Professional Documents
Culture Documents
Exome Sequencing Identifies MPL As A Causative Gen
Exome Sequencing Identifies MPL As A Causative Gen
Uploaded by
Muhammad DahryanOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Exome Sequencing Identifies MPL As A Causative Gen
Exome Sequencing Identifies MPL As A Causative Gen
Uploaded by
Muhammad DahryanCopyright:
Available Formats
See discussions, stats, and author profiles for this publication at: https://www.researchgate.
net/publication/51898968
Exome sequencing identifies MPL as a causative gene in familial Aplastic
anemia
Article in Haematologica · December 2011
DOI: 10.3324/haematol.2011.052787 · Source: PubMed
CITATIONS READS
39 182
8 authors, including:
Amanda Walne Arran Dokal
Queen Mary, University of London Barts Cancer Institute
70 PUBLICATIONS 3,940 CITATIONS 10 PUBLICATIONS 80 CITATIONS
SEE PROFILE SEE PROFILE
Josu de la Fuente Tom Vulliamy
Imperial College Healthcare NHS Trust Queen Mary, University of London
34 PUBLICATIONS 1,406 CITATIONS 178 PUBLICATIONS 11,950 CITATIONS
SEE PROFILE SEE PROFILE
Some of the authors of this publication are also working on these related projects:
WAIT - Wheeze and Intermittent treatment study View project
Pyridinium crosslinks in arthritis View project
All content following this page was uploaded by Amanda Walne on 22 May 2014.
The user has requested enhancement of the downloaded file.
Articles and Brief Reports Aplastic Anemia
Exome sequencing identifies MPL as a causative gene in familial aplastic anemia
Amanda J. Walne,1 Arran Dokal,1 Vincent Plagnol,2 Richard Beswick,1 Michael Kirwan,1 Josu de la Fuente,3
Tom Vulliamy,1 and Inderjeet Dokal1
1
Centre for Paediatrics, Blizard Institute, Barts and The London School of Medicine and Dentistry, Queen Mary University
of London, London, UK; 2University College London Genetics Institute, Gower Place, London, UK; and 3St Mary’s Hospital
Paddington, London, UK
ABSTRACT
The primary cause of aplastic anemia remains unknown in patients. This study shows for the first time a link between
many patients. The aim of this study was to clarify the genetic homozygous MPL mutations and familial aplastic anemia. It
cause of familial aplastic anemia. Genomic DNA of an affected also highlights the important role of MPL in trilineage
individual from a multiplex consanguineous family was hematopoiesis.
hybridized to a Nimblegen exome library before being
sequenced on a GAIIx genome analyzer. Once the disease Key words: aplastic anemia, MPL, exome sequencing.
causing homozygous mutation had been confirmed in the
consanguineous family, this gene was then analyzed for muta- Citation: Walne AJ, Dokal A, Plagnol V, Beswick R, Kirwan M,
tion in 33 uncharacterized index cases of aplastic anemia (<13 de la Fuente J, Vulliamy T, and Dokal I. Exome sequencing iden-
years) using denaturing HPLC. Abnormal traces were con- tifies MPL as a causative gene in familial aplastic anemia.
firmed by direct sequencing. Exome sequencing identified a Haematologica 2012;97(4):524-528.
novel homozygous nonsense mutation in the thrombopoietin doi:10.3324/haematol.2011.052787
receptor gene MPL. An additional novel homozygous MPL
mutation was identified in the screen of 33 aplastic anemia ©2012 Ferrata Storti Foundation. This is an open-access paper.
Introduction compound heterozygous mutations cannot be identified
using these types of approach. Recent advances, such as next
Aplastic anemia (AA, MIM #609138) is a rare heteroge- generation sequencing, have made it possible to sequence
neous disorder affecting 2-7 people per million per year.1 In the entire genome within weeks or, more easily, the entire
50-70% of cases, the etiology is unknown. In the remainder, coding exome within a few days.7 This new technology now
it is either inherited (10-20%) or is secondary to a particular allows the identification of causative mutations in families
insult (10-20%2,3). Some of the inherited cases can be linked and disorders that are not amenable to linkage-based posi-
to various syndromes, for example, Fanconi anemia (FA) or tional cloning.8
dyskeratosis congenita (DC), but others remain uncharacter- The aim of this study was to use exome sequencing to
ized.4 Familial AA is an extremely rare inherited subtype identify the cause of familial AA in a consanguineous family
affecting multiple individuals in a family. Patients typically with 2 affected children. This would then identify a gene to
only have features of AA; the absence of any somatic fea- screen in other uncharacterized AA cases.
