Professional Documents
Culture Documents
Justin Lammersdorf Exploring Gases
Justin Lammersdorf Exploring Gases
Uploaded by
Melk KidOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Justin Lammersdorf Exploring Gases
Justin Lammersdorf Exploring Gases
Uploaded by
Melk KidCopyright:
Available Formats
Exploring the behavior of Gases
Name: Justin lammersdorf Date: 10/16/2023
Get Started: Click on this link to open the gas simulation
Part I
1. Go to intro. Choose only one type of particle.
2. Record the pressure, temperature and volume.
3. Give one pump of gas and observe the behavior. How would you describe this?
Record the pressure, temperature and volume.
pressure-2.5 atm temp-300k
4. Hold volume and temperature constant and give one more pump.
Record the number of particles and the pressure in the data table below.
Describe what you saw.
Repeat this a few times, either increasing or decreasing the number of the same type of particles.
Number Pressure (atm)
of
50 5.8
particles
75 9.1
100 11.6
125 15.2
150 19.5
5. Is there a relationship between the number of particles and the pressure? Briefly describe this.
the more particles there are the pressure also rises.
What is the pressure in the container due to? (what assumption are we making?)
the more particles in the container the more the pressure goes up.
Downloaded by Melk Kid (kidmelk@gmail.com)
Part II:
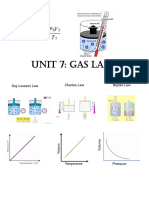
There are 3 parameters that need to be specified when describing a specific quantity of a gas. They are:
Pressure, Volume and Temperature. We will keep the number of particles constant in each “experiment”
and explore the effect (if any) a change in any of these parameters may have on the behavior of the gas.
Choose the Laws option on the right. See picture
Experiment 1- Volume
1. Give one pump of gas into the chamber.
2. Choose to hold the volume constant by selecting that option in the
upper right-hand corner. See the picture.
What is the initial temperature (in K) and pressure (in atm) in the chamber?
5.8atm
3. Use the slider at the bottom of the simulator to add heat and double the temperature.
Did the pressure go up or go down? What is the new pressure in the chamber?
the pressure rose up to 11.7atm
4. Keeping the volume constant (and the number of particles constant), change the temperature and
record the pressure. Repeat 4 times and record your data. Sketch a graph to the right of the
table. Be sure to give a title to your graph and label the axis completely.
Independent variable is : pressure
Dependent Variable: temperture
Constants: volume
Temperature (K) Pressure (atm)
600 11.7 atm
550 10.7
500 9.7
450 8.7
400 7.8
Describe the graph and relationship:
12
pressure(atm)
10
8
6
4
2
Honors Chemistry 400 450 500 550 600 2
Srikanthan
temperture (k)
Downloaded by Melk Kid (kidmelk@gmail.com)
Experiment 2 - Temperature
1. Reset the simulator by selecting the reset button in the bottom right corner of the
simulation.
2. Give one pump of gas into the chamber.
3. Choose to hold the temperature constant by selecting that
option in the upper right-hand corner. See the picture.
What is the initial pressure (in atm) in the chamber?
4.2 atm
4. Locate the handle on the left of the chamber and slide it to the right as far as it will go.
What units are used to measure the width of the chamber? nm^3
Does the volume go up or go down when you slide it to the right? down , Did the pressure go up or
go down? up ; Why? there is less space for the particles so the pressure will increase due to more
tension that builds up in a smaller container.
5. Slide the handle all the way to the left as far as it will go.
Did the pressure go up or go down? down ; Why?
becuase the volume increased creating more space for the pressure to relase.
6. Keeping the temperature constant (and the number of particles constant), change the volume and
record the pressure. Repeat 4 times and record your data. Sketch a graph to the right of the
table. Be sure to give a title to your graph and label the axis completely
pressure
Independent variable is :
Dependent Variable: volume
Constants: temperture
Volume (nm3) Pressure (atm)
10.0 5.9
5.3
11.0
12.0 4.9
13.0 4.5
14.0
4.2
Describe the graph and relationship:
15
volume (nm³
10
Honors Chemistry 3
1 2 3 4 5 6
Downloaded by Melk Kid (kidmelk@gmail.com)
Experiment 3 - Pressure
1. Reset the simulator by selecting the reset button in the bottom right corner of the
simulation.
