Professional Documents
Culture Documents
Biophysics of The Respiratory System
Biophysics of The Respiratory System
Uploaded by
Ife AzeezCopyright:
Available Formats
You might also like
- Chapter 48: Nursing Care of A Family When A Child Has An Endocrine or A Metabolic DisorderDocument22 pagesChapter 48: Nursing Care of A Family When A Child Has An Endocrine or A Metabolic DisorderAlyssaGrandeMontimorNo ratings yet
- Respiratory SystemDocument32 pagesRespiratory Systemapi-202513022No ratings yet
- Magnet Therapy and Protocols For Osteoporosis 2004 (Pag 18) by William H. Philpott - Negative Field On Brain and Spine For Pain, TreatmentsDocument25 pagesMagnet Therapy and Protocols For Osteoporosis 2004 (Pag 18) by William H. Philpott - Negative Field On Brain and Spine For Pain, Treatmentsitetejac100% (2)
- Respiratory Physiology PostedDocument25 pagesRespiratory Physiology PostedYeni PuspitaNo ratings yet
- Anatomi Fisiologi Pernapasan Dr. MDocument87 pagesAnatomi Fisiologi Pernapasan Dr. MARIF BSNo ratings yet
- Respiratory PhysiologyDocument68 pagesRespiratory PhysiologyDjNo ratings yet
- SapDocument5 pagesSapajminon16No ratings yet
- Respiratory System/3: Physical Principles of Gas ExchangeDocument27 pagesRespiratory System/3: Physical Principles of Gas ExchangeMohammad AlomariNo ratings yet
- Lecture - 6 - Airflow Breathing Mechanism - LectureDocument31 pagesLecture - 6 - Airflow Breathing Mechanism - LectureAmr KasemNo ratings yet
- Anatomi Fisiologi PernapasanDocument87 pagesAnatomi Fisiologi PernapasanAndre ChundawanNo ratings yet
- Respiratory SystemDocument23 pagesRespiratory SystembettyNo ratings yet
- University of Guyana School of Medicine Med 1106 - Physiology I DR Kalima ThompsonDocument66 pagesUniversity of Guyana School of Medicine Med 1106 - Physiology I DR Kalima ThompsonKNo ratings yet
- Respiratory System: Departement of Physiology Medical Faculty of Universitas Sumatera UtaraDocument142 pagesRespiratory System: Departement of Physiology Medical Faculty of Universitas Sumatera Utara292Vien HardiyantiNo ratings yet
- Respiratory System - Week 3Document66 pagesRespiratory System - Week 3Reentsha EllyNo ratings yet
- Lecture 1.6 Mechanics of Breathing II - Dr. AlkhateebDocument37 pagesLecture 1.6 Mechanics of Breathing II - Dr. AlkhateebJohn KashNo ratings yet
- Airflow: Group 4 - Respiratory System VentilationDocument18 pagesAirflow: Group 4 - Respiratory System VentilationNathalieCaracaNo ratings yet
- Breathing and Exchange of GasesDocument40 pagesBreathing and Exchange of GasesPranesh ShandilyaNo ratings yet
- Respiratory System: Dr. Agung Kurniawan, MkesDocument31 pagesRespiratory System: Dr. Agung Kurniawan, MkesJojo BodhoNo ratings yet
- H.circulatory SystemDocument11 pagesH.circulatory SystemElleNo ratings yet
- Pulmonary Physiology Pulmonary PhysiologyDocument45 pagesPulmonary Physiology Pulmonary PhysiologyAhmad Harissul IbadNo ratings yet
- Pulmonary PhysiologyDocument18 pagesPulmonary Physiologylezibells100% (2)
- Respiratory Physiology IIDocument83 pagesRespiratory Physiology IIDeea LobonțiuNo ratings yet
- Knes 313 Chapter 6 ReviewDocument3 pagesKnes 313 Chapter 6 Reviewюрий локтионовNo ratings yet
- Ani̇mal Respiratory System PhysiologyDocument18 pagesAni̇mal Respiratory System PhysiologyNajafova SuadaNo ratings yet
- M8 - Investigations and Monitoring in The ICUDocument68 pagesM8 - Investigations and Monitoring in The ICUVishal KumarNo ratings yet
- Res PH Dental - 2023Document82 pagesRes PH Dental - 2023shozainab2003No ratings yet
- Alterations in OxygenationDocument104 pagesAlterations in OxygenationMelchor Felipe Salvosa100% (1)
- Pulmonary PhysioDocument52 pagesPulmonary PhysiodjokerNo ratings yet
