Professional Documents
Culture Documents
9) Paper Chromatography
9) Paper Chromatography
Uploaded by
TushitaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
9) Paper Chromatography
9) Paper Chromatography
Uploaded by
TushitaCopyright:
Available Formats
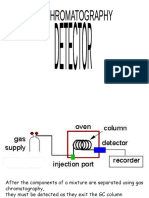
PAPER CHROMATOGRAPHY
- Chromatography is a family of analytical chemistry techniques for the separation of
mixtures.
- It was the Russian botanist Mikhail Tsvet (Mikhail Semyonovich Tsvet) who invented the
first chromatography technique in 1901.
- The separation of molecules depends on differences of 1- size 2- shape 3- mass 4- charges
5- solubility and 6- adsorption.
Types of Chromatography:
1- Adsorption chromatography.
2- Partition chromatography e.g. paper chromatography
3- Gel-filtration chromatography.
Uses of chromatography:
- Government laboratories used to check
for approved dyes in food
that vegetables contained tiny amounts of pesticides and herbicides
Advantages of using chromatography:
1. Require very minute amount for identification.
2. Can be used to identify substances that cannot be easily melted or distilled.
1 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
All types of chromatography involve interaction between:
1- The mixture to be separated.
2- The stationary phase.
3- The mobile phase.
Principle of Paper Chromatography:
- Method of separating and identifying both colored and colorless mixtures.
- Mixtures can be solids, liquids or gases but their components must be able to dissolve in
the same solvent to different extents.
- Generally involves 2 phases:
stationary phase solid support e.g. water on paper
mobile phase solvent or a gas
- Test mixture is applied onto the chromatography paper and a solvent is then allowed to
pass over the paper. As the solvent does so, the components of the mixture travel along with
it.
- The stationary phase retards the passage of the components of the sample. When
components pass through the system at different rates they become separated in time.
The solvent used depends on the substance to be separated
The components will travel at different rates over paper depending on:
their solubility in the solvent
how well the dyes adsorb on the chromatography paper
Generally, the more soluble the component is in the solvent and the less it adsorb onto the
chromatography paper, the faster it would move with the solvent on the paper and hence
the spot appears further up the paper
2 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
Result of chromatography is known as the >>>> Chromatogram
Types of Paper Chromatography:
- There are three types of paper chromatography:
I- Ascending Paper Chromatography:
- Solvent running up the paper by capillary action.
II- Descending Paper Chromatography:
- Solvent running down the paper by both capillary action and gravity.
Advantage of the descending method over the ascending method:
- Good for long pieces of paper thus better separation.
- Aided by gravity thus faster.
III- Two-Dimensional Paper Chromatography:
- The mixture is separated in the first solvent which should be volatile then after drying the
paper is turned through 90 and separation is carried out in the second solvent. After
location, a map is obtained and compounds can be identified by comparing their position
with a map of known compounds developed under the same conditions.
Stationary Phase:
- In paper chromatography, cellulose in the form of paper sheets makes an identical
support medium. >>>> WHY?
- Because it has the ability to adsorb water molecules between cellulose fibers and forms a
stationary hydrophilic phase.
3 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
- Paper: Watman No. 1 of high quality is the paper most frequently used for analytical
purposes.
Mobile Phase:
- In paper chromatography, mobile phase is a mixture of solvents.
- The choice of solvent depends on the mixture investigated:
1- If the compounds move close to solvent (A) front >>>>> these compounds are highly
soluble in solvent A
2- If the compounds are crowded around the origin >>>>> these compounds are not
sufficiently soluble in solvent B.
Therefore, a suitable solvent for separation would be an appropriate mixture of both
solvent A & B. As a result R f values of the components of the mixture are spread across
the length of the paper.
