Professional Documents
Culture Documents
P2 Memo 2013
P2 Memo 2013
Uploaded by
Bonga DubeCopyright:
Available Formats
You might also like
- Mark Scheme: Sample Assessment Material 2018Document17 pagesMark Scheme: Sample Assessment Material 2018謝利米No ratings yet
- Unofficial Mark SchemeDocument6 pagesUnofficial Mark SchemeRodolph SmithNo ratings yet
- Transmission Line PresentationDocument55 pagesTransmission Line PresentationAshwani Dogra89% (9)
- P2 Memo 2012Document9 pagesP2 Memo 2012Bonga DubeNo ratings yet
- PHY SC GR 12 June 2019 P2 and MemoDocument20 pagesPHY SC GR 12 June 2019 P2 and Memoamahlezitha91No ratings yet
- P2 Memo 2011Document8 pagesP2 Memo 2011Bonga DubeNo ratings yet
- Gce A Level Marking Scheme: AUTUMN 2021Document24 pagesGce A Level Marking Scheme: AUTUMN 2021NonuNo ratings yet
- Chemistry Answers PDFDocument126 pagesChemistry Answers PDFNurafiqah FarhaniNo ratings yet
- P2 Memo 2017Document12 pagesP2 Memo 2017Bonga DubeNo ratings yet
- Chem D SolvedDocument18 pagesChem D SolvedVkrt SharmaNo ratings yet
- Wjec PaperDocument16 pagesWjec PaperNonuNo ratings yet
- P2 Memo 2015Document10 pagesP2 Memo 2015Bonga DubeNo ratings yet
- SHCC - 2023 - Chem Paper2 - AnnaDocument8 pagesSHCC - 2023 - Chem Paper2 - AnnaOof GucciNo ratings yet
- Chemistry XII Practice Paper BOOK 1Document95 pagesChemistry XII Practice Paper BOOK 1hembrampriyanka07No ratings yet
- Chem B SolvedDocument17 pagesChem B SolvedVkrt SharmaNo ratings yet
- Physical Sciences P2 Nov 2012 EngDocument18 pagesPhysical Sciences P2 Nov 2012 Engthembekilethusi9No ratings yet
- Physical Sciences P2 Sep 2017 Memo GR 10 (14.9.2017)Document10 pagesPhysical Sciences P2 Sep 2017 Memo GR 10 (14.9.2017)toygm672No ratings yet
- s18 Chemistry - 2410u10 1 Wjec As Chemistry Un1 MsDocument14 pagess18 Chemistry - 2410u10 1 Wjec As Chemistry Un1 MsAngelika LauNo ratings yet
- National Senior CertificateDocument11 pagesNational Senior CertificateVassiliNo ratings yet
- Physical Sciences P2 Feb-March 2013 EngDocument19 pagesPhysical Sciences P2 Feb-March 2013 Engthembekilethusi9No ratings yet
- QC F6 Chem Mock 201920 P2Document10 pagesQC F6 Chem Mock 201920 P2ckt.123lolNo ratings yet
- Prelims 2 XII ISC Dec 2023Document7 pagesPrelims 2 XII ISC Dec 2023Nicxon Jeryl SolomonNo ratings yet
- QP CH 491 Assignment 4 060217 For 26 Jul 2023Document6 pagesQP CH 491 Assignment 4 060217 For 26 Jul 2023Chisama SichoneNo ratings yet
- 2.ce3019 Exam Paper Questions 21-22 - v2Document17 pages2.ce3019 Exam Paper Questions 21-22 - v2suhel ahmadNo ratings yet
- Physical Sciences P2 Nov 2018 Eng PDFDocument19 pagesPhysical Sciences P2 Nov 2018 Eng PDFAmmaarah RabanNo ratings yet
- Rates Review Questions ANSWERSDocument3 pagesRates Review Questions ANSWERSnadia sykesNo ratings yet
- Ese Test 3Document8 pagesEse Test 3Sheelendra kumarNo ratings yet
- Gce A Level Marking Scheme: AUTUMN 2021Document15 pagesGce A Level Marking Scheme: AUTUMN 2021NonuNo ratings yet
- October 2021 MS - Component 3 Eduqas Chemistry A-LevelDocument15 pagesOctober 2021 MS - Component 3 Eduqas Chemistry A-LevelNonuNo ratings yet
- Physical Sciences P2 Memo May 2022Document8 pagesPhysical Sciences P2 Memo May 2022thabangaphanebusiness1No ratings yet
- 4 Skema Pemarkahan Rate of ReactionDocument47 pages4 Skema Pemarkahan Rate of ReactionJun QiangNo ratings yet
- Chem DSE 2022 2 - E MarkingDocument7 pagesChem DSE 2022 2 - E MarkingSpeaker StrongNo ratings yet
- Major Test 4 2021 FinalDocument8 pagesMajor Test 4 2021 FinalLevi JohnsonNo ratings yet
- MARK SCHEME For The November 2005 Question PaperDocument5 pagesMARK SCHEME For The November 2005 Question PaperKarim AbdellatifNo ratings yet
- Gce A Level Marking Scheme: SUMMER 2022Document17 pagesGce A Level Marking Scheme: SUMMER 2022NonuNo ratings yet
- June 2018 MS - Unit 1 (F) WJEC Biology GCSEDocument26 pagesJune 2018 MS - Unit 1 (F) WJEC Biology GCSEAsni Tunjung ArantikiNo ratings yet
- g11 PSC p2 Jun 2019 and MemoDocument20 pagesg11 PSC p2 Jun 2019 and MemoKholofelo SebakeNo ratings yet
- 12 Chemistry SQP 1Document8 pages12 Chemistry SQP 1Prashanth 070No ratings yet
- Chemistry Paper 2Document6 pagesChemistry Paper 2Ngai Ivan CHANNo ratings yet
- Physical Sciences P2 May-June 2022 EngDocument20 pagesPhysical Sciences P2 May-June 2022 EngSimphiwe MpanzaNo ratings yet
- Physical Sciences P2 2022Document32 pagesPhysical Sciences P2 2022sanindlovu27No ratings yet
