Professional Documents
Culture Documents
Carryover Report
Carryover Report
Uploaded by
Riad ManamanniCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Carryover Report
Carryover Report
Uploaded by
Riad ManamanniCopyright:
Available Formats
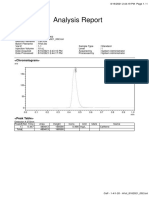
CARRYOVER REPORT
AC Q U I SI TI O N I N F O R M ATI O N
System Name: PATROL with PDA Acquired By: Jennb
Project Name: SQT_PATrol_PDA_CM_rev D Column serial Number: 11581505610K
Sample Set Method: 090 Carryover Sample Kit 1 Expiration date: 10/22/2010 12:00:00 AM
Processing Method Carryover PM, Carryover Clean Sample Kit 1 Lot Number: N070015
Report Method Name: Carryover Report Build Version: Empower 2 Software Build 2154 SPs
User Name: System Installed:
Sample Set Feature Release 5
Id: 11954 DB ID:
Result 847558692
Set Id 11966
Sample Set Name: Carryover Sample Set Start Date: 11/4/2010 9:31:58 PM
In the Carryover qualification, two pre-blank injections are performed to determine if the chromatograph is
clean enough to measure carryover. Those injections are followed by three Caffeine calibration injections at a
concentration of (0.2 ppm) which is 0.005 percent of the Challenge injection concentration (4000 ppm).
The Challenge injection is made and is followed by three post-blank injections.
Carryover is reported as a percent of the Challenge concentration. If the % Carryover of first post-blank injection
is less than 0.010% the qualification passes.
Carryover Peak Results
Carryover Carryover
SampleName Peak Type % Carryover
Specification Pass Fail
1 PostBlank1 < or = 0.010% Found 0.009 PASS
Printed 7:59:22 AM US/Eastern 11/17/2010 Page: 1 of 3
CARRYOVER REPORT
AC Q U I SI TI O N I N F O R M ATI O N
System Name: PATROL with PDA Acquired By: Jennb
Project Name: SQT_PATrol_PDA_CM_rev D Column serial Number: 11581505610K
Sample Set Method: 090 Carryover Sample Kit 1 Expiration date: 10/22/2010 12:00:00 AM
Processing Method Carryover PM, Carryover Clean Sample Kit 1 Lot Number: N070015
Report Method Name: Carryover Report Build Version: Empower 2 Software Build 2154 SPs
User Name: System Installed:
Sample Set Feature Release 5
Id: 11954 DB ID:
Result 847558692
Set Id 11966
Sample Set Name: Carryover Sample Set Start Date: 11/4/2010 9:31:58 PM
Caffeine
Caffeine
0.004 0.004 0.004
Caffeine
AU
AU
AU
0.002 0.002 0.002
0.000 0.000 0.000
0.00 1.00 2.00 0.00 1.00 2.00 0.00 1.00 2.00
Minutes Minutes Minutes
PreBlank; Vial: 12; Inj: 1 PreBlank; Vial: 12; Inj: 2 Standard; Vial: 8; Inj: 1
0.004 0.004 0.004
Caffeine
Caffeine
AU
AU
AU
0.002 0.002 0.002
0.000 0.000 0.000
0.00 1.00 2.00 0.00 1.00 2.00 0.00 5.00 10.00 15.00
Minutes Minutes Minutes
Standard; Vial: 8; Inj: 2 Standard; Vial: 8; Inj: 3 Challenge; Vial: 9; Inj: 1
Caffeine
Caffeine
Caffeine
0.004 0.004 0.004
AU
AU
AU
0.002 0.002 0.002
0.000 0.000 0.000
0.00 1.00 2.00 0.00 1.00 2.00 0.00 1.00 2.00
Minutes Minutes Minutes
PostBlank1; Vial: 12; Inj: 1 PostBlank2; Vial: 12; Inj: 1 PostBlank3; Vial: 12; Inj: 1
Printed 7:59:22 AM US/Eastern 11/17/2010 Page: 2 of 3
CARRYOVER REPORT
AC Q U I SI TI O N I N F O R M ATI O N
System Name: PATROL with PDA Acquired By: Jennb
Project Name: SQT_PATrol_PDA_CM_rev D Column serial Number: 11581505610K
