Professional Documents
Culture Documents
Fundamentals of Nuclear Reaction
Fundamentals of Nuclear Reaction
Uploaded by
Tonmoy SinghaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Fundamentals of Nuclear Reaction
Fundamentals of Nuclear Reaction
Uploaded by
Tonmoy SinghaCopyright:
Available Formats
Fundamentals of Nuclear Reaction
Structure of atom
A A
Z X or X Z
•X is the symbol, Z is the atomic number (no of protons), A is the mass
number (no of protons and neutrons)
•The radius of a nucleus is of the order 10-16 m and that of an atom is 10-11
m.
•Neutron mass 1.674×10-27 kg, proton mass 1.673×10-27 kg and electron
mass 9.109×10-31 kg
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 2
Terminologies
• Atoms with nuclei having the same number of protons have
similar chemical and physical properties and differ mainly in
their masses. They are called isotopes.
• Natural Uranium is composed of 99.282% U238, 0.712%
U235, and 0.006% U234, atomic number 92.
• Deuterium, an isotope of hydrogen, called heavy hydrogen.
Hydrogen produce water when combined with oxygen and
Deuterium produces heavy water D2O.
• Positron is a positively charged electron, symbols e+, +1e0, or
β+. Symbol of electron e-, -1e0, or β-
• Neutrino is a tiny neutral particle ejected during nuclear
fission carries 5% of the total energy produced in fission.
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 3
Chemical and nuclear reactions
• Atoms are combined or separated in a chemical reaction,
C+O2 = CO2
Here, atoms participates as a whole and retain identity,
only the valence electrons are shared or exchanged.
The nuclei do not change.
The number of atoms in the products equals the number in
the reactants.
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 4
Nuclear reactions
• The products do not have the reactants nuclei but some other
nuclei
• The number of nucleons in the products are the same as
those in the reactants.
Z1 A A1
Z2 B A2
Z3 C A3
Z4 D A4
where, Z1 Z 2 Z 3 Z 4 and A1 A2 A3 A4
• Sometimes EM radiations (γ rays) and neutrino (ν) emitted
but do not affect the balance given above, since they carry
only energy and have zero Z and A.
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 5
Binding energy
• The sum of masses of protons and neutrons that comprises
the nucleus exceeds the mass of the atomic nucleus, this
difference in mass is called mass defect. It is converted to
energy according to
E m.C 2
• The energy associated with the mass defects is called
binding energy of the nucleus.
• 1g of mass is equivalent to 9×1013J and 1 amu equals
14.94×10-11J = 9.31×108 eV = 931 MeV.
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 6
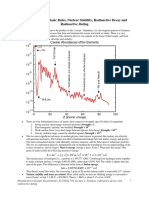
Radioactive decay and half life
• Most isotopes that occur in nature is stable, isotopes of heavier
elements e.g. polonium are not stable, the binding energy per
nucleon being small, and emits radiation until a more stable
nucleus is reached.
• This spontaneous disintegration process is called radioactive
decay.
• The resulting nucleus is called the daughter and the original is
called the parent.
• The daughter may be stable or radioactive
• Radioactive isotopes both natural and man made are called
radioisotopes.
• Radioactivity is always accompanied by a decrease of mass or
liberation of energy.
• The energy emits in the form of KE of the emitted particles and as
EM radiation.
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 7
Radioactive half life
If N be the number of radioactive atoms of one species at any
time t, the rate of decay
dN
N
dt
Where λ is decay constant. By integrating we get
N N 0 e t
Where, N0 is the radioactive atoms at time t=0 and N is the
radioactive atoms at time t.
N 1 ln 2 0.6931
e t1 / 2 t1/ 2
N0 2
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 8
Half life of some radioisotopes
isotopes t1/2 Activity
Uranium 233 1.65×105 yr β and γ
Uranium 235 7.1×108 yr α and γ
Uranium 238 4.51×109 yr α and γ
Plutonium 239 2.44×104 yr α and γ
Thorium 232 1.41×1010 yr β
Thorium 233 22.1 min α and γ
Radium 223 11.43 days α and γ
The unit of radioactivity is curie (Ci) equal 3.615× 1010 dis/s, SI unit is becquerel
(Bq) equals 1 dis/s
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 9
Emissions from isotopes
• Alpha decay- alpha particles are helium nuclei, alpha decay
accompanied by γ radiation
94 Pu
239
92 U 235 2 He 4
• Beta decay- emission of an electron and raises the atomic
number by one while the mass number remains the same.
82 Pb
214
83 Bi 214 1 e 0
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 10
Emissions from isotopes
• Gamma radiation- em radiation of short wavelength and high
energy, it originates from the nucleus while X-rays originate
from the atom as the orbital electrons changing energy labels
or shells.