- Gce A Level Marking SchemeDocument26 pagesGce A Level Marking SchemeNonuNo ratings yet
- Chem Set 2Document12 pagesChem Set 2ashishkumarberia016No ratings yet
- Weight Differs From Mass Due To : Page No 1 Open Book Model Exam OneDocument24 pagesWeight Differs From Mass Due To : Page No 1 Open Book Model Exam Onedmc constructionNo ratings yet
- 5721a223c7100bcebd631b2fd46cf5e5Document19 pages5721a223c7100bcebd631b2fd46cf5e5Deepender KashyapNo ratings yet
- Mock Che1Document6 pagesMock Che1mnyambo edsonNo ratings yet
- Chem Ms 2Document13 pagesChem Ms 2shirinNo ratings yet
- Physical Sciences p2 Nov 2019 MemoDocument11 pagesPhysical Sciences p2 Nov 2019 MemoMasimo SeforaNo ratings yet
- Final Chemistry SSC-I 2023Document5 pagesFinal Chemistry SSC-I 2023MuhammahRamzanNo ratings yet
- Chemistry 2022Document11 pagesChemistry 2022Dibyajyoti ChakrabortyNo ratings yet
- 2003 Paper 1 Marking SchemeDocument10 pages2003 Paper 1 Marking Schemeapi-3734333100% (1)
- Universiti Teknologi Mara Final Test: Confidential AS/JUL 2022/CHM421Document4 pagesUniversiti Teknologi Mara Final Test: Confidential AS/JUL 2022/CHM421Natasha AdreenaNo ratings yet
- CH4404 Process Technology 1Document6 pagesCH4404 Process Technology 1hannahNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument18 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- 2015 Dse Chem 2 1Document8 pages2015 Dse Chem 2 1Henry NgNo ratings yet
- 22-23 A2PRELIM JC2 Chemistry P4 MS FBPDocument6 pages22-23 A2PRELIM JC2 Chemistry P4 MS FBPluyanxin20062No ratings yet
- Physical Sciences P2 June-July 2015 EngDocument22 pagesPhysical Sciences P2 June-July 2015 EngLevi JohnsonNo ratings yet
- Electrical Technology Exemplar GR 12 2018 (Electronics) Memo EngDocument12 pagesElectrical Technology Exemplar GR 12 2018 (Electronics) Memo EngTauya MakandaNo ratings yet
- Physical Sciences P2 June-July 2015 EngDocument22 pagesPhysical Sciences P2 June-July 2015 EngLevi JohnsonNo ratings yet
- Classifying Spaces of Degenerating Polarized Hodge Structures. (AM-169)From EverandClassifying Spaces of Degenerating Polarized Hodge Structures. (AM-169)No ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument15 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P1 Memo 2015Document13 pagesP1 Memo 2015Bonga DubeNo ratings yet
- P1 Memo 2017Document11 pagesP1 Memo 2017Bonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument19 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument16 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P2 Memo 2019Document10 pagesP2 Memo 2019Bonga DubeNo ratings yet
- P2 Memo 2014Document15 pagesP2 Memo 2014Bonga DubeNo ratings yet
- P2 Memo 2012Document9 pagesP2 Memo 2012Bonga DubeNo ratings yet
- P1 Memo 2012Document12 pagesP1 Memo 2012Bonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument16 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- P2 Memo 2016Document9 pagesP2 Memo 2016Bonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument14 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P1 Memo 2013Document13 pagesP1 Memo 2013Bonga DubeNo ratings yet
- P1 Memo 2016Document12 pagesP1 Memo 2016Bonga DubeNo ratings yet
- P2 Memo 2015Document10 pagesP2 Memo 2015Bonga DubeNo ratings yet
- P1 Memo 2022Document12 pagesP1 Memo 2022Bonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument17 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument18 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P2 Memo 2010Document10 pagesP2 Memo 2010Bonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument14 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument16 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- Grade 11 Final Poetry BookletDocument76 pagesGrade 11 Final Poetry BookletBonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument14 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P1 Memo 2008Document10 pagesP1 Memo 2008Bonga DubeNo ratings yet
- P1 Memo 2010Document13 pagesP1 Memo 2010Bonga DubeNo ratings yet
- Openedge Abl Develop Soap ClientsDocument70 pagesOpenedge Abl Develop Soap ClientsLucas BicalhoNo ratings yet
- LDF4RKP-50A-136 Product SpecificationsDocument5 pagesLDF4RKP-50A-136 Product SpecificationsPhi FeiNo ratings yet
- Worksheet Momentum Force QuestionsDocument2 pagesWorksheet Momentum Force QuestionsBenjamin WatsonNo ratings yet
- Micro Teaching Lesson Plan For The Skill of Probing Question (Teach)Document3 pagesMicro Teaching Lesson Plan For The Skill of Probing Question (Teach)Riya Elizabeth J 13No ratings yet
