Professional Documents
Culture Documents
DPP 2
DPP 2
Uploaded by
ashutosh99878Copyright:
Available Formats
You might also like
- 123Document8 pages123Dalberto Ramirez50% (2)
- Team Bootcamp - Reaction Summary Sheet PDFDocument30 pagesTeam Bootcamp - Reaction Summary Sheet PDFDagne PovedaNo ratings yet
- Experiment To Find The Optimal Design For A Carbon Dioxide Propelled TorpedoDocument6 pagesExperiment To Find The Optimal Design For A Carbon Dioxide Propelled TorpedoJakeLeviNo ratings yet
- Extra ProblemDocument2 pagesExtra ProblemradwaelhadadNo ratings yet
- Ms Chouhan & Team: Organic ChemistryDocument16 pagesMs Chouhan & Team: Organic ChemistryPrithviraj GhoshNo ratings yet
- Hydrocarbons Jumbo Sheet by MKA SirDocument44 pagesHydrocarbons Jumbo Sheet by MKA SirRahul SinghNo ratings yet
- BOC Complete 1 To 5 DPPDocument5 pagesBOC Complete 1 To 5 DPPBhawna SharmaNo ratings yet
- 13 DPP 03g Goc Excel Acid+BaseDocument5 pages13 DPP 03g Goc Excel Acid+BasekljNo ratings yet
- Raj7 PDFDocument22 pagesRaj7 PDFAvdhoot GautamNo ratings yet
- Chemistry Advanced Level Problem Solving (ALPS-5) - SolutionDocument12 pagesChemistry Advanced Level Problem Solving (ALPS-5) - SolutionAnanmay Chauhan100% (1)
- Complete Course in Organic Chemistry by Vineet Khatri Sir: Class: Xi Time: 35 Min. DPP. NO.17Document4 pagesComplete Course in Organic Chemistry by Vineet Khatri Sir: Class: Xi Time: 35 Min. DPP. NO.17Arnab KumarNo ratings yet
- This DPP Gives You An Idea About Products Obtained in Elimination Reaction (Including Stereochemistry)Document3 pagesThis DPP Gives You An Idea About Products Obtained in Elimination Reaction (Including Stereochemistry)Aaryan KeshanNo ratings yet
- Reaction Mechanism Jeemain - GuruDocument12 pagesReaction Mechanism Jeemain - Guruprexa indiaNo ratings yet
- 3.AcidBases FinalDocument35 pages3.AcidBases FinalSoham RaneNo ratings yet
- Bakliwal Tutorials: Topic: Inductive Effect Part - A: SubjectiveDocument3 pagesBakliwal Tutorials: Topic: Inductive Effect Part - A: SubjectiveRitesh SonawaneNo ratings yet
- Carbonyl Compound WorksheetDocument25 pagesCarbonyl Compound WorksheetOmendra SinghNo ratings yet
- 5 DPP - 56to81 - FinalDocument44 pages5 DPP - 56to81 - FinalArnab KumarNo ratings yet
- CH CH CHCH CH H CH CH CH CH CH CH H CH: Byvineet Khatri SirDocument13 pagesCH CH CHCH CH H CH CH CH CH CH CH H CH: Byvineet Khatri Sirsarvesh goyalNo ratings yet
- 02 H.D.A. SN1 and SN2 Reaction 10-08-2021Document2 pages02 H.D.A. SN1 and SN2 Reaction 10-08-2021tejas naigaonkarNo ratings yet
- KKHKDocument29 pagesKKHKjaspreet singhNo ratings yet
- Alkenes Alkynes Oxidation PDFDocument32 pagesAlkenes Alkynes Oxidation PDFRamuNo ratings yet
- Ape Assignment 3Document7 pagesApe Assignment 3Atharva KulkarniNo ratings yet
- Micro - OC - DPP Without Answer (110-116)Document24 pagesMicro - OC - DPP Without Answer (110-116)Ayush JaiswalNo ratings yet