Z
A *
X A
Z X
• Positron decay- when a radioactive nucleus contains an
excess of protons, positron (positive beta) decay occurs,
converting a proton into a neutron.
15 P 30
14 Si 30
1 e 0
• Daughter nucleus has one less proton, to maintain charge
neutrality one orbital electron has to be released.
1 e 0
1 e 0
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 11
Emission from isotopes
• K Capture- when a nucleus has an excess of proton but does not have the
threshold energy to emit a positron, it captures an orbital electron from
the K shell. A proton in the nucleus changes into a neutron by K capture.
The vacancy in the k shell is filled by another electron falling from the
higher orbit.
• K capture is accompanied by x ray emission from the atom.
29 Cu 1 e 28 Ni
64 0 64
• Neutron emission- If a nucleus possesses an extremely high excitation
energy it may emit a neutron.
54 Xe 54 Xe 0 n
137 136 1
2/7/2024 Professor Dr S M Abdur Razzak, EEE, RUET 12
You might also like
- Nuclear Medicine Physics The Basics 6 Edn (2004) PDFDocument190 pagesNuclear Medicine Physics The Basics 6 Edn (2004) PDFseda oktriyani0% (1)
- Pool Canvas: Creation SettingsDocument9 pagesPool Canvas: Creation SettingsVlad Calaunan LugoNo ratings yet
- Lecture 36Document27 pagesLecture 36Jaya SharmaNo ratings yet
- Introduction To Nuclear Chemistry: September 2020Document45 pagesIntroduction To Nuclear Chemistry: September 2020Hesham IbrahemNo ratings yet
- Lec - Nuclear-PhysicsDocument49 pagesLec - Nuclear-PhysicsNovi Ann Yap LiboonNo ratings yet
- Nuclear Chemistry: The BasicsDocument44 pagesNuclear Chemistry: The BasicsRahul SainiNo ratings yet
- Dasar-Dasar Radiofarmasi: Lecturer: Prof. Resmi Mustarichie MSC, PH.D, AptDocument25 pagesDasar-Dasar Radiofarmasi: Lecturer: Prof. Resmi Mustarichie MSC, PH.D, Aptkiki ikrimaNo ratings yet
- Fisika Inti Nuclear PhysicsDocument35 pagesFisika Inti Nuclear PhysicsRian Galileo MawonNo ratings yet
- Nuclear PowerplantDocument35 pagesNuclear Powerplantasuf skakNo ratings yet
- Lesson 4 Nuclear ChemistryDocument7 pagesLesson 4 Nuclear ChemistrySheilora DagusNo ratings yet
- Atomic Nucleus NucleusDocument11 pagesAtomic Nucleus NucleusAbrar Mubasshir RahmanNo ratings yet
- Nuclear Chemistry: P. Nagaraja Assistant Professor in Chemistry Ap Iiit, RK Valley, RguktDocument27 pagesNuclear Chemistry: P. Nagaraja Assistant Professor in Chemistry Ap Iiit, RK Valley, RguktGORIPARTHI PENCHALA PRASADNo ratings yet
- Review 402Document50 pagesReview 402Serdar CharyyevNo ratings yet
- 22 NuclearDocument24 pages22 Nuclearosborne kachajeNo ratings yet
- Nuclear Physics - Sample MaterialDocument13 pagesNuclear Physics - Sample MaterialUma SinghNo ratings yet
- 9-Lecture 9Document29 pages9-Lecture 9george.1023077No ratings yet
- Nuclear ChemistryDocument42 pagesNuclear ChemistryJann Romene Decena100% (1)
- Radiation PhysicsDocument38 pagesRadiation PhysicsMaheshwar KumarNo ratings yet
- Atoms Nucleus and Radiation - To Be Filled inDocument15 pagesAtoms Nucleus and Radiation - To Be Filled inNick RandazzoNo ratings yet
- Lecture 1Document33 pagesLecture 1fatimaasiriNo ratings yet
- Introduction To RadiochemistryDocument28 pagesIntroduction To RadiochemistryAfifah NadiaNo ratings yet
- Nuclei Chapter 12 Class12 PhysicsDocument30 pagesNuclei Chapter 12 Class12 Physicsharikeshsingh45497No ratings yet
- Lecture 9Document19 pagesLecture 9WilliamNo ratings yet
- CHEM1104 Nuclear ChemistryDocument46 pagesCHEM1104 Nuclear ChemistryPaul Jhon EugenioNo ratings yet
- Atomic Structure - Dr. MahbubDocument50 pagesAtomic Structure - Dr. Mahbubfarhanfuad220534No ratings yet