tures making it distinct from other inherited aplastic ane-
mias. The primary cause of these uncharacterized familial Design and Methods
cases is of interest to the scientist and can present a diagnos-
tic conundrum to the clinician. Patient selection
Until recently the main ways to identify new disease caus- DNA samples were received from 10 family members of a
ing genes was to identify candidate gene regions using link- large consanguineous family from Tunisia (2 had severe AA:
age analysis or by a direct candidate gene approach based on Family 1). Other DNA samples came from individuals aged
similar biological function. Often success in such traditional under 13 years presenting with AA which was not associated
approaches required the recruitment of large families with with any other bone marrow (BM) failure syndrome. All sam-
many affected and unaffected individuals.5,6 These approach- ples were obtained with informed consent and with the
es have a low success rate in identifying new disease genes, approval of our local ethics committee.
especially when there are no current genes associated with a
particular disease phenotype. Inheritance type and family Homozygosity mapping
size also significantly affect the likelihood of success and Genome wide microsatellite analysis was performed on the
The online version of this article has a Supplementary Appendix.
Acknowledgments: we would like to thank all the families and clinicians for biological samples, specifically Dr. Abdelkefi (Tunisia) and Dr. Richards (UK).
Funding: this work was funded by The Wellcome Trust.
Manuscript received on July 29, 2011. Revised version arrived on November 29, 2011. Manuscript accepted on November 30, 2011.
Correspondence: Amanda Walne, Centre for Paediatrics, Blizard Institute of Cell and Molecular Science, Barts and The London School of Medicine and
Dentistry, Queen Mary University of London, 4 Newark Street, London, UK. Phone: international +44.207.8822458. E-mail: a.walne@qmul.ac.uk
524 haematologica | 2012; 97(4)
MPL mutations in AA
2 affected children (Family 1: 4-I and 4-III) and where iden- Telomere length measurement
tical homozygous markers were identified, all family mem- Telomere lengths were measured using a multiplex real
bers were typed at this and flanking markers. Areas of time PCR method as described previously.9-10 T/S ratios
homozygosity which were consistent with linkage were obtained for the patient samples were compared with
noted. healthy control samples and also patients with known
TERC mutations as examples of short telomeres.
Exome sequencing
Approximately 180,000 coding exons from 5µg of Results and Discussion
genomic DNA from patient 4-III (Family 1) were sequenced
on the Illumina Genome Analyser IIx after enrichment Following microsatellite analysis, several regions of
using the NimblGen SeqCap EZ exome library. Sequencing homozygosity shared by the 2 affected individuals were
data were processed through the Illumina pipeline and identified (Family 1, Online Supplementary Figure S1). Given
unique homozygous changes identified by filtering the the large number of genes in these intervals, DNA from
resultant data set against variations reported on dbSNP patient 4-III (Figure 1A) underwent exome sequencing. We
(www.ncbi.nlm.nih.gov/snp/), the 1000 genome project filtered the resultant data set against: i) known variations in
(www.1000genomes.org/) and an in-house data set. published databases; ii) an in-house database; iii) excluding
any changes not in a homozygous interval; iv) based on
MPL mutation detection and gene screening gene function; and v) precedence of a disease with similar
Primers were designed to all coding exons (Online clinical features being caused by mutations in the same
Supplementary Table S1). The mutation c.1248G>A gene. We were able to reduce the number of variations to
p.W416X in exon 8 of MPL, was confirmed by direct study to a homozygous mutation c.1248 G>A,
sequencing using ABI BigDye v3.1. For the other 33 sam- p.Trp416Stop in MPL. MPL (myeloproliferative leukemia
ples, all exons of MPL were screened by denaturing high virus oncogene) encodes the thrombopoietin (TPO) recep-
performance liquid chromatography (dHPLC) using the tor. The interaction of thrombopoietin with its receptor is
Transgenomic Wave DNA fragment analysis system to largely responsible for megakaryopoiesis, platelet activa-
detect heteroduplexes following pair-wise pooling. All tion, as well as the maintenance of hematopoietic stem cells
abnormal elution patterns were sequenced as described (HSC). This also initiates intracellular signaling, including
above. Any coding variations identified were compared to activation of the JAK/STAT, RAS/MAPK and P13K/AKT
dbSNP and 1000 genome databases to determine that they pathways.11 Clinically, biallelic constitutional mutations in
had not been described as a rare variation. MPL have been described in congenital amegakaryocytic
Table 1. Clinical features of the patients with biallelic MPL mutations and a comparison with features of CAMT.