- ADV. I 57 - 64 (Exercise 3)Document8 pagesADV. I 57 - 64 (Exercise 3)Aditya ShahNo ratings yet
- Kota Worksheet Chemical BondingDocument16 pagesKota Worksheet Chemical BondingYash GargNo ratings yet
- R IS IR: Iupac & NomenclatureDocument11 pagesR IS IR: Iupac & NomenclatureDhruv KuchhalNo ratings yet
- Alcohols Ethers and Phenol-03 - Assignments (New)Document26 pagesAlcohols Ethers and Phenol-03 - Assignments (New)Raju SinghNo ratings yet
- Aldol Reaction - ChemistryDocument7 pagesAldol Reaction - ChemistryGamer HelperNo ratings yet
- Jitendra Hirwani: Previous Year Problem Solving Iit Jee Main + Advanced Physical ChemistryDocument16 pagesJitendra Hirwani: Previous Year Problem Solving Iit Jee Main + Advanced Physical ChemistryAshish gurjarNo ratings yet
- Etoos 9 PDFDocument24 pagesEtoos 9 PDFB. P. A Music INDIA100% (1)
- Roadmap Problem - 9Document1 pageRoadmap Problem - 9abhyudaipathwayNo ratings yet
- Isomerism - Handwritten NotesDocument7 pagesIsomerism - Handwritten Notesgovind_galamNo ratings yet
- Structural Identification & Poc-1: Topic Page NoDocument35 pagesStructural Identification & Poc-1: Topic Page Nosiddansh100% (1)
- Organic Chemistry: Daily Practice ProblemsDocument35 pagesOrganic Chemistry: Daily Practice ProblemsRaju SinghNo ratings yet
- 6.DAY-8 CHE - Organic Chemistry Electron Migration Effects & Reagents - 25-05-2020 PDFDocument7 pages6.DAY-8 CHE - Organic Chemistry Electron Migration Effects & Reagents - 25-05-2020 PDFRamakrishna ReddyNo ratings yet
- IIT JEE Main Advanced Physical Chemistry 12th Volumetric Analysis PDFDocument38 pagesIIT JEE Main Advanced Physical Chemistry 12th Volumetric Analysis PDFGopal PenjarlaNo ratings yet
- 03 Reaction Mechanism (Solutions)Document19 pages03 Reaction Mechanism (Solutions)Anup KNo ratings yet
- Atomic StructureDocument18 pagesAtomic StructureAshish gurjarNo ratings yet
- Prince Singh: Physical & Inorganic ChemistryDocument5 pagesPrince Singh: Physical & Inorganic ChemistryJatin Singla100% (1)
- Carbonyl CompoundsDocument45 pagesCarbonyl CompoundsYogesh BansalNo ratings yet
- Solution - Colligative Properties Solutions PDFDocument25 pagesSolution - Colligative Properties Solutions PDFGOURISH AGRAWALNo ratings yet
- Cls Jeead-18-19 Xii Che Target-7 Set-2 Chapter-12Document47 pagesCls Jeead-18-19 Xii Che Target-7 Set-2 Chapter-12DxNo ratings yet
- Concentration Terms and Eudiometry: (Physical Chemistry) Exercise (O-I) Introduction of Concentration Terms 1Document59 pagesConcentration Terms and Eudiometry: (Physical Chemistry) Exercise (O-I) Introduction of Concentration Terms 1Jayarj singh100% (1)
- Reaction IntermediatesDocument32 pagesReaction Intermediatestechno studioNo ratings yet
- (@bohring - Bot) Special Batch Assignment-6Document30 pages(@bohring - Bot) Special Batch Assignment-6lash73752No ratings yet
- CLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFDocument40 pagesCLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFThavasimariselvam N100% (1)
- Inorganic Chemistry: Target: JEE (MAIN + ADVANCE) 2020Document30 pagesInorganic Chemistry: Target: JEE (MAIN + ADVANCE) 2020sarvesh goyalNo ratings yet
- 03ElectronicdisplacementEffects Exercise Send1Document33 pages03ElectronicdisplacementEffects Exercise Send1Aaryan Keshan100% (1)
- Alkyl Halides & Aryl Halides-02 - Solved ProblemsDocument13 pagesAlkyl Halides & Aryl Halides-02 - Solved ProblemsRaju SinghNo ratings yet
- DPP No.A1 To A11 Hints Solution Physical InorganicDocument51 pagesDPP No.A1 To A11 Hints Solution Physical InorganicAkkaldevi Saivinayak CRNo ratings yet
- IsomerismDocument62 pagesIsomerismsubesinghNo ratings yet
- Quantitative and QualitativeDocument15 pagesQuantitative and QualitativesquadralsupremeNo ratings yet
- G R Reduction AlkaneDocument43 pagesG R Reduction AlkaneManthan HaritashNo ratings yet
- S.No. Molecular Formula Number of Structural Isomers: Organic ChemistryDocument3 pagesS.No. Molecular Formula Number of Structural Isomers: Organic ChemistryLakshya wardhan singh ShekhawatNo ratings yet
- Solved Example: Chemistry For Neet & AiimsDocument24 pagesSolved Example: Chemistry For Neet & AiimsAnup KNo ratings yet
- Alcohol EtherDocument39 pagesAlcohol Etheraryamandas321100% (1)
- H.D.A. 2021Document54 pagesH.D.A. 2021Every Time Chemistry [ ETC]No ratings yet
- Chemistry Advanced Level Problem Solving (ALPS-6) - PaperDocument14 pagesChemistry Advanced Level Problem Solving (ALPS-6) - PaperAnanmay ChauhanNo ratings yet
- Assignment: Organic ChemistryDocument6 pagesAssignment: Organic ChemistryWalid EbaiedNo ratings yet
- DPP 02 Periodic Table JH Sir-3579Document8 pagesDPP 02 Periodic Table JH Sir-3579AmitSharmaNo ratings yet
- Aromaticity DPP 4Document4 pagesAromaticity DPP 4SubhadeepNo ratings yet
- Acidity, Basicity and TautomerismDocument16 pagesAcidity, Basicity and TautomerismShreyansh PatelNo ratings yet
- General Organic Chemistry - DPP 01 - Goc-dpp-01-Lakshya (Jee)Document4 pagesGeneral Organic Chemistry - DPP 01 - Goc-dpp-01-Lakshya (Jee)SS MENTOR100% (3)
- Thermo NotesDocument129 pagesThermo NotesManoj MallickNo ratings yet
- Farrell Ethanol Science 012706Document3 pagesFarrell Ethanol Science 012706brayanNo ratings yet
- Galvanized Rebar Vs Epoxy RebarDocument4 pagesGalvanized Rebar Vs Epoxy RebarEdwin53No ratings yet
- 18 CheesDocument17 pages18 CheesAnonymousNo ratings yet
- 1 Organic Compounds HydrocarbonsDocument29 pages1 Organic Compounds HydrocarbonsatNo ratings yet
- Epxy CLR TdsDocument4 pagesEpxy CLR TdsliebofreakNo ratings yet
- Virial Equation of State2Document46 pagesVirial Equation of State2ShainaBagonNo ratings yet
