Professional Documents
Culture Documents
Divalproex Tablets 500 MG Dissolution Data
Divalproex Tablets 500 MG Dissolution Data
Uploaded by
syedburhanuddinmoin0 ratings0% found this document useful (0 votes)
5 views1 pageThis document summarizes dissolution test results for 3 validation batches of 500 mg Divalproex Sodium ER Tablets at different time points. The results are reported as a percentage of drug released and meet the release specification of 15-35% at 2 hours, 40-65% at 6 hours, 65-90% at 12 hours, and not less than 80% at 24 hours. All 30 individual results from the 3 validation batches were within the release specifications at each time point tested.
Original Description:
500 mg parameters
Original Title
Copy of Divalproex tablets 500 mg dissolution data
Copyright
© © All Rights Reserved
Available Formats
XLSX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document summarizes dissolution test results for 3 validation batches of 500 mg Divalproex Sodium ER Tablets at different time points. The results are reported as a percentage of drug released and meet the release specification of 15-35% at 2 hours, 40-65% at 6 hours, 65-90% at 12 hours, and not less than 80% at 24 hours. All 30 individual results from the 3 validation batches were within the release specifications at each time point tested.
Copyright:
© All Rights Reserved
Available Formats
Download as XLSX, PDF, TXT or read online from Scribd
Download as xlsx, pdf, or txt
0 ratings0% found this document useful (0 votes)
5 views1 pageDivalproex Tablets 500 MG Dissolution Data
Divalproex Tablets 500 MG Dissolution Data
Uploaded by
syedburhanuddinmoinThis document summarizes dissolution test results for 3 validation batches of 500 mg Divalproex Sodium ER Tablets at different time points. The results are reported as a percentage of drug released and meet the release specification of 15-35% at 2 hours, 40-65% at 6 hours, 65-90% at 12 hours, and not less than 80% at 24 hours. All 30 individual results from the 3 validation batches were within the release specifications at each time point tested.
Copyright:
© All Rights Reserved
Available Formats
Download as XLSX, PDF, TXT or read online from Scribd
Download as xlsx, pdf, or txt
You are on page 1of 1
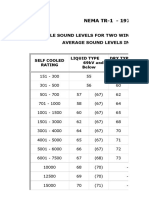
Divalproex Sodium ER Tablets USP 500 mg
Dissolution medium: pH 6.8 phosphate Buffer with 2% SLS
Release Specification (L1 criteria): 2hrs: 15 to 35%, 6hrs: 40-65%, 12hrs: 65-90%, 24hrs: NLT 80%
1st Validation batch 2nd Validation batch 3rd Validation batch
Time point B. No. DSB19001 DSB19001A DSB19001B DSB20001 DSB20001A DSB20001B DSB20002 DSB20002A DSB20002B
Lot1 (L3) Lot2 (L3) (100's count) (500's count) Lot1 (L2) Lot2 (L1) (100's count) (500's count) Lot1 (L2) Lot2 (L2) (100's count) (500's count)
63% (58-70) 65% (56-72) 69% (63-73) 67% (62-71) 69% (62-77) 66% (65-68) 68% (64-73) 71% (63-79) 65% (58-70) 65% (61-74) 66% (61-70) 67% (59-72)
(67,62,60,63,61,67, (67,62,56,62,67,61, (71,73,65,71,71,67, (71,69,69,68,67,66, (67,62,72,69,67,69, (66,65,66,67,65,68) (68,68,73,67,69,66, (79,70,71,75,70,73, (60,67,68,62,67,67, (62,62,61,65,62,62, (69,68,68,67,65,65, (62,64,70,70,69,70,