- Geometry, Mechanics, and Dynamics: Dong Eui Chang Darryl D. Holm George Patrick Tudor Ratiu EditorsDocument506 pagesGeometry, Mechanics, and Dynamics: Dong Eui Chang Darryl D. Holm George Patrick Tudor Ratiu EditorsEmiRB 99No ratings yet
- F1-Chapter 1 KSSMDocument207 pagesF1-Chapter 1 KSSMCikgu NitaNo ratings yet
- Using and Interpreting CronbachDocument5 pagesUsing and Interpreting CronbachMay Angelica TenezaNo ratings yet
- Rockwool Slab SL930Document1 pageRockwool Slab SL930Lee Kok RoyNo ratings yet
- Unit 3Document21 pagesUnit 3Akash Varma JampanaNo ratings yet
- ADC Module 3 FinalDocument44 pagesADC Module 3 FinalAnkit KumarNo ratings yet
- Web Dynpro: Context Mapping and Model Binding: January 2006Document9 pagesWeb Dynpro: Context Mapping and Model Binding: January 2006Carlos SilvaNo ratings yet
- Conduit BendDocument11 pagesConduit Bendngyan100% (1)
- Measuring and Monitoring The Efficiency of Central Chiller PlantsDocument14 pagesMeasuring and Monitoring The Efficiency of Central Chiller PlantsAqilaAndiNo ratings yet
- Conveyor Belt Scale MaintenanceDocument33 pagesConveyor Belt Scale MaintenanceDSGNo ratings yet
- "Cascading Style Sheets" For Styling WWW Information: Mike PangburnDocument24 pages"Cascading Style Sheets" For Styling WWW Information: Mike PangburnRihab Ait AzzhraNo ratings yet
- NG300 ESales Spec SheetDocument2 pagesNG300 ESales Spec SheetSamrandNo ratings yet
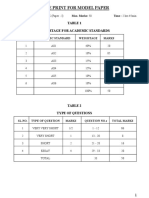
- Blue Print For Model Paper: Table 1 Weightage For Academic StandardsDocument5 pagesBlue Print For Model Paper: Table 1 Weightage For Academic StandardsKrishna VeniNo ratings yet
- bài tập vận dụng chap 1 2 m4bDocument12 pagesbài tập vận dụng chap 1 2 m4bconkienlua2005No ratings yet
- A Novel Kind of Concrete Superplasticizer Based On LigniteDocument8 pagesA Novel Kind of Concrete Superplasticizer Based On Lignitemoustafa hadj-doulaNo ratings yet
- Coding Interview Java (PART-2)Document207 pagesCoding Interview Java (PART-2)Ankush TiwariNo ratings yet
- Din 2458Document0 pagesDin 2458student_blNo ratings yet
- Homemade Jigsaw Cutting Station Free PLANS: Mazay DIY 3D Model Video TutorialDocument20 pagesHomemade Jigsaw Cutting Station Free PLANS: Mazay DIY 3D Model Video TutorialFranklin AvendañoNo ratings yet
- Permutations CombinationsDocument8 pagesPermutations CombinationsaryanmcywaliaNo ratings yet
- Radio Receiver: Malahit-Dsp1 Malahit-Dsp2 Malahit-Dsp3Document56 pagesRadio Receiver: Malahit-Dsp1 Malahit-Dsp2 Malahit-Dsp3ol ok100% (1)
- All PastPapersDocument320 pagesAll PastPapersDsquaredNo ratings yet
- Aplaya National High School - Annex: Quarter 1 Regino M - Adato Mathematics 9 Learning TasksDocument4 pagesAplaya National High School - Annex: Quarter 1 Regino M - Adato Mathematics 9 Learning TasksKRISTER ANN JIMENEZ50% (2)
- MFL37562519 English PDFDocument21 pagesMFL37562519 English PDFCărunta-Crista CristinaNo ratings yet
- Daiwa CS-401 RF Coaxial Switch SpecificationsDocument1 pageDaiwa CS-401 RF Coaxial Switch Specifications240GL guy100% (1)
- Innovations in Derivatives MarketsDocument446 pagesInnovations in Derivatives Marketsshrikantmsd100% (1)
P2 Memo 2013
P2 Memo 2013
Uploaded by
Bonga DubeCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
P2 Memo 2013
P2 Memo 2013
Uploaded by
Bonga DubeCopyright:
Available Formats
NATIONAL SENIOR CERTIFICATE EXAMINATION
NOVEMBER 2013
PHYSICAL SCIENCES: PAPER II
MARKING GUIDELINES
Time: 3 hours 200 marks
These marking guidelines are prepared for use by examiners and sub-examiners,
all of whom are required to attend a standardisation meeting to ensure that the
guidelines are consistently interpreted and applied in the marking of candidates'
scripts.
The IEB will not enter into any discussions or correspondence about any marking
guidelines. It is acknowledged that there may be different views about some
matters of emphasis or detail in the guidelines. It is also recognised that,
without the benefit of attendance at a standardisation meeting, there may be
different interpretations of the application of the marking guidelines.
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 2 of 10
QUESTION 1
1.1 B 1.6 B
1.2 D 1.7 A
1.3 D 1.8 C
1.4 D 1.9 D
1.5 A 1.10 C
[10 × 2 = 20]
QUESTION 2
2.1 2.1.1 Hypothesis – Catalyst A will decompose hydrogen peroxide faster than
Catalyst B.