- Anatomi Dan Fisiologi RespirasiDocument22 pagesAnatomi Dan Fisiologi RespirasinortonaingNo ratings yet
- Respiratory PhysiologyDocument98 pagesRespiratory PhysiologySurya SuryaNo ratings yet
- 1.airway, Airflow, Ventilation and DiffusionDocument64 pages1.airway, Airflow, Ventilation and DiffusionEbin EbenezerNo ratings yet
- Respiratory SystemDocument192 pagesRespiratory SystemQuolette Constante100% (1)
- M132 Respiratory 2022Document28 pagesM132 Respiratory 2022Bana ItanNo ratings yet
- Giu 2986 62 16130 2024-02-22T14 46 59Document18 pagesGiu 2986 62 16130 2024-02-22T14 46 59amoraaloushNo ratings yet
- Basics of Respiratory Function: Mee Wah NG RSO Dubai 2002Document43 pagesBasics of Respiratory Function: Mee Wah NG RSO Dubai 2002chanlalNo ratings yet
- Fundamentalof Ventilation & PacemakerDocument22 pagesFundamentalof Ventilation & PacemakerLucila Lugo100% (1)
- Respiratory System-1Document84 pagesRespiratory System-1pranjal raiNo ratings yet
- Faal Paru - RingkasDocument102 pagesFaal Paru - RingkasdhaneswaraawrNo ratings yet
- OLM. Respiratory System Functional Anatomy, Muscles and Pressure - WholeDocument58 pagesOLM. Respiratory System Functional Anatomy, Muscles and Pressure - Wholedagehi6880No ratings yet
- Respiratoryfunctionandimportancetoanesthesia Final 140103050711 Phpapp02Document62 pagesRespiratoryfunctionandimportancetoanesthesia Final 140103050711 Phpapp02rijjorajooNo ratings yet
- L - 1 Physiology of Respiration IDocument28 pagesL - 1 Physiology of Respiration IshahabsworldNo ratings yet
- Chapter 22Document12 pagesChapter 22Jenn ParksNo ratings yet
- 16 Pulmonary Ventsilation CH16 2Document34 pages16 Pulmonary Ventsilation CH16 2Brodsky MacedoNo ratings yet
- Breathing & Exchange of Gases NotesDocument7 pagesBreathing & Exchange of Gases NotesKankana Biswas100% (1)
- L - 1 Physiology of Respiration IDocument25 pagesL - 1 Physiology of Respiration Ikaukab azimNo ratings yet
- 06 Respiratory System PhysiologyDocument43 pages06 Respiratory System PhysiologyKaye Alyssa EnriquezNo ratings yet
- Ventilation Dead Space Ventilation Physiology of RespirationDocument55 pagesVentilation Dead Space Ventilation Physiology of RespirationkuhutansittinurhalizaNo ratings yet
- CH 25 Respiratory AssessmentDocument14 pagesCH 25 Respiratory Assessmentهدوء النسمةNo ratings yet
- Chapter 12Document28 pagesChapter 12Ammar SmadiNo ratings yet
- Chapter 12Document28 pagesChapter 12براءة أحمد السلاماتNo ratings yet
- Resp Physio SAQsDocument25 pagesResp Physio SAQsanaeshkl100% (1)
- The Respiratory SystemDocument2 pagesThe Respiratory SystemconnievelardeNo ratings yet
- Resp CH 1Document53 pagesResp CH 1Ahmed khanNo ratings yet
- Ventilation and Respiratory VolumesDocument20 pagesVentilation and Respiratory VolumesJessa mae CabredoNo ratings yet
- Compliance of Lung 2003Document13 pagesCompliance of Lung 2003Narendra Bhattarai100% (1)
- 11 Respiratory SystemDocument19 pages11 Respiratory SystemSenthereng MoaisiNo ratings yet
- Pulmonary VentilationDocument24 pagesPulmonary VentilationAreeba KhanNo ratings yet
- Pulmonary Ventilation: Dr. Mohamed HassanDocument29 pagesPulmonary Ventilation: Dr. Mohamed HassanSAKARIYE MAXAMEDNo ratings yet
- Breathing and Exchange of GasesDocument25 pagesBreathing and Exchange of GasesSureshNo ratings yet
- Textbook ReadingDocument275 pagesTextbook ReadingNovitasari EkaNo ratings yet
- Coronavirus Disease 2019Document46 pagesCoronavirus Disease 2019nimer sheikhNo ratings yet
- Ayurvedic Management of Ulcerative Colitis W.S.R To Grahni Dusti-A Case StudyDocument6 pagesAyurvedic Management of Ulcerative Colitis W.S.R To Grahni Dusti-A Case StudyDrHassan Ahmed ShaikhNo ratings yet