Feature CAMT * Family 1 Family 2
Ethnic origin Tunisia Pakistan
Relationship of parents Consanguineous Non consanguineous but parents
from same village
Age at presentation Typically at birth/infancy 4 years (index case) 3 years 18 months (index case) 3 years
Presenting feature Reduced/absent Severe aplastic anemia Aplastic anemia Rotavirus infection
megakaryocytes with no other (abnormal blood
and platelets clinical abnormalities count noted) Family study
Blood counts (At age 6 yrs) Thrombocytopenia
Hb (g/dL) 7.1 NA 11.3 only
WBC (¥109/L) 2.5 Low
Platelets (¥109/L) 7 14 60
Retriculocytes (¥109/L) 15
Neutrophils (¥109/L) 0.4 1.56
Exclude inherited Yes FA excluded FA excluded FA excluded FA excluded
forms of
thrombocytopenia
Bone marrow Normal cellularity Hypocellular, Hypocellular Hypocellular, mild Hypocellular
cellularity (initially) no dysmyelopoiesis dyserythropoesis
Other non Minor heart, None None None None
hematologic eye and brain abnormalities
abnormalities reported in some
Treatment Various including Successful HLA Successful HLA Close surveillance Close surveillance
platelet transfusions, identical sib BMT identical sib BMT + supportive care
matched BMT (2 yrs post transplant (engraftment by day 11)
full donor chimerism)
MPL mutations Homozygous or c.1248 G>A p.Trp416Stop c.1180 C>T p. Pro394Ser (homozygous)
compound heterozygous (homozygous)
*Definition from Ballmaier and Germeshausen19; BMT: bone marrow transplantation; FA: Fanconi anemia; Hb: hemoglobin; NA: not available; WBC: white blood cells.
haematologica | 2012; 97(4) 525
a.J. walne et al.
thrombocytopenia (CAMT). Patients with CAMT typically (Figure 1A). We then extended our screen to patients that
present with thrombocytopenia in the first few were reported to have AA in childhood but not necessarily
weeks/months of life.12 Knockout studies in mice for either in the neonatal period. From the clinical information avail-
Mpl or Tpo show a significant decrease in the numbers of able, the diagnosis of CAMT was not considered by the
myeloid, erythroid, megakaryocytic and burst colony form- referring physician and it was not always clear if a throm-
ing units, but apart from a reduction in platelet numbers, no bocytopenic phase preceded the presentation of the AA. A
reduction in other peripheral blood counts was observed. total of 33 patients were selected (mean age at presentation
When progenitor assays are performed on BM from 4.5 years; range 3 months-11 years). All coding exons of
patients with CAMT, there is a significant reduction in the MPL were screened and a novel missense mutation was
clonogenic cells of all three lineages when compared with identified in one patient. This patient (Family 2, from
BM from healthy individuals, suggesting that the thrombo- Pakistan) had a homozygous mutation c.1180 C>T
cytopenia and pancytopenia observed in these patients may p.Pro394Ser. A study of additional family members showed
result from loss of function mutations in MPL.13 that a second affected brother was also homozygous for
Sequencing the c.1248 G>A, p.Trp416Stop variation in all this mutation (Figure 1B). Both mutations identified in this
Family 1 members confirmed this novel MPL mutation seg- study were clustered in exon 8. This exon is not reported to
regated with disease in an autosomal recessive manner be widely mutated, and the majority of published muta-
A B
Figure 1. Genetic evaluation of MPL mutations in aplastic anemia. (A) Segregation of the c.1248G>A MPL mutation in Family 1 as identified
by exome sequencing. Filled shapes: affected individuals; arrow: affected base. (B) The homozygous missense mutation c.1180C>T
p.Pro394Ser segregates with disease in Family 2. Filled shapes: affected individuals; arrow: affected base. (C) Relative locations of the
mutations identified in MPL from this study (marked by arrows) and previously published studies. The exon structure and derived protein
structure with the functional domains are shown in the lower panel. Filled diamonds: nonsense mutations; open diamond: missense muta-
tions; filled triangles: frame shift mutations; open triangle: 7bp deletion; S: splice site mutations; *: recurrent mutation with <5 mutated
alleles reported; †: recurrent mutation with 5-10 mutated alleles reported; ‡: recurrent mutation with >10 mutated alleles reported; SP;
signal peptide; TM; transmembrane domain. Adapted from Ballamaier et al.19
526 haematologica | 2012; 97(4)
MPL mutations in AA
P<0.0001 this classification it is apparent that the affected individuals
2.0 in Family 1 have a different clinical course from the more
usual presentation of CAMT type I (Table 1). This cannot
be easily explained as both the nonsense mutations usually
associated with CAMT type I and the one reported here
1.5
would be predicted not to produce any functional protein;
therefore, there would be an absence of receptors, which
would impact on the downstream signaling.
T/S Ratio
1.0 In retrospect, Family 2 could be considered to be similar
to type II CAMT but this diagnosis only became possible
after the homozygous missense mutation had been identi-
0.5 fied. The age of onset is more variable in type II CAMT and
presentation tends to be less severe. Clinically, type II
CAMT can be defined by platelet count within the first year
of life as transiently increasing to over 50¥109/L or by devel-
0.0
Controls Family 2 TERC
oping pancytopenia over the age of six years.