- VIP Nuclear BasicsDocument166 pagesVIP Nuclear BasicsgetachewNo ratings yet
- Nuclear Power Engineering: Aiub Dr. M. Tanseer Ali NPWR Lec 03 /1Document43 pagesNuclear Power Engineering: Aiub Dr. M. Tanseer Ali NPWR Lec 03 /1Naushed NihalNo ratings yet
- Nuclear ChemistryDocument78 pagesNuclear Chemistrynagendra_rdNo ratings yet
- CFELabA1 Nuclear ChemistryDocument88 pagesCFELabA1 Nuclear ChemistryAllyson OrpiaNo ratings yet
- Nuclear Physics: Powerpoint Lectures ForDocument25 pagesNuclear Physics: Powerpoint Lectures ForRns Naresh KumarNo ratings yet
- Nuclear Power PlantsDocument107 pagesNuclear Power PlantsManvendra Pratap Singh Bisht50% (2)
- Atomic Nucleus and RadioactivityDocument59 pagesAtomic Nucleus and Radioactivitymladen lakic100% (2)
- Particle Physics 221216 121646Document44 pagesParticle Physics 221216 121646Nur Ainna LiyanaNo ratings yet
- Physics NuclearDocument26 pagesPhysics NuclearPASSAWAT BOONCHITSIRIKULNo ratings yet
- General Physics IIDocument20 pagesGeneral Physics IINasrinTalebpourNo ratings yet
- Nuclear Physics: An OverviewDocument29 pagesNuclear Physics: An OverviewabdulbaseerNo ratings yet
- Nuclear PhysicsDocument46 pagesNuclear PhysicsAmit PaulNo ratings yet
- Nuclear Power Engineering: Aiub Dr. M. Tanseer Ali NPWR Lec 02 /1Document20 pagesNuclear Power Engineering: Aiub Dr. M. Tanseer Ali NPWR Lec 02 /1Naushed NihalNo ratings yet
- Nuclear Chemistry: Symbol or Symbol - Mass #Document18 pagesNuclear Chemistry: Symbol or Symbol - Mass #simpanan hitamNo ratings yet
- Nuclear PhysicsDocument24 pagesNuclear PhysicsCalvin LabialNo ratings yet
- CFE - 11 Nuclear ChemistryDocument37 pagesCFE - 11 Nuclear ChemistryChristian C. Suase100% (1)
- Nuclear ExitDocument35 pagesNuclear Exitdavididosa40No ratings yet
- Atomic Structure PDFDocument40 pagesAtomic Structure PDFAnkit MaanNo ratings yet
- TRM 12Document96 pagesTRM 12KayKhaing OoNo ratings yet
- (NIT Rourkela) Nuclear PhysicsDocument46 pages(NIT Rourkela) Nuclear PhysicsAditya JagadalaNo ratings yet
- The Atomic NucleusDocument85 pagesThe Atomic NucleusARUNA RESHMINo ratings yet
- Nuclear PhysicsDocument42 pagesNuclear PhysicsJennie Rose KomalaNo ratings yet
- 12.744/12.754 The Basic Rules, Nuclear Stability, Radioactive Decay and Radioactive DatingDocument8 pages12.744/12.754 The Basic Rules, Nuclear Stability, Radioactive Decay and Radioactive Datingshawnah jamesNo ratings yet
- 13 ChangesInTheNucleus 2bDocument14 pages13 ChangesInTheNucleus 2bmainethemaineNo ratings yet
- Nuclear Chemistry: Topic 3bDocument50 pagesNuclear Chemistry: Topic 3bJeam Russell AlfaroNo ratings yet
- Radiation Detection and Measurement: Tuesday September 12, 2017Document33 pagesRadiation Detection and Measurement: Tuesday September 12, 2017hannan alnziNo ratings yet
- Radioactivity: Background RadiationDocument13 pagesRadioactivity: Background RadiationSuresh SenanayakeNo ratings yet
- Chapter 17: Nuclear PhysicsDocument30 pagesChapter 17: Nuclear PhysicsMukit Hasan JimNo ratings yet
- CBSE Class 11 Chemistry Notes: Nuclear Chemistry: Nucleons and Nuclear ForcesDocument11 pagesCBSE Class 11 Chemistry Notes: Nuclear Chemistry: Nucleons and Nuclear ForcesAjay Kumar GantiNo ratings yet
- BCHEM 264 Mar. 05, 2015 Radioisotope Techniques The Nature of Radioactivity Atomic StructureDocument16 pagesBCHEM 264 Mar. 05, 2015 Radioisotope Techniques The Nature of Radioactivity Atomic StructureDuodu StevenNo ratings yet
- Topic 1 Basic Physics For Nuclear Medicine PDFDocument66 pagesTopic 1 Basic Physics For Nuclear Medicine PDFJeeShaoMinNo ratings yet