- Jovr 6 3 192Document8 pagesJovr 6 3 192Felicia JesslynNo ratings yet
- Neurologic System AssessmentDocument22 pagesNeurologic System Assessmentroy_linaoNo ratings yet
- Polycythemi A: Presented By: Palaran, Nina Mae CDocument16 pagesPolycythemi A: Presented By: Palaran, Nina Mae CJichutreasureNo ratings yet
- Pediatric CataractDocument8 pagesPediatric CataractDayang Sayalam100% (1)
- Acute and Chronic PancreatitisDocument1 pageAcute and Chronic PancreatitisMaryam FadahNo ratings yet
- Biological Science Class 11 SyllabusDocument10 pagesBiological Science Class 11 SyllabusSwapnil Das HCS - 46No ratings yet
- ICU protocol 2015 قصر العيني by mansdocsDocument227 pagesICU protocol 2015 قصر العيني by mansdocsWalaa YousefNo ratings yet
- Homoeopathy BookletDocument66 pagesHomoeopathy BookletNafisa AhmadNo ratings yet
- Neurological ExaminationDocument13 pagesNeurological Examinationsaveetha purushothamanNo ratings yet
- ch01 PDFDocument5 pagesch01 PDFHugsNo ratings yet
- Reading Activity: Newspaper ArticleDocument2 pagesReading Activity: Newspaper ArticleAlba etceteraNo ratings yet
- Estradiol 10mcg (Vagifen)Document13 pagesEstradiol 10mcg (Vagifen)asdwasdNo ratings yet
- Unit 7 - Digestive System Guided Notes - VaughanDocument9 pagesUnit 7 - Digestive System Guided Notes - VaughanJeanette IrambonaNo ratings yet
- Continence Tools For Residential Aged Care: An Education GuideDocument27 pagesContinence Tools For Residential Aged Care: An Education GuideAlfred SamuelNo ratings yet
- Sonographic Diagnosis of Pneumonia and BronchopneumoniaDocument8 pagesSonographic Diagnosis of Pneumonia and BronchopneumoniaCaitlynNo ratings yet
- Antibióticos para La Neumonía Asociada Al Ventilador.Document96 pagesAntibióticos para La Neumonía Asociada Al Ventilador.Julio GonzalesNo ratings yet
- Oral Pathology in Clinical Dentistry: A Systematic Approach: Nvited EviewDocument5 pagesOral Pathology in Clinical Dentistry: A Systematic Approach: Nvited EviewRoxana OneaNo ratings yet
- Topic 2Document2 pagesTopic 2Joshua PanganNo ratings yet
- Pertemuan 15 - Epidemiologi Penyakit New Emerging DiseaseDocument79 pagesPertemuan 15 - Epidemiologi Penyakit New Emerging DiseasePutti AnnisaNo ratings yet
- ASTMHExam Brochure 16 FNLV 2Document17 pagesASTMHExam Brochure 16 FNLV 2esraaNo ratings yet
- Functional Syndrome in NeurologyDocument11 pagesFunctional Syndrome in Neurologyidno1008No ratings yet
- Hepatitis B VaccineDocument6 pagesHepatitis B VaccineAziil LiizaNo ratings yet
- Borax Booklet Doctors Without BordersDocument12 pagesBorax Booklet Doctors Without Bordersfdartee100% (3)
- Module 4 - Death Certification and Cause of Death - 2Document21 pagesModule 4 - Death Certification and Cause of Death - 2anton_susanto10No ratings yet
- Eov PDFDocument2 pagesEov PDFabhishekmla007No ratings yet
Biophysics of The Respiratory System
Biophysics of The Respiratory System
Uploaded by
Ife AzeezOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Biophysics of The Respiratory System
Biophysics of The Respiratory System
Uploaded by
Ife AzeezCopyright:
Available Formats
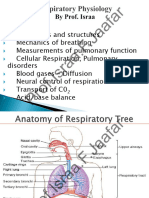
The Respiratory System
Physiological Functions of Lung
- Exchange of O2 and CO2
- Heat exchange
- Keeping the acidity of blood on a constant level
- Fluid balance of body/warming and moisturizing the inhaled air
2 characteristics of the human respiratory system
1. Uses convective systems (circulatory and ventilatory systems) for long-distance transport of O2
and CO2
2. Reserves diffusion for short-distance transport of O2 and CO2
Convection = the circulatory motion that occurs in gas or liquid at a nonuniform temperature owing to