Short telomeres are also a recognized feature of patients
with AA.20 Mutations in genes encoding the telomerase
complex result in short telomeres and some of these have
Figure 2. Telomeres are not significantly short in patients with been described in patients with apparently acquired severe
aplastic anemia associated with biallelic MPL mutations compared
with controls. Telomere lengths are short in patients with TERC AA.21,22 Telomere lengths were assayed to determine if short
mutations compared to controls. telomeres were involved in the pathology of AA caused by
MPL mutations. Due to technical problems, we were only
able to assay telomeres from both affected individuals from
Family 2. For comparison, telomere lengths reported as T/S
ratios are included for healthy controls (n=95, mean 0.83;
tions tend to cluster in exons 2 and 3 (Figure 1C). range 0.32-1.86) and for patients with TERC mutations
Many mutations in MPL have been identified in relation (n=18, mean 0.36; range 0.27-0.86). The T/S ratios for the
to CAMT (Figure 1C12,14-16 ) and this in turn has allowed dif- patients with MPL mutations are above the 10th percentile
ferent subtypes of CAMT to be described, partially based for the normal range (Figure 2) and longer than those seen
on the mutation type. Type I is the most severe form with in patients with TERC mutations (which are significantly
early onset pancytopenia, decreased BM activity and very shorter than controls). This suggests that the AA associated
low platelet count. The mutations associated with this sub- with MPL mutations is due more to a reduction of stem
type involve premature stop codons or frame shift muta- cells, resulting in an inability to maintain the pool of blood
tions that eliminate receptor signaling by deletion of all or cells, rather than short telomeres causing increased cellular
most of the intracellular domain. Type II CAMT is a milder apoptosis and/or senescence leading to the same reduction
form of the disease with transient increases of platelet count in blood cells, as seen in AA patients with TERC mutations.
up to nearly normal values in the first year of life and a later In this study, we have demonstrated exome sequencing
onset of BM failure (between 3-6 years). The mutations of a single individual from a consanguineous family is pow-
associated with this subtype include missense mutations or erful enough to identify the causative recessive disease
splicing defects resulting in reduced, residual receptor activ- gene, particularly when coupled with previous linkage data.
ity. A third type has also been described in which patients This reduces the number of potential variations, as any not
have ineffective megakaryopoiesis with no apparent in a previously identified homozygous region could be
defects in the MPL gene.15,17,18 excluded. We identified the causative homozygous MPL
In a systematic review of the literature, Ballmaier and mutation in a family with familial AA. This gene has been
Gereshausen report on all the cases of CAMT published implicated in a similar disease phenotype CAMT, but due
since 1990.19 Of the 71 cases where the age of onset of pan- to the differences in presentation and course of the disease
cytopenia or BM hypocellularity was reported, the average this diagnosis was not considered. An additional novel MPL
age was 5.3 months (range 0-96 months). Our patient group homozygous mutation was also identified in another fami-
differs from the more usual presentation of CAMT, suggest- ly following screening of 33 AA cases. We have shown for
ing they could be part of a different subtype of disease ( key the first time that homozygous MPL mutations can be
clinical features are shown in Table 1). As a diagnosis of found (albeit rarely) in children with familial AA in whom
CAMT was not suspected in the larger group of patients CAMT was not diagnosed or suspected. Additional studies
with AA, MPL was not an obvious candidate gene to screen will be needed to further clarify the relationship between
without the information obtained from the exome sequenc- CAMT, AA and MPL.
ing data from Family 1. In Family 2, the thrombocytopenia
was only identified when the children were undergoing
clinical investigations for a viral infection. None of these Authorship and Disclosures
patients had the classical presentation of CAMT.
In Family 1, although having a premature stop mutation The information provided by the authors about contributions from
that is usually associated with type I CAMT, the age of dis- persons listed as authors and in acknowledgments is available with
ease onset is later. Clinically, CAMT patients can be classi- the full text of this paper at www.haematologica.org.
fied as type I if the platelet count in the first year of life is Financial and other disclosures provided by the authors using the
50¥109/L or under or, if this information is not present, pan- ICMJE (www.icmje.org) Uniform Format for Disclosure of
cytopenia develops before the age of three years.15 From Competing Interests are also available at www.haematologica.org.
haematologica | 2012; 97(4) 527
a.J. walne et al.
References avenues in the analysis of monogenic disor- bocytopenia: the type of mutation predicts
ders. Hum Mutat. 2011;32(2):144-51. the course of the disease. Hum Mutat.
9. Cawthon RM. Telomere length measure- 2006;27(3):296.
1. Young NS, Scheinberg P, Calado RT. ment by a novel monochrome multiplex 16. Ballmaier M, Germeshausen M, Schulze H,
Aplastic anemia. Curr Opin Hematol. quantitative PCR method. Nuc Acids Res. Cherkaoui K, Lang S, Gaudig A, et al. c-mpl
2008;15(3):162-8. 2009;37(3):e21. mutations are the cause of congenital
2. Dokal I. Inherited aplastic anaemia/bone 10. Walne AJ, Vulliamy T, Beswick R, Kirwan amegakaryocytic thrombocytopenia.
marrow failure syndromes. In: Hoffbrand M and Dokal I. Mutations in C16orf57 and Blood. 2001;97(1):139-46.
