Professional Documents
Culture Documents
CMV Igm 2018-10 v9-1
CMV Igm 2018-10 v9-1
Uploaded by
ابو حمزةOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
CMV Igm 2018-10 v9-1
CMV Igm 2018-10 v9-1
Uploaded by
ابو حمزةCopyright:
Available Formats
ms_04784618190V9.
Elecsys CMV IgM
MODULAR ANALYTICS E170
cobas e 411
04784618 190 100
cobas e 601
cobas e 602
English CMV IgG avidity is used as an aid.2,4,5,12 A positive IgM result in
combination with a low avidity index for IgG is a strong indication of a recent
System information primary CMV infection.4,5,12 Seroconversion to CMV IgM and IgG may also
For cobas e 411 analyzer: test number 580 indicate a recent CMV infection.2,3,4,5,12
For MODULAR ANALYTICS E170, cobas e 601 and cobas e 602
analyzers: Application Code Number 011 Test principle
µ-Capture test principle. Total duration of assay: 18 minutes.
Intended use
Immunoassay for the in vitro qualitative determination of IgM antibodies to ▪ 1st incubation: 10 µL of sample are automatically prediluted 1:20 with
cytomegalovirus in human serum and plasma. Diluent Universal. Biotinylated monoclonal anti‑h‑IgM‑specific antibodies
are added.
Results obtained with this assay are used as an aid in the diagnosis of
recent CMV infections. ▪ 2nd incubation: CMV‑specific recombinant antigen labeled with a
The electrochemiluminescence immunoassay “ECLIA” is intended for use ruthenium complexa) and streptavidin-coated microparticles are added.
on Elecsys and cobas e immunoassay analyzers. Anti‑CMV IgM antibodies present in the sample react with the
ruthenium-labeled CMV‑specific recombinant antigen. The complex
Regulatory status becomes bound to the solid phase via interaction of biotin and
This assay has been CE marked according to Directive 98/79/EC. Test streptavidin.
performance has been established for diagnostic use and for testing of ▪ The reaction mixture is aspirated into the measuring cell where the
blood donations. microparticles are magnetically captured onto the surface of the
Summary electrode. Unbound substances are then removed with
ProCell/ProCell M. Application of a voltage to the electrode then induces
Cytomegalovirus (CMV), a member of the herpes virus family, is ubiquitous chemiluminescent emission which is measured by a photomultiplier.
in all human populations, causing infections which are followed by life-long
latency in the host with occasional reactivations.1,2 The seroprevalence of ▪ Results are determined automatically by the software by comparing the
antibodies in adults ranges from 40‑100 % with inverse correlation to electrochemiluminescence signal obtained from the reaction product of
socioeconomic status.1,2,3 CMV is transmitted through body fluids, including the sample with the signal of the cutoff value previously obtained by
blood, genital secretions and breast milk. Saliva and urine of infected calibration.
individuals also represent a prominent source of infection, and children, a) Tris(2,2'-bipyridyl)ruthenium(II)-complex (Ru(bpy) )
especially those attending day care facilities, are an important vector for
viral spread.2,3,4,5,6 In immunocompetent individuals primary CMV infection Reagents - working solutions
is usually mild or asymptomatic.2,5 Patients commonly present with a The reagent rackpack (M, R1, R2) is labeled as CMVIGM.
mononucleosis-like syndrome, including fever, sore throat, cervical
lymphadenopathy, malaise, headache, muscle ache and joint pains.2,3,5,7 M Streptavidin-coated microparticles (transparent cap), 1 bottle, 6.5 mL:
During pregnancy, CMV can cause congenital infection which may result in Streptavidin-coated microparticles 0.72 mg/mL; preservative.
permanent physical and/or neurological sequelae to the child.5 CMV
infection can be primary, i.e. newly acquired, or secondary, i.e. due to R1 Anti-h-IgM-Ab~biotin (gray cap), 1 bottle, 9 mL:
reactivation of the latent virus or re-infection with a different viral strain.3,5 Biotinylated monoclonal anti‑h‑IgM antibody (mouse) > 500 µg/L,
Primary CMV infection is reported in 1‑4 % of seronegative women during
pregnancy and the risk of transmission to the fetus is estimated to be about MES buffer 50 mmol/L, pH 6.5; preservative.
30‑40 %.3,4 Reactivation of CMV infection during pregnancy is reported in R2 CMV-Ag~Ru(bpy) (black cap), 1 bottle, 9 mL:
10‑30 % of seropositive women and, in this circumstance, the risk of
transmission of the virus is about 1‑3 %.3,4,5 Overall, prenatal CMV infection CMV‑specific antigen (recombinant, E. coli) labeled with ruthenium
occurs in 0.6‑0.7 % of all life births in the developed world.4,5,8 The majority complex > 50 µg/L; MES buffer 50 mmol/L, pH 5.5; preservative.
of babies born with congenital CMV infection are asymptomatic at birth.8,9,10
Of these 5‑15 % still develop irreversible impairments, most frequently CMVIGM Cal1 Negative calibrator 1 (white cap), 2 bottles of 1.0 mL
hearing loss, that can occur several months or even years after birth.5,8,9,10
For babies symptomatic at birth, prognosis is very poor, and the vast each:
majority will develop severe mental impairment and/or hearing loss.5,8,9,10 Human serum, negative for anti‑CMV IgM; preservative.
Different studies have shown that the risk of symptomatic congenital
disease in the fetus or newborn infant is high, when maternal primary CMVIGM Cal2 Positive calibrator 2 (black cap), 2 bottles of 1.0 mL each:
infection takes place in early pregnancy before week 20 of gestation, and Anti‑CMV IgM (human serum) in HEPES buffer, pH 7.4;
lower thereafter.4,5 The congenital CMV infection caused by recurrent
maternal infection seldom leads to symptomatic disease at birth.4,5 bovine albumin; preservative.
