒ 2Fe(OH)3 + 3CaSO4
c) Ferric chloride
Ferric chloride is extremely corrosive and difficult to handle, store and apply. It is therefore
not commonly used.
Ferric Chloride reacts with natural or added alkalinity. See reactions below.
Reaction with natural alkalinity
2FeCl 3 + 3Ca(HCO3)2 ⇒ 2Fe(OH) 3 + 3CaCl2 + 6CO2
Reaction with added alkalinity
2Fe Cl3 + 3Ca(OH)2 ⇒ 2Fe(OH)3 + 3CaCl 2
For all calculations involving coagulation reactions a balanced will be provided.
Lime
Lime is used with other coagulants as described above. It is available as CaO (quicklime) or
Ca(OH)2 (slaked lime or hydrated lime). Quicklime is stronger and cheaper than hydrated lime but
is difficult to treat and handle. It is normally used when hydrated lime is not readily available.
Example
A treatment plant is treating 10 000m3 /d of water using 20 mg/l of alum.
Calculate
a) The daily requirement of alum (in kg).
b) The amount of natural alkalinity (in mg/l) required to react with the alum.
Institute of Water and Sanitation Development 37 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Answer
a) Raw water supply = 10 000m3 /d
Alum dosage = 20 mg/l
= 20 x 1 000mg /m3 = 20 000mg/m3 = 20 g/m3
The daily requirement of alum = 20 x 10 000g/d
= 200 000 g/d
= 200kg/day
b) Al2(SO4)3 14.3H2O + 3Ca(HCO3)2 ⇒ 2Al(OH)3 + 3CaSO4 + 14.3H2O + 6CO2
I mol of alum requires 3 mols of natural alkalinity as Ca(HCO3)2
600 mg/l of alum requires 3 x 162 mg/l of Ca(HCO3)2
1 mg/l of alum requires (3 x 162) mg/l of Ca(HCO3)2
600
20 mg/l of alum requires (3 x 162)x20 mg/l of Ca(HCO3)2
600
Therefore the amount of natural alkalinity (in mg/l) required to react with the alum is 16.2 mg/l as
Ca(HCO3)2 which is equivalent to 10 mg/l as Ca(CO3).
Example
A ferrous sulphate dosage of 35mg/l and lime are used for coagulation in a plant
treating 10 000m3/d of raw water.
Calculate:
a) the daily requirement of ferrous sulphate.
b) the dosage of lime (in mg/l) required to react with Fe SO .
c) the dosage of added alkalinity as CaO(70% purity) in mg/l.
d) the production of ferric hydroxide sludge.
Institute of Water and Sanitation Development 38 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Answer
2a) Daily amount of raw water = 10 000m3/d
Ferrous sulphate dosage = 5mg/l = 35 x 1000 mg/m3 = 35 000 mg/m3
= 35 g/m3
The daily requirement of ferrous sulphate = 35 x 10 000 g/d
= 350 000 g/d
= 350 kg/d
b) 2FeSO4 .7H2 O + 2Ca(OH)2 + ½O2 = 2Fe(OH)3 + 2CaSO4 + 13H2O
2 molecules of Ferrous sulphate require 2 molecules of calcium hydroxide
2 x 278 mg/l ferrous sulphate reacts with 2 x 74 mg/l of calcium hydroxide
1mg/l of ferrous sulphate requires 2 x 74 = 0.27 mg/l of calcium hydroxide (lime)
2 x 278
The dosage of lime required to react with 35 mg/l ferrous sulphate = 35 x 0.27 mg/l
1
The dosage of lime required = 9.45mg/l
c) The dosage of lime (calcium hydroxide) = 9.45 mg/l
Equivalent mass of Calcium hydroxide = 37
Equivalent mass of Calcium oxide = 28
Equivalent dosage of calcium oxide = 28 x 9.45 = 7.2 mg/l
37
Dosage of calcium oxide (70% purity) = 7.2 x 100
70
= 10.29 mg/l
d) 2Fe2SO4 .7H2O + 2Ca(OH)2 + ½O2 = 2Fe(OH)3 + 2CaSO4 + 13H2O
2 molecules ferrous sulphate produce 2 molecules ferrous hydroxide
2 x 278 kg produce 2 x 107 kg
Amount of ferric hydroxide sludge produced
= 2 x 107 x 350 kg/d = 134,7 kg/day
2 x 278
Coagulant Aids
There are several types of coagulant aids, which may be used to improve the characteristics of the
floc.
Institute of Water and Sanitation Development 39 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Polymers (also known as poly-electrolytes) are the most commonly used coagulant aids. Polymers
are synthetic, high molecular weight organic compounds. Their main application is the
improvement of floc size, strength, and settlability under difficult treatment conditions. In a few
cases, polymers have been used alone (that is, as the primary coagulant) but these cases are rare.
Several hundred different polymers are available commercially, in both liquid and solid form.
Polymers are classified into three groups, depending on the type of electrical charge present on the
polymer molecules:
1. Anionic : having a negative charge.
2. Cationic : having a positive charge
3. Nonionic: having no charge.
The only way to determine the best polymer to use is by jar testing and observation of the effect of
different types of polymer on plant conditions. Some polymers will have little or no effect, or may
even make conditions worse. But in many cases, the right polymer used under the proper
conditions can make a very dramatic improvement.
1.13.5. Factors Affecting The Coagulation Process
There are many factors that affect the coagulation process.
Following are some of the most important:
1. The coagulant used.
Most water treatment plants use aluminium sulphate as the coagulant. In some cases, iron salts
such as ferric sulphate or ferric chloride are used. Different coagulants may give different results.
2. The coagulant dosage.
Under any given set of conditions, a particular water requires a certain optimum dosage of
coagulant for coagulation to be successful. If the coagulant dosage is either too high or too low,
coagulation will be incomplete and the results of treatment will be unsatisfactory.
3. the mineral content of the water.
Effectiveness of coagulation will depend on minerals present in the water. Treatment with alum is
more effective if the water contains abundant amounts of certain minerals, like calcium and
magnesium, to provide alkalinity. Other chemicals, like phosphate compounds, will inhibit
coagulation, and may require larger doses of coagulant to be used.
4 The water temperature.
Most chemical reactions will proceed faster when the water is warm than when it is cold.
5. the time allowed for coagulation.
Institute of Water and Sanitation Development 40 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
The time allowed for mixing and for coagulation to occur is important.
6. pH Effect
At a pH between 5 and 8, aluminium sulphate reacts with the alkalinity of the water to form a
gelatinous precipitate of aluminium hydroxide. If the pH is less than 5 or over 8 no precipitate will
form and any that has already formed will tend to redissolve. Furthermore, at a pH of over 6,8
hydroxide precipitate will have a negative electrostatic charge and will therefore be ineffective in
countering the negative charge of colloids. The optimum pH varies with the coagulant used and
other characteristics of the water.
7. Alkalinity effect
If insufficient alkalinity is present to react with the acidic coagulant, the following reaction takes
place between alum and water;
Al2 (S04) + 6H2 O- ---- > 2AI(OH)3 + 3H2SO4
The sulphuric acid (H2S04) so formed will quickly overcome the buffering capacity of the water, the
pH will drop and the aluminium hydroxide will redissolve. The same reaction will take place if an
excessive dose of coagulant if added, thereby destroying all alkalinity present and creating acidic
conditions. Not only is this wasteful in chemicals but when the pH is subsequently raised for
corrosion control, iron or aluminium hydroxide will re-precipitate in the reservoirs and mains,
causing consumer complaints.