- AISI 1045 Medium Carbon Steel - AZoMDocument4 pagesAISI 1045 Medium Carbon Steel - AZoMLuis NunesNo ratings yet
- FEM: Dynamic ProblemDocument3 pagesFEM: Dynamic ProblemAlexander VovaNo ratings yet
- Herbagreen Slide Show040213Document75 pagesHerbagreen Slide Show040213Wan Ayie100% (1)
- As 2212-1979 Elastomeric Hose and Hose Assemblies For Transferring Anhydrous AmmoniaDocument7 pagesAs 2212-1979 Elastomeric Hose and Hose Assemblies For Transferring Anhydrous AmmoniaSAI Global - APACNo ratings yet
- Shree CV - 2019Document4 pagesShree CV - 2019Anonymous QE8HMXSNo ratings yet
- Measurements of Water-Holding Capacity and ColorDocument10 pagesMeasurements of Water-Holding Capacity and ColorFaysa UtbaNo ratings yet
- CC-STEADY STATE Tutorial: CHEMCAD TutorialsDocument22 pagesCC-STEADY STATE Tutorial: CHEMCAD TutorialsDwi RamadhaniNo ratings yet
- Draft Resti..Document63 pagesDraft Resti..Leni LismayantiNo ratings yet
- TAPPI/ANSI T 454 Om-15: CautionDocument4 pagesTAPPI/ANSI T 454 Om-15: CautionramonNo ratings yet
- SVKM J.V. Parekh International School 2018-2019 Cycle Test 2 Portion Class: IXDocument3 pagesSVKM J.V. Parekh International School 2018-2019 Cycle Test 2 Portion Class: IXNyonikaNo ratings yet
- NucTheor&Applic Proc 2011Document500 pagesNucTheor&Applic Proc 2011Valeriy PostnikovNo ratings yet
- B.Tech ME 2016 v2Document123 pagesB.Tech ME 2016 v2hakeemniyasNo ratings yet
- LDPE-Low Density Polyethylene LTM 2185/47: Typical DataDocument3 pagesLDPE-Low Density Polyethylene LTM 2185/47: Typical DataMohamadMostafaviNo ratings yet
- P266001coll1 4860Document82 pagesP266001coll1 4860Jemicah YumenaNo ratings yet
- Estimating The Shielding Requirements For A Newly Planned PET-CT FacilityDocument6 pagesEstimating The Shielding Requirements For A Newly Planned PET-CT FacilitySelim HossainNo ratings yet
- Abs MTG 204&abs 0604Document1 pageAbs MTG 204&abs 0604同道文档中心100% (1)
- Aqa Phy3h W Ms Jun12Document14 pagesAqa Phy3h W Ms Jun12Rubab ZahraNo ratings yet
- Arktite CPP - CPS Plugs and ReceptaclesDocument4 pagesArktite CPP - CPS Plugs and ReceptaclesHoangNo ratings yet
- Chapter 6 Metallic Waveguide and Cavity ResonatorsDocument34 pagesChapter 6 Metallic Waveguide and Cavity ResonatorsRasheed Mohammed AbdulNo ratings yet
- Conplast SP430SRVDocument2 pagesConplast SP430SRVVenkata Raju KalidindiNo ratings yet
DPP 2
DPP 2
Uploaded by
ashutosh99878Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
DPP 2
DPP 2
Uploaded by
ashutosh99878Copyright:
Available Formats
1
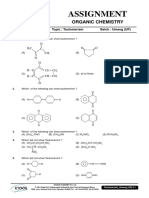
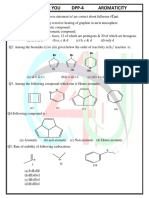
ARJUNA JEE 2023
GOC-Isomerism DPP-02
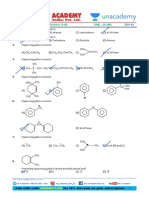
1. The inductive effect of the groups: –NH3⊕; –D; 5. Arrange the following groups in the order of
–CO2Θ; –COOH are respectively decreasing (–I) effect.