2. Give one pump of gas into the chamber.
3. Choose to hold the pressure constant by selecting that option in the upper right-
hand corner. See the picture.
What is the initial temperature (in K) in the chamber? 300
4. Use the slider at the bottom of the simulator to add heat and increase the
temperature.
Did the volume go up or go down? up
5. Keeping the pressure constant (and the number of particles constant), change the
temperature and record the volume. Repeat 4 times and record your data. Sketch a graph to the
right of the table. Be sure to give a title to your graph and label the axis completely
Independent variable is : volume
Dependent Variable: temperture
Constants: pressure and particles
Temperature (K) Volume (nm3)
150 5
200 6.7
250 8.4
10
300
350 11.7
Describe the graph and relationship:
350
temperture (k)
300
250
200
150
Downloaded by Melk Kid (kidmelk@gmail.com)
Analysis
findings
In each ofof
the your experiments
experiments, byoneusing
you hold arrows
parameter (T orwhile
constant to represent
!) changing what
the other happened.
2. Summarize the
Experiment 1: When temperature went , the pressure went up .
up
Possible reason?
the temperture increases and creates more movement in the particles that increases the pressure,
Real world example?
pressure cooker when you expand the pot gas is realesed that redices the pressure insdie a pressure cooker
Experiment 2: When volume went down , the pressure went up .
Possible reason?
the size of the container gave the particles less space to move increasing the pressure inside the container,
Real world example?
when you squeez a ball it gets harder and harder to squeeze the more you push it.
up
Experiment 3: When temperature went , the volume went up .
Possible reason?
it gave room for the particles to move without building up the pressure
Real world example?
popcorn when you pop popcorn the temp rises and buiilds up the pressure however the bigger the pot the longer it takes
for the pressure to increase due to the volume of the container.
Downloaded by Melk Kid (kidmelk@gmail.com)
You might also like
- Exploring Pressure With PhETDocument3 pagesExploring Pressure With PhETRobert Campillo100% (1)
- 5-1: Boyle's Law: Pressure and VolumeDocument5 pages5-1: Boyle's Law: Pressure and VolumeEliana Hwang100% (1)
- MIME 3450 Energy Laboratory-1Document6 pagesMIME 3450 Energy Laboratory-1shafahatNo ratings yet
- HEAT PUMP REPORT ReportDocument8 pagesHEAT PUMP REPORT ReportShan MuthuNo ratings yet
- Boyles Law PhETDocument7 pagesBoyles Law PhETShally RahmawatyNo ratings yet
- Exact Constraint (ME Magazine Sep'09)Document7 pagesExact Constraint (ME Magazine Sep'09)aoeusnthidNo ratings yet
- Dr. H. S. Kalsi Dept. of Physics Guru Nanak Khalsa College Matunga, MumbaiDocument100 pagesDr. H. S. Kalsi Dept. of Physics Guru Nanak Khalsa College Matunga, MumbaiGAURI AGROTECH100% (1)
- From Maxwell's Equations To Free and Guided Electromagnetic Waves - An Introduction For First-Year UndergraduatesDocument71 pagesFrom Maxwell's Equations To Free and Guided Electromagnetic Waves - An Introduction For First-Year UndergraduatesNicolas MarinNo ratings yet