8 Rapidity of' Mixing
The ionic products formed on addition of the coagulant will adsorb to whatever they come into
contact with almost instantaneously, be it to electro-negative colloids or to each other or to neutral
metal hydroxides of colloidal size. The entire process including adsorption is extremely rapid and
is reported to take place in less than 1/10th of a second at normal temperatures. Thus the
coagulant should be dispersed almost instantaneously throughout the water to be treated, so that
all colloids can be exposed to the neutralising ions during the very short time that these are
available. Poor distribution or dispersion would result in the nearest colloids being overtreated
while a large percentage of the effective ions would be wasted due to adsorption to each other and
to metal hydroxide precipitates. This results in less than ideal coagulation or the need for
excessive dosing of coagulant to compensate.
Rapid dispersion may be achieved by introducing the coagulant in suitably dilute form, at some
point of high turbulence in the incoming water.
1.13.6. FLOCCULATION
Process Description
Flocculation is a slow stirring process which causes the particles of natural turbidity in the raw
water and the small floc particles produced during coagulation to clump together to form larger,
settleable particles.
Factors Affecting The Flocculation Process
Institute of Water and Sanitation Development 41 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
The purpose of all flocculators is to provide gentle mixing that will produce a quick-settling floc.
The success of flocculation is affected by only a few factors.
The main factor is the degree of mixing.
1. What do you think would happen if the mixing is too gentle?
2. What do you think would happen if the mixing is too violent?
Your answers are correct if they indicate the following:
If mixing is too gentle, the suspended particles will not be brought into contact with one
another, and there will be fewer opportunities for large clumps of floc to form.
Mixing that is too violent will tear apart the floc particles and prevent them from attaining the
proper size.
A second factor affecting flocculation is the time of mixing.
A minimum time of mixing is necessary for flocculation to be completed. In actual plant operation,
and depending on the temperature of the raw water, a period of 20-40 minutes is usually sufficient.
It is important that all portions of the water are kept in the flocculator for the required time, so the
container has to be one that is designed to prevent short-circuiting. Inlet baffles allow for better
flocculation, since the baffle will help prevent short-circuiting through the container.
We mentioned the degree of mixing and the time of mixing as two factors affecting flocculation.
A third factor affecting flocculation is the number of particles.
A relatively clear water is harder to flocculate than a turbid water containing a lot of suspended
matter. The difference is that the greater number of particles in the turbid water collide with one
another more often.
Coagulation And Flocculation Mixers
Mixing is the most important part of the coagulation-flocculation process. Different types of mixers
are used for each purpose.
Let's look at the coagulation mixers first. Because coagulation takes place within the first few
seconds after the coagulant is added to the water, rapid, instant and high speed mixing is needed.
This process is called flash mixing.
Institute of Water and Sanitation Development 42 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Following are diagrams of three types of flash mixers commonly used. In each case, notice that
the point of addition of the coagulant is close to the point of rapid mixing.
A different kind of mixing is needed for flocculation. In the flocculation stage, we are attempting to
move neutralized particles around so that they collide and clump together.
Although the floc formed contains most of the suspended matter in the water, it is still made up of
approximately 95% water. This means that the floc is very fragile and has to be treated gently.
High speed flocculation must be avoided. Instead, flocculation requires slow speed mixing.
Following are diagrams of commonly used flocculators.
Institute of Water and Sanitation Development 43 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
1.14. CLARIFIERS / SEDIMENTATION BASINS.
Sedimentation usually occurs after coagulation and flocculation. It is usually the last operation
before water goes on to filtration.
Settling is an economical way of clarifying water. Therefore this treatment step is usually used
whenever the water contains even a moderate amount of suspended matter.
(There is an exception. If there is very little suspended matter in the water and there is not much
floc, it may not be economical or necessary for water to go through sedimentation. The water may
flow directly to filtration, and that process is called direct filtration.)
As we said, the purpose of settling or sedimentation is to remove as much floc and other
suspended material as possible before the water flows on to the next treatment stage,
which is usually filtration.
In settling, we want heavier materials in the water to settle out by gravity, as shown in this diagram:
Settling is the process of holding water in a quiet, low flow condition so that suspended matter and
particles can be settled out by gravity to the bottom of the tank and removed as sludge.
In treatment plants, sedimentation takes place in sedimentation basins or tanks. These tanks can
be circular, square or rectangular. The particular shape of the tank depends on many factors, like
the land area available, and the type of sludge removal equipment to be used.
The important point about any sedimentation tank is that it is designed so that the water
can enter the tank, pass through, and leave without creating much turbulence. This allows
the maximum settling of suspended solids.
To understand the operation of a settling tank, it is helpful to think of it as having four
zones, as shown in the diagram below:
Institute of Water and Sanitation Development 44 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Please realise that the size of each of the zones varies, and that the boundaries between
zones are vague and indefinite rather than sharp and well defined.
Clarifier design should facilitate removal of sludge and this is best achieved under hydraulic head
for vertical flow tanks. For horizontal flow tanks some form of mechanical scrapper system is
needed to move the sludge to a collection point, where it may be withdrawn hydraulically.
Clarification removes up to 90% or turbidity in water. Clear water drawn off from the top 15cm in
the tank by weirs or perforated pipes or troughs and then fed to filters.
Typical clarifier loading rates in Zimbabwe:
Vertical flow: 1,25 – 2,5 m/hr (m3m-2hr-1)
Horizontal flow: 0,5 – 1,25 m /hr
Small circular clarifiers: 0,25m/hr
For smaller works lower loading rates are recommended. With a polyelectrolyte type of ‘floc-aid’
used in addition to the simple chemical flocculant, much higher loading rates are obtained. But
flocculant aids are normally expensive and imported. Therefore their use must be fully justified.
1.15. FILTRATION
Filtration may be defined as the process of passing a liquid through a porous or open-textured
medium in order to remove suspended matter by retaining it in the pores of the filtering medium or
at the surface of the filter.
1.15.1. Slow Sand Filters:
The action in a slow sand filter is basically one of simplest straining on the surface of the sand. A
(schmutzdecke) layer of particles larger than the interstices between the sand, forms on the
surface and in its turn arrests smaller particles. This mat of arrested substances also contains
biological organisms which induce bio-flocculation of suspended solids and colloids, oxidize
organic matter and reduce bacterial counts. This layer takes two weeks to form therefore
Institute of Water and Sanitation Development 45 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
commissioning a slow sand filter requires at least two weeks while the water is being lost.
Since filtration takes place on the surface only, the sand bed is comparatively shallow (0,4 m). To
avoid penetration of solid material into the sand, the filtration rate is kept low (1,3 to 3,7 m3 /m2 /d)
and the sand should be fairly fine and natural,
not evenly graded like that of a rapid gravity filter. Very fine particles should be washed out,
leaving a grading of 0,15 to 0,85 mm. The sand bed is supported on layers of gravel and stone,
graded in ascending size so that there is minimum penetration of small stone into the interstices of
the underlying layer.