(1) –I, +I, +I, –I (1) CN > F > Br > Cl > COOH > I > H
(2) –I, –I, –I, +I (2) COOH > CN > F > Br > Cl > I > H
(3) +I, No effect, –I, –I (3) H > COOH > CN > I > Cl > F > Br
(4) +I, –I, –I, –I (4) CN > COOH > F > Cl > Br > I > H
2. Choose the correct statement 6. The +I.E. (inductive effect) is shown by:
(1) I effect operates in both σ and π bonds (1) –CH3 (2) –OH
(2) I effect creates net charge in molecule (3) –F (4) –C6H5
(3) I effect transfers electron from one carbon to
another 7. The –I effect is shown by:
(4) I effect creates partial charges and it is distance (1) –COOH (2) –CH3
dependent (3) –CH2CH3 (4) –CHR2
3. The most stable carbocation among the following is: 8. The increasing order of +ve I-effect shown by H,
CH3, C2H5 and C3H7 is:
(1)
(1) H < CH3 < C2H5 < C3H7
(2) (2) H > CH3 > C2H5 >C3H7
(3) H < C2H5 < CH3 < C3H7
(3)
(4) None of the above
9. The most acidic compound among following is:
(4)
(1) F – CH2 – COOH
(2) Cl – CH2 – COOH
(3) Br – CH2 – COOH
4. Order of stability of given free radical is: (4) I – CH2 – COOH
10. Which order is correct regarding the –I effect of the
substituents?
(1) –NR2 < –OR < –F
(2) –NR2 > –OR < –F
(3) NR2 > –F > –OR
(1) III > I > II (2) I > II > III
(4) –NR2 > –OR > –F
(3) II > I > III (4) III > II > I
2
Note: Kindly find the Video Solution of DPPs Questions in the DPPs Section.
Answer Key
1. (1) 6. (1)
2. (4) 7. (1)
3. (3) 8. (1)
4. (4) 9. (1)
5. (4) 10. (1)
For more questions, kindly visit the library section: Link for app: https://links.physicswallah.live/vyJw
For more questions, kindly visit the library section: Link for web: https://physicswallah.live/tabs/tabs/library-tab
PW Mobile APP: https://physicswala.page.link/?type=contact-us&data=open
For PW Website: https://www.physicswallah.live/contact-us
You might also like
- 123Document8 pages123Dalberto Ramirez50% (2)
- Team Bootcamp - Reaction Summary Sheet PDFDocument30 pagesTeam Bootcamp - Reaction Summary Sheet PDFDagne PovedaNo ratings yet
- Experiment To Find The Optimal Design For A Carbon Dioxide Propelled TorpedoDocument6 pagesExperiment To Find The Optimal Design For A Carbon Dioxide Propelled TorpedoJakeLeviNo ratings yet
- Extra ProblemDocument2 pagesExtra ProblemradwaelhadadNo ratings yet
- Ms Chouhan & Team: Organic ChemistryDocument16 pagesMs Chouhan & Team: Organic ChemistryPrithviraj GhoshNo ratings yet
- Hydrocarbons Jumbo Sheet by MKA SirDocument44 pagesHydrocarbons Jumbo Sheet by MKA SirRahul SinghNo ratings yet
- BOC Complete 1 To 5 DPPDocument5 pagesBOC Complete 1 To 5 DPPBhawna SharmaNo ratings yet
- 13 DPP 03g Goc Excel Acid+BaseDocument5 pages13 DPP 03g Goc Excel Acid+BasekljNo ratings yet
- Raj7 PDFDocument22 pagesRaj7 PDFAvdhoot GautamNo ratings yet
- Chemistry Advanced Level Problem Solving (ALPS-5) - SolutionDocument12 pagesChemistry Advanced Level Problem Solving (ALPS-5) - SolutionAnanmay Chauhan100% (1)
- Complete Course in Organic Chemistry by Vineet Khatri Sir: Class: Xi Time: 35 Min. DPP. NO.17Document4 pagesComplete Course in Organic Chemistry by Vineet Khatri Sir: Class: Xi Time: 35 Min. DPP. NO.17Arnab KumarNo ratings yet