Retention Factor(R f ):
- The retention is measured as the retention factor Rf, the run length of the compound
divided by the run length of the solvent front:
- Unknown substances could be identified by the Rf values
Rf = dist. Moved by the substance
dist. Moved by the solvent
- The Rf of a compound often differs considerably between experiments and
laboratories due to variations of the solvent, the stationary phase, temperature, and the
4 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
setup. It is therefore important to compare the retention of the test compound to that of
one or more standard compounds under absolutely identical conditions.
Detection of Spots:
- After development, the spots corresponding to different compounds may be located by
their color
- However, most compounds are colorless and are visualized by:
1- Spraying the paper with specific reagents.
2- Dipping the paper in a solution of the reagent in a volatile solvent.
3- Fluorescent substances can be visualized by ultraviolet (UV) light.
SEPARATION OF AMINO ACIDS BY
PAPER CHROMATOGRAPHY
- Separation and identification of amino acids are operations that must be performed
frequently by biochemists. The 20 amino acids present in proteins have similar structures.
5 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
However, each amino acid is unique in polarity and ionic characteristics. In this
experiment, we will use paper chromatography to separate and identify the components of
an unknown amino acid mixture.
- The solvent mixture contains several components, one of which is usually water and
another of which is a more non-polar solvent. As the solvent mixture moves up the paper
by capillary action, the water in the mixture binds to the hydrophilic paper (cellulose) and
creates a liquid stationary phase of many small water droplets. The non-polar solvent
continues to move up the paper forming a liquid mobile phase. Since amino acids have
different R-groups, they also have different degrees of solubility in water vs. the non-polar
solvent. An amino acid with a polar R-group will be more soluble in water than in the non-
polar solvent, so it will dissolve more in the stationary water phase and will move up the
paper only slightly. An amino acid with a hydrophobic R-group will be more soluble in the
mobile non-polar solvent than in water, so it will continue to move up the paper. Different
amino acids will move different distances up the paper depending upon their relative
solubilities in the two solvents, allowing for separation of amino acid mixtures.
- The movement of amino acids can be defined by a quantity known as Rf value, which
measures the movement of an amino acid compared to the movement of the solvent. At the
start of the chromatography, the amino acid is spotted at what is called the origin. The
chromatography is then performed, and the procedure is stopped before the solvent runs
all the way up the paper. The level to which the solvent has risen is called the solvent front.
The Rf value of an amino acid is the ratio of the distance traveled by the amino acid from
the origin to the distance traveled by the solvent from the origin.
- Since Rf value for an amino acid is constant for a given chromatography system, an
unknown amino acid can be identified by comparing its Rf value to those of known amino
acids.
Application:
Materials:
1- Filter paper: Watman No.1.
2- Solvent system: Butanol: glacial acetic acid: water.
6 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
3- Ninhydrine reagent.
4- Standard amino acids and mixture of unknown.
Procedure:
1- Draw a light pencil line 1-2cm from the bottom of the paper.
2- Place a single drop of compound at intervals 2cm.
3- Dry with hair dryer.
4- Dip the paper in the jar with one of the edges of the paper to which the sample of the
spot is adjacent into the solvent.
5- Allow to run.
6- Remove the paper.
7- Determine the solvent front.
8- Dry.
9- Spray the paper with ninhydrin.
10- Dry the paper.
After some time
References:
www.wikipedia.org
7 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
RESULTS & LAB REPORT
- Calculate the R f and then identify the unknown amino acids in the mixture.
- Present your results in a good and full lab report.