To clean the filter, it is allowed to dry out
and the shrivelled-up layer of dirty on the
surface is swept off. Water is then
allowed to filter to waste until a
schmutzdecke is formed. This means
that some two-thirds of the total filter
area will always be in various stages of
draining and drying out for cleaning. It is
obvious, therefore, that three times the
theoretical filter area must be provided,
making slow sand filters impractical for
anything but a very small works.
Their advantage lies in cheapness of construction, ease of operation with reliable results.
1.15.2. Rapid Gravity Sand Filters:
Design
The rapid gravity filter has a deep bed (0,75 to 1 m) filled with relatively coarse, but carefully graded
sand. The terms "Effective Size" and "Uniformity Coefficient" are used in relation to sand grading.
Effective Size: is the size of aperture through which 10% of the sand by weight will pass.
Uniformity Coefficient is the size of aperture through which 60% of the sand, by weight, will pass,
divided by the size aperture through which 10% of the sand by weight will pass.
Institute of Water and Sanitation Development 46 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
The effective size of the sand may vary from 0,4 mm to 2,0 mm depending on the design of the
filter, the most important factor is its uniformity. Uniformity coefficients are generally specified to fall
between 1,0 and 1,5. Solids penetrate a long way into the sand but because of the depth of the
bed there is little chance of break-through.
The working sand of a filter may be supported on layers of coarser sand and stones which
separate it from the system of nozzles, laterals and headers which go to form the filter floor.
However, the most recent trend is to lay the sand directly on the filter floor, which is provided with a
large number of evenly spaced nozzles designed to prevent ingress of sand particles. The wash
water distribution system naturally also serves as the filtered water collecting system when the filter
is in use.
Filtration rates are higher than in slow sand filters, on average 60 to 180 m3 /m2/d. On occasion
filters may be designed for much higher rates (up to 1 000 m3 /m2 /d) providing the water is suitable
and if the depth of bed is increased or effective sand size decreased. In practice the use of rates
greater than 600 m3 /m2 /d has little advantage because of the frequency of back washing, the
quantity of backwash water and special supervision required.
Filter Cleaning:
Before the filter effluent noticeably deteriorates, the clogging of the interstices will cause a loss of
head through the filter, usually indicated on a gauge. When the loss of head reaches a certain
figure, the filter must be washed. Cleaning is carried out by back-washing which process converts
the filter media into quick sand and any solid object near the surface will sink right through it. At the
end of a filter run, the uppermost 150 to 200 mm of the sand is bound into a fairly solid cake by the
solid matter caught in it. If the wash water is applied at the normal rate while the surface is in this
condition, the latter will break up into lumps which will immediately sink through the loose sand.
This phenomenon is known as "mud-balling" and is best prevented by blowing air upwards through
the media prior to backwashing: water jets or mechanical rakes are sometimes used instead of air
scour.
Institute of Water and Sanitation Development 47 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Diagram of a rapid gravity sand filter
1.15.3. Pressure Filters:
There is no intrinsic difference between rapid gravity and pressure filters and the operating
principles are identical for the two. Average filtration rates are generally higher, some 140
to 350 m3 /m2 /d. High rates may be used in swimming pool filters for instance as the quality
of water is usually good.
The method of back washing is similar with the exception that mechanically driven rakes
are frequently used instead of air scour to break up the compacted dirty sand.
The real difference lies in the method whereby the head on the filter is produced. The gravity filter
is open and there is an actual layer of water of required depth on top of the sand, while the
pressure filter is totally enclosed and the head is produced by pumping water into it, or by running it
Institute of Water and Sanitation Development 48 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
in from an elevated position.
While the outlet of a gravity filter is always at approximately atmospheric pressure, the outlet of a
pressure filter can be at virtually any pressure. It is possible to pump water through a pressure filter
and deliver filtered water at a level far above that of the filter itself.
One disadvantage of pressure filters is that the backwashing process cannot be watched and it is
difficult to establish at any time whether the sand is in a satisfactory condition. Pressure filters are
normally used on a small works, where the water has to be pumped in any case (swimming pools
being a good example) and on larger works sited so that the hydraulic gradients make it difficult to
install gravity filters.
1.16. DISINFECTION OR STERILISATION
Disinfection ensures drinking water that is free of pathogenic organisms. Water with low turbidities
may be disinfected without any additional treatment for pathogen removal.
Purpose
- destruction of all disease causing organisms in water
- disinfection without rendering the water toxic or carcinogenic
- to give a residual concentration which acts as a safeguard against recontamination.
Sterilization is the destruction of all pathogens in water.
A good disinfectant should permit simple and quick monitoring of the strength and concentration in
water, permit safe handling and application and be readily available and at a reasonable cost.
Institute of Water and Sanitation Development 49 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Methods
- Physical means e.g. removal of bacteria by SSF, microstraining, boiling, storage, etc
- Irradiation e.g. UV-light
- Use of metal irons, e.g. Ag, Cu.
- Chemical treatment e.g. use of oxidants (halogens and halogens compounds - chlorine,
iodine, bromine), ozone, potassium permanganate, hydrogen peroxide)
1.16.1. Chlorination
Chlorination: Simply it is the addition of chlorine to water.
When chlorine is added to water it reacts to produce various compounds. Some of these
compounds are effective disinfectants whereas others represent the end product of a reaction that
has removed an undesirable constituent from the water.
Reaction In Pure Water
When chlorine is added to pure water it reacts as follows:
Cl2 + H2 O ---- HCCl + HCl Eq. 1
Chlorine Water Hypochlorous Hydrochloric
Acid Acid
In this reaction the chlorine combined with water to produce hypochlorous acid (HOCl). This is one
of the two free available chlorine residual forms. Due to the ease with which HOCl penetrates into
and kills bacteria it is the most effective form of chlorine for disinfection. However some of the
HOCl (weak acid) dissociates as follows:
HOCl -----> H+ + OCl- Eq. 2
Hypochlorous Hydrogen Hypochlorite
acid ion
As shown by Equation 1, this dissociation produces hydrogen (which neutralizes alkalinity or
lowers pH) and hypochlorite ion (OCl- ), the second type of free available chlorine residual. The
OCl is a relatively poor disinfectant compared with HOCl, primarily because of its inability to
penetrate into the bacteria.
Equation 2 shows what happens to the hydrochloric acid (a strong acid) formed in the first reaction.
HCl -------> H+ + Cl- Eq. 3
Hydrochloric Hydrogen Chlorine
acid ion
Notice it also dissociates, forming hydrogen (which neutralizes alkalinity or lowers pH) and chloride
ions, one of the same ions formed when common table salt is dissolved in water. Neither the
hydrogen nor the chlorine ion act as disinfectants.
Institute of Water and Sanitation Development 50 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
The effectiveness of chlorination is based on five important factors:
Ø pH
Ø concentration
Ø temperature
Ø contact time
Ø other substances in water
The pH strongly influences the ratio of HOCl to OCl- low pH values favour the formation of HOCl,
the more effective free residual. (Equation 1), while high pH values favour the formation of OCl- ,
the less effective free residual form Equation 2). As pH increases from 7,0 to 10,7, the OCl- form
begins to predominate and the time required for the free residual to effectively disinfect increases.
The added time is barely detectable in the pH range 7,0 - 8,5 but is markedly longer for a pH
greater than 8,5.