At risk for CMV infection and disease are also immunocompromised Precautions and warnings
patients such as transplant recipients and HIV infected patients where the For in vitro diagnostic use.
virus can cause life-threatening diseases.11,12 The CMV status of transplant Exercise the normal precautions required for handling all laboratory
donors and recipients is very important, as it will determine prophylactic and reagents.
pre-emptive treatment strategies against CMV. CMV‑negative transplant Disposal of all waste material should be in accordance with local guidelines.
recipients should receive donations from CMV‑negative individuals or Safety data sheet available for professional user on request.
leukocyte depleted blood products. CMV can reside in latency in infected
cells and free viral DNA load is usually low. The CMV status can still be All human material should be considered potentially infectious.
determined by testing for CMV IgG antibodies. All products derived from human blood (CMVIGM Cal1, CMVIGM Cal2) are
Within the appropriate clinical context, the first step in diagnosing acute prepared exclusively from the blood of donors tested individually and shown
primary CMV infection is most commonly made by the detection of to be free from HBsAg and antibodies to HCV and HIV.
anti‑CMV‑specific IgG and IgM antibodies.5 Samples being reactive for IgM The serum containing anti‑CMV IgM (CMVIGM Cal2) was sterile filtrated.
antibodies indicate an acute, recent or reactivated infection.2,4,5,12 For The testing methods used assays approved by the FDA or cleared in
further analysis of a primary CMV infection the determination of the compliance with the European Directive 98/79/EC, Annex II, List A.
2018-10, V 9.0 English 1/5
ms_04784618190V9.0
Elecsys CMV IgM
However, as no testing method can rule out the potential risk of infection Stable for 4 weeks at 2‑8 °C, 7 days at 25 °C, 3 months at ‑20 °C (± 5 °C).
with absolute certainty, the material should be handled with the same level The samples may be frozen 5 times.
of care as a patient specimen. In the event of exposure, the directives of the The sample types listed were tested with a selection of sample collection
responsible health authorities should be followed.13,14 tubes or systems that were commercially available at the time of testing, i.e.
Avoid foam formation in all reagents and sample types (specimens, not all available tubes of all manufacturers were tested. Sample collection
calibrators and controls). systems from various manufacturers may contain differing materials which
could affect the test results in some cases. When processing samples in
Reagent handling primary tubes (sample collection systems), follow the instructions of the
The reagents in the kit are ready‑for‑use and are supplied in bottles tube manufacturer.
compatible with the system.
Specimens should not be subsequently altered with additives (biocides,
cobas e 411 analyzer: The calibrators should only be left on the analyzer anti‑oxidants or substances that could possibly change the pH of the
during calibration at 20‑25 °C. After use, close the bottles as soon as sample) in order to avoid erroneous findings.
possible and store upright at 2‑8 °C.
Pooled samples and other artificial material may have different effects on
Due to possible evaporation effects, not more than 5 calibration procedures different assays and thus may lead to discrepant findings.
per bottle set should be performed.
Centrifuge samples containing precipitates and frozen samples before
MODULAR ANALYTICS E170, cobas e 601 and cobas e 602 analyzers: performing the assay. Lyophilized samples can be used.
Unless the entire volume is necessary for calibration on the analyzers,
transfer aliquots of the ready‑for‑use calibrators into empty snap-cap bottles Do not use heat‑inactivated samples.
(CalSet Vials). Attach the supplied labels to these additional bottles. Store Ensure the samples, calibrators and controls are at 20‑25 °C prior to
the aliquots at 2‑8 °C for later use. measurement.
Perform only one calibration procedure per aliquot. Due to possible evaporation effects, samples, calibrators and controls on
All information required for correct operation is read in from the respective the analyzers should be analyzed/measured within 2 hours.
reagent barcodes. The performance of the Elecsys CMV IgM assay has not been established
Please note: Both the vial labels, and the additional labels (if available) with cadaveric samples or body fluids other than serum and plasma.
contain 2 different barcodes. The barcode between the yellow markers is Materials provided
for cobas 8000 systems only. If using a cobas 8000 system, please turn See “Reagents – working solutions” section for reagents.
the vial cap 180° into the correct position so the barcode can be read by the
system. Place the vial on the instrument as usual. Materials required (but not provided)
Storage and stability ▪ 04784626190, PreciControl CMV IgM, 16 x 1.0 mL
Store at 2‑8 °C. ▪ 11732277122, Diluent Universal, 2 x 16 mL sample diluent or
Do not freeze. 03183971122, Diluent Universal, 2 x 36 mL sample diluent
Store the Elecsys reagent kit upright in order to ensure complete ▪ 11776576322, CalSet Vials, 2 x 56 empty snap-cap bottles
availability of the microparticles during automatic mixing prior to use. ▪ General laboratory equipment
Stability of the reagent rackpack ▪ MODULAR ANALYTICS E170 or cobas e analyzer
Accessories for cobas e 411 analyzer:
unopened at 2‑8 °C up to the stated expiration date
▪ 11662988122, ProCell, 6 x 380 mL system buffer
after opening at 2‑8 °C 12 weeks
▪ 11662970122, CleanCell, 6 x 380 mL measuring cell cleaning
on MODULAR ANALYTICS E170 2 weeks or solution
and cobas e 6 weeks if stored alternately in the
▪ 11930346122, Elecsys SysWash, 1 x 500 mL washwater additive
refrigerator and on the analyzers
(up to 80 hours on the analyzer) ▪ 11933159001, Adapter for SysClean
▪ 11706802001, AssayCup, 60 x 60 reaction cups
Stability of the calibrators ▪ 11706799001, AssayTip, 30 x 120 pipette tips
unopened at 2‑8 °C up to the stated expiration date ▪ 11800507001, Clean‑Liner
after opening at 2‑8 °C 8 weeks Accessories for MODULAR ANALYTICS E170, cobas e 601 and
cobas e 602 analyzers:
on cobas e 411 at 20‑25 °C up to 5 hours
▪ 04880340190, ProCell M, 2 x 2 L system buffer
on MODULAR ANALYTICS E170, use only once
cobas e 601 and cobas e 602 at ▪ 04880293190, CleanCell M, 2 x 2 L measuring cell cleaning
solution
20‑25 °C
▪ 03023141001, PC/CC‑Cups, 12 cups to prewarm ProCell M and
Store calibrators upright in order to prevent the calibrator solution from CleanCell M before use
adhering to the snap‑cap.