- Neutronlvhfgth 1561665Document12 pagesNeutronlvhfgth 1561665johnny deppNo ratings yet
- Atomic Structure NotesDocument8 pagesAtomic Structure NotesAayNo ratings yet
- RevisionDocument13 pagesRevisionrajayush67780No ratings yet
- NuclearDocument117 pagesNuclearsarfaraz khanNo ratings yet
- Chapter 11 Nuclear StructureDocument38 pagesChapter 11 Nuclear StructureAimi NabilaNo ratings yet
- Inverse Square LawDocument70 pagesInverse Square LawEngr Rab Nawaz ButtNo ratings yet
- Chapter 23 Nuclear ChemistryDocument44 pagesChapter 23 Nuclear ChemistryArienta NiadisyaNo ratings yet
- Radioisotope TechniquesDocument63 pagesRadioisotope TechniquesPallavi Kanungo UpritNo ratings yet
- Handout of Physical Chemistry: Course: Vikaas (Ja) Target: Jee (Advanced)Document5 pagesHandout of Physical Chemistry: Course: Vikaas (Ja) Target: Jee (Advanced)Naunidh Singh MadhokNo ratings yet
- Chapter 3 Nuclear Chemistry and EnergyDocument54 pagesChapter 3 Nuclear Chemistry and EnergyBruce MendozaNo ratings yet
- Radproduction Chapter 2-9Document276 pagesRadproduction Chapter 2-9Christian DioNo ratings yet
- The Stark Arc ReactorDocument7 pagesThe Stark Arc ReactorKleberMottaNo ratings yet
- Nuclear and Radioactivity NotesDocument41 pagesNuclear and Radioactivity NotesDamesha ManoharanNo ratings yet
- PC Chapter 44Document85 pagesPC Chapter 44ultimuNo ratings yet
- Module 3Document24 pagesModule 3Jessalyn PaclebNo ratings yet
- Errata To "Introduction To Nuclear Engineering", 3rd Edition (Lamarsh)Document4 pagesErrata To "Introduction To Nuclear Engineering", 3rd Edition (Lamarsh)Jose MarquesNo ratings yet
- Nuclear Stability and RadioactivityDocument35 pagesNuclear Stability and RadioactivityAmirHakimRusliNo ratings yet
- Neutrons Are Not Fundamental ParticlesDocument8 pagesNeutrons Are Not Fundamental ParticlesKeith D. FooteNo ratings yet
- NCERT Solutions Physics Chapter 13 NucleiDocument16 pagesNCERT Solutions Physics Chapter 13 NucleiVidyakulNo ratings yet
- UNIT 14-PHY 131-Chapter 29-Nuclear PhysicsDocument23 pagesUNIT 14-PHY 131-Chapter 29-Nuclear Physicsmakhobasibusiso89No ratings yet
- Radiation Safety Officers Handbook A PDFDocument100 pagesRadiation Safety Officers Handbook A PDFAlejandro Zubiate100% (1)
- Unit 9Document26 pagesUnit 9Amol ShindeNo ratings yet
- Nuclear Binding EnergyDocument7 pagesNuclear Binding EnergyWhite HeartNo ratings yet
- ZChapter18 Nuclear ChemistryDocument15 pagesZChapter18 Nuclear ChemistryViha AncilliaNo ratings yet
- CH186 Nuclear Chemistry Exam Questions From Spring 2001 SemesterDocument11 pagesCH186 Nuclear Chemistry Exam Questions From Spring 2001 SemesterAllizwell OSNo ratings yet
- Chapter 19: The Nucleus: A Chemist's ViewDocument13 pagesChapter 19: The Nucleus: A Chemist's ViewIron ManNo ratings yet
- Nuclear Energy (Radioactivity, Nuclear Fission and Fusion)Document14 pagesNuclear Energy (Radioactivity, Nuclear Fission and Fusion)Mohammad Irfan YousufNo ratings yet
- Environmental Isotopes in The Hydrological Cycle Vol 1 PDFDocument185 pagesEnvironmental Isotopes in The Hydrological Cycle Vol 1 PDFNoreida PRIETO MILLAN100% (1)
- Chapter 19 - New PDFDocument4 pagesChapter 19 - New PDFalaa al sahmaraniNo ratings yet
- Chem Lec ReviewerDocument16 pagesChem Lec ReviewerSophia EspirituNo ratings yet
- Rso PDFDocument120 pagesRso PDFjohn shepardNo ratings yet
- 2022 FDG-PET-CT and PET-MR in CVDsDocument382 pages2022 FDG-PET-CT and PET-MR in CVDsEnzo Tejada Rivera100% (1)
- S11 12PS-IIIa-2 Star Formation and EvolutionDocument18 pagesS11 12PS-IIIa-2 Star Formation and EvolutionMarArizala0% (1)