AV, Catovsky, D. Tuddenham E.G.D and normal-length telomeres unify a subset of 17. Geddis AE. Congenital amegakaryocytic
Green, A.R, ed. Postgraduate Haematology patients with dyskeratosis congenita, poik- thrombocytopenia. Pediatr Blood Cancer.
(ed 6th): Wiley-Blackwell; 2011:186-205. iloderma with neutropenia and Rothmund- 2011;57(2):199-203.
3. Marsh J and Young N. Acquired aplastic Thomson syndrome. Hum Mol Genet. 18. Al-Qahtani FS. Congenital amegakaryocyt-
anaemia. In: Hoffbrand AV, Catovsky, D. 2010;19(22):4453-61. ic thrombocytopenia: a brief review of the
Tuddenham E.G.D and Green, A.R, ed. 11. Kaushansky K. Molecular mechanisms of literature. Clin Med Insights Pathol.
Postgraduate Haematology (ed 6th): Wiley- thrombopoietin signaling. J Thromb 2010;3:25-30.
Blackwell; 2011:206-25. Haemost. 2009;7(Suppl 1)235-8. 19. Ballmaier M, Germeshausen M. Advances
4. Dokal I, Vulliamy T. Inherited bone mar- 12. Ihara K, Ishii E, Eguchi M, Takada H, in the understanding of congenital
row failure syndromes. Haematologica. Suminoe A, Good RA, et al. Identification amegakaryocytic thrombocytopenia. Br J
2010;95(8):1236-40. of mutations in the c-mpl gene in congeni- Haematol. 2009;146(1):3-16.
5. Knight SW, Vulliamy TJ, Heiss NS, Matthijs tal amegakaryocytic thrombocytopenia. 20. Ball SE, Gibson FM, Rizzo S, Tooze JA,
G, Devriendt K, Connor JM, et al. 1.4 Mb Proc Natl Acad Sci USA. 1999;96(6):3132-6. Marsh JC, Gordon-Smith EC. Progressive
candidate gene region for X linked dysker- 13. Chou FS, Mulloy JC. The thrombopoi- telomere shortening in aplastic anemia.
atosis congenita defined by combined hap- etin/MPL pathway in hematopoiesis and Blood. 1998;91(10):3582-92.
lotype and X chromosome inactivation leukemogenesis. J Cell Biochem. 2011;112 21. Yamaguchi H, Calado RT, Ly H, Kajigaya S,
analysis. J Med Genet. 1998;35(12):993-6. (6):1491-8. Baerlocher GM, Chanock SJ, et al.
6. Lander ES, Botstein D. Homozygosity 14. Savoia A, Dufour C, Locatelli F, Noris P, Mutations in TERT, the gene for telomerase
mapping: a way to map human recessive Ambaglio C, Rosti V, et al. Congenital reverse transcriptase, in aplastic anemia. N
traits with the DNA of inbred children. amegakaryocytic thrombocytopenia: clini- Engl J Med. 2005;352(14):1413-24.
Science. 1987;236(4808):1567-70. cal and biological consequences of five 22. Marrone A, Sokhal P, Walne A, Beswick R,
7. Mardis ER. Next-generation DNA sequenc- novel mutations. Haematologica. 2007;92 Kirwan M, Killick S, et al. Functional char-
ing methods. Annu Rev Genomics Hum (9):1186-93. acterization of novel telomerase RNA
Genet. 2008;9387-402. 15. Germeshausen M, Ballmaier M, Welte K. (TERC) mutations in patients with diverse
8. Kuhlenbaumer G, Hullmann J, Appenzeller MPL mutations in 23 patients suffering clinical and pathological presentations.