▪ 03005712190, ProbeWash M, 12 x 70 mL cleaning solution for run
Specimen collection and preparation finalization and rinsing during reagent change
Only the specimens listed below were tested and found acceptable. ▪ 12102137001, AssayTip/AssayCup, 48 magazines x 84 reaction
Serum collected using standard sampling tubes or tubes containing cups or pipette tips, waste bags
separating gel. ▪ 03023150001, WasteLiner, waste bags
Li‑heparin, Na‑heparin, K2‑EDTA, K3‑EDTA, ACD, CPD, CP2D, CPDA and ▪ 03027651001, SysClean Adapter M
Na‑citrate plasma.
Accessories for all analyzers:
Criterion: Mean recovery of serum value: negative samples ± 0.2 COI
(cutoff-index); boderline/reactive samples: 80‑120 %. ▪ 11298500316, ISE Cleaning Solution/Elecsys SysClean,
Sampling devices containing liquid anticoagulants have a dilution effect 5 x 100 mL system cleaning solution
resulting in lower COI values for individual patient specimens. Assay
In order to minimize dilution effects it is essential that respective sampling For optimum performance of the assay follow the directions given in this
devices are filled completely according to manufacturer’s instructions. document for the analyzer concerned. Refer to the appropriate operator’s
manual for analyzer‑specific assay instructions.
2/5 2018-10, V 9.0 English
ms_04784618190V9.0
Elecsys CMV IgM
Resuspension of the microparticles takes place automatically prior to use. Samples with a cutoff index between ≥ 0.7 and < 1.0 are considered
Read in the test‑specific parameters via the reagent barcode. If in indeterminate. The sample should be retested. In case the result is still
exceptional cases the barcode cannot be read, enter the 15‑digit sequence indeterminate, a second sample should be tested e.g. within the following
of numbers (except for the cobas e 602 analyzer). 2‑3 weeks.
Bring the cooled reagents to approximately 20 °C and place on the reagent Samples with a cutoff index ≥ 1.0 are reactive in the Elecsys CMV IgM
disk (20 °C) of the analyzer. Avoid foam formation. The system assay.
automatically regulates the temperature of the reagents and the The magnitude of the measured result above the cutoff is not indicative of
opening/closing of the bottles. the total amount of antibody present in the sample.
Place the calibrators in the sample zone. The anti‑CMV IgM results in a given specimen, as determined by assays
All the information necessary for calibrating the assay is automatically read from different manufacturers, can vary due to differences in reagents and
into the analyzer. assay methods.
After calibration has been performed, store the calibrators at 2‑8 °C or Limitations - interference
discard (MODULAR ANALYTICS E170, cobas e 601 and cobas e 602 A negative CMV IgM test result, also in combination with a positive
analyzers). CMV IgG result, does not completely rule out the possibility of an acute
Calibration infection with cytomegalovirus:
Traceability: This method has been standardized against a Roche standard. ▪ Individuals at the early stage of acute infection may not exhibit
The units have been selected arbitrarily. detectable amounts of CMV IgM antibodies. In some of these individuals
Calibration frequency: Calibration must be performed once per reagent lot an indeterminate or low positive result with the Elecsys CMV IgG assay
using CMVIGM Cal1, CMVIGM Cal2 and fresh reagent (i.e. not more than may be found and indicate an early acute infection. A second sample
24 hours since the reagent kit was registered on the analyzer). should be tested e.g. within 2 weeks. The detection of CMV IgM and/or
Renewed calibration is recommended as follows: a significant increase of the Elecsys CMV IgG antibody titer in the
second sample supports the diagnosis of acute CMV infection.15,16
Calibration interval may be extended based on acceptable verification of
calibration by the laboratory. ▪ The individual immune response following CMV infection varies
considerably.
▪ after 1 month (28 days) when using the same reagent lot
In some individuals non‑reactive results may occur in the late phase of
▪ after 7 days (when using the same reagent kit on the analyzer) acute infection by the Elecsys CMV IgM assay.
▪ as required: e.g. quality control findings with PreciControl CMV IgM The detection of IgM antibodies against CMV in a single sample is not
outside the defined limits sufficient to prove an acute CMV infection. In single cases elevated IgM
antibody levels may persist even for years after initial infection. For further
▪ more frequently when this is required by pertinent regulations clarification additional laboratory tests (e.g. CMV IgG and IgG avidity) or
Range for the electrochemiluminescence signals (counts) for the combination of tests should be performed and results should be assessed
calibrators: in conjunction with the patient’s medical history and clinical symptoms.
Negative calibrator (CMVIGM Cal1): 500‑1600
Positive calibrator (CMVIGM Cal2): 2800‑16000 The results in HIV patients, in patients undergoing immunosuppressive
therapy, or in patients with other disorders leading to immune suppression,
Quality control should be interpreted with caution.