Temperature Has Two Influences
Very high temperatures (for example, boiling) speed the killing of organisms. However, within the
range of temperatures normally found in water, the lower the temperature the more effective the
chlorination. There are two reasons lower temperatures favour the formation of HOCl, which is
more effective than OCl-. Secondly lower temperatures promote quick dissipation.
The destruction of organisms (kill) is directly related to the contact time and concentration of
chlorine. For example to accomplish a given kill in a given period of contact time you might need a
certain concentration of chlorine. Providing other conditions remain constant (such as pH and
temperature), if the contact time is increased, less chlorine concentration is needed to accomplish
the same kill. As dosage concentrations are increased the contact time needed can be decreased.
Equations 1-3 represent what happens when chlorine is added to pure water. However, the water
treated at a treatment plant is far from pure. The organic materials in raw water supplies can and
do take part in the reaction with chlorine in water and these include:
Ø Ammonia (NH3)
Ø Iron (Fe)
Ø Manganese (MN)
Ø Hydrogen sulphide (H2S)
Ø Dissolved organic materials
REACTION WITH AMMONIA
One of the most common reactions of chlorine in raw water involves ammonia, Ammonia (NH3 ) is
an inorganic compound occurring naturally due to decaying vegetation or artificially from domestic
and industrial waste water discharges. Chlorine reacts with ammonia to form chloramine,
compounds containing both nitrogen and chloride ions. As shown in Equation 55 -57, chloramine
are formed in three successive steps.
Institute of Water and Sanitation Development 51 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
NH3 + HOCl ----> NH2 Cl + H2O Eq. 4
Ammonia Hypochlorous Monochloramine Water
acid
NH2 Cl + HOCl ----> NHCl2 + H2 Eq. 5
Monochloramine Hypochlorous Dichloramine
acid
NHCl2 + HOCl -----> NCl3 + H2 O Eq. 6
Dichloramine Hypochlorous Trichloramine
acid
Whether one chloramine compound or a combination is formed depends on the pH of water and
on the presence of enough ammonia. Monochloramine and dichloramine are effective disinfecting
agents, but they are commonly not as effective as free chlorine.
SOURCES OF CHLORAMINE
There are three types of materials commonly used as a source of chloramine:
(1) gaseous chlorine
(2) calcium hypochlorite
(3) and sodium hypochlorite.
Liquid chlorine is a compressed amber-coloured gas containing 99,5 percent pure chlorine. At
room temperature and pressure 1 cm3 of liquid will expand to approximately 500 cm3 of gas.
Chlorine gas is greenish in colour and visible at high concentrations. It is highly toxic even at
concentration as low as 0,1 percent by volume. As demonstrated in Equation 7, plain chlorine
mixed with water produces hypochlorous acid.
Cl2 + H2 O ----> HOCl + HCl Eq. 7
Chlorine gas Water Hypochlorous Hydrochloric
or liquid acid acid
Chlorine liquid or gas is neither explosive nor flammable but it will support combustion.
The liquid changes easily to a gas at normal temperatures and pressures. If chlorine
remains dry it will not corrode metal. However mixed with some moisture it is extremely
corrosive. Chlorine liquid is approximately 1,5 times the weight of water. Gas is
approximately 2,5 times the weight of air.
Calcium hypochlorite, Ca(OCl)2 is dry, white or yellow-white, granular material. The granular
material contains 65 percent available chlorine by weight.
When added to water Ca (OCl)2 reacts as follows:-
Ca(OCl)2 + 2H2O -----> 2HOCl + Ca(OH) 2 Eq. 8
Calcium Water Hypochlorous Lime
acid
Calcium hypochlorite should be stored carefully to avoid contact with easily oxidized material
because this type of chlorine can cause fires when brought into contact with many types of organic
compounds.
Institute of Water and Sanitation Development 52 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Sodium hypochlorite (NaOCl) is a clear, greenish-yellow liquid chlorine solution normally used in
bleaching. Normally household bleach is an example of sodium hypochlorite. It contains 5 percent
available chlorine.
Practical application
Chlorination should not be done prior to SSF as the residual chlorine destroys biological
agents. The sedimentation and filtration preceding chlorination enhance the disinfection
process. Addition can be batch chlorination, as tablets (chlorine), diffusion chlorination or
continuous chlorination.
1.17. OTHER TREATMENT PROCESSES
Activated carbon
Activated carbon is used for removal of tastes and odours. If this is dosed as a powder it must
itself be removed by filtration.
Complete removal of dissolved solids for high pressure boilers in power stations and industrial
plants requires a demineralizing plant which is specific to user industry.
Water softening
Calcium and magnesium salts react with soap and so lather cannot form between these
neutralized. There are 2 basic water softening methods:
• precipitation methods which depend on the addition of lime or soda ash resulting in the
formation of non-soluble carbonates which can be settled out or filtered.
• Base exchange whereby calcium and magnesium salts are replaced by sodium salts
(which do not cause hardness). Calcium and magnesium chlorides are discharged to
waste. This is simple to implement and operate and is therefore best for communities.
However a lot of salt is generated and there is need for storage and a method of disposal.
Iron and Manganese removal
Iron and manganese cause water discolouring, laundry staining, tastes and growth of certain
bacteria. Iron removal is by introducing oxygen and raising pH. This changes iron from the ferrous
state which is soluble to the insoluble ferric state as hydroxide, which precipitates and can be
settled out or filtered.
Manganese removal requires a high pH, plus oxidation by aeration or chlorination. The most
appropriate is potassium permanganese which will precipitate the manganese when the pH is
above 6,5. The precipitate is then removed by a normal sand filter.
Control of flourides (dental effects)
Flourides are beneficial around 1mg/l but hazardous above 3 - 5 mg/l. Removal is achieved by
passing water through tricalcium phosphate, certain iron exchange compounds or activated
alumina. The process is expensive. An alternative is to abandon the water source and find water
elsewhere.
Addition of flouride is in the form of sodium flouride on small works, and Na2SiF6 on bigger works
Institute of Water and Sanitation Development 53 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
(>5ml/day); H2SiF6 acid where cheapness is desired. Careful operation of the doses is necessary.
Algae and Algal control
Algae are minute organisms classified as plants which proliferate in rivers and reservoirs. Algae
outbreaks are sporadic and severe and coincide with a rise in temperature on seasonal basis and
are encouraged by open reservoirs since sunlight is required for photosynthesis. Heavy pollution
inhibits algal growth by redusing the oxygen content of water.
Removal of algae by settling basins is difficult. Since algae floats upward flow tanks are best for its
removal especially with blanket principle. Algae is also best destroyed by pre-chlorination before
reaching basins with a dosage of 1mg/l being fairly effective. Algae growth is inhibited by cooper
sulphate, CuSO4 with which a dose of 0,3mg/l is sufficient. A dosage of 5kg/ha is recommended
when this is applied on to a water surface but this is toxic to some fish types. Strainers are also
used to remove algae as microstrainers or as a rapid filter running without coagulants.
1.18. OPERATION AND MAINTENANCE FOR TREATMENT PLANT
1.18.1. Maintenance
Preventive maintenance encompasses all the essential activities in order to ensure effective and
continuos utilisation of equipment and processes so as to prevent breakdowns. Repair is work to
restore broken down equipment back to working order.
