Professional Documents
Culture Documents
S 017 Azac
S 017 Azac
Uploaded by
rendra boyke2023Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
S 017 Azac
S 017 Azac
Uploaded by
rendra boyke2023Copyright:
Available Formats
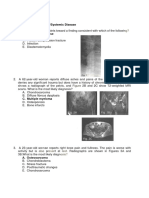
AZACITIDINE
CAS No. 320-67-2
First Listed in the Eighth Report on Carcinogens
NH2
N N
O N
HO
H H
H H
OH OH
CARCINOGENICITY
Azacitidine (5-Azacytidine; 5-AzaC) is reasonably anticipated to be a human carcinogen
based on evidence of malignant tumor formation at multiple tissue sites in multiple species of
experimental animals (NCI 1978, Luz and Murray 1988, IARC 1990).
5-AzaC, when administered by intraperitoneal (i.p.) injection, induced lymphoreticular
neoplasms and skin and lung tumors in male and/or female mice (NCI 1978, Luz and Murray
1988, IARC 1990), and leukemia, lymphoma, and tumors of the liver and lung in offspring of
treated pregnant dams (IARC 1990). In male rats, 5-AzaC administered i.p. induced squamous
cell carcinoma of the skin and interstitial-cell tumors of the testes, and appeared to increase the
incidence of non-testis tumors in male offspring of treated dams (IARC 1990).
No data were available to evaluate the carcinogenicity of 5-AzaC in humans (IARC
1990).
ADDITIONAL INFORMATION RELEVANT TO CARCINOGENESIS OR POSSIBLE
MECHANISMS OF CARCINOGENESIS
Using an initiation-promotion experimental design, chronic i.p. treatment of male rats,
previously acutely administered N-nitrosodiethylamine (DEN) and partially hepatectomized, with
5-AzaC resulted in a synergistic increase in the frequency of liver, lung, and skin tumors (Carr et
al. 1988, IARC 1990).
The carcinogenic/enhancement activity of 5-AzaC has been postulated to result directly or
indirectly from its ability to inhibit DNA methylation (Harrison et al. 1983, Kerbel et al. 1984,
Kerbel et al. 1986, Takenaga 1986, Glover et al. 1987, Glover and Leyland-Jones 1987, Jones
and Buckley 1990, Haaf 1995). Altered levels of DNA methylation can affect gene expression
(Cedar 1988, IARC 1990, Fajkus et al. 1992, Velge et al. 1995), with hypomethylation being
associated with the expression of genes that are normally silent or downregulated (Jones et al.
1983, Nyce et al. 1983, Riggs and Jones 1983, Collard et al. 1989, Jones and Buckley 1990,
Pascale et al. 1993). In addition, 5-AzaC in the absence of metabolic activation is positive in a
wide variety of prokaryotic, lower eukaryotic, and mammalian in vitro test systems, inducing
REASONABLY ANTICIPATED TO BE A HUMAN CARCINOGEN TENTH REPORT ON CARCINOGENS
Azacitidine (Continued)
DNA damage, mutations (base-pair substitution mutations only) in prokaryote systems; mitotic
recombination, gene conversion, and gene mutations in somatic and germ cells of lower
eukaryotes (yeast, Drosophila, plants), and DNA damage, chromosomal aberrations, mutations
(but not point), and morphological transformation in cultured mammalian cells. Studies to
evaluate the genetic activity of 5-AzaC in somatic cells of mammals have not been reported;
however, it was reported as negative for dominant lethal mutations in mice (IARC 1990).
No data were available to suggest that the mechanisms thought to account for tumor
induction by 5-AzaC in experimental animals would not also operate in humans.
PROPERTIES
5-AzaC is a white crystalline powder with a melting point of 228 to 230oC. It is soluble
in water, ethanol, acetone, chloroform, hexane, and dimethyl sulfoxide. 5-AzaC is very unstable
in aqueous media, with rapid degradation to complex products occurring within hours of
dissolution in intravenous solutions at room temperature. The chemical may discolor upon
exposure to light and is probably combustible (NTP 2001).
USE
5-AzaC is an antineoplastic agent that has been used as an investigational drug since the
l970s for the treatment of patients with acute myeloblastic anemia, acute lymphoblastic leukemia,
and myelodysplastic syndromes (IARC 1990, NTP 2001, Santini et al. 2001, Pharmion 2001). 5-
AzaC has been used clinically in cancer treatment trial protocols in combination with other
antineoplastic agents such as vincristine, prednisone, vinblastine, cytarabine, or amsacrine (IARC
1990). 5-AzaC (alone or in combination with phenylbutyrate or amifostine) is currently being
tested in at least seven clinical trials for the following conditions: beta thalassemia, acute myeloid
leukemia, myelodysplastic syndrome, advanced or metastatic solid tumors, non-Hodgkins
lymphoma, multiple myeloma, non-small cell lung cancer, and prostate cancer
(ClinicalTrials.gov 2001).
PRODUCTION
5-AzaC is synthesized in Germany. It can be prepared by synthetic methods or can be
isolated from a culture of the bacterium Streptoverticillium ladakanus (IARC 1990). No data on
imports or exports of 5-AzaC were available. Chem Sources (2001) listed 11 U.S. suppliers of 5-
AzaC.
EXPOSURE
The primary routes of potential human exposure to 5-AzaC are intravenous and
intramuscular injection and intravenous infusion at daily doses of 40 to 750 mg/m2 (IARC 1990).