For quality control, use PreciControl CMV IgM. Specimens from neonates, cord blood, pretransplant patients or body fluids
Controls for the various concentration ranges should be run individually at other than serum and plasma, such as urine, saliva or amniotic fluid have
least once every 24 hours when the test is in use, once per reagent kit, and not been tested.
following each calibration. The assay is unaffected by icterus (bilirubin ≤ 342 µmol/L or ≤ 20 mg/dL),
The control intervals and limits should be adapted to each laboratory’s hemolysis (Hb ≤ 0.310 mmol/L or ≤ 0.500 g/dL), lipemia (Intralipid
individual requirements. Values obtained should fall within the defined ≤ 1500 mg/dL) and biotin (≤ 410 nmol/L or ≤ 100 ng/mL).
limits. Each laboratory should establish corrective measures to be taken if Criterion: Mean recovery of positive samples within ± 20 % of serum value.
values fall outside the defined limits. Samples should not be taken from patients receiving therapy with high
If necessary, repeat the measurement of the samples concerned. biotin doses (i.e. > 5 mg/day) until at least 8 hours following the last biotin
Follow the applicable government regulations and local guidelines for administration.
quality control. No interference was observed from rheumatoid factors up to a
Note: The controls are not barcode-labeled and therefore the controls must concentration of 2000 IU/mL.
run on all instruments as non-Roche controls. The control values and In vitro tests were performed on 18 commonly used pharmaceuticals and in
ranges have to be entered manually (except for the cobas e 602 analyzer). addition on ganciclovir and valganciclovir. No interference with the assay
Please refer to the corresponding section in the operator’s manual. was found.
The exact lot-specific target values and ranges are printed on the value Sera from patients with primary EBV infections can demonstrate positive
sheet which is included in the control kit or reagent kit (or electronically results in the Elecsys CMV IgM assay. This is not unexpected as both
available). viruses belong to the herpes virus family and this potential interference is
Calculation known for CMV IgM assays.16 As with many μ‑capture assays, an
interference with unspecific IgM is observed. Increasing amounts of
The analyzer automatically calculates the cutoff based on the measurement unspecific IgM may lead to a decrease in the recovery of positive samples
of CMVIGM Cal1 and CMVIGM Cal2. The result of a sample is given either with the Elecsys CMV IgM assay.
as reactive or non-reactive as well as in the form of a cutoff index (signal
sample/cutoff). In rare cases, interference due to extremely high titers of antibodies to
analyte‑specific antibodies, streptavidin or ruthenium can occur. These
Interpretation of the results effects are minimized by suitable test design.
Results obtained with the Elecsys CMV IgM assay can be interpreted as For diagnostic purposes, the results should always be assessed in
follows: conjunction with the patient’s medical history, clinical examination and other
Non-reactive: < 0.7 COI findings.
Indeterminate: ≥ 0.7 - < 1.0 COI Specific performance data
Reactive: ≥ 1.0 COI Representative performance data on the analyzers are given below.
Samples with a cutoff index < 0.7 are non-reactive in the Elecsys CMV IgM Results obtained in individual laboratories may differ.
assay.
2018-10, V 9.0 English 3/5
ms_04784618190V9.0
Elecsys CMV IgM
Precision Site N Sensitivity (%)
Precision was determined using Elecsys reagents, human sera and controls Elecsys CMV IgM assay Comparison CMV IgM test
in a protocol (EP5‑A2) of the CLSI (Clinical and Laboratory Standards
Institute): 2 runs per day in duplicate each for 21 days (n = 84); repeatability 3 39e) 91.2 (31/34) 79.4 (27/34)
n = 21. The following results were obtained:
35f) 93.1 (27/29) 100 (29/29)
cobas e 411 analyzer
4g) 54 92.3 (48/52) 98.1 (51/52)
Repeatability Intermediate precision
d) 66 samples were excluded due to high/moderate avidity.
Sample Mean SD CV Mean SD CV e) 5 samples were excluded due to moderate avidity and concordant negative results for CMV
COI COI % COI COI % IgM.
f) 6 samples were excluded due to moderate/high avidity and concordant negative results for
HSb), negative 0.188 0.004 2.1 0.175 0.004 2.4 CMV IgM.
HS, low positive 1.52 0.032 2.1 1.62 0.056 3.4 g) 2 samples were excluded due to moderate avidity and discrepant CMV IgM results.
HS, high positive 14.2 0.295 2.1 13.9 0.438 3.2 Specificity in past infection
A total of 158 frozen samples from pregnant women with past CMV
PCc) CMV IgM 1 0.185 0.006 3.1 0.171 0.005 2.8 infection, analyzed by commercial CMV assays, were tested with the
PC CMV IgM 2 2.04 0.052 2.6 1.98 0.103 5.3 Elecsys CMV IgM assay at 4 different sites. All samples were pre‑selected
positive for CMV IgG, negative for CMV IgM and a high avidity index
b) HS = human serum showing absence of acute infection.
c) PC = PreciControl Relative specificity in past infections
MODULAR ANALYTICS E170, cobas e 601 and cobas e 602 analyzers Site N Relative specificity (%)
Repeatability Intermediate precision Elecsys CMV IgM assay Comparison CMV IgM test
Sample Mean SD CV Mean SD CV 1h) 48 98 100
COI COI % COI COI %
2 30 100 100
HS, negative 0.172 0.004 2.0 0.184 0.010 5.2
3i) 50 84 86
HS, low positive 1.17 0.027 2.3 1.68 0.082 4.9
4 30 100 100
HS, high positive 15.0 0.297 2.0 14.0 0.524 3.8
h) 1 sample was found indeterminate with the Elecsys CMV IgM assay and negative with the
PC CMV IgM 1 0.164 0.003 1.8 0.182 0.010 5.2 comparison assay.
PC CMV IgM 2 1.97 0.033 1.7 1.96 0.119 6.1 i) 8 samples were positive with the Elecsys CMV IgM assay; 7 samples were equivocal with the
comparison assay.
Analytical specificity Capability to discriminate the persisting IgM after CMV infection
433 potentially cross reacting samples were tested with the Elecsys A total of 68 frozen samples from pregnant women analyzed by commercial
CMV IgM assay and a comparison CMV IgM assay comprising the following CMV IgM assays were tested with the Elecsys CMV IgM assay at 3
specimens: different sites. All samples were pre‑selected positive for CMV IgG and
CMV IgM and a high avidity index showing absence of acute infection.