12th Hour 64,61,67,67,70,64, 58,66,58,62,58,62, 72,67,65,67,63,72) 69,62,63,66,71,67) 69,66,66,71,72,77) 64,71,68,69,66,65) 66,63,63,68,77,74) 62,69,70,63,69,58) 74,62,70,66,62,66) 67,61,61,65,70,66) 59,59,69,70,72,67)
58,58,63,63,64,62, 71,65,70,59,68,68,
59,59,65,60,62,65) 70,69,68,69,72,62)
You might also like
- Dival Discussion WordDocument1 pageDival Discussion WordsyedburhanuddinmoinNo ratings yet
- PRT - 02 (27-02-2024) SolDocument14 pagesPRT - 02 (27-02-2024) Solsarthakyedlawar04No ratings yet
- Unexpected Molecular Sieving Properties of Zeolitic Imidazolate Framework-8Document34 pagesUnexpected Molecular Sieving Properties of Zeolitic Imidazolate Framework-8brianNo ratings yet
- Supplementary Material For Multi-Faceted Distillation of Base-Novel Commonality For Few-Shot Object DetectionDocument6 pagesSupplementary Material For Multi-Faceted Distillation of Base-Novel Commonality For Few-Shot Object DetectionAli Eren KaradağNo ratings yet
- SS - FTS - 70 (Online) - (Main) C - 2021-06-13 - 2020 - ADocument9 pagesSS - FTS - 70 (Online) - (Main) C - 2021-06-13 - 2020 - AAbhiNo ratings yet
- Word AsistensiDocument2 pagesWord AsistensikegitankuliahbisaNo ratings yet
- SS-JCT - Unit Test-5 - (Code-A) - 27-08-2022 - SolutionsDocument11 pagesSS-JCT - Unit Test-5 - (Code-A) - 27-08-2022 - Solutionsmohitabochare2039No ratings yet
- Test - 30: Final Test Series (Online) For JEE (Main) - 2021Document9 pagesTest - 30: Final Test Series (Online) For JEE (Main) - 2021Vishal kumar MauryaNo ratings yet
- Test - 03 - AIATS - JEE (M) - 2024 - FS - GR - 01 & 02 - (Code-A) - 29-01-2023 - Sol.Document12 pagesTest - 03 - AIATS - JEE (M) - 2024 - FS - GR - 01 & 02 - (Code-A) - 29-01-2023 - Sol.A S MOHAMED ASILNo ratings yet
- المحاسبة الاداريةDocument307 pagesالمحاسبة الاداريةSami AhmedNo ratings yet
- Test 03 AIATS JEE (M) 2023 FS (Code-A) 19-12-2021 Sol.Document11 pagesTest 03 AIATS JEE (M) 2023 FS (Code-A) 19-12-2021 Sol.Akshay KumarNo ratings yet
- RTS - Demo Paper-2 - JEE Mains - SolutionsDocument11 pagesRTS - Demo Paper-2 - JEE Mains - Solutionsgyanchandrapradhan711No ratings yet
- Audible Sound Levels For Two Winding Transformers Average Sound Levels in DeciblesDocument4 pagesAudible Sound Levels For Two Winding Transformers Average Sound Levels in DeciblesipraoNo ratings yet
- SPC Example 1Document5 pagesSPC Example 1christliu1024No ratings yet
- 30-09-2023 - SR - Super60 - NUCLEUS & STERLING - BT - Jee-Main-RPTM-09 - KEY & Sol'SDocument14 pages30-09-2023 - SR - Super60 - NUCLEUS & STERLING - BT - Jee-Main-RPTM-09 - KEY & Sol'Stmyalt0No ratings yet
- 21-01-2023 - SR - Super60 - NUCLEUS & ALL - BT - Jee-Main-GTM-11 - KEY & Sol'SDocument20 pages21-01-2023 - SR - Super60 - NUCLEUS & ALL - BT - Jee-Main-GTM-11 - KEY & Sol'SkasalachinuNo ratings yet
- Nilai Ujian Mata Pelajaran Ipa Dari 100 Siswa Kelas Xi Nama: Rifatun Nisak Kelas: MPI B3Document5 pagesNilai Ujian Mata Pelajaran Ipa Dari 100 Siswa Kelas Xi Nama: Rifatun Nisak Kelas: MPI B3Rifatun NisakNo ratings yet
- Aiats-3 Ss Xii Jee Main Test-03!20!11-2022 SolDocument14 pagesAiats-3 Ss Xii Jee Main Test-03!20!11-2022 SolAditya PetkarNo ratings yet