- Laboratory Exercise 2 Exploring The Behavior of Gases-1Document10 pagesLaboratory Exercise 2 Exploring The Behavior of Gases-1imaniNo ratings yet
- Gas Law ActivityDocument7 pagesGas Law Activitydaniel tabuzoNo ratings yet
- Simulation 2Document10 pagesSimulation 2Myeisha Williams100% (1)
- Phet - Gas Laws in html5Document5 pagesPhet - Gas Laws in html5api-573214664No ratings yet
- LC+HL+Physics+Solutions 2015Document21 pagesLC+HL+Physics+Solutions 2015Marwa Saad SherifNo ratings yet
- Lab 6 Heat of VaporizationDocument6 pagesLab 6 Heat of Vaporizationapi-458764744No ratings yet
- Orca Share Media1644931117177 6899341164500579200Document6 pagesOrca Share Media1644931117177 6899341164500579200Lae RamirezNo ratings yet
- Lab 6Document9 pagesLab 6Dilshad S FaisalNo ratings yet
- Real Gas LabDocument10 pagesReal Gas LabRaymond BaldelovarNo ratings yet
- Physics Thursday WorkDocument17 pagesPhysics Thursday WorkAndrew AndersonNo ratings yet
- Mechanical Part BDocument30 pagesMechanical Part BAhmad HashmiNo ratings yet
- Density of LiquidsDocument11 pagesDensity of LiquidsStephen Mirdo100% (3)
- Gases Properties Phet LabDocument3 pagesGases Properties Phet LabNelvin Rivera Nool100% (1)
- Experiment 2. Vapor Pressure.Document4 pagesExperiment 2. Vapor Pressure.Esmeralda A OcampoNo ratings yet
- Marcet Boiler Experiment LabsheetDocument8 pagesMarcet Boiler Experiment LabsheetWan NurdyanaNo ratings yet
- Exp Boyles Law 15-08-22Document4 pagesExp Boyles Law 15-08-22Phil100% (1)
- Pressure and TemperatureDocument2 pagesPressure and TemperatureTanish bossNo ratings yet
- Act B4 GAS-SI LabReport F2022Document7 pagesAct B4 GAS-SI LabReport F2022Hassan ZakariaNo ratings yet
- Key - 8.1 Gas Law Lab PDFDocument6 pagesKey - 8.1 Gas Law Lab PDFzhuzaiNo ratings yet
- HM4 Salikhov AmirDocument7 pagesHM4 Salikhov Amirsalikhovamir77No ratings yet
- Phet Gas PropertiesDocument4 pagesPhet Gas Propertiesapi-715452941No ratings yet
- Chem 101 Lab 12 Gas LawsDocument12 pagesChem 101 Lab 12 Gas LawsT Josiah RichardsonNo ratings yet
- Lab 9 - Pressure Vs Temperature.Document5 pagesLab 9 - Pressure Vs Temperature.Omar AlkhadraNo ratings yet
- Gas Phase KineticsDocument7 pagesGas Phase KineticsBrio Chemical0% (1)
- LAB - Period of A Spring (Phet)Document8 pagesLAB - Period of A Spring (Phet)Nicholas WongNo ratings yet
- Appendix A: Laboratory Report FormatDocument10 pagesAppendix A: Laboratory Report Formatahsan aliNo ratings yet
- States of Matter Worksheet - TaggedDocument6 pagesStates of Matter Worksheet - Taggedguiang.michaelaNo ratings yet
- Optimization of Capillary Tube in Air Conditioning SystemDocument11 pagesOptimization of Capillary Tube in Air Conditioning SystemFauzi Hussin LeoNo ratings yet
- Experiment: Ideal Gas Law (Manual+ Worksheet) : UsingDocument7 pagesExperiment: Ideal Gas Law (Manual+ Worksheet) : UsingMazanda YalinduaNo ratings yet
- Key - 2544434 - 2024-03-15 09 - 56 - 27 +0000Document24 pagesKey - 2544434 - 2024-03-15 09 - 56 - 27 +0000P.S.S AmulyaNo ratings yet