▪ containing antibodies against HBV, HAV, HCV, HIV*, HTLV, EBV**,
HSV***, VZV, Parvo B19***, Rubella****, Treponema pallidum, Capability to discriminate the persisting CMV IgM
Toxoplasma gondii Site N Elecsys CMV IgM assay Comparison CMV IgM test
▪ containing autoantibodies***** (ANA, anti-tissue, RF) tested/reactive tested/reactive
* HIV: 8 discordant samples were found out of 70 samples.
** EBV: 13 discordant samples were found out of 48 samples. 1j) 20 20/6 20/20
*** HSV, Parvo B19: 1 discordant sample was found in each group. 2k) 28 28/4 28/28
**** Rubella: 2 discordant samples were found.
***** Autoantibodies: 7 discordant samples were found out of 73 samples. 3l) 20 20/7 20/20
An overall agreement of 92.3 % (381/413) was found in these specimens j) 14 samples were discrepant negative with the Elecsys CMV IgM assay.
with the Elecsys CMV IgM assay and the comparison test. 377 samples k) 24 samples were discrepant negative with the Elecsys CMV IgM assay.
were found concordantly negative and 4 samples were found positive. l) 12 samples were discrepant negative with the Elecsys CMV IgM assay.
20 samples were found indeterminate either with the Elecsys CMV IgM Specificity in pre‑selected negative samples
assay or the comparison test.
A total of 173 frozen samples from pregnant women in which a CMV
Clinical sensitivity infection was excluded and analyzed by commercial CMV IgM assays were
Sensitivity in primary infections tested with the Elecsys CMV IgM assay at 4 different sites.
A total of 365 frozen samples from pregnant women with primary CMV
infection including sequential and single samples analyzed by commercially Site N Specificity (%)
available CMV IgM assays were tested with the Elecsys CMV IgM assay at Elecsys CMV IgM assay Comparison CMV IgM test
4 different sites. For calculation of sensitivity confirmed positive samples
had a low avidity index or were clinically characterized. Indeterminate 1 50 100 100
samples were counted as positives. Samples with a high avidity index and 2m) 50 98 100
all discrepant samples or concordant negative samples with a moderate
avidity index were excluded. 3n) 23 100 95.7
4 50 100 100
Site N Sensitivity (%)
m) 1 sample was found positive with the Elecsys CMV IgM assay.
Elecsys CMV IgM assay Comparison CMV IgM test
n) 1 sample was found equivocal with the comparison assay.
1d) 180 93.0 (106/114) 94.7 (108/114)
Clinical specificity
2 57 96.5 (55/57) 96.5 (55/57) A total of 1646 fresh samples from clinical routine (blood donors, site 1)
were tested at 3 different sites (pregnancy testing, site 2 and 3) with the
4/5 2018-10, V 9.0 English
ms_04784618190V9.0
Elecsys CMV IgM
Elecsys CMV IgM assay in comparison to competitor assays. The For further information, please refer to the appropriate operator’s manual for
IgG avidity test was used to resolve discrepant results. the analyzer concerned, the respective application sheets, the product
Relative specificity after resolution information and the Method Sheets of all necessary components (if
available in your country).
Site N Relative Lower Relative Lower A point (period/stop) is always used in this Method Sheet as the decimal
specificity confidence specificity confidence separator to mark the border between the integral and the fractional parts of
Elecsys limit Comparison limit a decimal numeral. Separators for thousands are not used.
CMV IgM % CMV IgM test % Symbols
assay % Roche Diagnostics uses the following symbols and signs in addition to
% those listed in the ISO 15223‑1 standard (for USA: see
https://usdiagnostics.roche.com for definition of symbols used):
1o) 511 98.8 (495/501) 97.4 96.6 (484/501) 94.6
2p) 616 97.1 (574/591) 95.4 93.4 (552/591) 91.1 Contents of kit
3q) 519 97.0 (492/507) 95.2 92.9 (471/507) 90.3 Analyzers/Instruments on which reagents can be used
o) 10 samples were excluded due to moderate avidity or missing avidity result. Reagent
p) 10 samples were confirmed positive by low avidity index; 15 samples were excluded due to Calibrator
moderate or missing avidity result.
q) 3 samples were confirmed positive by low avidity index; 9 samples were excluded due to Volume after reconstitution or mixing
moderate or missing avidity result.
GTIN Global Trade Item Number
References
1 Cannon MJ, Schmid DS, Hyde TB. Review of cytomegalovirus COBAS, COBAS E, ELECSYS and PRECICONTROL are trademarks of Roche. INTRALIPID is a trademark of
seroprevalence and demographic characteristics associated with Fresenius Kabi AB.
infection. Rev Med Virol 2010;20:202-213. All other product names and trademarks are the property of their respective owners.
Additions, deletions or changes are indicated by a change bar in the margin.
2 Johnson J, Anderson B, Pass RF. Prevention of maternal and
© 2017, Roche Diagnostics
congenital cytomegalovirus infection. Clin Obstet Gynecol
2012;55:521-530.
3 Yinon Y, Farine D, Yudin MH. Screening, diagnosis, and management
of cytomegalovirus infection in pregnancy. Obstet Gynecol Surv
2010;65:736-743. Roche Diagnostics GmbH, Sandhofer Strasse 116, D-68305 Mannheim
www.roche.com
4 Buonsenso D, Serranti D, Gargiullo L, et al. Congenital
cytomegalovirus infection: current strategies and future perspectives.
Eur Rev Med Pharmacol Sci 2012;16:919-935.
5 Coll O, Benoist G, Ville Y, et al. WAPM Perinatal Infections Working
Group. Guidelines on CMV congenital infection. J Perinat Med
2009;37:433-445.