S. Novel genomic techniques open new from congenital amegakaryocytic throm- Haematologica. 2007;92(8):1013-20.
528 haematologica | 2012; 97(4)
View publication stats
You might also like
- IEC 60950 Rev1Document3 pagesIEC 60950 Rev1Rip_BarNo ratings yet
- Computer System Validation Risk Assessment ToolDocument3 pagesComputer System Validation Risk Assessment Toolcpkakope100% (1)
- Holistic Self Health CareDocument80 pagesHolistic Self Health Careadkittipong100% (1)
- Pre-Commissioning & Commissioning For ETPDocument28 pagesPre-Commissioning & Commissioning For ETPprasad reddy100% (2)
- SGCE and Myoclonus Dystonia: Motor Characteristics, Diagnostic Criteria and Clinical Predictors of GenotypeDocument9 pagesSGCE and Myoclonus Dystonia: Motor Characteristics, Diagnostic Criteria and Clinical Predictors of GenotypeTalib AdilNo ratings yet
- FPDMM2Document9 pagesFPDMM2sccNo ratings yet
- Fced GenetikDocument33 pagesFced GenetikyosuanugrahapratamaNo ratings yet
- De Novo Mutations in YWHAG Cause Early Onset EpilepsyDocument11 pagesDe Novo Mutations in YWHAG Cause Early Onset EpilepsyGiselle Costa Daniel HonoratoNo ratings yet
- AJKD Furlano Autosomal Dominant Alport 2021Document12 pagesAJKD Furlano Autosomal Dominant Alport 2021Alice Porto Guerra VasconcelosNo ratings yet
- New England Journal Medicine: The ofDocument16 pagesNew England Journal Medicine: The ofMauricio FemeníaNo ratings yet
- 1 s2.0 S1071909198800077 MainDocument8 pages1 s2.0 S1071909198800077 MainamandaribeirouNo ratings yet
- Wu Et Al 2016Document11 pagesWu Et Al 2016MiaNo ratings yet
- Molec Gen Gen Med - 2020 - Luquetti - MYT1 Role in The Microtia Craniofacial Microsomia SpectrumDocument5 pagesMolec Gen Gen Med - 2020 - Luquetti - MYT1 Role in The Microtia Craniofacial Microsomia SpectrumVanessa KowalskaNo ratings yet
- Epidemiology and Etiology of Infantile Developmental andDocument8 pagesEpidemiology and Etiology of Infantile Developmental andyavuzatas23No ratings yet
- © Simtipro SRL: A New Blood Group System: Scientific Relevance and Media ResonanceDocument2 pages© Simtipro SRL: A New Blood Group System: Scientific Relevance and Media ResonanceMoncef PechaNo ratings yet
- 32Document8 pages32Guhan KANo ratings yet
- Familial Chronic Myeloproliferative Disorders: Clinical Phenotype and Evidence of Disease AnticipationDocument6 pagesFamilial Chronic Myeloproliferative Disorders: Clinical Phenotype and Evidence of Disease Anticipationayu_cicuuNo ratings yet
- 2002 LD DistichiasisDocument6 pages2002 LD DistichiasisDANIELANo ratings yet
- Migraña Con Aura: Una Mirada Molecular A Un Problema HereditarioDocument7 pagesMigraña Con Aura: Una Mirada Molecular A Un Problema Hereditariojucrf1218No ratings yet
- Consensus On The Rational Use of Antithrombotics in Veterinary Critical Care Curative Domain1defining Populations at Risk VeccDocument12 pagesConsensus On The Rational Use of Antithrombotics in Veterinary Critical Care Curative Domain1defining Populations at Risk VecchematologiaNo ratings yet
- Paper Seminario Jose EpulefDocument11 pagesPaper Seminario Jose EpulefJL EulefiNo ratings yet
- Churpek Et Al. - 2015 - Genomic Analysis of Germ Line and Somatic Variants in Familial Myelodysplasiaacute Myeloid Leukemia Key PointsDocument7 pagesChurpek Et Al. - 2015 - Genomic Analysis of Germ Line and Somatic Variants in Familial Myelodysplasiaacute Myeloid Leukemia Key Pointstefod30422No ratings yet
- DIAGNÓSTICO INTEGRAL DEL CÁNCER Clase 5Document15 pagesDIAGNÓSTICO INTEGRAL DEL CÁNCER Clase 5Nerea OteguiNo ratings yet
- La Mellarin D2Document2 pagesLa Mellarin D2JESUS DAVID BOLA‹O JIMENEZNo ratings yet
- Modpathol 201783Document14 pagesModpathol 201783Tian Nopita SariNo ratings yet
- Combined Analysis of Linkage and Whole Exome Sequencing Reveals Cic As A Candidate Gene For Isolated DystoniaDocument7 pagesCombined Analysis of Linkage and Whole Exome Sequencing Reveals Cic As A Candidate Gene For Isolated DystoniaSalih ÇayırNo ratings yet
- This Content Downloaded From 120.126.70.24 On Wed, 29 Mar 2023 11:59:10 UTCDocument8 pagesThis Content Downloaded From 120.126.70.24 On Wed, 29 Mar 2023 11:59:10 UTCmarcia suNo ratings yet