(Any reasonable answer as a statement - There must be a relationship
between two variables )
Alternatives : “ Catalyst A will produce oxygen faster ( at a faster rate)
than catalyst B”
2.1.2 Investigative question – which of the two catalysts, A or B, will
decompose hydrogen peroxide faster?
(Any reasonable answer as a question) (4)
1 mark for it in question format
1 mark for correct variables used in the question
2.2 The variable that is changed in a controlled way during the experiment by the
experimenter. (2)
Alternative : A manipulated variable which does not depend on the outcome
variable
DO NOT ACCEPT – variable that is controlled in the experiment
2.3 This is the variable that is being measured in the experiment. (2)
Can accept : volume of oxygen produced is dependent on the efficiency of the
catalyst
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 3 of 10
2.4
Graphs to show relationship between
volume of 02 produced and time for
Catalysts A and B.
Catalyst A
VOLUME OF 02 (dm3)
Catalyst B
Description Mark allocated
Heading Full description between at least two
variables (1)
Axes labelled with units (1)
Scale correct on both axes (Half page) (1)
Points plotted correctly All points plotted correctly for both (2)
Points incorrect (1 – 2) (1)
More than 2 points incorrect (0)
Line of best fit Correct smooth shape for both graphs (2)
Do not accept “dot to dot”
Must check that line of best fit for catalyst
A joins the last two points or -1
(7)
If axes are swapped around – max 4 out of 7
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 4 of 10
2.5 Rate = gradient
volume
=
time
1, 25 0
=
1,5 0
Rate = 0,83 dm3·min–1 (3)
2.6 4,2 dm3
Same number of moles (amount) of H2O2 (g) being converted to O2 (g) – only
slower. (3)
Alternative – same amount of reactant used in both experiments
[21]
QUESTION 3
3.1 3.1.1 These are carbon based side branches attached to the main carbon
chain. (hydrocarbon side branches obeying the general formula (CnH2n+1)) (2)
3.1.2 Organic compounds consisting of C and H atoms only and have only single
bonds to the C atoms in the chain. (All four bonds to the C in chain
singularly occupied) (2)
CH3
3.2 3.2.1 CH3 CHC(CH3) CH2 CH3 (2)
CH3 – CH =C – CH2 – CH3
3.2.3 H Cℓ Cℓ H
| | | |
H–C – C – C – C–H 1 mark for Cl in correct position
| | | |
H H H H 1 mark for correct chain and structure (2)
3.3 3.3.1 B If B with another letter …… 1 mark only (2)
3.3.2 D and G (2)
If other letters used , mark only the first two
3.3.3 A, E and F (any two) (2)
3.3.4 Haloalkanes (alkyl halides) halogenoalkanes (1)
3.3.5 CH3CH2CH2CH2CH3 Pentane
CH3C(CH3)2CH3 2,2 – dimethylpropane (4)
3.3.6 E – Has – OH which causes hydrogen bonding intermolecular forces
between molecules
A – Is polar causing dipole – dipole intermolecular forces between
molecules which are not as strong as hydrogen bonds. (4)
1 mark : E has H- bonding intermolecular forces
1 mark : A has dipole-dipole/van der Waals/Keesom IM forces
1 mark : stating that H-bonding stronger than dipole-dipole
1 mark : more energy needed to overcome IM forces between
molecules E
[23]
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 5 of 10
QUESTION 4
4.1 4.1.1 I Substitution bromination halogenation
II Elimination dehydration
III Esterification/Elimination
IV Combustion (4)
4.1.2 (a) Propyl ethanoate (2)
(b) Ethanoic acid (2)
(c) Acting as a dehydrating agent the concentrated form must be used to
allow rapid dehydration( must have no water present to be more
effective) to produce the ester, thus speeding up the reaction giving
it catalytic properties as well. If dilute , water already present thus
will not be as effective and reduce the rate at which reaction
takes place (4)
(d) Pentanoic acid (2)
4.2 4.2.1 This is a single (small) molecular unit that is repeated throughout the
polymer structure (2)
4.2.2 Addition (1)
4.2.3 Initiation
Propagation
Termination (3)
4.2.4
(3)
4.2.5 Advantage – strong and durable, thus can last over long periods of time.
- Can be recycled
- unreactive with oxygen and water
Disadvantage – will not decompose naturally, thus a pollutant.
- weakened by UV rays (2)
4.2.6 1. Will clog landfill sites
2. Unsightly
3. Release hazardous gases into environment if burnt (CO2 and CO)
Any two (4)
- danger to animals
- unsightly litter
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 6 of 10
QUESTION 5
5.1 In an isolated (closed) chemical system the rate of the forward reaction will equal the
rate of the reverse reaction. (3)
Alternative : forward and reverse reaction continue to take place at equal
rates
5.2
H2 I2
m m
n= n=
M M
3,5 635
= =
2 254
= 1,75 mol = 2,5 mol
(4)
5.3 2,5 – 1,45 carry over error
= 1,05 mol (2)
5.4 5.4.1 H2 : I2
1:1 can use H2 : HI ratio of 1 : 2 to get 1,05mol
1,05 : 1,05
n = 1,05 mol of H2 used (2)
5.4.2 H2 left : 1,75 – 1,05 carry over error
= 0,7 mol (2)
5.5
H2 I2 HI
n n n
c= carry over error c = c=
v v v
0, 7 1, 45 2,1
= = =
1,5 1,5 1,5
= 0,466 mol·dm–3 = 0,966 mol·dm–3 = 1,4 mol·dm–3
(4)
HI carry over error if K
2
5.6 Kc = used correctly with incorrect values from above
H 2 I2
c
1, 4
2
=
0, 466 0,966
Kc = 4,35 (3)
5.7 Increase in temperature will cause forward reaction to be favoured.
Will increase amount of HI and decrease the amount of H2 and I2.