6 Joseph S, Beliveau C, Muecke C, et al. Cytomegalovirus as an
occupational risk in daycare educators. Paediatr Child Health
2006;11:401-407.
7 Lazzarotto T, Guerra B, Lanari M, et al. New advances in the diagnosis
of congenital cytomegalovirus infection. J Clin Virol 2008;41:192-197.
8 Lombardi G, Garofoli F, Stronati M. Congenital cytomegalovirus
infection: treatment, sequelae and follow-up. J Matern Fetal Neonatal
Med 2010;23Suppl3:45-48.
9 Elliott SP. Congenital cytomegalovirus infection: an overview. Infect
Disord Drug Targets 2011;11:432-436.
10 Lazzarotto T, Guerra B, Gabrielli L, et al. Update on the prevention,
diagnosis and management of cytomegalovirus infection during
pregnancy. Clin Microbiol Infect 2011;17:1285-1293.
11 Plosa EJ, Esbenshade JC, Fuller MP, et al. Cytomegalovirus infection.
Pediatr Rev 2012;33:156-163.
12 Ross SA, Novak Z, Pati S, et al. Overview of the diagnosis of
cytomegalovirus infection. Infect Disord Drug Targets 2011;11:466-474.
13 Occupational Safety and Health Standards: Bloodborne pathogens.
(29 CFR Part 1910.1030). Fed. Register.
14 Directive 2000/54/EC of the European Parliament and Council of
18 September 2000 on the protection of workers from risks related to
exposure to biological agents at work.
15 Revello MG, Gerna G. Diagnosis and Management of Human
Cytomegalovirus Infection in the Mother, Fetus, and Newborn Infant.
Clin Microbiol Rev 2002;15(4):680-715.
16 Genser B, Truschnik-Wilders M, Stünzner D, et al. Evaluation of Five
Commercial Enzyme Immunoassays for the Detection of Human
Cytomegalovirus-Specific IgM Antibodies in the Absence of a
Commercially Available Gold Standard. Clin Chem Lab Med
2001;39(1): 62-70.
2018-10, V 9.0 English 5/5
You might also like
- Foods To Fight Cancer - What To Eat PDFDocument242 pagesFoods To Fight Cancer - What To Eat PDFdokundot86% (7)
- Gen 5 SoftwareDocument452 pagesGen 5 Softwareابو حمزةNo ratings yet
- ChlorhexidineDocument21 pagesChlorhexidineKristin CurtisNo ratings yet
- CMV Igg 2018-12 v10Document5 pagesCMV Igg 2018-12 v10ابو حمزةNo ratings yet
- Insert - Elecsys CMV IgG.09118551500.V1.EnDocument6 pagesInsert - Elecsys CMV IgG.09118551500.V1.EnVegha NedyaNo ratings yet
- CMV Igm: Igm Antibodies To Cytomegalovirus 04784618 190 100 TestsDocument4 pagesCMV Igm: Igm Antibodies To Cytomegalovirus 04784618 190 100 TestsModestusNo ratings yet
- Toxo Igm 2018-12 v11Document5 pagesToxo Igm 2018-12 v11ابو حمزةNo ratings yet
- Rubella Igm 2018-10 v10Document5 pagesRubella Igm 2018-10 v10ابو حمزةNo ratings yet
- Immulite: Anti-Hbc IgmDocument28 pagesImmulite: Anti-Hbc IgmpinoponiNo ratings yet
- Insert - Elecsys Syphilis - Ms 07802960190.V3.EnDocument5 pagesInsert - Elecsys Syphilis - Ms 07802960190.V3.EnGuneyden GuneydenNo ratings yet
- Virapid Mono M&G - VR001 - enDocument4 pagesVirapid Mono M&G - VR001 - enCristinaNo ratings yet
- Viral DiagnosisDocument37 pagesViral Diagnosissalamshakir56No ratings yet
- Toxo Igg 2018-09 v13Document5 pagesToxo Igg 2018-09 v13ابو حمزةNo ratings yet
- Rubella Igg 2018-10 v9Document5 pagesRubella Igg 2018-10 v9ابو حمزةNo ratings yet
- 17 - Serological DiagnosisDocument9 pages17 - Serological DiagnosisTamda ShitakeNo ratings yet
- Cytomegalovirus (CMV) Igm: ELISA Kit ProtocolDocument11 pagesCytomegalovirus (CMV) Igm: ELISA Kit ProtocolSandyOctavianusLNo ratings yet
- CMV Igg: SystemDocument7 pagesCMV Igg: Systemtesteste testeNo ratings yet
- 34 FullDocument8 pages34 FullCherryNo ratings yet
- Laboratory Diagnosis of CMV Infection: A ReviewDocument6 pagesLaboratory Diagnosis of CMV Infection: A ReviewrendyjiwonoNo ratings yet
- 4014 HSV1 IgM Elisa KitDocument2 pages4014 HSV1 IgM Elisa KitHéctor CastelánNo ratings yet
- Epstein-Barr Virus Super FinalDocument6 pagesEpstein-Barr Virus Super FinalStill DollNo ratings yet
- Human Cytomegalovirus and Kidney Transplantation: A Clinician's UpdateDocument9 pagesHuman Cytomegalovirus and Kidney Transplantation: A Clinician's UpdateFlorinda CarranzaNo ratings yet
- Ca 125 IiDocument4 pagesCa 125 IiHassan GillNo ratings yet
- 819 FullDocument5 pages819 FullMinerva StanciuNo ratings yet
- VIRAL-DeTECTION Dxvirology AacbungayDocument102 pagesVIRAL-DeTECTION Dxvirology AacbungayDominic Bernardo100% (1)
- Jurnal Imun 4Document7 pagesJurnal Imun 4dian.esterinaNo ratings yet
- Insert - Elecsys CA 125 II - Ms - 11776223161 - USA.V2.enDocument5 pagesInsert - Elecsys CA 125 II - Ms - 11776223161 - USA.V2.enonedarigirlNo ratings yet