8 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
`1
9 Page Organized by Lecturers: Sharifa Al-Ghamdi& Huda Al-Shaibi
You might also like
- Paper ChromatographyDocument34 pagesPaper ChromatographyShaise Jacob100% (6)
- PAPER+CHROMATOGRAPHY PpsDocument35 pagesPAPER+CHROMATOGRAPHY PpsjasyeelelooNo ratings yet
- Identification of Amino Acids-Paper ChromatographyDocument9 pagesIdentification of Amino Acids-Paper ChromatographySharanya Srinivasan50% (2)
- Analytical Techniques Final Note NADocument121 pagesAnalytical Techniques Final Note NAyuvi78312No ratings yet
- Paper ChromatographyDocument5 pagesPaper ChromatographyDr. P.S.SenguptaNo ratings yet
- 37 Analytical Techniques PDFDocument225 pages37 Analytical Techniques PDFKomal EhsanNo ratings yet
- Paper ChromatographyDocument5 pagesPaper ChromatographyMaria Elena PascualNo ratings yet
- ChromatographyDocument11 pagesChromatographyAmrit KoiralaNo ratings yet
- Sugars-TLC-method 2 - 2024Document5 pagesSugars-TLC-method 2 - 2024yateesahu2002No ratings yet
- Paper Chromatography AND Thin Layer ChromatographyDocument28 pagesPaper Chromatography AND Thin Layer Chromatographyraja singamNo ratings yet
- Bioc 211Document6 pagesBioc 211Femina ArgonzaNo ratings yet
- Lab Report Exp 6Document5 pagesLab Report Exp 6api-384913960No ratings yet
- ChromatographyDocument18 pagesChromatographyYerram Raju BeharaNo ratings yet
- Paper & Gas ChromatographyDocument21 pagesPaper & Gas ChromatographyusamaNo ratings yet
- 083 - Chromatography and Its Uses in BiologyDocument3 pages083 - Chromatography and Its Uses in Biologylastjoe71No ratings yet
- Jadavpur University: CollegeDocument14 pagesJadavpur University: CollegekuntaljuNo ratings yet
- Che ChromatographyDocument15 pagesChe ChromatographyArpit MauryaNo ratings yet
- Chem ChromatographyDocument15 pagesChem ChromatographyArpit MauryaNo ratings yet
- EXP10 ChromatographyDocument27 pagesEXP10 ChromatographyKiara Sy0% (1)
- Paper ChromatographyDocument5 pagesPaper ChromatographyKarishmaNo ratings yet
- Chromatography Class 12Document12 pagesChromatography Class 12kirti7666anand15No ratings yet
- Experiment 2 ChromatographyDocument6 pagesExperiment 2 ChromatographyFranz CandidoNo ratings yet
- Experiment 2 Written ReportDocument4 pagesExperiment 2 Written ReportJoachimNo ratings yet
- Chromatography SLT SpyDocument6 pagesChromatography SLT SpyAbdulnafiu AhmadNo ratings yet
- Chromatographic Techniques 1200328060603035050Document12 pagesChromatographic Techniques 1200328060603035050KIRAN ALLUNo ratings yet
- Paper ChromatographyDocument13 pagesPaper ChromatographyAlex MagatNo ratings yet
- Paper ChromatographyDocument31 pagesPaper ChromatographyPearl MirandaNo ratings yet
- Chromatography: Rayaan Ahmed 9-YellowDocument16 pagesChromatography: Rayaan Ahmed 9-YellowRayaan Ahmed MemonNo ratings yet
- Chromatography: Rayaan Ahmed 9-YellowDocument16 pagesChromatography: Rayaan Ahmed 9-YellowRayaan Ahmed MemonNo ratings yet
- Paper ChromatographyDocument6 pagesPaper ChromatographyManohar PattarNo ratings yet
- Experimental Techniques (Chapter 2) : Presented By: Mrs. SaimaDocument16 pagesExperimental Techniques (Chapter 2) : Presented By: Mrs. SaimaMohamed MunirNo ratings yet