- This DPP Gives You An Idea About Products Obtained in Elimination Reaction (Including Stereochemistry)Document3 pagesThis DPP Gives You An Idea About Products Obtained in Elimination Reaction (Including Stereochemistry)Aaryan KeshanNo ratings yet
- Reaction Mechanism Jeemain - GuruDocument12 pagesReaction Mechanism Jeemain - Guruprexa indiaNo ratings yet
- 3.AcidBases FinalDocument35 pages3.AcidBases FinalSoham RaneNo ratings yet
- Bakliwal Tutorials: Topic: Inductive Effect Part - A: SubjectiveDocument3 pagesBakliwal Tutorials: Topic: Inductive Effect Part - A: SubjectiveRitesh SonawaneNo ratings yet
- Carbonyl Compound WorksheetDocument25 pagesCarbonyl Compound WorksheetOmendra SinghNo ratings yet
- 5 DPP - 56to81 - FinalDocument44 pages5 DPP - 56to81 - FinalArnab KumarNo ratings yet
- CH CH CHCH CH H CH CH CH CH CH CH H CH: Byvineet Khatri SirDocument13 pagesCH CH CHCH CH H CH CH CH CH CH CH H CH: Byvineet Khatri Sirsarvesh goyalNo ratings yet
- 02 H.D.A. SN1 and SN2 Reaction 10-08-2021Document2 pages02 H.D.A. SN1 and SN2 Reaction 10-08-2021tejas naigaonkarNo ratings yet
- KKHKDocument29 pagesKKHKjaspreet singhNo ratings yet
- Alkenes Alkynes Oxidation PDFDocument32 pagesAlkenes Alkynes Oxidation PDFRamuNo ratings yet
- Ape Assignment 3Document7 pagesApe Assignment 3Atharva KulkarniNo ratings yet
- Micro - OC - DPP Without Answer (110-116)Document24 pagesMicro - OC - DPP Without Answer (110-116)Ayush JaiswalNo ratings yet
- ADV. I 57 - 64 (Exercise 3)Document8 pagesADV. I 57 - 64 (Exercise 3)Aditya ShahNo ratings yet
- Kota Worksheet Chemical BondingDocument16 pagesKota Worksheet Chemical BondingYash GargNo ratings yet
- R IS IR: Iupac & NomenclatureDocument11 pagesR IS IR: Iupac & NomenclatureDhruv KuchhalNo ratings yet
- Alcohols Ethers and Phenol-03 - Assignments (New)Document26 pagesAlcohols Ethers and Phenol-03 - Assignments (New)Raju SinghNo ratings yet
- Aldol Reaction - ChemistryDocument7 pagesAldol Reaction - ChemistryGamer HelperNo ratings yet
- Jitendra Hirwani: Previous Year Problem Solving Iit Jee Main + Advanced Physical ChemistryDocument16 pagesJitendra Hirwani: Previous Year Problem Solving Iit Jee Main + Advanced Physical ChemistryAshish gurjarNo ratings yet
- Etoos 9 PDFDocument24 pagesEtoos 9 PDFB. P. A Music INDIA100% (1)
- Roadmap Problem - 9Document1 pageRoadmap Problem - 9abhyudaipathwayNo ratings yet
- Isomerism - Handwritten NotesDocument7 pagesIsomerism - Handwritten Notesgovind_galamNo ratings yet
- Structural Identification & Poc-1: Topic Page NoDocument35 pagesStructural Identification & Poc-1: Topic Page Nosiddansh100% (1)
- Organic Chemistry: Daily Practice ProblemsDocument35 pagesOrganic Chemistry: Daily Practice ProblemsRaju SinghNo ratings yet
- 6.DAY-8 CHE - Organic Chemistry Electron Migration Effects & Reagents - 25-05-2020 PDFDocument7 pages6.DAY-8 CHE - Organic Chemistry Electron Migration Effects & Reagents - 25-05-2020 PDFRamakrishna ReddyNo ratings yet
- IIT JEE Main Advanced Physical Chemistry 12th Volumetric Analysis PDFDocument38 pagesIIT JEE Main Advanced Physical Chemistry 12th Volumetric Analysis PDFGopal PenjarlaNo ratings yet