- 25-11-2023 - SR - Super60 - NUCLEUS & STERLING - BT - Jee-Main-RPTM-16 - KEY & Sol'SDocument18 pages25-11-2023 - SR - Super60 - NUCLEUS & STERLING - BT - Jee-Main-RPTM-16 - KEY & Sol'Snareshsuja123No ratings yet
- MTA 7024: Set Up Product Qualityand Standards Process Nama: HistogramDocument1 pageMTA 7024: Set Up Product Qualityand Standards Process Nama: HistogramezziatunNo ratings yet
- Abu Batubara Tanpa Aktivasi - Theta - 2-Theta SmoothingDocument23 pagesAbu Batubara Tanpa Aktivasi - Theta - 2-Theta SmoothingNata NataNo ratings yet
- All India Aakash Test Series For JEE (Main) - 2021: Mock Test-12 Mock Test-6Document14 pagesAll India Aakash Test Series For JEE (Main) - 2021: Mock Test-12 Mock Test-6sandeepNo ratings yet
- SCORE300 Test Series-2024 - T02 (Code-A) - SolutionDocument17 pagesSCORE300 Test Series-2024 - T02 (Code-A) - Solutiondummymail6383No ratings yet
- Asis 1Document2 pagesAsis 1kegitankuliahbisaNo ratings yet
- Matriks Similaritas - Fenogram - Laporan Sementara - SisHew IIDocument2 pagesMatriks Similaritas - Fenogram - Laporan Sementara - SisHew IIBerliana AmeyliaNo ratings yet
- Unit Test For First Step (Group 1) 2025 - T01 (Code A) - 07 06 2023 - Sol.Document12 pagesUnit Test For First Step (Group 1) 2025 - T01 (Code A) - 07 06 2023 - Sol.RAJANI SAINo ratings yet
- 19 07 2021 SR Super60 & ICON All Jee Main GTM 40 Key & Sol'sDocument16 pages19 07 2021 SR Super60 & ICON All Jee Main GTM 40 Key & Sol'sTejas MagguNo ratings yet
- STC Denr Memo - Npcgazet - Philippine Local Codal RequirementDocument1 pageSTC Denr Memo - Npcgazet - Philippine Local Codal RequirementRoland ArimadoNo ratings yet
- हरियाणा ज्ञान दर्शन (virender) 8700178026Document234 pagesहरियाणा ज्ञान दर्शन (virender) 8700178026Virender Bhardwaj100% (3)
- TStsoln 3Document14 pagesTStsoln 3Vinay BelekarNo ratings yet
- 31-12-2022 - SR - Super60 - NUCLEUS & STERLING - BT - Jee-Main-PTM-16 - KEY & Sol'SDocument12 pages31-12-2022 - SR - Super60 - NUCLEUS & STERLING - BT - Jee-Main-PTM-16 - KEY & Sol'SSameena LoniNo ratings yet
- Test 01 AIATS JEEM 2025 FS Phase 1 Code B 27 08 2023 Sol 1Document12 pagesTest 01 AIATS JEEM 2025 FS Phase 1 Code B 27 08 2023 Sol 1singh4567tarunNo ratings yet
- Test - 02 - AIATS - JEE (M) - 2024 - FS - GR - 01 - 02 - (Code-A) - 11-12-2022 - SolDocument13 pagesTest - 02 - AIATS - JEE (M) - 2024 - FS - GR - 01 - 02 - (Code-A) - 11-12-2022 - Solujjwalsingh947.kv2No ratings yet
- FS-2 - AIATS - 07 (Main) - A - 2021-03-28 - 2020 - ADocument14 pagesFS-2 - AIATS - 07 (Main) - A - 2021-03-28 - 2020 - ATachyonNo ratings yet
- Considering: Rec. ITU-R M.493-10 1Document29 pagesConsidering: Rec. ITU-R M.493-10 13 GamerNo ratings yet
- JO HSE 028 Noise Monitoring ReportDocument2 pagesJO HSE 028 Noise Monitoring ReportIvankaNo ratings yet
- GR&R FormDocument1 pageGR&R Formoscarho2004No ratings yet
- Monitoring of Noise Levels at Selected Cities in North IndiaDocument9 pagesMonitoring of Noise Levels at Selected Cities in North IndiacpcbsktNo ratings yet
- SS - FTS - 68 (Online) - (Main) C - 2021-06-09 - 2020 - ADocument10 pagesSS - FTS - 68 (Online) - (Main) C - 2021-06-09 - 2020 - AAbhiNo ratings yet