Sample Set Method: 090 Carryover Sample Kit 1 Expiration date: 10/22/2010 12:00:00 AM
Processing Method Carryover PM, Carryover Clean Sample Kit 1 Lot Number: N070015
Report Method Name: Carryover Report Build Version: Empower 2 Software Build 2154 SPs
User Name: System Installed:
Sample Set Feature Release 5
Id: 11954 DB ID:
Result 847558692
Set Id 11966
Sample Set Name: Carryover Sample Set Start Date: 11/4/2010 9:31:58 PM
Peak Results
Sample Type: Unknown
SampleName Sample Type Vial Injection Name RT Area Height Peak Type % Carryover
1 PreBlank Unknow n 12 1 Caffeine 1.093 12212 5248 Found 0.0084
2 PreBlank Unknow n 12 2 Caffeine 1.099 11456 4924 Found 0.0079
Peak Results
Sample Type: Standard
SampleName Sample Type Vial Injection Name RT Area Height Peak Type % Carryover
1 Standard Standard 8 1 Caffeine 1.095 7356 3123 Found 0.0050
2 Standard Standard 8 2 Caffeine 1.095 7135 3032 Found 0.0050
3 Standard Standard 8 3 Caffeine 1.097 7278 3079 Found 0.0050
Peak Results
Sample Type: Unknown
SampleName Sample Type Vial Injection Name RT Area Height Peak Type % Carryover
1 PostBlank1 Unknow n 12 1 Caffeine 1.097 12752 5371 Found 0.0088
2 PostBlank2 Unknow n 12 1 Caffeine 1.093 11818 5040 Found 0.0081
3 PostBlank3 Unknow n 12 1 Caffeine 1.092 11832 4970 Found 0.0082
Printed 7:59:22 AM US/Eastern 11/17/2010 Page: 3 of 3
You might also like
- Di HybridDocument6 pagesDi HybridTaheera Khan100% (2)
- Math Preparedness WorkbookDocument26 pagesMath Preparedness WorkbookAnonymous czkmnf100% (2)
- Sociological Analysis For The Joy Luck ClubDocument5 pagesSociological Analysis For The Joy Luck ClubNathan Doesitmatter100% (2)
- flgx213 (2) Pkjun2015Document6 pagesflgx213 (2) Pkjun2015Jason Van Den HeeverNo ratings yet
- Vic 2019 CatalogDocument893 pagesVic 2019 CatalogAnna Mae Marantan100% (1)
- SB10 Chapter 8 AnswersDocument16 pagesSB10 Chapter 8 Answersanna jones20% (5)
- Denis Deletant, RomanianStudiesUKDocument9 pagesDenis Deletant, RomanianStudiesUKMarius SilvesanNo ratings yet
- Carryover ReportDocument3 pagesCarryover ReportRiad ManamanniNo ratings yet
- Carryover ReportDocument3 pagesCarryover ReportRiad ManamanniNo ratings yet
- Flow Rate Linearity ReportDocument3 pagesFlow Rate Linearity ReportRiad ManamanniNo ratings yet
- Carryover ReportDocument3 pagesCarryover ReportRiad ManamanniNo ratings yet
- Test Injections ReportDocument3 pagesTest Injections ReportRiad ManamanniNo ratings yet
- Flow Rate Linearity ReportDocument3 pagesFlow Rate Linearity ReportRiad ManamanniNo ratings yet
- Test Injections ReportDocument3 pagesTest Injections ReportRiad ManamanniNo ratings yet
- Dilutor Linearity ReportDocument3 pagesDilutor Linearity ReportRiad ManamanniNo ratings yet
- Online System PrecisionDocument3 pagesOnline System PrecisionRiad ManamanniNo ratings yet
- UV Test InjDocument4 pagesUV Test InjRiad ManamanniNo ratings yet
- UV System PrecDocument3 pagesUV System PrecRiad ManamanniNo ratings yet
- Online System PrecisionDocument3 pagesOnline System PrecisionRiad ManamanniNo ratings yet