Kc will thus increase. (3)
Alternative : Increase in temp will cause endothermic reaction to be favoured
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 7 of 10
5.8 Increasing volume will decrease pressure in the system.
Total molar volumes of both reactant and product the same.
Change in pressure will thus have no effect on equilibrium hence statement is
inaccurate. (3)
Alternative : rate of both forward and reverse reactions will decrease
rate of reverse reaction will decrease but rate of forward reaction
will decrease equally
[26]
QUESTION 6
6.1 6.1.1 Temperature 25 °C 298 K
Concentration of electrolyte = 1 mol·dm–3
Pressure of H2 gas = 1 atmosphere (101,3 kPa) ( 1,013 x 105 Pa) (3)
6.1.2 It is used as the reference electrode to which all other half cell potentials are
compared. (2)
6.1.3 (a) Platinum
It is an inert (unreactive) metal and will not become
electrochemically involved in the reaction. (2)
(b) Fe/Fe2+ has E = –0,44 V
Meaning Fe is a stronger reducing agent than ( Fe has a more negative
reduction potential) H2 thus hydrogen electrode will be the cathode (3)
6.1.4 Anode – oxidation of metal electrode increases cation concentration.
Cathode – reduction of cations in electrolyte reduces cation
concentration.
Salt bridge will act as an 'ion pump' by moving anions and cations from salt
bridge into half cells to balance out ionic charges. (3)
[13]
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 8 of 10
QUESTION 7
7.1 Alumina (1)
7.2 Lowers melting point. lowers the working temperature of the cell
Less energy needs to be consumed.
Less demand on power grid means less coal burned (less CO2/SO2 emissions) (3)
7.3 Cathode : Aℓ3+ + 3e- Aℓ - 1 mark if double arrows used (2)
7.4 O2 reacts with the carbon electrodes to form CO2. (2)
Accept : C + O2 CO2
7.5 7.5.1 Energy = 17 000 × 3 600 000
6,12 1010 J
=
1 000 000
= 61 200 MJ (3)
m
7.5.2 (a) n= - 1 mark for no conversion to grams
MR
225 000
=
12
= 18 750 mol of C (3)
(b) 2Aℓ2O3 + 3C 4Aℓ + 3CO2
C : Al
3:4
Alternative
18 750 : n C : Al
n = 25 000 mol. 3 : 4
36g.mol‐1 : 108g.mol‐1
m = n.MR 225 000 : x
= 25 000 × 27
= 6,75 × 105 g X = 6,75 x 105 g
6,75 105 X = 0,675 tonnes
1 000 1 000
M = 0,675 tonnes of Al (4)
7.6 (i) As bauxite is imported, ports offer easy transport to smelter.
(ii) Aluminium is exported – again ports offer easy transport to ships.
(lower/minimal transport costs) (2)
Accept : reduction of pollution due to minimal transport
IEB Copyright © 2013 PLEASE TURN OVER
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 9 of 10
QUESTION 8
8.1 Primary cell – non rechargeable
Secondary cell – rechargeable (2)
8.2 Cd will act as anode – has a more negative E value hence Cd is a better reducing
agent and will favour oxidation.
NiO(OH) will act as cathode – has more positive E value, hence NiO(OH) is a better
oxidising agent – favour reduction. (4)
1 each for correct identification of cathode and anode
2 marks for correct reasoning on one or the other
8.3 Cd + 2NiO(OH) + 2H2O Cd(OH)2 + 2Ni(OH)2 -1 for any double arrows
(–1 for any error) (2)
8.4 Ni – Cd cell converts chemical energy to electrical energy. (2)
Accept : cell has a positive emf
8.5 8.5.1 The total amountof charge that a cell is able to deliver. (2)
Alternative : the ability of a battery to deliver a specific amount of current
(electricity)over a specific amount of time
W
8.5.2 V=
Q
W
Q=
V
256 250
=
1, 25
= 205 000 C
Q = It
Q
I=
t
205 000
=
100 800
I = 2,03A (4)
8.6 1. Made from toxic substances (heavy metals).
2. Expensive to produce thus to sell.
(Any two reasonable answers) (2)
Accept : can explode
Loose capacity fast
Relatively low cell capacity
Poor “memory”
[18]
IEB Copyright © 2013
NATIONAL SENIOR CERTIFICATE: PHYSICAL SCIENCES: PAPER II – MARKING GUIDELINES Page 10 of 10
QUESTION 9
9.1 Brine (1)
9.2 Asbestos (1)
9.3 Electrical energy to chemical energy. (2)
9.4 9.4.1 2Cℓ– Cℓ2 + 2e– -1 for double arrows over whole question (2)
9.4.2 2H2O + 2e– H2 + 2OH– (2)
9.4.3 2Cℓ– + 2H2O Cℓ2 + H2 + 2OH– (2)
2NaCl + 2H2O Cl2 + H2 + 2NaOH
9.5 Creates a pressure gradient which allows Na+ ions and unreacted Cℓ– ions to migrate
into cathode compartment. (3)
Prevents OH- ions from flowing back into the anode compartment
9.6 Na+ is a much weaker oxidising agent than H2O. (2)
H2O is a stronger oxidising agent than Na+
9.7 9.7.1 Fluoro polymer – is ion selective thus will only allow Na+ ions to migrate
into cathode compartment.