- Lab Report Thermo - RealDocument12 pagesLab Report Thermo - Realazzatul amiraNo ratings yet
- EAT 339 Jan 2023 TCT QuestionsDocument10 pagesEAT 339 Jan 2023 TCT QuestionsOji Ifeanyi0% (1)
- Ellsworth, Patrick Jordan S.: The Pressure IncreasesDocument11 pagesEllsworth, Patrick Jordan S.: The Pressure IncreasesJohn NicolasNo ratings yet
- Pressure Temperature PhETDocument6 pagesPressure Temperature PhETMiNH HayatNo ratings yet
- Phet Gas Law Simulation 2010Document8 pagesPhet Gas Law Simulation 2010api-262304087No ratings yet
- Lab Iv. Graphing DataDocument4 pagesLab Iv. Graphing DataLiliana PerezNo ratings yet
- API Creep RuptureDocument3 pagesAPI Creep RuptureRavi Raj100% (1)
- Unit 1 Collaborative Notes - Models of The Particulate Nature of Matter SLDocument36 pagesUnit 1 Collaborative Notes - Models of The Particulate Nature of Matter SLRodrigo ErthalNo ratings yet
- 유체역학 2장Document63 pages유체역학 2장김진성No ratings yet
- Unit 7 Gas Laws Packet 2021Document40 pagesUnit 7 Gas Laws Packet 2021Alberto Laborte Jr.No ratings yet
- GEOG 323 - Lab 1Document5 pagesGEOG 323 - Lab 1Rajeev misraNo ratings yet
- Finite Element Numerical Simulation of A TIG Welding Test: A.Capriccioli, P.FrosiDocument46 pagesFinite Element Numerical Simulation of A TIG Welding Test: A.Capriccioli, P.Frosimeckup123No ratings yet
- WS Practice W GraphsDocument4 pagesWS Practice W GraphsgiyagirlsNo ratings yet
- Tugas FisikaDocument3 pagesTugas FisikabNo ratings yet
- SIM Comparison 100 MPa Hydraulic Gauge Pressure Balance PaperDocument6 pagesSIM Comparison 100 MPa Hydraulic Gauge Pressure Balance Paperargelio arroyoNo ratings yet
- Yoyo 9.1 Lab Report On Hooke S Law 1Document7 pagesYoyo 9.1 Lab Report On Hooke S Law 1Linda LiNo ratings yet
- Assignment 6 Solutions PDFDocument8 pagesAssignment 6 Solutions PDFKarim TouqanNo ratings yet
- Creep QuestionDocument8 pagesCreep QuestionWaris Nawaz KhanNo ratings yet
- 11 Boyles Law LABSu2020 Online VersionDocument4 pages11 Boyles Law LABSu2020 Online VersionAcademic NerdsNo ratings yet
- Fall 2021 CHEM301 Physical Chemistry Laboratory: EXPERIMENT 1 Report Writing GuideDocument3 pagesFall 2021 CHEM301 Physical Chemistry Laboratory: EXPERIMENT 1 Report Writing GuideFULL DİAMOND SET HONEYBADGERNo ratings yet
- 314problems NorthwesternUnivDocument8 pages314problems NorthwesternUnivAnonymous 9rJe2lOskxNo ratings yet
- States of Matter - Charles GuiangDocument6 pagesStates of Matter - Charles Guiangguiang.michaelaNo ratings yet
- The Realation Betewwn Press and TemperatureDocument7 pagesThe Realation Betewwn Press and TemperatureAyad DariNo ratings yet
- Ap World History 5.1Document10 pagesAp World History 5.1Melk KidNo ratings yet
- MYP2209 REM G09 U2B Kapuscinski LTE EwdDocument4 pagesMYP2209 REM G09 U2B Kapuscinski LTE EwdMelk KidNo ratings yet
- Berlatsky LTE EwdDocument2 pagesBerlatsky LTE EwdMelk KidNo ratings yet
- Bernoullisprinciple - 5 8 02 09 17 508Document34 pagesBernoullisprinciple - 5 8 02 09 17 508Melk KidNo ratings yet
- The Horned ToadDocument2 pagesThe Horned ToadMelk KidNo ratings yet
- Saddle Crack AnalysisDocument9 pagesSaddle Crack AnalysisSiva Prasad MamillapalliNo ratings yet