- System Precision ReportDocument3 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- Diff Operating Characteristic - Otf:: Test Object - Differential ParametersDocument3 pagesDiff Operating Characteristic - Otf:: Test Object - Differential ParametersDuna FlaviuNo ratings yet
- Dilutor Linearity ReportDocument3 pagesDilutor Linearity ReportRiad ManamanniNo ratings yet
- Caff - 1-3 - 1-14 - 11th - 8162021 - 001 PDFDocument1 pageCaff - 1-3 - 1-14 - 11th - 8162021 - 001 PDFAli RizviNo ratings yet
- System Precision ReportDocument3 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- Caff - 1-3 - 1-16 - 11th - 8162021 - 003Document1 pageCaff - 1-3 - 1-16 - 11th - 8162021 - 003Ali RizviNo ratings yet
- Prueba de EficienciaDocument11 pagesPrueba de EficienciaIván Andrés Benavides pajoyNo ratings yet
- Dilutor Accuracy and Precision ReportDocument5 pagesDilutor Accuracy and Precision ReportRiad ManamanniNo ratings yet
- LC Calibration Report: Peak: ScopoletinDocument7 pagesLC Calibration Report: Peak: Scopoletinnananana199103No ratings yet
- Test Injections UV RMDocument3 pagesTest Injections UV RMRiad ManamanniNo ratings yet
- Caff - 1-3 - 1-15 - 11th - 8162021 - 002Document1 pageCaff - 1-3 - 1-15 - 11th - 8162021 - 002Ali RizviNo ratings yet
- Ochnaflavone 4'-Methyl ether-COA-PRF21083044Document2 pagesOchnaflavone 4'-Methyl ether-COA-PRF21083044DhavalNo ratings yet
- Gradient Performance ReportDocument3 pagesGradient Performance ReportRiad ManamanniNo ratings yet
- HARTIN TRF 1 Sef LVDocument43 pagesHARTIN TRF 1 Sef LVzita aisyahNo ratings yet
- Comp AccDocument3 pagesComp AccRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- Test Injections UV RMDocument3 pagesTest Injections UV RMRiad ManamanniNo ratings yet
- Caff - 1-4 - 1-20 - H1st - 8162021 - 002Document1 pageCaff - 1-4 - 1-20 - H1st - 8162021 - 002Ali RizviNo ratings yet
- 长春花碱液相色谱Document3 pages长春花碱液相色谱Hoàng Ngọc AnhNo ratings yet
- Revised - PT Preliminary Report - Grape 25.08.2023Document16 pagesRevised - PT Preliminary Report - Grape 25.08.2023pratap.kadam43No ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- ISM SplitDocument4 pagesISM SplitRiad ManamanniNo ratings yet
- Chromatogram and Results: Injection DetailsDocument5 pagesChromatogram and Results: Injection DetailsNova SeptiNo ratings yet
- Wavelength Accuracy ReportDocument3 pagesWavelength Accuracy ReportRiad ManamanniNo ratings yet
- 015-Operation & Cleaning of Balance (100 KG)Document2 pages015-Operation & Cleaning of Balance (100 KG)Ashok Lenka100% (1)
- Diff Operating Characteristic Y-N PH in B11 - P643 - F87T FINAL SETTINGDocument1 pageDiff Operating Characteristic Y-N PH in B11 - P643 - F87T FINAL SETTINGItachi SaiyanNo ratings yet
- Carryover RMDocument4 pagesCarryover RMRiad ManamanniNo ratings yet
- CarryoverDocument3 pagesCarryoverRiad ManamanniNo ratings yet
- HPLCDocument12 pagesHPLCviravangsoNo ratings yet
- Diff Operating Characteristic in UNIT 2 - DIFF - CONDENSATE EXTRACTION PUMP A - 20BBB07Document6 pagesDiff Operating Characteristic in UNIT 2 - DIFF - CONDENSATE EXTRACTION PUMP A - 20BBB07MühâMméd SàhãdhNo ratings yet
- Gradient Performance ReportDocument3 pagesGradient Performance ReportRiad ManamanniNo ratings yet
- UV Injection Lin Acc RMDocument3 pagesUV Injection Lin Acc RMRiad ManamanniNo ratings yet