- Marsilio Et Al., 2023Document23 pagesMarsilio Et Al., 2023Charmila SouzaNo ratings yet
- Clinical Whole-Exome Sequencing For The Diagnosis of Mendelian DisordersDocument10 pagesClinical Whole-Exome Sequencing For The Diagnosis of Mendelian DisordersdssadkjlNo ratings yet
- Proceedings of The World Small Animal Veterinary Association Sydney, Australia - 2007Document7 pagesProceedings of The World Small Animal Veterinary Association Sydney, Australia - 2007Hanif WahyuNo ratings yet
- Role For Hedgehog Signaling in Cranial-Suture Development and ObesityDocument9 pagesRole For Hedgehog Signaling in Cranial-Suture Development and ObesityDanny Alvarez FocacciNo ratings yet
- Catarata Congenita HereditariaDocument13 pagesCatarata Congenita HereditariaMontze DíazNo ratings yet
- Zheng 2013Document6 pagesZheng 2013namiraNo ratings yet
- ALSDocument14 pagesALSZé CunhaNo ratings yet
- Review LESDocument16 pagesReview LESyoNo ratings yet
- Analysis of Chromosomal Aberrations and H2A - 2021 - Mutation Research GeneticDocument13 pagesAnalysis of Chromosomal Aberrations and H2A - 2021 - Mutation Research GeneticAURORA RENJANI KIRANANo ratings yet
- Genomics and Genes For ADME of Drugs in LiverDocument9 pagesGenomics and Genes For ADME of Drugs in LiverCutest guddyNo ratings yet
- Epi Genetica 2011Document11 pagesEpi Genetica 2011Laura Dragnic ToháNo ratings yet
- Autophagy Is Activated in Systemic Lupus Erythematosus and Required For Plasmablast DevelopmentDocument9 pagesAutophagy Is Activated in Systemic Lupus Erythematosus and Required For Plasmablast DevelopmentOliver Vebrian SampelilingNo ratings yet
- First HPSE2 Missense Mutation in Urofacial SyndromeDocument5 pagesFirst HPSE2 Missense Mutation in Urofacial SyndromeAmir Akram MasihNo ratings yet
- CMT WEGS Nejmoa0908094Document11 pagesCMT WEGS Nejmoa0908094MCuk2606No ratings yet
- Multiple Lipomas Linked To An RB1 Gene Mutation in A Large Pedigree With Low Penetrance RetinoblastomaDocument5 pagesMultiple Lipomas Linked To An RB1 Gene Mutation in A Large Pedigree With Low Penetrance RetinoblastomaMaria Burke-ChaussonNo ratings yet
- Benign Familial HematuriaDocument6 pagesBenign Familial Hematuriasiska_mariannaNo ratings yet
- Gen-Tieu XuowngDocument21 pagesGen-Tieu XuowngphungNo ratings yet
- Progressive Increase of The Mutated Mitochondria1 DNA Fraction in Kearns-Sayre SyndromeDocument6 pagesProgressive Increase of The Mutated Mitochondria1 DNA Fraction in Kearns-Sayre SyndromeGréta BotyánszkiNo ratings yet
- Sindrome NefroticoDocument8 pagesSindrome NefroticoRudy Espejo del ArcaNo ratings yet
- Gene Hunting: J. PongraczDocument15 pagesGene Hunting: J. PongracztahirnaeemkhanNo ratings yet
- Diagnosis and High Incidence of HyperornDocument7 pagesDiagnosis and High Incidence of HyperornnjdshdshuNo ratings yet
- Alloimmunization and Red Cellautoimmunization in Multitransfused Thalassemic of Indian OriginDocument5 pagesAlloimmunization and Red Cellautoimmunization in Multitransfused Thalassemic of Indian OriginFatimatuzzahra ShahabNo ratings yet
- FGS Ovarian CADocument12 pagesFGS Ovarian CAselcukorkmazNo ratings yet
- Involvement of MnSOD Ala16Val Polymorphism in Epilepsy A Relationship With Seizure TypeDocument6 pagesInvolvement of MnSOD Ala16Val Polymorphism in Epilepsy A Relationship With Seizure TypeDetector BotsNo ratings yet
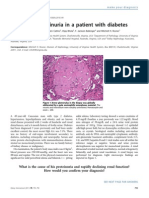
- The Case - Proteinuria in A Patient With DiabetesDocument2 pagesThe Case - Proteinuria in A Patient With Diabetesdrgawad2797No ratings yet
- PIIS1098360021012399Document10 pagesPIIS1098360021012399Yenifer Chipana QuispeNo ratings yet
- American J Hematol - 2006 - Takahashi - Methylprednisolone Pulse Therapy For Severe Immune Thrombocytopenia Associated WithDocument5 pagesAmerican J Hematol - 2006 - Takahashi - Methylprednisolone Pulse Therapy For Severe Immune Thrombocytopenia Associated WithMohan PrasadNo ratings yet
- Pti PurpuraDocument10 pagesPti PurpuradanielNo ratings yet
- Curs 8 - GenomicaDocument90 pagesCurs 8 - GenomicaNica PaulaNo ratings yet
- ICSH Guidelines For The Laboratory Diagnosis of Nonimmune Hereditary Red Cell Membrane DisordersDocument22 pagesICSH Guidelines For The Laboratory Diagnosis of Nonimmune Hereditary Red Cell Membrane DisordersSofia BujosaNo ratings yet