currents caused by differences in density
Lung Volumes
○ Tidal Volume = is the amount of air that can be inhaled or exhaled during one respiratory cycle
○ Inspiratory Reserve Volume = is the amount of air that can be forcibly inhaled after a normal tidal
volume
○ Residual Volume = is the volume of air remaining in the lungs after maximal exhalation
○ Expiratory Reserve Volume = is the volume of air that can be exhaled forcibly after exhalation of
normal tidal volume
○ Total Lung Capacity = is the maximum volume of air the lungs can accommodate or sum of all
volume compartments or volume of air in lungs after maximum inspiration
Vital Capacity and General Gas Law
- Boyle's Law tells us that the volume of gas increases as the pressure decreases.
- Charles' Law tells us that the volume of gas increases as the temperature increases.
- Avogadro's Law tell us that the volume of gas increases as the amount of gas increases.
- The ideal gas law is the combination of the three simple gas laws.
Dalton's Law of Partial Pressure = the total pressure exerted by a mixture of gases is equal to the sum
of separate or partial pressures that each gas would exert if it alone occupied the entire volume
For atmospheric air
The partial pressure of any gas can be calculated using the following formula:
partial pressure = fractional concentration (F) x total gas pressure
Water Vapor Pressure and Temperature
- Temperature
The higher the temperature is, the more molecules have enough energy to escape from the liquid or solid,
which leads to higher vapor pressure values.
temperature of a liquid increases → kinetic energy of its molecules increases → number of molecules
transitioning into a vapor increases → vapor pressure increases
At lower temperatures, fewer molecules have sufficient energy.
- Substance nature (types of molecules)
The vapor pressure will be relatively low for substances with stronger intermolecular forces. On the contrary,
the vapor pressure is relatively high for relatively weak forces.
The important thing to mention is the fact that the surface area of liquid/solid substance in contact with the gas
doesn't affect the vapor pressure. So it doesn't matter if we put our liquid into a wide flask or a thin graduated
cylinder - the vapor pressure remains the same.
The partial pressure of O2 is calculated using the formula
Biophysics of the Respiratory System Page 1
The Respiratory System
Henry's Law = the quantity of a dissolved gas in a liquid at a given temperature is almost
directly proportional to the partial pressure of that gas in the gas phase in equilibrium
Partial pressure of oxygen reflects the amount of oxygen gas dissolved in the blood
- Elevated level is associated with
○ Increased oxygen levels in the inhaled air
○ Polycythaemia
- Decreased levels is associated with
○ Decreased oxygen levels in the inhaled air
○ Anaemia
○ Heart decompensation
○ Chronic obstructive pulmonary disease
○ Restrictive pulmonary disease
○ Hypoventilation
Partial pressure of carbon dioxide reflects the amount of carbon dioxide is dissolved in the
blood
- Elevated levels are associated with
○ Pulmonary oedema
○ Obstructive lung disease
- Decreased levels are associated with
○ Hyperventilation
○ Hypoxia
○ Anxiety
○ Pregnancy
- 2 factors significantly impact the partial pressure of carbon dioxide
1. The depth and speed of breathing
Hyperventilating - lower pco2
Holding in breath - higher pco2
2. The lungs capacity for freely exchanging CO2 across the alveolar membrane
Pulmonary oedema - extra layer of fluid in the alveoli that interferes with the lung's
ability to get rid of CO2 - rise in pco2
Acute asthmatic attack - upper and middle airway obstruction to block alveolar