Requirements for effective maintenance
- knowledge of equipment and processes
- tools
- spares
- planned programme
The main purpose of preventative maintenance is:
- to prevent costly
- to prevent loss of service (water supply)
- to improve efficiency
Regular inspection and maintenance should be targeted at:
• electrical and mechanical equipment (chemical feeders, air compressors, sludge pumps,
standby generators, etc)
• structures (clarifiers, filters, reservoirs, etc)
• unit processes (clear well water levels system hydraulics, chemical dosing, backwashing,
filtration, pH and turbidity monitoring, etc.)
The major components of preventive maintenance include:
(a) Regular greasing
Lubrication oil changes
Cleaning
Replacement of worn parts
(b) Systematic scheduling of maintenance services (staggering the servicing of different pieces
of equipment)
Institute of Water and Sanitation Development 54 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
(c) Maintenance of a planning schedule (keep record to avoid need to rely on memory).
Revise schedule depending on experience if necessary
(d) Keep a record of the following:
- date of maintenance
- nature of work
- condition of equipment
- parts replaced or ordered
- time spent and costs
- serial numbers of spares replaced - this simplifies future ordering of similar parts.
(a) Maintain stocks of items which are required regularly like bearings, oil seals, gears, etc.
(b) consider stationing an electrician and a mechanic at the works to service electric motors
and switch gear, and diesel or petrol driven motors respectively regularly.
(c) For corrosion protection regularly clean and paint
- store rooms
- pump houses
- other buildings
Treatment plant record keeping
- useful because of high staff turnover, etc
- size
- age
- capacity of units
- daily working times for each unit
- daily working capacity of each section of plant
- essential maintenance
- repairs carried out
- storage
- etc
For plant and equipment record the full particulars:
- installation date
- repair works
- outages
- modifications
- additions
- etc
The following is given as a guide
• chemical feed pumps or equipment: type, make, etc
• clarification tank: number, size, shape, volume, sludge removal pipe system, etc.
• sand filter: size backwashing needs, etc
• reservoirs: size, volume etc
• pumps and motors: no, horsepower, capacity, etc.
• pipes: size, length, etc
• dosages and chemical quantities used
Institute of Water and Sanitation Development 55 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
• pumping plant and hours run
• production and metering of influent water, treated water, filter rates, water loss through
backwashing of filters, sludge wastage, etc.
• labour and stores
Also record water quality and quantity data and the treatment plant's operations. Carry our
regulatory water tests. Some regular tests involve caliform analysis, chemical analysis, organic
and inorganic chemical test, jar test, hardness tests for softening.
For groundwater, supplies record operational hours, pumping rate, static and pumping water levels
discharge pressure, power consumption and maintenance.
Annual report
Annually summarise operation, maintenance, business and cost data. The report should include:
• consumption data - minimum, mean and maximum values as total daily quantities and per
capita consumption
• comparison of quality data to drinking water standards
• major problems and their solutions, consumer complaints and their resolutions.
• Operating costs, personnel, improvements (both carried out and required)
Water and energy conservation are issues which should always be considered in the operation of a
water treatment plant.
It is of utmost importance to issue operations and maintenance manual to the senior works
operator.
Institute of Water and Sanitation Development 56 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Final exercise to Session IV
1. Why is setting alone not sufficient to remove suspended particles?
2. State the three types of forces that act on suspended particles.
3. Describe briefly what happens during coagulation.
4. What are coagulant aids?
5. Why is flash mixing necessary during coagulation?
6. List the advantages of using ferric sulphate as a coagulant over aluminium sulphate.
7. A water treatment plant treating 10 000m3 /d is using 20 mg/l of alum. How much natural
alkalinity will be required to react with alum? If there is no natural alkalinity, compute the
theoretical dosage of lime in mg/l and kg/d.
8. A raw water supply of 5 000 m3 /d is treated with an alum dosage of 25 mg/l. Calculate:
a) The daily requirement of alum (in kg)
b) The amount of natural alkalinity (in mg/l) required to react with the alum.
c) The volume of A1(OH)3 sludge produced per day.
9. State the purpose of a sedimentation tank
10. State thre characteristics of a particle that affect its settling rate (downward travel)
Note: Molecular mass:
Al2 (SO4 ) 3.14.3H2 O = 600; Al(OH)3 = 78; FeSO4 .7H 2O = 278
Ca(OH)2 = 74; FeCl3 = 162; Fe(OH)3 = 107; Ca(HCO3)2 = 162; CaCO3 = 100
Equivalent mass :
CaCO3 = 50; Ca(HCO3)2 = 81; CaO = 28; Ca(OH)2 = 37;
Na2CO3 = 53.
Institute of Water and Sanitation Development 57 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
SECTION V - WATER SUPPLY SERVICES
Objectives
At the end of the session you should be able to;
• state the purpose of reservoir
• state the minimum water storage requirements
• state the advantages and disadvantages of different pumps
1.18.2. STORAGE RESERVOIRS
Introduction
Storage for treated water in either ground or elevated reservoirs is essential to ensure continued
supply in case of breakdown or the need for maintenance of the treatment plant or pumping and
distribution system. Fluctuation in demand which would otherwise require different pumping rates
are also evened out.
The purpose of a storage reservoir is
• to balance daily pumping rates
• to equalize supply and demand over a long period of high demand, i.e. to balance out
fluctuating demand from the distribution system against the output from the source
• to maintain a constant residual pressure
• to supply emergency water for fire fighting, breakdowns, etc. They act as a safeguard for
the continuity of the supply in case of breakdown at the source or in the trunk mains and
maintain a fire-fighting reserve.
• to act as a safeguard against accidental pollution
Storage is essential
• where water is obtained by pumping
• where water is to be treated
• with reticulated supply - communal water points or individual water connections especially
district services centres and growth points.
Boreholes (with handpumps) and wells act as storage mediums provided abstraction is less than
the recharge rate over a 24 hour period.
The following are minimum storage requirements:
• Service reservoirs should have enough storage to allow for peaks (5 - 10 hrs storage is
Institute of Water and Sanitation Development 58 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
sufficient to even out fluctuations)
• Service reservoirs provide additional storage to ensure continuity of supply in case of
breakdown (a minimum of 12 hours supply). This contingency storage depends on the
organisation of the maintenance team (water authority).
• Service reservoirs may not be full when a breakdown occurs, i.e. if the above two
conditions coincide. To cover these, it is necessary to have a minimum of 24 hours
storage. [1 - 3 days storage is typical]. A storage of 48 hours is recommended. A storage
of 36 hours is recommended where a reservoir is supplied by a gravity main.
Location and Elevation of Service Reservoirs
• Service reservoirs should be as near as possible to demand area and within city
boundaries.
• Service reservoirs should be at a suitable elevation for sufficient pressure at all points in
distribution system so as to maintain the minimum pressure at the furthest point. Water
towers should be used to gain extra elevation though these are more expensive.
• The elevation depends on the distance of the furthest point in the distribution system and
on the elevation of the tallest building.
• If the distribution area varies widely in elevation, it should be split into zones, which should
then be supplied independently, otherwise high pressures will cause high leakage values.
• Break-pressure tanks can be used to break-up high pressures.
• Boaster pumps are an alternative to elevated storage BUT there is need for 100% standby
capacity.
• With combined ground and elevated storage, the combined capacity (storage) should be
twice average daily demand. Hospitals and other institutions should have their own storage
which should be at least 6 hours and preferably 12 hours the average demand.