- 1 s2.0 S0038092X1930355X Main PDFDocument9 pages1 s2.0 S0038092X1930355X Main PDFkrunalgangawane85No ratings yet
- Fatigue Failure Analysis of A Cooling Fan Blade: A Case StudyDocument7 pagesFatigue Failure Analysis of A Cooling Fan Blade: A Case StudyJay Dee11No ratings yet
- Wave Particle DualityWave-Particle DualityDocument4 pagesWave Particle DualityWave-Particle Dualityatom77966No ratings yet
- Symmetrical Fault AnalysisDocument81 pagesSymmetrical Fault AnalysisAshutoshSharma100% (1)
- Thin Walled Pressure VesselDocument13 pagesThin Walled Pressure VesselaalijanaabNo ratings yet
- Viva Questions in AnsysDocument5 pagesViva Questions in AnsyssharathNo ratings yet
- En 15377 - 3Document64 pagesEn 15377 - 3олегNo ratings yet
- Social Science Research Methods - PISU 2019Document111 pagesSocial Science Research Methods - PISU 2019Samaki AlsatNo ratings yet
- Extra Edge PhysicsDocument115 pagesExtra Edge PhysicsVishuNo ratings yet
- Modern Physics NotesDocument15 pagesModern Physics NotesSigwa Timo100% (1)
- CT-CH 1-Lecture Slides For Academic VersionDocument44 pagesCT-CH 1-Lecture Slides For Academic Versionarmando josueNo ratings yet
- Chapter 9: External Incompressible Viscous Flows: (Unbounded)Document133 pagesChapter 9: External Incompressible Viscous Flows: (Unbounded)matthewrjonesrugbyNo ratings yet
- CHM 17 Experiment 10 Mixed Melting PTDocument6 pagesCHM 17 Experiment 10 Mixed Melting PTwilkinson kimathiNo ratings yet
- Ep 10Document5 pagesEp 10Pratik BeheraNo ratings yet
- Question Bank OHE 22.07.2015 PDFDocument111 pagesQuestion Bank OHE 22.07.2015 PDFAdhir giriNo ratings yet
- B.Sc. Mechanical Engineering 2018 & OnwardDocument38 pagesB.Sc. Mechanical Engineering 2018 & OnwardMohtasham NaeemNo ratings yet
- Parameter Value: Inputs For Bend Scour OnlyDocument25 pagesParameter Value: Inputs For Bend Scour OnlysumitanuragNo ratings yet
- Partial Differential Equations and Fourier AnalysisDocument195 pagesPartial Differential Equations and Fourier AnalysischakfarmerNo ratings yet
- Momentum QuetionsDocument7 pagesMomentum Quetionsquami3quartsNo ratings yet
- Minimum Thermal FlowDocument4 pagesMinimum Thermal Flowdk4monjureNo ratings yet
- Physics 2D and 3D MotionDocument65 pagesPhysics 2D and 3D MotionMary Ann DimacaliNo ratings yet
- A Study On The Effect of Welding Sequence in Fabrication of Large Stiffened Plate Panels (Paper)Document8 pagesA Study On The Effect of Welding Sequence in Fabrication of Large Stiffened Plate Panels (Paper)pal_malayNo ratings yet
- Theory of Quantum EntanglementDocument103 pagesTheory of Quantum EntanglementMark M. Wilde33% (3)
- Vector Mechanics For Engineers: Dynamics: Twelfth EditionDocument100 pagesVector Mechanics For Engineers: Dynamics: Twelfth EditionFady MagedNo ratings yet
- Tank-API-650-SI-Manzanillo-30 mbls-API-650-Calculation-Report - 1Document52 pagesTank-API-650-SI-Manzanillo-30 mbls-API-650-Calculation-Report - 1Tony Lozada Mahuen100% (1)
- JEE Main Mock Test From IITian's TAPASYADocument15 pagesJEE Main Mock Test From IITian's TAPASYAAkash GoyalNo ratings yet