ventilation - CO2 retention
Medical Application of Henry Law
- Compensation of inadequate haemoglobin content by Hyperbaric Oxygen
→ acute poisoning of carbon dioxide
→ Gas gangrene
→ Loss of sexual vitality
- Decompression sickness
Breathing gases - mixture of gases used in breathing equipment and enclosed habitats
- Reduces the risk of decompression sickness
- Reduces the duration of decompression
- Reduces nitrogen narcosis
- Allowing safer deep diving
Features of a safe breathing gas:
- Contains sufficient oxygen to support life, consciousness and work rate of the breather
- Must not contain harmful gases
- Must not become toxic when being breathed at high pressure
Biophysics of the Respiratory System Page 2
The Respiratory System
Oxygen Toxicity
- Anaesthesiology
○ Free radicals damage cell membranes and damage of surfactants
○ Neonatal fibrosis
- Diving
- Depends on exposition time and personal susceptibility
Symptoms of oxygen toxicity
CNS
• V - visual disturbances
• E - ears (hearing abnormalities)
• N - nausea
• T - twitching
• I - irritability
• D - dizziness
Lungs
- Decrease in vital capacity
- Cough
- Increases respiratory effort
- Pain in sternum
- Dyspnea
- Lung oedema
- Lung fibrosis
Volume changes in the thoracic cavity and produces a pressure gradient which moves air in and out of the
alveoli
Total Respiratory Pulmonary Rate = Breathing frequency x Tidal Volume
Inspiration and Expiration
Inspiration = contraction of the diaphragm
- Expands the size of the thoracic cavity
- Decreases the intrapleural pressure
- Pulls the visceral pleura along with the parietal pleura
- Decreases the intrapulmonary (alveolar) pressure
- Draws air into the alveoli
Expiration = relaxation of the diaphragm and external intercostal muscles
- Permits recoil of the elastic fibers in the walls of the lungs and alveoli
- Increases the intrapulmonary (alveolar) pressure
- Air is forced out of the alveoli
- Pulls the parietal pleura along with the visceral pleura
- Decreases the size of the thoracic cavity
Intrapleural pressure
- The intrapleural pressure (which is usually -4 mmHg at rest) becomes more subatmospheric or more
negative.
- Due to the adhesive force of the pleural fluid, the expansion of the thoracic cavity forces the lungs to
stretch and expand as well.
- This increase in volume leads to a decrease in intra-alveolar pressure, creating a pressure lower than
atmospheric pressure.
What happens when intrapleural pressure is equal to atmospheric pressure (pneomothorax or collapsed
lung)
Biophysics of the Respiratory System Page 3
The Respiratory System
Compliance (C) = is a measure of the elasticity of the connective of airways ,lungs and
blood vessels and the surface tension of the alveolar gas-liquid interface)
- Refers to the distensibility of an elastic structure
- It is the difference between alveolar pressure and intrapleural pressure
Emphysema - many alveolar walls are lost, the lungs become so loose and floppy that only a
small pressure difference is necessary to maintain a large volume. Thus the lungs become
highly compliant, involving more difficult expiration - loss of elastic recoil
Fibrosis - the lung becomes stiff, making a large pressure necessary to maintain a moderate
volume - poorly compliant, more work required to breathe
Determinants of lung compliance
- Surface tension; Laplace law, pulmonary surfactants
- Elastic properties
Surface tension and Laplace law
Laplace's law states that the pressure inside an inflated elastic container with a curved
surface, e.g., a bubble or a blood vessel, is inversely proportional to the radius as long as the
surface tension is presumed to change little.