The recommended limits on residual pressure at consumer point should be observed and in
Zimbabwe these are:
• maximum residual : 60m
• minimum residual : 5m
The following storage reservoir appurtenances are essential:
• water level indicator
• ventilation for free entry or exit of air during filling and emptying. These openings should be
screened to prevent entry of insects and dirty.
• underfloor drains to take care of leakage
• access ladders and walkways and safety cages and hand rails
• inlet, with ball - valve or pressure switch.
• Outlet separated widely from the inlet to avoid short circuiting and dead areas
• Overflow, in case the inlet control system fails
• Washout or scour, for cleaning, inspection and maintenance.
Institute of Water and Sanitation Development 59 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
• Valves to allow for complete isolation of reservoirs.
1.18.3. Pumps - types, choice and maintenance
Types of pumps
Pumps fall into the following major classification:
A Positive displacement or reciprocating pumps
B Diaphragm pumps
C Air lift pumps
D Rotordynamic pumps, where the rotating element generates pressure and velocity
E Hydraulic ram pumps
A. Positive displacement or Reciprocating pumps
Positive displacement pumps have the following characteristics:
- operate on the positive displacement principle, with a ram, plunger or piston moving to and
fro in a cylinder
- discharge a definite quantity irrespective of head pumped against
- consist of a piston reciprocating in a cylinder, with one way valves on the suction and
delivery lines
- may be double action type, whereby both sides of piston are used for pumping, giving a
more even flow, but this requires a complex valve system
Disadvantages
- much larger and heavier than rotor dynamic ones of same capacity
- air vessel essential in rising main to prevent intermittent or pulsating flow
- solids in water interfere with valves
Advantages
Each stoke is equivalent to a fixed volume, therefore it is easy to compute volume in cases
where it is difficult to assess e.g. sludge with solids which can block normal water meters
B. Diaphragm pumps
Diaphragm pumps have a very small capacity and are normally hand operated for example
for emptying a tank. They are used for dosing chemical solutions where they can deliver
constant quantities.
C. Air lift pumps
With air lift pumps compressed air is forced down a pipe tube immersed in the liquid to be
pumped, which is then lifted. These have the advantage of low maintenance costs.
D. Rotor dynamic pumps
The operation principle of rotor dynamic pumps is that energy is imparted to the liquid by a
rotating propeller or impeller. The imparted energy (by acceleration) is converted into
pressure and velocity. This is achieved by rotating the impeller or propeller at a high speed
using an electric motor or internal combustion engine. The unit comprises the impeller,
outer fixed casing shaft, and driving motor.
Institute of Water and Sanitation Development 60 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
Rotor dynamic pumps fall into the following categories: centrifugal, axial flow or propeller
pumps, and mixed flow pumps.
Centrifugal pumps
Centrifugal pumps are the most common, generally with a discharge of 41/min to 30m3S-1
and a generated head of 1 - 120m. The efficiency may approach 90% in larger sizes. The
water enters at the centre or eye of impeller and is forced outwards radially. There is need
for priming (using foot valve or pump) to avoid over heating.
Axial flow pumps
With axial flow pumps the fluid remains axial (along axis of pump) as it passes through it.
This is achieved by using a propeller type of rotor fixed in the suction pipe (which is the
pump casing). These pumps are efficient and simple to construct and suitable where large
discharges are to be delivered against a low head e.g. in irrigation, land drainage and
sewerage. The maximum operating head is between 9 and 12 m. It is possible to use 2 or
3 impeller stages for higher heads, but this is uneconomical. Suction lift should be minimal
(or discouraged). It is best to arrange for the impeller to be immersed so that it is always
primed.
Mixed flow pumps
Mixed flow pumps are intermediate between centrifugal and axial. The flow is part radial
and part axial and the impeller is shaped accordingly. The fluid particle path is that of the
impeller on a cone. The head range is 25m per impeller. The advantage over axial pumps
is that the power demand on the motor is nearly constant.
Hydraulic ram pumps
The hydraulic ram is a simple device which uses water hammer oscillations in a pipe using
a valve arranged to close after flow commences. This is used for raising water by using the
energy of moving water of which a portion (3 - 25%) is to be raised. It is ideal for hilly or
mountainous areas with perennial streams and for supplying water to small rural
communities. It has low maintenance costs and no fuel or electricity is needed.
The selection of a pump is dependant on the pump characteristics (for rotordynamic pumps). The
design should be based on the required discharge and head. Performance curves are a set of
curves relating head, power input and efficiency to discharge while the pump is running at its
designed speed. These are useful also for operation in parallel and in series and are available from
the manufacturer. The design point is the ideal pumping point at maximum efficiency, and it
determines the total head under which a pump should operate and this also gives the power.
Power sources
Sources of power for pumping include:
- internal combustion engines
- electric motors
Institute of Water and Sanitation Development 61 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
- wind power
- solar power
- muscle
Booster stations are required when a pipeline is very long (high friction losses resulting in high
pumping head) i.e. when pumping head is greater than the capacity of available pumps and pipe
classes.
Institute of Water and Sanitation Development 62 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
ANSWERS SESSION
Answers to final Exercise of Session IV
1. Although large particles such as gravel, sand, and heavy silt can be settled by slowing the
velocity of the water smaller particles will not sink. Also colour which is caused by dissolved
rather than suspended material will not be removed. Coagulation and flocculation is necessary
to bring the particles together and increase their sizes so that they can settle to the bottom.
2. The three forces that act on suspended particles are the Brownian movement, turbulance and
electrical repulsion.
3. During coagulation some chemical or chemicals are introduced into the water. These
chemicals react with suspended and dissolved solids in the water to form particles which can
attract each other. Because of this, the particles are able to form bigger particles which can
settle to the bottom.
4. Coagulant aids are chemicals used to improve the characteristics of the floc. Poly-electrolytes
are the most common. They improve the floc size, strength and settleability.
5. The coagulant process which involves formation of ionic products and then an absorption
takes place in less than one tenth of a second. Flash mixing is therefore necessary to make sure
that in that instantaneous moment the coagulant chemical is distributed as much as possible to be
in contact with all suspended particles in the water.
6. (i) Ferric sulphate prefers a larger pH range compared to aluminium sulphate.
(ii) Ferric sulphate can be used for colour, Ca and Mg removal and softening of water.
(iii)The floc formed is heavier than for alum.
(iv) Ferric hydroxide floc does not re-dissolve at high pH values.