- 03 Reaction Mechanism (Solutions)Document19 pages03 Reaction Mechanism (Solutions)Anup KNo ratings yet
- Atomic StructureDocument18 pagesAtomic StructureAshish gurjarNo ratings yet
- Prince Singh: Physical & Inorganic ChemistryDocument5 pagesPrince Singh: Physical & Inorganic ChemistryJatin Singla100% (1)
- Carbonyl CompoundsDocument45 pagesCarbonyl CompoundsYogesh BansalNo ratings yet
- Solution - Colligative Properties Solutions PDFDocument25 pagesSolution - Colligative Properties Solutions PDFGOURISH AGRAWALNo ratings yet
- Cls Jeead-18-19 Xii Che Target-7 Set-2 Chapter-12Document47 pagesCls Jeead-18-19 Xii Che Target-7 Set-2 Chapter-12DxNo ratings yet
- Concentration Terms and Eudiometry: (Physical Chemistry) Exercise (O-I) Introduction of Concentration Terms 1Document59 pagesConcentration Terms and Eudiometry: (Physical Chemistry) Exercise (O-I) Introduction of Concentration Terms 1Jayarj singh100% (1)
- Reaction IntermediatesDocument32 pagesReaction Intermediatestechno studioNo ratings yet
- (@bohring - Bot) Special Batch Assignment-6Document30 pages(@bohring - Bot) Special Batch Assignment-6lash73752No ratings yet
- CLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFDocument40 pagesCLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFThavasimariselvam N100% (1)
- Inorganic Chemistry: Target: JEE (MAIN + ADVANCE) 2020Document30 pagesInorganic Chemistry: Target: JEE (MAIN + ADVANCE) 2020sarvesh goyalNo ratings yet
- 03ElectronicdisplacementEffects Exercise Send1Document33 pages03ElectronicdisplacementEffects Exercise Send1Aaryan Keshan100% (1)
- Alkyl Halides & Aryl Halides-02 - Solved ProblemsDocument13 pagesAlkyl Halides & Aryl Halides-02 - Solved ProblemsRaju SinghNo ratings yet
- DPP No.A1 To A11 Hints Solution Physical InorganicDocument51 pagesDPP No.A1 To A11 Hints Solution Physical InorganicAkkaldevi Saivinayak CRNo ratings yet
- IsomerismDocument62 pagesIsomerismsubesinghNo ratings yet
- Quantitative and QualitativeDocument15 pagesQuantitative and QualitativesquadralsupremeNo ratings yet
- G R Reduction AlkaneDocument43 pagesG R Reduction AlkaneManthan HaritashNo ratings yet
- S.No. Molecular Formula Number of Structural Isomers: Organic ChemistryDocument3 pagesS.No. Molecular Formula Number of Structural Isomers: Organic ChemistryLakshya wardhan singh ShekhawatNo ratings yet
- Solved Example: Chemistry For Neet & AiimsDocument24 pagesSolved Example: Chemistry For Neet & AiimsAnup KNo ratings yet
- Alcohol EtherDocument39 pagesAlcohol Etheraryamandas321100% (1)
- H.D.A. 2021Document54 pagesH.D.A. 2021Every Time Chemistry [ ETC]No ratings yet
- Chemistry Advanced Level Problem Solving (ALPS-6) - PaperDocument14 pagesChemistry Advanced Level Problem Solving (ALPS-6) - PaperAnanmay ChauhanNo ratings yet
- Assignment: Organic ChemistryDocument6 pagesAssignment: Organic ChemistryWalid EbaiedNo ratings yet
- DPP 02 Periodic Table JH Sir-3579Document8 pagesDPP 02 Periodic Table JH Sir-3579AmitSharmaNo ratings yet
- Aromaticity DPP 4Document4 pagesAromaticity DPP 4SubhadeepNo ratings yet
- Acidity, Basicity and TautomerismDocument16 pagesAcidity, Basicity and TautomerismShreyansh PatelNo ratings yet