- ChromatographyDocument3 pagesChromatographyAashiNo ratings yet
- Instrumental Methods of AnalysisDocument56 pagesInstrumental Methods of AnalysisPriya Bardhan RayNo ratings yet
- Lesson 1 IntroductionDocument17 pagesLesson 1 IntroductionGemma Wrigley100% (1)
- ChromaaaaaalateDocument7 pagesChromaaaaaalateVanessaOlgaJ.DagondonNo ratings yet
- Chromatography: Is A Technique Used To Separate and Identify The Components of A MixtureDocument72 pagesChromatography: Is A Technique Used To Separate and Identify The Components of A MixtureMustafa KhandgawiNo ratings yet
- Paper Chromatography 2023 - 230901 - 173633Document35 pagesPaper Chromatography 2023 - 230901 - 173633p.ishaanpawarNo ratings yet
- Kuliah Kromatografi Kertas 2011Document55 pagesKuliah Kromatografi Kertas 2011Ekka Ramayanti AqueaNo ratings yet
- 3.0 Seperation of Sugars by Paper Chromatography PrincipleDocument4 pages3.0 Seperation of Sugars by Paper Chromatography PrincipleMurugan M100% (1)
- Paper ChromatographyDocument7 pagesPaper ChromatographySEHAR KHAN100% (1)
- ASSIGNMENT NO 1 Chromatography EXACTDocument4 pagesASSIGNMENT NO 1 Chromatography EXACTRashid RashidNo ratings yet
- Principle of ChromatographyDocument8 pagesPrinciple of ChromatographyMuhammad ZeeshanNo ratings yet
- Paper ChromatographyDocument3 pagesPaper ChromatographyArnab SadhuNo ratings yet
- Chromatography Student 2021-2022Document17 pagesChromatography Student 2021-2022Nickoye WilliamsNo ratings yet
- Chromatography of Amino Acids Lab ReportDocument2 pagesChromatography of Amino Acids Lab ReportAjagwu EustaceNo ratings yet
- HrmtograhiDocument8 pagesHrmtograhiALJOREY LAZARITONo ratings yet
- Paper ChromatographyDocument5 pagesPaper Chromatographys8903082No ratings yet
- Lab 3 - Separation of PH Indicators Using Paper ChromatographyDocument6 pagesLab 3 - Separation of PH Indicators Using Paper ChromatographyJesiann SmithNo ratings yet
- Paper Chromatography: Assignment Report ONDocument14 pagesPaper Chromatography: Assignment Report ONKaran BhojwaniNo ratings yet
- Paper Chromatography Model and Practical LabDocument4 pagesPaper Chromatography Model and Practical Labdebbie bongNo ratings yet
- Chromatography SirkeDocument36 pagesChromatography SirkeAkash RoyNo ratings yet
- B.tech. Biotechnology NotesDocument2 pagesB.tech. Biotechnology NotesMudit MisraNo ratings yet
- Paper Chromatography: Members: Gay Marie Mallari Amelia Mabunot Ronie Lumot Junix Milad Jezrille Manzano Florante OlonanDocument18 pagesPaper Chromatography: Members: Gay Marie Mallari Amelia Mabunot Ronie Lumot Junix Milad Jezrille Manzano Florante OlonanCriminegrology TvNo ratings yet
- Paper ChromatographyDocument6 pagesPaper ChromatographysamNo ratings yet
- 5 ChromatographyDocument7 pages5 ChromatographyAntonio CharismaNo ratings yet
- BEP1021 - Group 3 Experiment 4Document16 pagesBEP1021 - Group 3 Experiment 4Tasmea sultanaNo ratings yet
- SBCC 1106-2Document17 pagesSBCC 1106-2opolla nianorNo ratings yet
- Practical Handbook of Pharmaceutical Chemistry for M.PharmFrom EverandPractical Handbook of Pharmaceutical Chemistry for M.PharmNo ratings yet
- Vitamin C LcmsDocument1 pageVitamin C LcmsdewiNo ratings yet