Institute of Water and Sanitation Development 63 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
7. Data
Molecular mass Al2(SO4) 14.3H2O = 600
Molecular mass Ca(HCO3)2 = 162
Amount of water treated = 10 000 m3/d
Dosage of alum = 20 mg/l
a) Al2(SO4)3.14.3H2O + 3Ca(HCO3)2 ⇒ 2Al(OH)3 + 3CaSO4 + 14,3H2O + 6CO2
One molecule of aluminium sulphate reacts with 3 molecules of calcium hydrogen carbonate
(natural alkalinity)
1 x 600 mg/l of alum reacts with 3 x 162 mg/l of natural alkalinity
1mg /l alum reacts with 3 x 162 = 0.81mg/l of alkalinity as Ca(HCO)
600
Conversion to Calcium Carbonate equivalent
Calcium carbonate equivalent weight = 50
Calcium hydrogen carbonate equivalent weight = 81
The equivalent alkalinity as CaCo3 = 0.81 x 50 = 0.5mg/l
81
This means that 1 mg/l of alum requires 0.5mg/l of natural alkalinity as CaCO3
20 mg/l of alum will require 0.5 x 20 = 10 mg/l of natural alkalinity as CaCO3
ii)Added alkalinity as lime : Ca(OH)2
Equivalent weights CaCO3 = 50
Ca(OH)2 = 37
So Ca(OH)2 dosage = 10 x 37 = 7.4 mg/l
50
Theoretical dosage of lime = 7.4 mg/l
iii)The quantity of Ca(OH)2 required = 7,4 x 10 000 x 1000 kg/d
1000 x 1000
Theoretical dosage per day = 74 kg/d
Institute of Water and Sanitation Development 64 2001
Post Graduate Diploma in Water Sully and Sanitation Water Supply Systems
8. Raw water supply = 5 000 m /d
Alum dosage = 25 mg/l
a) Daily requirement of alum =25 x 5 000 x 1000 kg /d = 25kg/d
1000 x 1000
b) From Q. 7 it was seen that 1 mg/l of Alum requires 0.5 mg/l of natural alkalinity as CaCO3 .
25 mg/l of alum requires 25 x 0.5 = 12.5 mg/l of added alkalinity as Ca(OH)2 .
c) Al2 (SO 4)3 14.3H 2O + 3Ca(OH)2 = 2Al(OH)3 + 3CaSO4 + 14.3H 2O
Molecular mass of Al 2(SO4 ) 14.3H 2O = 6CO
Molecular mass of Al(OH)3 = 78
600g (alum) produces 2 x 78g (Al(OH)3 .
125kg of alum produces 2 x 78 x 125kg/d = 32.5kg/day
600
9. The sedimentation tank separates suspended particles from water by gravity to reduce the
solids loading on filters so that longer filter runs can be achieved.
10. Settling of a particle depends on specific gravity, the shape and size of the particle and the
temperature of the water.
Institute of Water and Sanitation Development 65 2001
You might also like
- Ra 9275Document19 pagesRa 9275Calvin Jr. Wong100% (2)
- Handbook of Thin Film TechnologyDocument384 pagesHandbook of Thin Film TechnologyDavid100% (1)
- Wastewater and Excreta DisposalDocument95 pagesWastewater and Excreta DisposalRyeanKRumanoNo ratings yet
- Thesis Final Last PDFDocument46 pagesThesis Final Last PDFDeepalNo ratings yet
- Lake Tana Water Quality MonitoringDocument24 pagesLake Tana Water Quality MonitoringAlfatah muhumedNo ratings yet
- Module 1 IrrigationDocument20 pagesModule 1 IrrigationCole Jade DayoNo ratings yet
- Ehs 409Document207 pagesEhs 409Amanda BowersNo ratings yet
- WSP ToR - WSP - BiT 2Document7 pagesWSP ToR - WSP - BiT 2Tilik Tena WondimNo ratings yet
- Report UNESCODocument188 pagesReport UNESCOChanna SamarasingheNo ratings yet
- Muy Muy BuenoDocument165 pagesMuy Muy BuenoOrlando ArayaNo ratings yet
- Modeling of Water QualityDocument146 pagesModeling of Water QualityMartin Daniel Palacios QuevedoNo ratings yet
- 7 Training Module Water Quality Control and ManagementDocument107 pages7 Training Module Water Quality Control and ManagementHas GomNo ratings yet
- Draft Final School Sanitation Standrads and GuidelinesDocument37 pagesDraft Final School Sanitation Standrads and GuidelinesJoseph ZimbaNo ratings yet
- Water Conservation & Management (3677) : Supplementary Material Spring, 2021Document12 pagesWater Conservation & Management (3677) : Supplementary Material Spring, 2021Muhammad AbdullahNo ratings yet
- Water Purifier For Unclean Water On FaucetsDocument8 pagesWater Purifier For Unclean Water On FaucetsKenneth CanoNo ratings yet
- Water Conservation & Management (3677) : Supplementary Material Autumn, 2021Document12 pagesWater Conservation & Management (3677) : Supplementary Material Autumn, 2021Moix KhanNo ratings yet
- Solar Pasteurizer Project OverviewDocument5 pagesSolar Pasteurizer Project OverviewwaseemNo ratings yet
- M11 WashDocument36 pagesM11 WashRoNo ratings yet
- SAMWEL - Charles - FYP - Progressive ReportDocument13 pagesSAMWEL - Charles - FYP - Progressive Reportnas alawiNo ratings yet
- Water Borne Disease: For The Ethiopian Health Center TeamDocument96 pagesWater Borne Disease: For The Ethiopian Health Center Teamgirma shimelisNo ratings yet
- Mir ProjectDocument22 pagesMir ProjectAyush DuttaNo ratings yet
- "Online Examination For Hawassa University Informatics Department" Project School of InformaticsDocument13 pages"Online Examination For Hawassa University Informatics Department" Project School of InformaticsAnonymous 5Hk0VoIFh100% (1)
- ChE 3131 MODULE1Document31 pagesChE 3131 MODULE1Poison PinkNo ratings yet
- New Suliyat SeminarDocument21 pagesNew Suliyat SeminarHola MiNo ratings yet
- Drinkingujoter: Principles and PracticesDocument10 pagesDrinkingujoter: Principles and PracticesOmar CaruanaNo ratings yet
- Group 9 Final ReportDocument7 pagesGroup 9 Final Reportziziphomkosana2003No ratings yet
- Plan ActionDocument7 pagesPlan Actionterefa1234No ratings yet
- Water Sampling For Bacteriological Exam Water District GroupDocument12 pagesWater Sampling For Bacteriological Exam Water District GroupBen Fraizal UkailNo ratings yet
- NOTICE: This PDF File Was Adapted From An On-Line Training Module of The EPA's Watershed Academy Web, Found atDocument35 pagesNOTICE: This PDF File Was Adapted From An On-Line Training Module of The EPA's Watershed Academy Web, Found atRenato AguilarNo ratings yet
- ReportDocument17 pagesReportibrahimNo ratings yet
- Capstone Project Report Vishal 2Document26 pagesCapstone Project Report Vishal 266 vishal madhukar sonawaneNo ratings yet
- Chapter 4thDocument15 pagesChapter 4thSapna 2704No ratings yet
- Mad Report BhuvanaDocument29 pagesMad Report BhuvanaTejas SNo ratings yet
- Toolkit WaterDocument55 pagesToolkit WaterDaisyNo ratings yet
- A Step-By-Step Approach in Dhanusha Community and Water Supply Development Project (DCWDP)Document8 pagesA Step-By-Step Approach in Dhanusha Community and Water Supply Development Project (DCWDP)Emmanuel BettNo ratings yet
- Infant'S Serum TrackerDocument85 pagesInfant'S Serum TrackerANDRU PRADHEEPNo ratings yet