- Test Injections ReportDocument5 pagesTest Injections ReportRiad ManamanniNo ratings yet
- Noise ReportDocument2 pagesNoise ReportRiad ManamanniNo ratings yet
- Operation and Calibration of HPLC SystemDocument12 pagesOperation and Calibration of HPLC SystemMubarak PatelNo ratings yet
- Diff Trip Time Characteristic:: Test Object - Device SettingsDocument4 pagesDiff Trip Time Characteristic:: Test Object - Device SettingsDuna FlaviuNo ratings yet
- Calibration of HPLCDocument12 pagesCalibration of HPLCprashanth50% (6)
- Noise ReportDocument2 pagesNoise ReportRiad ManamanniNo ratings yet
- Diff Operating CharacteristicDocument6 pagesDiff Operating CharacteristicMühâMméd SàhãdhNo ratings yet
- ISM Split PrecDocument4 pagesISM Split PrecRiad ManamanniNo ratings yet
- Basic Instrumental Analysis Experiment 2: Uv-Visible Determination of An Unknown Concentration of Kmno4 SolutionDocument7 pagesBasic Instrumental Analysis Experiment 2: Uv-Visible Determination of An Unknown Concentration of Kmno4 SolutionSiti Maizatul AkmaNo ratings yet
- Nursing Lab Articles & Advance Skill Labs For GNM As Per Inc - DSDDocument5 pagesNursing Lab Articles & Advance Skill Labs For GNM As Per Inc - DSDRahul Kashyap100% (1)
- Chlopheniramine STDDocument2 pagesChlopheniramine STDIrvinn NhutsNo ratings yet
- Flow Rate Linearity ReportDocument3 pagesFlow Rate Linearity ReportRiad ManamanniNo ratings yet
- Test Injections ReportDocument5 pagesTest Injections ReportRiad ManamanniNo ratings yet
- Procedimento para Calibração de Cola PDFDocument1 pageProcedimento para Calibração de Cola PDFDeyler SaquettoNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- ELS Detector Response ReportDocument5 pagesELS Detector Response ReportRiad ManamanniNo ratings yet
- SQ Detector Operators GuideDocument220 pagesSQ Detector Operators GuideRiad ManamanniNo ratings yet
- System IQDocument3 pagesSystem IQRiad ManamanniNo ratings yet
- System PrecisionDocument3 pagesSystem PrecisionRiad ManamanniNo ratings yet
- Excitation Wavelength AccDocument2 pagesExcitation Wavelength AccRiad ManamanniNo ratings yet
- Sampler Temp AccDocument1 pageSampler Temp AccRiad ManamanniNo ratings yet
- MS Detector Lin Sens RMDocument4 pagesMS Detector Lin Sens RMRiad ManamanniNo ratings yet
- Wavelength Accuracy ReportDocument3 pagesWavelength Accuracy ReportRiad ManamanniNo ratings yet
- Carryover RMDocument4 pagesCarryover RMRiad ManamanniNo ratings yet
- Gradient RMDocument6 pagesGradient RMRiad ManamanniNo ratings yet
- Qualification Summary RMDocument2 pagesQualification Summary RMRiad ManamanniNo ratings yet
- Test Injections UV RMDocument3 pagesTest Injections UV RMRiad ManamanniNo ratings yet
- Acq SM MP FRMDocument1 pageAcq SM MP FRMRiad ManamanniNo ratings yet
- Test Injections RMDocument3 pagesTest Injections RMRiad ManamanniNo ratings yet
- AcqQSM BSMOQDocument3 pagesAcqQSM BSMOQRiad ManamanniNo ratings yet
- UV Injection Lin Acc RMDocument3 pagesUV Injection Lin Acc RMRiad ManamanniNo ratings yet
- Acq Samp Org OQDocument3 pagesAcq Samp Org OQRiad ManamanniNo ratings yet
- Amounts TUVPDA2996Document4 pagesAmounts TUVPDA2996Riad ManamanniNo ratings yet
- Acq QSM MP FRMDocument1 pageAcq QSM MP FRMRiad ManamanniNo ratings yet