- General Organic Chemistry - DPP 01 - Goc-dpp-01-Lakshya (Jee)Document4 pagesGeneral Organic Chemistry - DPP 01 - Goc-dpp-01-Lakshya (Jee)SS MENTOR100% (3)
- Thermo NotesDocument129 pagesThermo NotesManoj MallickNo ratings yet
- Farrell Ethanol Science 012706Document3 pagesFarrell Ethanol Science 012706brayanNo ratings yet
- Galvanized Rebar Vs Epoxy RebarDocument4 pagesGalvanized Rebar Vs Epoxy RebarEdwin53No ratings yet
- 18 CheesDocument17 pages18 CheesAnonymousNo ratings yet
- 1 Organic Compounds HydrocarbonsDocument29 pages1 Organic Compounds HydrocarbonsatNo ratings yet
- Epxy CLR TdsDocument4 pagesEpxy CLR TdsliebofreakNo ratings yet
- Virial Equation of State2Document46 pagesVirial Equation of State2ShainaBagonNo ratings yet
- AISI 1045 Medium Carbon Steel - AZoMDocument4 pagesAISI 1045 Medium Carbon Steel - AZoMLuis NunesNo ratings yet
- FEM: Dynamic ProblemDocument3 pagesFEM: Dynamic ProblemAlexander VovaNo ratings yet
- Herbagreen Slide Show040213Document75 pagesHerbagreen Slide Show040213Wan Ayie100% (1)
- As 2212-1979 Elastomeric Hose and Hose Assemblies For Transferring Anhydrous AmmoniaDocument7 pagesAs 2212-1979 Elastomeric Hose and Hose Assemblies For Transferring Anhydrous AmmoniaSAI Global - APACNo ratings yet
- Shree CV - 2019Document4 pagesShree CV - 2019Anonymous QE8HMXSNo ratings yet
- Measurements of Water-Holding Capacity and ColorDocument10 pagesMeasurements of Water-Holding Capacity and ColorFaysa UtbaNo ratings yet
- CC-STEADY STATE Tutorial: CHEMCAD TutorialsDocument22 pagesCC-STEADY STATE Tutorial: CHEMCAD TutorialsDwi RamadhaniNo ratings yet
- Draft Resti..Document63 pagesDraft Resti..Leni LismayantiNo ratings yet
- TAPPI/ANSI T 454 Om-15: CautionDocument4 pagesTAPPI/ANSI T 454 Om-15: CautionramonNo ratings yet
- SVKM J.V. Parekh International School 2018-2019 Cycle Test 2 Portion Class: IXDocument3 pagesSVKM J.V. Parekh International School 2018-2019 Cycle Test 2 Portion Class: IXNyonikaNo ratings yet
- NucTheor&Applic Proc 2011Document500 pagesNucTheor&Applic Proc 2011Valeriy PostnikovNo ratings yet
- B.Tech ME 2016 v2Document123 pagesB.Tech ME 2016 v2hakeemniyasNo ratings yet
- LDPE-Low Density Polyethylene LTM 2185/47: Typical DataDocument3 pagesLDPE-Low Density Polyethylene LTM 2185/47: Typical DataMohamadMostafaviNo ratings yet
- P266001coll1 4860Document82 pagesP266001coll1 4860Jemicah YumenaNo ratings yet
- Estimating The Shielding Requirements For A Newly Planned PET-CT FacilityDocument6 pagesEstimating The Shielding Requirements For A Newly Planned PET-CT FacilitySelim HossainNo ratings yet
- Abs MTG 204&abs 0604Document1 pageAbs MTG 204&abs 0604同道文档中心100% (1)
- Aqa Phy3h W Ms Jun12Document14 pagesAqa Phy3h W Ms Jun12Rubab ZahraNo ratings yet
- Arktite CPP - CPS Plugs and ReceptaclesDocument4 pagesArktite CPP - CPS Plugs and ReceptaclesHoangNo ratings yet
- Chapter 6 Metallic Waveguide and Cavity ResonatorsDocument34 pagesChapter 6 Metallic Waveguide and Cavity ResonatorsRasheed Mohammed AbdulNo ratings yet
- Conplast SP430SRVDocument2 pagesConplast SP430SRVVenkata Raju KalidindiNo ratings yet