Asbestos – non-ion selective (2)
9.7.2 Asbestos is toxic to humans causing disease (asbestosis).
●
Asbestos is carcinogenic causing cancer. (4)
(accept answers which may come from the Table used in Q9.8)
9.8 Membrane cell
Highest purity of NaOH produced.
Least amount of H2O used. any two
Less energy consumed. (3)
V
9.9 n=
Vo
11 000
=
22, 4
= 491,1 mol of Cℓ2
2Cℓ– Cℓ2 + 2e–
1:2
ne– = 491,1 × 2
= 982,2 mol of e–
Amount of change for 1 mol e– = 96 500 C•mol–1
Q = 96 500 × 982,2 OR
= 94 782 300 C
Q = 982,2 × 6,02 × 1023 × 1,6 × 10–19
Q = It Q = 94 605 504 C
Q Q
I = I=
t t
94 782 300 94 605 504
= =
63 000 63 000
I = 1 504,39 A I = 1 501,76 A (6)
[30]
Total: 200 marks
IEB Copyright © 2013
You might also like
- Mark Scheme: Sample Assessment Material 2018Document17 pagesMark Scheme: Sample Assessment Material 2018謝利米No ratings yet
- Unofficial Mark SchemeDocument6 pagesUnofficial Mark SchemeRodolph SmithNo ratings yet
- Transmission Line PresentationDocument55 pagesTransmission Line PresentationAshwani Dogra89% (9)
- P2 Memo 2012Document9 pagesP2 Memo 2012Bonga DubeNo ratings yet
- PHY SC GR 12 June 2019 P2 and MemoDocument20 pagesPHY SC GR 12 June 2019 P2 and Memoamahlezitha91No ratings yet
- P2 Memo 2011Document8 pagesP2 Memo 2011Bonga DubeNo ratings yet
- Gce A Level Marking Scheme: AUTUMN 2021Document24 pagesGce A Level Marking Scheme: AUTUMN 2021NonuNo ratings yet
- Chemistry Answers PDFDocument126 pagesChemistry Answers PDFNurafiqah FarhaniNo ratings yet
- P2 Memo 2017Document12 pagesP2 Memo 2017Bonga DubeNo ratings yet
- Chem D SolvedDocument18 pagesChem D SolvedVkrt SharmaNo ratings yet
- Wjec PaperDocument16 pagesWjec PaperNonuNo ratings yet
- P2 Memo 2015Document10 pagesP2 Memo 2015Bonga DubeNo ratings yet
- SHCC - 2023 - Chem Paper2 - AnnaDocument8 pagesSHCC - 2023 - Chem Paper2 - AnnaOof GucciNo ratings yet
- Chemistry XII Practice Paper BOOK 1Document95 pagesChemistry XII Practice Paper BOOK 1hembrampriyanka07No ratings yet
- Chem B SolvedDocument17 pagesChem B SolvedVkrt SharmaNo ratings yet
- Physical Sciences P2 Nov 2012 EngDocument18 pagesPhysical Sciences P2 Nov 2012 Engthembekilethusi9No ratings yet
- Physical Sciences P2 Sep 2017 Memo GR 10 (14.9.2017)Document10 pagesPhysical Sciences P2 Sep 2017 Memo GR 10 (14.9.2017)toygm672No ratings yet
- s18 Chemistry - 2410u10 1 Wjec As Chemistry Un1 MsDocument14 pagess18 Chemistry - 2410u10 1 Wjec As Chemistry Un1 MsAngelika LauNo ratings yet
- National Senior CertificateDocument11 pagesNational Senior CertificateVassiliNo ratings yet
- Physical Sciences P2 Feb-March 2013 EngDocument19 pagesPhysical Sciences P2 Feb-March 2013 Engthembekilethusi9No ratings yet
- QC F6 Chem Mock 201920 P2Document10 pagesQC F6 Chem Mock 201920 P2ckt.123lolNo ratings yet
- Prelims 2 XII ISC Dec 2023Document7 pagesPrelims 2 XII ISC Dec 2023Nicxon Jeryl SolomonNo ratings yet
- QP CH 491 Assignment 4 060217 For 26 Jul 2023Document6 pagesQP CH 491 Assignment 4 060217 For 26 Jul 2023Chisama SichoneNo ratings yet
- 2.ce3019 Exam Paper Questions 21-22 - v2Document17 pages2.ce3019 Exam Paper Questions 21-22 - v2suhel ahmadNo ratings yet
- Physical Sciences P2 Nov 2018 Eng PDFDocument19 pagesPhysical Sciences P2 Nov 2018 Eng PDFAmmaarah RabanNo ratings yet
- Rates Review Questions ANSWERSDocument3 pagesRates Review Questions ANSWERSnadia sykesNo ratings yet
- Ese Test 3Document8 pagesEse Test 3Sheelendra kumarNo ratings yet
- Gce A Level Marking Scheme: AUTUMN 2021Document15 pagesGce A Level Marking Scheme: AUTUMN 2021NonuNo ratings yet
- October 2021 MS - Component 3 Eduqas Chemistry A-LevelDocument15 pagesOctober 2021 MS - Component 3 Eduqas Chemistry A-LevelNonuNo ratings yet
- Physical Sciences P2 Memo May 2022Document8 pagesPhysical Sciences P2 Memo May 2022thabangaphanebusiness1No ratings yet
- 4 Skema Pemarkahan Rate of ReactionDocument47 pages4 Skema Pemarkahan Rate of ReactionJun QiangNo ratings yet
- Chem DSE 2022 2 - E MarkingDocument7 pagesChem DSE 2022 2 - E MarkingSpeaker StrongNo ratings yet
- Major Test 4 2021 FinalDocument8 pagesMajor Test 4 2021 FinalLevi JohnsonNo ratings yet
- MARK SCHEME For The November 2005 Question PaperDocument5 pagesMARK SCHEME For The November 2005 Question PaperKarim AbdellatifNo ratings yet