- DX de Las Enfermedades No Inmunologicas de La Membrana Del EritorcitoDocument22 pagesDX de Las Enfermedades No Inmunologicas de La Membrana Del EritorcitoMiguel Angel Hernandez SerratoNo ratings yet
- ICSH Guidelines For The Laboratory Diagnosis of Nonimmune Hereditary Red Cell Membrane DisordersDocument22 pagesICSH Guidelines For The Laboratory Diagnosis of Nonimmune Hereditary Red Cell Membrane DisordersSofia BujosaNo ratings yet
- Ejhg 201324Document7 pagesEjhg 201324sssssNo ratings yet
- CHAP NO4 Results (Abbas) Rewritten FileDocument12 pagesCHAP NO4 Results (Abbas) Rewritten FileRahid KhanNo ratings yet
- Carriers of COL3A1 Pathogenic Variants in DenmarkDocument10 pagesCarriers of COL3A1 Pathogenic Variants in Denmarkserrano.infinixNo ratings yet
- 2023 Ying Ung Chuin 1680579 Ethesis CompressedDocument281 pages2023 Ying Ung Chuin 1680579 Ethesis Compressedpilar gabriela hernandez fernandezNo ratings yet
- Chimerism: A Clinical GuideFrom EverandChimerism: A Clinical GuideNicole L. DraperNo ratings yet
- The Development of A Heat Wave Vulnerability Index For London-2013Document10 pagesThe Development of A Heat Wave Vulnerability Index For London-2013gilberto777No ratings yet
- Call History 640273efa1d48Document2 pagesCall History 640273efa1d48Krishnapriya GovindNo ratings yet
- Long Quiz Earth Sci 11Document2 pagesLong Quiz Earth Sci 11Jesha mae MagnoNo ratings yet
- Environmental Geotechniques: Theories of Ion ExchangeDocument21 pagesEnvironmental Geotechniques: Theories of Ion ExchangeTenkurala srujanaNo ratings yet
- Women Mental Health:: Postpartum DepressionDocument62 pagesWomen Mental Health:: Postpartum DepressionTickablingbling Alwaysshine Likeatenglarious100% (1)
- Expense 12-18 Agustus 2022Document4 pagesExpense 12-18 Agustus 2022Rizal RisqiNo ratings yet
- Feasibility of Screening For Preschool Behavioral and Emotional Problems in Primary Care Using The Early Childhood Screening AssessmentDocument9 pagesFeasibility of Screening For Preschool Behavioral and Emotional Problems in Primary Care Using The Early Childhood Screening AssessmentAhmed RamzyNo ratings yet
- BWT - HandbookDocument16 pagesBWT - HandbookDarko DuiloNo ratings yet
- Vespa S 125 3V Ie 150 3V Ie UPUTSTVODocument90 pagesVespa S 125 3V Ie 150 3V Ie UPUTSTVOdoughstoneNo ratings yet
- Vegetarian Meals On College CampusesDocument14 pagesVegetarian Meals On College CampusesVegan FutureNo ratings yet
- OSH SeminarDocument2 pagesOSH SeminarSahar Ulu JeruasNo ratings yet
- Autoconceito ShavelsonDocument15 pagesAutoconceito ShavelsonJuliana SchwarzNo ratings yet
- Vikas BhagwanDocument13 pagesVikas BhagwanhappysinNo ratings yet
- Frenkel's ExerciseDocument40 pagesFrenkel's ExerciseManiu EmeseNo ratings yet
- Mitsubishi Motors Vs Chrysler Philippines Labor UnionDocument2 pagesMitsubishi Motors Vs Chrysler Philippines Labor UnionKobe BullmastiffNo ratings yet
- ME Lab 2 LectureDocument5 pagesME Lab 2 LectureIan Paul TongolNo ratings yet
- Electrical and Electronic Troubleshooting For Industrial EnginesDocument25 pagesElectrical and Electronic Troubleshooting For Industrial EnginespanddyanNo ratings yet
- Cruciferous VegetablesDocument13 pagesCruciferous Vegetablesapi-271257230No ratings yet
- Group3 Dilemmac pr6031Document8 pagesGroup3 Dilemmac pr6031api-576539858No ratings yet
- Pipe System Friction Loss CalculationDocument4 pagesPipe System Friction Loss Calculationtorbenchr100% (1)
- Why Aws Is Even Better With CloudcheckrDocument7 pagesWhy Aws Is Even Better With CloudcheckrPardha SaradhiNo ratings yet
- Wagner and Traud ArticleDocument13 pagesWagner and Traud Articlegrimaguil100% (1)
- Muay Thai Beginners GuideDocument44 pagesMuay Thai Beginners GuideAmar Cooper67% (3)
- Building An Effective Safety Management System For Airlines: James J.H. Liou, Leon Yen, Gwo-Hshiung TzengDocument7 pagesBuilding An Effective Safety Management System For Airlines: James J.H. Liou, Leon Yen, Gwo-Hshiung Tzengzatul hasniNo ratings yet
- Chapter 05 Dental Caries - SlidesDocument27 pagesChapter 05 Dental Caries - SlidesNabila ApriyanNo ratings yet
- Inorganic Chemistry HomeworkDocument3 pagesInorganic Chemistry HomeworkAlpNo ratings yet