- 1 PB PDFDocument6 pages1 PB PDFJayantiNo ratings yet
- Anti-HAV IgM 12176513001 - enDocument3 pagesAnti-HAV IgM 12176513001 - enArnaz AdisaputraNo ratings yet
- Medi 97 E12380Document8 pagesMedi 97 E12380Mohamed NabilNo ratings yet
- Diagnostic Microbiology: Identification of MicrobesDocument20 pagesDiagnostic Microbiology: Identification of MicrobesMarl EstradaNo ratings yet
- Tes Lab Penyakit Infeksi Dan Tropis (April 2011)Document143 pagesTes Lab Penyakit Infeksi Dan Tropis (April 2011)Fajrul AnsarNo ratings yet
- HAV IgMDocument8 pagesHAV IgMCinthia Usagui Ancari RamirezNo ratings yet
- Covid-19 Igg/Igm Rapid Test Cassette (WB/S/P) (Principle) : EnglishDocument2 pagesCovid-19 Igg/Igm Rapid Test Cassette (WB/S/P) (Principle) : Englishniluh suwasantiNo ratings yet
- Elecsys® Anti-HAV IgM Immunoassay For The Qualitative Detection of IgM Antibodies Against HAVDocument4 pagesElecsys® Anti-HAV IgM Immunoassay For The Qualitative Detection of IgM Antibodies Against HAVLAMA LAMANo ratings yet
- Interpretation of Serology Tests in Selected InfectionsDocument35 pagesInterpretation of Serology Tests in Selected InfectionsAnees AdamsNo ratings yet
- Acta Paediatrica - 2020 - Lüsebrink - Retrospectively Diagnosing Congenital Cytomegalovirus Infections in SymptomaticDocument6 pagesActa Paediatrica - 2020 - Lüsebrink - Retrospectively Diagnosing Congenital Cytomegalovirus Infections in Symptomaticdossantoselaine212No ratings yet
- CA 125 II 12176645001 - enDocument4 pagesCA 125 II 12176645001 - enModestusNo ratings yet
- Alinity Rubella IgGDocument8 pagesAlinity Rubella IgGLoloNo ratings yet
- Avm CeDocument8 pagesAvm CeMaría Gabriela Ocanto DNo ratings yet
- Mumps Laboratory Case DefinitionDocument9 pagesMumps Laboratory Case Definitionauliya nidaNo ratings yet
- Documento Con Títulos y Abstracts de Los Artículos EncontradosDocument15 pagesDocumento Con Títulos y Abstracts de Los Artículos EncontradosDaniel NietoNo ratings yet
- Entamoeba HistolyticaDocument5 pagesEntamoeba Histolyticaعائش العموديNo ratings yet
- Applications of Hetergenous EIA (Measles and Rubella) : by Harkeerat KaurDocument7 pagesApplications of Hetergenous EIA (Measles and Rubella) : by Harkeerat KaurHarkeerat KaurNo ratings yet
- CA 125 II: Cancer Antigen 125Document4 pagesCA 125 II: Cancer Antigen 125hairiNo ratings yet
- Rubella Igm Elisa Test System: Intended UseDocument7 pagesRubella Igm Elisa Test System: Intended UsestandinginAWENo ratings yet
- Elecsys CA 15-3 II: A) Tris (2,2'-Bipyridyl) Ruthenium (II) - Complex (Ru (Bpy) )Document5 pagesElecsys CA 15-3 II: A) Tris (2,2'-Bipyridyl) Ruthenium (II) - Complex (Ru (Bpy) )Douglas MirandaNo ratings yet
- Epstein BarrDocument21 pagesEpstein BarrkamalNo ratings yet
- Tell Me Fast Novel Coronavirus (Covid-19) Igg/Igm Antibody TestDocument8 pagesTell Me Fast Novel Coronavirus (Covid-19) Igg/Igm Antibody TestDortmunderNo ratings yet
- 115 Syphilis-V3.0-en-EUDocument4 pages115 Syphilis-V3.0-en-EUPierina DonayreNo ratings yet
- Eurosurv 27 42 5Document13 pagesEurosurv 27 42 5Eben Leonel Albano MaiopueNo ratings yet
- IFA Produktinfo Infectious Diseases GB 10.2015 SK 1Document28 pagesIFA Produktinfo Infectious Diseases GB 10.2015 SK 1siddhi divekarNo ratings yet
- South Africa CMV Viral Load Testing May2015Document4 pagesSouth Africa CMV Viral Load Testing May2015EnergadeNo ratings yet
- HEV IgMDocument7 pagesHEV IgMMaría Gabriela Ocanto DNo ratings yet
- Congenital Cytomegalovirus Infection in The Absence of Maternal Cmv-Igm Antibodies: A Case ReportDocument3 pagesCongenital Cytomegalovirus Infection in The Absence of Maternal Cmv-Igm Antibodies: A Case ReportVictoria CyntiaNo ratings yet
- JCM 43 8 4194-4196 2005Document3 pagesJCM 43 8 4194-4196 2005Sumesh ShresthaNo ratings yet
- Lab DiagnosisDocument42 pagesLab DiagnosisNestley TiongsonNo ratings yet
- A Rapid Test For Detection of Dengue FeverDocument2 pagesA Rapid Test For Detection of Dengue FeverYvette TiongsonNo ratings yet
- Anti-HAV IgMDocument3 pagesAnti-HAV IgMHassan GillNo ratings yet
- COVID-19: Instructions For UseDocument2 pagesCOVID-19: Instructions For UseTheresia IlyanNo ratings yet
- Laboratory Diagnosis of CMV Infection: A ReviewDocument6 pagesLaboratory Diagnosis of CMV Infection: A ReviewAchmad ArrizalNo ratings yet