- Gce A Level Marking Scheme: SUMMER 2022Document17 pagesGce A Level Marking Scheme: SUMMER 2022NonuNo ratings yet
- June 2018 MS - Unit 1 (F) WJEC Biology GCSEDocument26 pagesJune 2018 MS - Unit 1 (F) WJEC Biology GCSEAsni Tunjung ArantikiNo ratings yet
- g11 PSC p2 Jun 2019 and MemoDocument20 pagesg11 PSC p2 Jun 2019 and MemoKholofelo SebakeNo ratings yet
- 12 Chemistry SQP 1Document8 pages12 Chemistry SQP 1Prashanth 070No ratings yet
- Chemistry Paper 2Document6 pagesChemistry Paper 2Ngai Ivan CHANNo ratings yet
- Physical Sciences P2 May-June 2022 EngDocument20 pagesPhysical Sciences P2 May-June 2022 EngSimphiwe MpanzaNo ratings yet
- Physical Sciences P2 2022Document32 pagesPhysical Sciences P2 2022sanindlovu27No ratings yet
- Gce A Level Marking SchemeDocument26 pagesGce A Level Marking SchemeNonuNo ratings yet
- Chem Set 2Document12 pagesChem Set 2ashishkumarberia016No ratings yet
- Weight Differs From Mass Due To : Page No 1 Open Book Model Exam OneDocument24 pagesWeight Differs From Mass Due To : Page No 1 Open Book Model Exam Onedmc constructionNo ratings yet
- 5721a223c7100bcebd631b2fd46cf5e5Document19 pages5721a223c7100bcebd631b2fd46cf5e5Deepender KashyapNo ratings yet
- Mock Che1Document6 pagesMock Che1mnyambo edsonNo ratings yet
- Chem Ms 2Document13 pagesChem Ms 2shirinNo ratings yet
- Physical Sciences p2 Nov 2019 MemoDocument11 pagesPhysical Sciences p2 Nov 2019 MemoMasimo SeforaNo ratings yet
- Final Chemistry SSC-I 2023Document5 pagesFinal Chemistry SSC-I 2023MuhammahRamzanNo ratings yet
- Chemistry 2022Document11 pagesChemistry 2022Dibyajyoti ChakrabortyNo ratings yet
- 2003 Paper 1 Marking SchemeDocument10 pages2003 Paper 1 Marking Schemeapi-3734333100% (1)
- Universiti Teknologi Mara Final Test: Confidential AS/JUL 2022/CHM421Document4 pagesUniversiti Teknologi Mara Final Test: Confidential AS/JUL 2022/CHM421Natasha AdreenaNo ratings yet
- CH4404 Process Technology 1Document6 pagesCH4404 Process Technology 1hannahNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument18 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- 2015 Dse Chem 2 1Document8 pages2015 Dse Chem 2 1Henry NgNo ratings yet
- 22-23 A2PRELIM JC2 Chemistry P4 MS FBPDocument6 pages22-23 A2PRELIM JC2 Chemistry P4 MS FBPluyanxin20062No ratings yet
- Physical Sciences P2 June-July 2015 EngDocument22 pagesPhysical Sciences P2 June-July 2015 EngLevi JohnsonNo ratings yet
- Electrical Technology Exemplar GR 12 2018 (Electronics) Memo EngDocument12 pagesElectrical Technology Exemplar GR 12 2018 (Electronics) Memo EngTauya MakandaNo ratings yet
- Physical Sciences P2 June-July 2015 EngDocument22 pagesPhysical Sciences P2 June-July 2015 EngLevi JohnsonNo ratings yet
- Classifying Spaces of Degenerating Polarized Hodge Structures. (AM-169)From EverandClassifying Spaces of Degenerating Polarized Hodge Structures. (AM-169)No ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument15 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P1 Memo 2015Document13 pagesP1 Memo 2015Bonga DubeNo ratings yet
- P1 Memo 2017Document11 pagesP1 Memo 2017Bonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument19 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument16 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P2 Memo 2019Document10 pagesP2 Memo 2019Bonga DubeNo ratings yet
- P2 Memo 2014Document15 pagesP2 Memo 2014Bonga DubeNo ratings yet
- P2 Memo 2012Document9 pagesP2 Memo 2012Bonga DubeNo ratings yet
- P1 Memo 2012Document12 pagesP1 Memo 2012Bonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument16 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- P2 Memo 2016Document9 pagesP2 Memo 2016Bonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument14 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P1 Memo 2013Document13 pagesP1 Memo 2013Bonga DubeNo ratings yet
- P1 Memo 2016Document12 pagesP1 Memo 2016Bonga DubeNo ratings yet
- P2 Memo 2015Document10 pagesP2 Memo 2015Bonga DubeNo ratings yet
- P1 Memo 2022Document12 pagesP1 Memo 2022Bonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument17 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument18 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P2 Memo 2010Document10 pagesP2 Memo 2010Bonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument14 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- Physical Sciences: Paper I: Please Turn OverDocument16 pagesPhysical Sciences: Paper I: Please Turn OverBonga DubeNo ratings yet