- The pressure inside the bubble is greater to stop it from imploding
Surfactants - surface active agents
Characteristics of surfactant
- Serves to minimize the surface tension
- As the surface area of the film is reduced, the surface tension decreases
- Contains both hydrophobic and hydrophilic groups (amphipathic)
- Contains phosphatidylcholine
Functions of surfactants
- Lines alveoli
- Reduces surface tension between water molecules
- Increases compliance
- Prevents oedema = movement of fluid into the alveoli
Physiological Advantages of Lung Surfactants
- Reduction of the work of breathing; reduces muscular effort needed to expand the lung
- Decrease in the elastic recoil at low lung volumes - helps to prevent the lung from
collapsing at the end of each respiration
- Stabilization of alveoli
Resistance to Airflow
Gas flow during breathing is opposed by:
- Viscous resistance caused by tissue friction during breathing
- Airway resistance caused by friction between gas molecules and the walls of airways
and internal friction between gas molecules
- Inertial forces
To calculate the resistance to airflow
Biophysics of the Respiratory System Page 4
The Respiratory System
Determinants of Gas Diffusion in Lungs
- The physiochemical characteristics of transferred gas
○ Molecular weight
○ Gas temperature
○ Solubility coefficient
- The driving pressure across the alveolocapillary membrane
○ Alveolar ventilation
○ Chemical reactions
○ Pulmonary capillary blood flow
- The characteristics of the lung
○ Membrane area
○ Diffusion distance
○ Liquid viscosity
Biophysics of the Respiratory System Page 5
You might also like
- Chapter 48: Nursing Care of A Family When A Child Has An Endocrine or A Metabolic DisorderDocument22 pagesChapter 48: Nursing Care of A Family When A Child Has An Endocrine or A Metabolic DisorderAlyssaGrandeMontimorNo ratings yet
- Respiratory SystemDocument32 pagesRespiratory Systemapi-202513022No ratings yet
- Magnet Therapy and Protocols For Osteoporosis 2004 (Pag 18) by William H. Philpott - Negative Field On Brain and Spine For Pain, TreatmentsDocument25 pagesMagnet Therapy and Protocols For Osteoporosis 2004 (Pag 18) by William H. Philpott - Negative Field On Brain and Spine For Pain, Treatmentsitetejac100% (2)
- Respiratory Physiology PostedDocument25 pagesRespiratory Physiology PostedYeni PuspitaNo ratings yet
- Anatomi Fisiologi Pernapasan Dr. MDocument87 pagesAnatomi Fisiologi Pernapasan Dr. MARIF BSNo ratings yet
- Respiratory PhysiologyDocument68 pagesRespiratory PhysiologyDjNo ratings yet
- SapDocument5 pagesSapajminon16No ratings yet
- Respiratory System/3: Physical Principles of Gas ExchangeDocument27 pagesRespiratory System/3: Physical Principles of Gas ExchangeMohammad AlomariNo ratings yet
- Lecture - 6 - Airflow Breathing Mechanism - LectureDocument31 pagesLecture - 6 - Airflow Breathing Mechanism - LectureAmr KasemNo ratings yet
- Anatomi Fisiologi PernapasanDocument87 pagesAnatomi Fisiologi PernapasanAndre ChundawanNo ratings yet
- Respiratory SystemDocument23 pagesRespiratory SystembettyNo ratings yet
- University of Guyana School of Medicine Med 1106 - Physiology I DR Kalima ThompsonDocument66 pagesUniversity of Guyana School of Medicine Med 1106 - Physiology I DR Kalima ThompsonKNo ratings yet
- Respiratory System: Departement of Physiology Medical Faculty of Universitas Sumatera UtaraDocument142 pagesRespiratory System: Departement of Physiology Medical Faculty of Universitas Sumatera Utara292Vien HardiyantiNo ratings yet
- Respiratory System - Week 3Document66 pagesRespiratory System - Week 3Reentsha EllyNo ratings yet
- Lecture 1.6 Mechanics of Breathing II - Dr. AlkhateebDocument37 pagesLecture 1.6 Mechanics of Breathing II - Dr. AlkhateebJohn KashNo ratings yet
- Airflow: Group 4 - Respiratory System VentilationDocument18 pagesAirflow: Group 4 - Respiratory System VentilationNathalieCaracaNo ratings yet