- Water SupplyDocument331 pagesWater Supplya95450469No ratings yet
- Ehs 503Document103 pagesEhs 503Has GomNo ratings yet
- Kent Mineral WaterDocument63 pagesKent Mineral WaterPrince Kumar100% (1)
- Edited CorrectionsDocument64 pagesEdited CorrectionsAkmad Ali AbdulNo ratings yet
- Flooding Problems and Surface Water DraiDocument124 pagesFlooding Problems and Surface Water DraiEmily WillisNo ratings yet
- Leonardo Alfonso MSCDocument112 pagesLeonardo Alfonso MSCHelder CarvalhoNo ratings yet
- Improvised Pneumatic Operated Can Crusher For Enhancing Recycling Practices 1.1Document40 pagesImprovised Pneumatic Operated Can Crusher For Enhancing Recycling Practices 1.1Andrea PepitoNo ratings yet
- Selamawit WorkinehDocument131 pagesSelamawit WorkinehDebergeNo ratings yet
- Bachelors Ordinances 2022023subsequentsessionsDocument42 pagesBachelors Ordinances 2022023subsequentsessionsahmadtaiyabiNo ratings yet
- Design, Implementation, and Evaluation of An Online Water Quality Monitoring System in Lake Saimaa, FinlandDocument118 pagesDesign, Implementation, and Evaluation of An Online Water Quality Monitoring System in Lake Saimaa, FinlandAaqib Hussain ShahNo ratings yet
- Methods For ConsiderationsDocument10 pagesMethods For ConsiderationssimbiroNo ratings yet
- Course Out Line For EIADocument2 pagesCourse Out Line For EIAAbdanur JihadNo ratings yet
- RESEARCH TOPIC EuniceDocument38 pagesRESEARCH TOPIC EuniceShariff Oluoch OdiwuorNo ratings yet
- MOB Mid AssignmentDocument11 pagesMOB Mid AssignmentMd Dewan MahmudNo ratings yet
- Proposal - Lab - Environment - G4 EditedDocument40 pagesProposal - Lab - Environment - G4 EditedMuhammad Afiq Bin KamalNo ratings yet
- Summer Traninig Report (Thangamalar)Document47 pagesSummer Traninig Report (Thangamalar)thangamalarNo ratings yet
- Water Quality WSO Student Workbook Water Supply OperationsDocument69 pagesWater Quality WSO Student Workbook Water Supply OperationsdikeNo ratings yet
- "Student Management System": Project ReportDocument32 pages"Student Management System": Project ReportDiksha KashyapNo ratings yet
- Prepare A Report On Household Water Filtera Ons UnitDocument12 pagesPrepare A Report On Household Water Filtera Ons UnitChaitanya KurandaleNo ratings yet
- Tutorial Letter 101/0/2020: Reticulation Design Management (Project)Document31 pagesTutorial Letter 101/0/2020: Reticulation Design Management (Project)sachenNo ratings yet
- Ashiedu James UcheDocument32 pagesAshiedu James UcheBelieve AdamsNo ratings yet
- Method and Practice of Community Water PurificationDocument51 pagesMethod and Practice of Community Water PurificationUsman Ahmad TijjaniNo ratings yet
- MDP PR S1G6 012021Document32 pagesMDP PR S1G6 012021Sukhjeet SinghNo ratings yet
- Design of Wastewater and Stormwater Pumping Stations MOP FD-4, 3rd EditionFrom EverandDesign of Wastewater and Stormwater Pumping Stations MOP FD-4, 3rd EditionRating: 5 out of 5 stars5/5 (1)
- Beginner's Ultimate Guide to In-Ground Pool Maintenance: Tips to Keep Your Pool SparklingFrom EverandBeginner's Ultimate Guide to In-Ground Pool Maintenance: Tips to Keep Your Pool SparklingNo ratings yet
- 1.2 Device Overall Sketch & Installation Sketch: Process DescriptionDocument1 page1.2 Device Overall Sketch & Installation Sketch: Process DescriptionSAJID SHAHIDNo ratings yet
- Orifice CalculationsDocument23 pagesOrifice CalculationsYatish Kumar Jain0% (1)
- State of The Art CCS Technologies 2022Document81 pagesState of The Art CCS Technologies 2022reda100% (1)
- Fluids Long ReportDocument23 pagesFluids Long ReportjameswattNo ratings yet
- Fluid Mechanics-1: Pressure Distribution in A FluidDocument63 pagesFluid Mechanics-1: Pressure Distribution in A FluidNoman ButtNo ratings yet
- Ex 2-Volatility Distillation and Determination of Boiling PointDocument7 pagesEx 2-Volatility Distillation and Determination of Boiling PointGlister Diadem Dollera100% (1)
- CAMEOTips - Aloha Decision KeysDocument20 pagesCAMEOTips - Aloha Decision KeysCass WillardNo ratings yet
- Gas Field Compression Jet Compressor Gea 170587Document4 pagesGas Field Compression Jet Compressor Gea 170587Ayesha ShaikhNo ratings yet
- FB Plasma NotesDocument155 pagesFB Plasma NotesNeena JanNo ratings yet
- Pro/Ii Excel - Engine Unitid XXX: NcalDocument32 pagesPro/Ii Excel - Engine Unitid XXX: NcalYves-donald MakoumbouNo ratings yet
- Chapter 2 PDFDocument14 pagesChapter 2 PDFJean-Louis KouassiNo ratings yet
- Sewer DesignDocument41 pagesSewer DesignRaihan Amzad75% (4)
- Guide For VesselDocument41 pagesGuide For Vesseltutuionut100% (1)
- Name: Balazon, Krissel Joy: Describe Changes That Happen To Water During Each ProcessDocument7 pagesName: Balazon, Krissel Joy: Describe Changes That Happen To Water During Each ProcessMyrna ParasNo ratings yet
- Design of Boiler Forced Draft FanDocument5 pagesDesign of Boiler Forced Draft FanAu TagolimotNo ratings yet
- Gravitational Settling ChambersDocument6 pagesGravitational Settling ChambersVinod KumarNo ratings yet
- Pressure Drop of Y-StrainerDocument3 pagesPressure Drop of Y-StrainerAnantawut RakkaewNo ratings yet
- Talon Bsre2-1 Eng - Utilities C2a3Document3 pagesTalon Bsre2-1 Eng - Utilities C2a3Kimberly Joy TalonNo ratings yet
- (Mech) Gland Steam SystemDocument7 pages(Mech) Gland Steam SystemThanh Son100% (2)
- Calculadora Indice de LangelierDocument6 pagesCalculadora Indice de LangelierFábio SenaNo ratings yet
- Gases: Behavior & PropertiesDocument35 pagesGases: Behavior & PropertiesAdelia SetianingsihNo ratings yet
- MIG WeldingDocument17 pagesMIG WeldingAnonymous mRBbdopMKfNo ratings yet
- Paper - Effects of Blade Number On Characteristics of Centrifugal Pumps - 2009Document6 pagesPaper - Effects of Blade Number On Characteristics of Centrifugal Pumps - 2009Lucas Rizzardi SaldanhaNo ratings yet
- Sylvania Mercury Lamps Ordering Guide Rev. 4Document2 pagesSylvania Mercury Lamps Ordering Guide Rev. 4Alan MastersNo ratings yet
- Belajar KaznaeDocument277 pagesBelajar KaznaewahyuNo ratings yet
- Agitator Power Requirement and Mixing Intensity CalculationDocument27 pagesAgitator Power Requirement and Mixing Intensity CalculationElizabeth Allende CéspedesNo ratings yet
- Unit-3 MHD Part-1Document12 pagesUnit-3 MHD Part-1amit621988No ratings yet
- Water Sampling Plan and ProcedureDocument3 pagesWater Sampling Plan and ProcedureMotenaNo ratings yet