- Acq Els MP FRMDocument1 pageAcq Els MP FRMRiad ManamanniNo ratings yet
- Wavelength Accuracy ReportDocument3 pagesWavelength Accuracy ReportRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- Acq BSM MP FRMDocument1 pageAcq BSM MP FRMRiad ManamanniNo ratings yet
- Compositional Accuracy ReportDocument2 pagesCompositional Accuracy ReportRiad ManamanniNo ratings yet
- Gain Linearity ReportDocument3 pagesGain Linearity ReportRiad ManamanniNo ratings yet
- Test Equip CalDocument2 pagesTest Equip CalRiad ManamanniNo ratings yet
- Final ApprovalDocument1 pageFinal ApprovalRiad ManamanniNo ratings yet
- Detector Linearity & Sensitivity ReportDocument4 pagesDetector Linearity & Sensitivity ReportRiad ManamanniNo ratings yet
- ProcSmMpFrm 715002173rBDocument1 pageProcSmMpFrm 715002173rBRiad ManamanniNo ratings yet
- Jepretan Layar 2023-08-16 Pada 3.51.45 PMDocument47 pagesJepretan Layar 2023-08-16 Pada 3.51.45 PMFenni PrisiliaNo ratings yet
- Study Session 7 - Understanding Balance Sheets - 2022Document54 pagesStudy Session 7 - Understanding Balance Sheets - 2022IhuomacumehNo ratings yet
- MR 11plct Frevzfeb12Document496 pagesMR 11plct Frevzfeb12charles_boyle_3No ratings yet
- Data AlatDocument29 pagesData AlatRefi Opti faniNo ratings yet
- HP Smart Update Firmware DVD 9.20 What's New and Release NotesDocument13 pagesHP Smart Update Firmware DVD 9.20 What's New and Release NotesDavid SaralidzeNo ratings yet
- Thesis Statement For BiodieselDocument8 pagesThesis Statement For Biodieselkualxkiig100% (2)
- Nutrition in Animals NotesDocument5 pagesNutrition in Animals NotesGowri YadavNo ratings yet
- Ibm Food Trust Value Prop Pamphlets - Dec2018 - 46022546USENDocument14 pagesIbm Food Trust Value Prop Pamphlets - Dec2018 - 46022546USENRoxanaNo ratings yet
- 555 TimerDocument48 pages555 TimeracidreignNo ratings yet
- BS ConsolidationDocument25 pagesBS ConsolidationKamukwema johnNo ratings yet
- Hindi E XDocument19 pagesHindi E XTamboli Shaikh Muaavvir AkbarNo ratings yet
- Sonia Baltodano ResumeDocument1 pageSonia Baltodano Resumesbaltoda100% (2)
- Module IDocument14 pagesModule IDianna Rose BelenNo ratings yet
- Aluminium FormworkDocument14 pagesAluminium FormworkebinVettuchirayil100% (6)
- TDS BLR 896 EN ScreenDocument1 pageTDS BLR 896 EN ScreenLuis Angel MendozaNo ratings yet
- Imam Ash Shafi'EeDocument3 pagesImam Ash Shafi'EeAmalia AmiraNo ratings yet
- 42 Items Q and ADocument2 pages42 Items Q and AJoy NavalesNo ratings yet
- Understanding The Self Syllabus 2nd Sem 2021-22Document7 pagesUnderstanding The Self Syllabus 2nd Sem 2021-22Mary Crisza RentoriaNo ratings yet
- Vibracord. Vibration Meter Summary Sheet (Graphic) : Record Time: 4 SDocument1 pageVibracord. Vibration Meter Summary Sheet (Graphic) : Record Time: 4 SSadettin BağdatlıNo ratings yet
- Tle Freezing and Defrosting MeatDocument17 pagesTle Freezing and Defrosting MeatEnna PaulaNo ratings yet
- Scadapack 4203 PDFDocument7 pagesScadapack 4203 PDFFelix VelasquezNo ratings yet
- Emad 21522379 Type A KWH PDFDocument8 pagesEmad 21522379 Type A KWH PDFEMAD ABDULRAHMAN ABDULLAH HASAN MASHRAH -No ratings yet
- Mama AnnDocument2 pagesMama AnnVon JimenezNo ratings yet