- Breathing and Exchange of GasesDocument40 pagesBreathing and Exchange of GasesPranesh ShandilyaNo ratings yet
- Respiratory System: Dr. Agung Kurniawan, MkesDocument31 pagesRespiratory System: Dr. Agung Kurniawan, MkesJojo BodhoNo ratings yet
- H.circulatory SystemDocument11 pagesH.circulatory SystemElleNo ratings yet
- Pulmonary Physiology Pulmonary PhysiologyDocument45 pagesPulmonary Physiology Pulmonary PhysiologyAhmad Harissul IbadNo ratings yet
- Pulmonary PhysiologyDocument18 pagesPulmonary Physiologylezibells100% (2)
- Respiratory Physiology IIDocument83 pagesRespiratory Physiology IIDeea LobonțiuNo ratings yet
- Knes 313 Chapter 6 ReviewDocument3 pagesKnes 313 Chapter 6 Reviewюрий локтионовNo ratings yet
- Ani̇mal Respiratory System PhysiologyDocument18 pagesAni̇mal Respiratory System PhysiologyNajafova SuadaNo ratings yet
- M8 - Investigations and Monitoring in The ICUDocument68 pagesM8 - Investigations and Monitoring in The ICUVishal KumarNo ratings yet
- Res PH Dental - 2023Document82 pagesRes PH Dental - 2023shozainab2003No ratings yet
- Alterations in OxygenationDocument104 pagesAlterations in OxygenationMelchor Felipe Salvosa100% (1)
- Pulmonary PhysioDocument52 pagesPulmonary PhysiodjokerNo ratings yet
- Anatomi Dan Fisiologi RespirasiDocument22 pagesAnatomi Dan Fisiologi RespirasinortonaingNo ratings yet
- Respiratory PhysiologyDocument98 pagesRespiratory PhysiologySurya SuryaNo ratings yet
- 1.airway, Airflow, Ventilation and DiffusionDocument64 pages1.airway, Airflow, Ventilation and DiffusionEbin EbenezerNo ratings yet
- Respiratory SystemDocument192 pagesRespiratory SystemQuolette Constante100% (1)
- M132 Respiratory 2022Document28 pagesM132 Respiratory 2022Bana ItanNo ratings yet
- Giu 2986 62 16130 2024-02-22T14 46 59Document18 pagesGiu 2986 62 16130 2024-02-22T14 46 59amoraaloushNo ratings yet
- Basics of Respiratory Function: Mee Wah NG RSO Dubai 2002Document43 pagesBasics of Respiratory Function: Mee Wah NG RSO Dubai 2002chanlalNo ratings yet
- Fundamentalof Ventilation & PacemakerDocument22 pagesFundamentalof Ventilation & PacemakerLucila Lugo100% (1)
- Respiratory System-1Document84 pagesRespiratory System-1pranjal raiNo ratings yet
- Faal Paru - RingkasDocument102 pagesFaal Paru - RingkasdhaneswaraawrNo ratings yet
- OLM. Respiratory System Functional Anatomy, Muscles and Pressure - WholeDocument58 pagesOLM. Respiratory System Functional Anatomy, Muscles and Pressure - Wholedagehi6880No ratings yet
- Respiratoryfunctionandimportancetoanesthesia Final 140103050711 Phpapp02Document62 pagesRespiratoryfunctionandimportancetoanesthesia Final 140103050711 Phpapp02rijjorajooNo ratings yet
- L - 1 Physiology of Respiration IDocument28 pagesL - 1 Physiology of Respiration IshahabsworldNo ratings yet
- Chapter 22Document12 pagesChapter 22Jenn ParksNo ratings yet
- 16 Pulmonary Ventsilation CH16 2Document34 pages16 Pulmonary Ventsilation CH16 2Brodsky MacedoNo ratings yet
- Breathing & Exchange of Gases NotesDocument7 pagesBreathing & Exchange of Gases NotesKankana Biswas100% (1)
- L - 1 Physiology of Respiration IDocument25 pagesL - 1 Physiology of Respiration Ikaukab azimNo ratings yet
- 06 Respiratory System PhysiologyDocument43 pages06 Respiratory System PhysiologyKaye Alyssa EnriquezNo ratings yet
- Ventilation Dead Space Ventilation Physiology of RespirationDocument55 pagesVentilation Dead Space Ventilation Physiology of RespirationkuhutansittinurhalizaNo ratings yet
- CH 25 Respiratory AssessmentDocument14 pagesCH 25 Respiratory Assessmentهدوء النسمةNo ratings yet
- Chapter 12Document28 pagesChapter 12Ammar SmadiNo ratings yet
- Chapter 12Document28 pagesChapter 12براءة أحمد السلاماتNo ratings yet
- Resp Physio SAQsDocument25 pagesResp Physio SAQsanaeshkl100% (1)
- The Respiratory SystemDocument2 pagesThe Respiratory SystemconnievelardeNo ratings yet