- Non Ventilation PneumoniaeDocument24 pagesNon Ventilation PneumoniaentnquynhproNo ratings yet
- 68th AACC Annual Scientific Meeting Abstract eBookFrom Everand68th AACC Annual Scientific Meeting Abstract eBookNo ratings yet
- CMV Igg 2018-12 v10Document5 pagesCMV Igg 2018-12 v10ابو حمزةNo ratings yet
- Manual Inspire 2 en USDocument65 pagesManual Inspire 2 en USابو حمزةNo ratings yet
- Ra081013ldf enDocument4 pagesRa081013ldf enابو حمزةNo ratings yet
- OvenDocument23 pagesOvenابو حمزةNo ratings yet
- Manual Charge 5 en USDocument76 pagesManual Charge 5 en USابو حمزةNo ratings yet
- Roche Accu Chek Compact Plus ManualDocument111 pagesRoche Accu Chek Compact Plus Manualابو حمزةNo ratings yet
- Assets PDF EMEA Brochure CL V3600 EMEA PR-06134Document4 pagesAssets PDF EMEA Brochure CL V3600 EMEA PR-06134ابو حمزةNo ratings yet
- Cybex 530T Pro Plus Stableflex TreadmillDocument110 pagesCybex 530T Pro Plus Stableflex Treadmillابو حمزةNo ratings yet
- Brc1e62 1Document28 pagesBrc1e62 1ابو حمزةNo ratings yet
- EncoderDocument72 pagesEncoderابو حمزةNo ratings yet
- Bipolar Disorder: SeminarDocument7 pagesBipolar Disorder: SeminarZhi Wu BhâvaNo ratings yet
- Mcce QDocument139 pagesMcce QBahaa LotfyNo ratings yet
- Research FinaaaaaalDocument76 pagesResearch FinaaaaaalAbdulhakeem AmirNo ratings yet
- The Respiratory System I.: Subject: Subject TeacherDocument4 pagesThe Respiratory System I.: Subject: Subject TeacherHyacinth FarinasNo ratings yet
- DR Saman Questions of Moodle Final 2019Document3 pagesDR Saman Questions of Moodle Final 2019Miki AberaNo ratings yet
- Metabolic SyndromeDocument19 pagesMetabolic SyndromebradleyrrNo ratings yet
- Child AbuseDocument22 pagesChild AbuseSharon Umblas RubinoNo ratings yet
- B. Sc. 2 YearDocument84 pagesB. Sc. 2 YearSreelekha Ramtenki100% (1)
- 7.7 Strategies - Questions and Answers Q1.: BK Ch7 S7.7 Q1Document2 pages7.7 Strategies - Questions and Answers Q1.: BK Ch7 S7.7 Q1educator56No ratings yet
- Cardiotonics & Inotropic Drugs PDFDocument10 pagesCardiotonics & Inotropic Drugs PDFZehra AmirNo ratings yet
- Gadtc PHCDocument246 pagesGadtc PHCshawnNo ratings yet
- Anemia in PregnancyDocument24 pagesAnemia in PregnancyAzwanie Ali100% (1)
- Preoperative Assessment of The Patient With Heart Disease Case FileDocument2 pagesPreoperative Assessment of The Patient With Heart Disease Case Filehttps://medical-phd.blogspot.comNo ratings yet
- Gynae FCPS 1 Guide-1Document24 pagesGynae FCPS 1 Guide-1Lubna MushtaqNo ratings yet
- Poster Presentations: 2009 The Authors Journal Compilation 2009 Blackwell Verlag GMBH - Mycoses, 52 (Suppl. 1), 29-123Document95 pagesPoster Presentations: 2009 The Authors Journal Compilation 2009 Blackwell Verlag GMBH - Mycoses, 52 (Suppl. 1), 29-123Flávio VianiNo ratings yet
- What Does High Potassium in Blood Work MeanDocument2 pagesWhat Does High Potassium in Blood Work MeanMathumathi BalakrishnanNo ratings yet
- Imci Repaired NotesDocument136 pagesImci Repaired NotesGladys WarigheNo ratings yet
- MudrasDocument8 pagesMudrasKishore CheralaNo ratings yet
- Chemistry Project: Study of Adultrants in FoodDocument21 pagesChemistry Project: Study of Adultrants in Foodmohit pandeyNo ratings yet
- Clinical Differentiation of Upper Extremity Pain EtiologiesDocument9 pagesClinical Differentiation of Upper Extremity Pain EtiologiesmarcosgabNo ratings yet
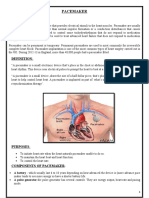
- PACEMAKERDocument5 pagesPACEMAKERPoonam ThakurNo ratings yet
- My Revision Notes Edexcel As History The Triumph of Fascism in Italy, 1896-1943Document81 pagesMy Revision Notes Edexcel As History The Triumph of Fascism in Italy, 1896-1943Alyan HanifNo ratings yet
- Vaginal MassDocument12 pagesVaginal MassShem DelolaNo ratings yet
- Assignment For Endocrine SystemDocument5 pagesAssignment For Endocrine SystemMoheeddin KababNo ratings yet
- Pathogenesis and Management of Renal Failure and Replacement Therapy For Stage V Chronic Renal DiseaseDocument30 pagesPathogenesis and Management of Renal Failure and Replacement Therapy For Stage V Chronic Renal DiseaseByzantine Wulandari ParubakNo ratings yet
- Abortion BSN 0PR-2: Case Study of BelleDocument27 pagesAbortion BSN 0PR-2: Case Study of BellekervinNo ratings yet
- Gels 10 00188Document46 pagesGels 10 00188mkhubaibNo ratings yet
- Topical Immunomodulators in DermatologyDocument9 pagesTopical Immunomodulators in DermatologyRifky Budi TriyatnoNo ratings yet