- 18CY1003-Lab ExperimentsDocument15 pages18CY1003-Lab ExperimentsRam MNo ratings yet
- Simultaneous Extraction of Bioactive Limonoid Aglycones and Glucoside From Citrus Aurantium L. Using HydrotropyDocument5 pagesSimultaneous Extraction of Bioactive Limonoid Aglycones and Glucoside From Citrus Aurantium L. Using Hydrotropyjohn4jNo ratings yet
- Titration Level 1: Krizzi AimsDocument4 pagesTitration Level 1: Krizzi AimsKrizzi Dizon GarciaNo ratings yet
- Bio Analytical Application of LC-MSDocument45 pagesBio Analytical Application of LC-MSDulal Mahato100% (1)
- LAB514Document16 pagesLAB514Imran MusaNo ratings yet
- Experiment 1: Biochemical CalculationsDocument81 pagesExperiment 1: Biochemical CalculationsTrishia Lauren SegismarNo ratings yet
- Atomic Mass SpectrosDocument40 pagesAtomic Mass Spectrosjohnpaul varonaNo ratings yet
- Acid Base & Ionic Equilibrium (English)Document38 pagesAcid Base & Ionic Equilibrium (English)rewantreddygajrabhanushaliNo ratings yet
- Characterization of Given Compound by UV VIS SpectrosDocument2 pagesCharacterization of Given Compound by UV VIS Spectrosرجاء صدیقیNo ratings yet
- 2a - Packed ColumnsDocument55 pages2a - Packed ColumnsDoaa Badarneh100% (1)
- Buffers Complete Handout 2020 With Answer KeyDocument14 pagesBuffers Complete Handout 2020 With Answer KeyRadhika RaniNo ratings yet
- MentholDocument4 pagesMentholLipsi Merchán100% (1)
- Purification of AcetanilideDocument3 pagesPurification of AcetanilideHeinrich SolivenNo ratings yet
- Group I Cations: This Centrifuge Is Saved For Analysis of Group II-VDocument1 pageGroup I Cations: This Centrifuge Is Saved For Analysis of Group II-VpixiedustNo ratings yet
- Lab ManualDocument35 pagesLab ManualOla Eman100% (1)
- 01 Solid State Physics 02 (Compatibility Mode) (Repaired)Document54 pages01 Solid State Physics 02 (Compatibility Mode) (Repaired)aristiawanNo ratings yet
- GC DetectorDocument34 pagesGC Detectorhanif fNo ratings yet
- CHM 141-Practice QuestionsDocument7 pagesCHM 141-Practice QuestionsayomideajeniyaNo ratings yet
- Equilibrium ShiftDocument2 pagesEquilibrium ShiftCatalina PerryNo ratings yet
- Mineral OilDocument2 pagesMineral OilHelena ConnollyNo ratings yet
- Aoac 979.08Document1 pageAoac 979.08blink scientificNo ratings yet
- Analytical ChemistryDocument58 pagesAnalytical ChemistryJohn Joshua Protacio100% (1)
- Analytical Separation MethodDocument2 pagesAnalytical Separation MethodNajmin RoslanNo ratings yet
- Introduction To Chemistry FacCh 101Document116 pagesIntroduction To Chemistry FacCh 101lolo.moussa159No ratings yet
- SCION 436-GC: Specification SheetDocument5 pagesSCION 436-GC: Specification SheetGuille ñatoNo ratings yet
- Terræ Sistemas Cristalinos: Nomenclatura e Convenções: Introdução FundamentaçãoDocument7 pagesTerræ Sistemas Cristalinos: Nomenclatura e Convenções: Introdução FundamentaçãoMauro AbnnerNo ratings yet
- Wu, Chi-San Handbook of Size Exclusion Chromatography and Related Techniques Revised and ExpandedDocument697 pagesWu, Chi-San Handbook of Size Exclusion Chromatography and Related Techniques Revised and ExpandedIoana Ruxandra AroşculeseiNo ratings yet
- 201 L 4 Gravimetric AnalysisDocument24 pages201 L 4 Gravimetric AnalysisJawad AhmadNo ratings yet