- Resp CH 1Document53 pagesResp CH 1Ahmed khanNo ratings yet
- Ventilation and Respiratory VolumesDocument20 pagesVentilation and Respiratory VolumesJessa mae CabredoNo ratings yet
- Compliance of Lung 2003Document13 pagesCompliance of Lung 2003Narendra Bhattarai100% (1)
- 11 Respiratory SystemDocument19 pages11 Respiratory SystemSenthereng MoaisiNo ratings yet
- Pulmonary VentilationDocument24 pagesPulmonary VentilationAreeba KhanNo ratings yet
- Pulmonary Ventilation: Dr. Mohamed HassanDocument29 pagesPulmonary Ventilation: Dr. Mohamed HassanSAKARIYE MAXAMEDNo ratings yet
- Breathing and Exchange of GasesDocument25 pagesBreathing and Exchange of GasesSureshNo ratings yet
- Textbook ReadingDocument275 pagesTextbook ReadingNovitasari EkaNo ratings yet
- Coronavirus Disease 2019Document46 pagesCoronavirus Disease 2019nimer sheikhNo ratings yet
- Ayurvedic Management of Ulcerative Colitis W.S.R To Grahni Dusti-A Case StudyDocument6 pagesAyurvedic Management of Ulcerative Colitis W.S.R To Grahni Dusti-A Case StudyDrHassan Ahmed ShaikhNo ratings yet
- Jovr 6 3 192Document8 pagesJovr 6 3 192Felicia JesslynNo ratings yet
- Neurologic System AssessmentDocument22 pagesNeurologic System Assessmentroy_linaoNo ratings yet
- Polycythemi A: Presented By: Palaran, Nina Mae CDocument16 pagesPolycythemi A: Presented By: Palaran, Nina Mae CJichutreasureNo ratings yet
- Pediatric CataractDocument8 pagesPediatric CataractDayang Sayalam100% (1)
- Acute and Chronic PancreatitisDocument1 pageAcute and Chronic PancreatitisMaryam FadahNo ratings yet
- Biological Science Class 11 SyllabusDocument10 pagesBiological Science Class 11 SyllabusSwapnil Das HCS - 46No ratings yet
- ICU protocol 2015 قصر العيني by mansdocsDocument227 pagesICU protocol 2015 قصر العيني by mansdocsWalaa YousefNo ratings yet
- Homoeopathy BookletDocument66 pagesHomoeopathy BookletNafisa AhmadNo ratings yet
- Neurological ExaminationDocument13 pagesNeurological Examinationsaveetha purushothamanNo ratings yet
- ch01 PDFDocument5 pagesch01 PDFHugsNo ratings yet
- Reading Activity: Newspaper ArticleDocument2 pagesReading Activity: Newspaper ArticleAlba etceteraNo ratings yet
- Estradiol 10mcg (Vagifen)Document13 pagesEstradiol 10mcg (Vagifen)asdwasdNo ratings yet
- Unit 7 - Digestive System Guided Notes - VaughanDocument9 pagesUnit 7 - Digestive System Guided Notes - VaughanJeanette IrambonaNo ratings yet
- Continence Tools For Residential Aged Care: An Education GuideDocument27 pagesContinence Tools For Residential Aged Care: An Education GuideAlfred SamuelNo ratings yet
- Sonographic Diagnosis of Pneumonia and BronchopneumoniaDocument8 pagesSonographic Diagnosis of Pneumonia and BronchopneumoniaCaitlynNo ratings yet
- Antibióticos para La Neumonía Asociada Al Ventilador.Document96 pagesAntibióticos para La Neumonía Asociada Al Ventilador.Julio GonzalesNo ratings yet
- Oral Pathology in Clinical Dentistry: A Systematic Approach: Nvited EviewDocument5 pagesOral Pathology in Clinical Dentistry: A Systematic Approach: Nvited EviewRoxana OneaNo ratings yet
- Topic 2Document2 pagesTopic 2Joshua PanganNo ratings yet
- Pertemuan 15 - Epidemiologi Penyakit New Emerging DiseaseDocument79 pagesPertemuan 15 - Epidemiologi Penyakit New Emerging DiseasePutti AnnisaNo ratings yet
- ASTMHExam Brochure 16 FNLV 2Document17 pagesASTMHExam Brochure 16 FNLV 2esraaNo ratings yet
- Functional Syndrome in NeurologyDocument11 pagesFunctional Syndrome in Neurologyidno1008No ratings yet
- Hepatitis B VaccineDocument6 pagesHepatitis B VaccineAziil LiizaNo ratings yet
- Borax Booklet Doctors Without BordersDocument12 pagesBorax Booklet Doctors Without Bordersfdartee100% (3)
- Module 4 - Death Certification and Cause of Death - 2Document21 pagesModule 4 - Death Certification and Cause of Death - 2anton_susanto10No ratings yet
- Eov PDFDocument2 pagesEov PDFabhishekmla007No ratings yet