- Grade 11 Final Poetry BookletDocument76 pagesGrade 11 Final Poetry BookletBonga DubeNo ratings yet
- Physical Sciences: Paper Ii: Please Turn OverDocument14 pagesPhysical Sciences: Paper Ii: Please Turn OverBonga DubeNo ratings yet
- P1 Memo 2008Document10 pagesP1 Memo 2008Bonga DubeNo ratings yet
- P1 Memo 2010Document13 pagesP1 Memo 2010Bonga DubeNo ratings yet
- Openedge Abl Develop Soap ClientsDocument70 pagesOpenedge Abl Develop Soap ClientsLucas BicalhoNo ratings yet
- LDF4RKP-50A-136 Product SpecificationsDocument5 pagesLDF4RKP-50A-136 Product SpecificationsPhi FeiNo ratings yet
- Worksheet Momentum Force QuestionsDocument2 pagesWorksheet Momentum Force QuestionsBenjamin WatsonNo ratings yet
- Micro Teaching Lesson Plan For The Skill of Probing Question (Teach)Document3 pagesMicro Teaching Lesson Plan For The Skill of Probing Question (Teach)Riya Elizabeth J 13No ratings yet
- Geometry, Mechanics, and Dynamics: Dong Eui Chang Darryl D. Holm George Patrick Tudor Ratiu EditorsDocument506 pagesGeometry, Mechanics, and Dynamics: Dong Eui Chang Darryl D. Holm George Patrick Tudor Ratiu EditorsEmiRB 99No ratings yet
- F1-Chapter 1 KSSMDocument207 pagesF1-Chapter 1 KSSMCikgu NitaNo ratings yet
- Using and Interpreting CronbachDocument5 pagesUsing and Interpreting CronbachMay Angelica TenezaNo ratings yet
- Rockwool Slab SL930Document1 pageRockwool Slab SL930Lee Kok RoyNo ratings yet
- Unit 3Document21 pagesUnit 3Akash Varma JampanaNo ratings yet
- ADC Module 3 FinalDocument44 pagesADC Module 3 FinalAnkit KumarNo ratings yet
- Web Dynpro: Context Mapping and Model Binding: January 2006Document9 pagesWeb Dynpro: Context Mapping and Model Binding: January 2006Carlos SilvaNo ratings yet
- Conduit BendDocument11 pagesConduit Bendngyan100% (1)
- Measuring and Monitoring The Efficiency of Central Chiller PlantsDocument14 pagesMeasuring and Monitoring The Efficiency of Central Chiller PlantsAqilaAndiNo ratings yet
- Conveyor Belt Scale MaintenanceDocument33 pagesConveyor Belt Scale MaintenanceDSGNo ratings yet
- "Cascading Style Sheets" For Styling WWW Information: Mike PangburnDocument24 pages"Cascading Style Sheets" For Styling WWW Information: Mike PangburnRihab Ait AzzhraNo ratings yet
- NG300 ESales Spec SheetDocument2 pagesNG300 ESales Spec SheetSamrandNo ratings yet
- Blue Print For Model Paper: Table 1 Weightage For Academic StandardsDocument5 pagesBlue Print For Model Paper: Table 1 Weightage For Academic StandardsKrishna VeniNo ratings yet
- bài tập vận dụng chap 1 2 m4bDocument12 pagesbài tập vận dụng chap 1 2 m4bconkienlua2005No ratings yet
- A Novel Kind of Concrete Superplasticizer Based On LigniteDocument8 pagesA Novel Kind of Concrete Superplasticizer Based On Lignitemoustafa hadj-doulaNo ratings yet
- Coding Interview Java (PART-2)Document207 pagesCoding Interview Java (PART-2)Ankush TiwariNo ratings yet
- Din 2458Document0 pagesDin 2458student_blNo ratings yet
- Homemade Jigsaw Cutting Station Free PLANS: Mazay DIY 3D Model Video TutorialDocument20 pagesHomemade Jigsaw Cutting Station Free PLANS: Mazay DIY 3D Model Video TutorialFranklin AvendañoNo ratings yet
- Permutations CombinationsDocument8 pagesPermutations CombinationsaryanmcywaliaNo ratings yet
- Radio Receiver: Malahit-Dsp1 Malahit-Dsp2 Malahit-Dsp3Document56 pagesRadio Receiver: Malahit-Dsp1 Malahit-Dsp2 Malahit-Dsp3ol ok100% (1)
- All PastPapersDocument320 pagesAll PastPapersDsquaredNo ratings yet
- Aplaya National High School - Annex: Quarter 1 Regino M - Adato Mathematics 9 Learning TasksDocument4 pagesAplaya National High School - Annex: Quarter 1 Regino M - Adato Mathematics 9 Learning TasksKRISTER ANN JIMENEZ50% (2)
- MFL37562519 English PDFDocument21 pagesMFL37562519 English PDFCărunta-Crista CristinaNo ratings yet
- Daiwa CS-401 RF Coaxial Switch SpecificationsDocument1 pageDaiwa CS-401 RF Coaxial Switch Specifications240GL guy100% (1)
- Innovations in Derivatives MarketsDocument446 pagesInnovations in Derivatives Marketsshrikantmsd100% (1)