- SS - AIATS - 11 (Main) - A - 2021-03-28 - 2020 - ADocument13 pagesSS - AIATS - 11 (Main) - A - 2021-03-28 - 2020 - Aasdfghzxchtfc0987No ratings yet
- Data Untuk SPSSDocument21 pagesData Untuk SPSSAhmad FahruddinNo ratings yet
- Mock Test 02 - (Hints & Solution) - Lakshya JEE 2024Document11 pagesMock Test 02 - (Hints & Solution) - Lakshya JEE 2024Akash KumarNo ratings yet
- JEEM Mock 2 SolDocument11 pagesJEEM Mock 2 SolYuvarajNo ratings yet
- Test - 32: Final Test Series (Online) For JEE (Main) - 2021Document9 pagesTest - 32: Final Test Series (Online) For JEE (Main) - 2021Vishal kumar Maurya100% (2)
- 23Document12 pages23Mohamed HamdallahNo ratings yet
- 23Document12 pages23Mohamed HamdallahNo ratings yet
- L - L 20 Log R RDocument12 pagesL - L 20 Log R RMohamed HamdallahNo ratings yet
- Performance of A Double-Pipe Heat Exchanger Experiment: Group 7Document10 pagesPerformance of A Double-Pipe Heat Exchanger Experiment: Group 7RonaldNo ratings yet
- 26-08-23 JR - Super60 Nucleus BT Jee-Main Wtm-06 Key & Sol'sDocument12 pages26-08-23 JR - Super60 Nucleus BT Jee-Main Wtm-06 Key & Sol'sSanthosh KasulavadhaNo ratings yet
- SS - FTS - 85 (Online) - Main (C) - 2021-07-19 - 2020 - ADocument13 pagesSS - FTS - 85 (Online) - Main (C) - 2021-07-19 - 2020 - ADhirendra Pratap SinghNo ratings yet
- FTS 06 Code A SolutionDocument26 pagesFTS 06 Code A SolutionSachin VermaNo ratings yet
- FTS - Test-2 - (Code-B) - JEE (Main) - 2023 (27-12-2022) Sol-1Document12 pagesFTS - Test-2 - (Code-B) - JEE (Main) - 2023 (27-12-2022) Sol-1Anoushka KanchanNo ratings yet
- SM N9208 Pspec 2Document37 pagesSM N9208 Pspec 2psugiartonoNo ratings yet
- Lighting SpecificationDocument89 pagesLighting SpecificationGalfarqatar MEP100% (1)
- 1 SSS 1 Art Grade Sheet Cont ResentDocument89 pages1 SSS 1 Art Grade Sheet Cont ResentAbubakarr SwarayNo ratings yet
- Effect of Roasting Condition On The Physicochemical Properties of Rice Flour and The Quality Characteristics of Tara UkDocument9 pagesEffect of Roasting Condition On The Physicochemical Properties of Rice Flour and The Quality Characteristics of Tara Ukgio ferryNo ratings yet
- All India Aakash Test Series For JEE (Main) - 2020Document12 pagesAll India Aakash Test Series For JEE (Main) - 2020deepNo ratings yet
- Scts 2Document15 pagesScts 2KillerpkNo ratings yet
- TABELA DO EMS 55 AdvancedDocument2 pagesTABELA DO EMS 55 AdvancedFabio MagalhãesNo ratings yet
- The JPEG 2000 SuiteFrom EverandThe JPEG 2000 SuitePeter SchelkensNo ratings yet
- Divalproex Tablets 250mg Process ParametersDocument3 pagesDivalproex Tablets 250mg Process ParameterssyedburhanuddinmoinNo ratings yet
- API Excip PackagingDocument13 pagesAPI Excip PackagingsyedburhanuddinmoinNo ratings yet
- BudgetDocument2 pagesBudgetsyedburhanuddinmoinNo ratings yet
- Budget US+ QueryDocument6 pagesBudget US+ QuerysyedburhanuddinmoinNo ratings yet
- CostingDocument2 pagesCostingsyedburhanuddinmoinNo ratings yet
- Facility CostingDocument10 pagesFacility CostingsyedburhanuddinmoinNo ratings yet
- Injection ProposalDocument15 pagesInjection ProposalsyedburhanuddinmoinNo ratings yet