Occupational exposure may occur for workers formulating or packaging the solutions and for
health care professionals administering the drug. The National Occupational Exposure Survey
(1981-1983) indicated that 1,069 workers, including 699 women, potentially were exposed to 5-
AzaC (NIOSH 1990). This estimate was derived from total observations of the use of the actual
compound (48%) and trade name products (52%).
REASONABLY ANTICIPATED TO BE A HUMAN CARCINOGEN TENTH REPORT ON CARCINOGENS
Azacitidine (Continued)
REGULATIONS
According to a monograph in the 1996 Handbook on Injectable Drugs (HID) entitled
“Azacitidine Investigational” (Trissel 1996), azacitidine is still an investigational drug. Its
investigational number is NSC-102816. No new information regarding drug status was found.
OSHA regulates azacitidine under the Hazard Communication Standard and as a
chemical hazard in laboratories. Regulations are summarized in Volume II, Table 17.
REFERENCES
Carr, B.I., S. Rahbar, Y. Asmeron, A. Riggs, and C.D. Winberg. Carcinogenicity and
Haemoglobin Synthesis Induction by Cytidine Analogues. Br. J. Cancer Vol. 57, 1988, pp. 395-
402.
Cedar, H. DNA Methylation and Gene Activity. Cell Vol. 53, 1988, pp. 3-4.
Chem Sources. Chemical Sources International, Inc. http://www.chemsources.com, 2001.
ClinicalTrials.gov. A service of the National Institutes of Health. Developed by the National
Library of Medicine, 2001 (http://www.clinicaltrials.gov and search "azacytidine").
Collard, J.G., G.G.M. Habets, R. van der Kammen, and E. Scholtes. Genetic Basis of T-
Lymphoma Invasion. Invasion Metastasis Vol. 9, 1989, pp. 379-390.
Fajkus, J., B. Vyskot, and M. Bezdik. Changes in Chromatin Structure Due to Hypomethylation
Induced with 5-Azacytidine or DL-Ethionine. FEBS Vol. 314, No. 1, 1992, pp. 13-16.
Glover, A.B., and B. Leyland-Jones. Biochemistry of Azacytidine: A Review. Cancer Treat.
Rep. Vol. 71, No. 10, 1987, pp. 959-964.
Glover, A.B., B.R. Leyland-Jones, H.G. Chun, B. Davies, and D.F. Hoth. Azacytidine: 10 Years
Later. Cancer Treat. Rep. Vol. 71, No. 7/8, 1987, pp. 737-746.
Haaf, T. The Effects of 5-Azacytidine and 5-Azadeoxycytidine on Chromosome Structure and
Function: Implications for Methylation-Associated Cellular Processes. Pharmacol. Ther. Vol.
65, 1995, pp. 19-46.
Harrison, J.J., A. Anisowicz, I.K. Gadi, M. Raffeld, and R. Sager. Azacytidine-Induced
Tumorigenesis of CHEF/18 Cells: Correlated DNA Methylation and Chromosome Changes.
Proc. Natl. Acad. Sci. Vol. 80, 1983, pp. 6606-6610.
IARC. International Agency for Research on Cancer. IARC Monographs on the Carcinogenic
Risk of Chemicals to Humans. Pharmaceutical Drugs. Vol. 50. 415 pp. Lyon, France: IARC,
1990.
Jones, P.A., and J.D. Buckley. The Role of DNA Methylation in Cancer. Adv. Cancer Res. Vol.
54, 1990, pp. 1-23.
Jones, P.A., S.M. Taylor, and V. Wilson. DNA Modification, Differentiation, and
Transformation. J. Exp. Zool. Vol. 228, 1983, pp. 287-295.
REASONABLY ANTICIPATED TO BE A HUMAN CARCINOGEN TENTH REPORT ON CARCINOGENS
Azacitidine (Continued)
Kerbel, R.S., P. Frost, R. Liteplo, D.A. Carlow, and B.E. Elliot. Possible Epigenetic Mechanism
of Tumor Progression: Induction of High-Frequency Heritable but Phenotypically Unstable
Changes in the Tumorigenic and Metastatic Properties of Tumor Cell Populations by 5-
Azacytidine Treatment. J. Cell. Physiol. Suppl. 3, 1984, pp. 87-97.
Kerbel, R.S., R. Liteplo, and P. Frost. On the Possible Contribution of DNA Hypomethylation to
the Induction of High Frequency and Heritable Drug-Induced Alterations in the Malignant
Phenotype. Prog. Clin. Biol. Res. Vol. 212, 1986, pp. 292-303.
Luz, A., and A.B. Murray. Sudden Outbreak of a Leukemia-Like Lesion in Female CBA Mice
After Repeated Injections of 5-Azacytidine. J. Cancer Res. Clin. Oncol. Vol. 114, 1988, pp. 525-
527.
NCI. National Cancer Institute. Carcinogenesis, Technical Report Series No. 42. Bioassay of 5-
Azacytidine for Possible Carcinogenicity (CAS No. 320-67-2). DHEW (NIH) Publication No.
78-842. National Institutes of Health, Bethesda, MD, 1978.
NIOSH. National Institute for Occupational Safety and Health. National Occupational Exposure
Survey (1981-83). Unpublished provisional data as of 7/1/90. Cincinnati, OH: Department of
Health and Human Services, 1990.
NTP. National Toxicology Program. NTP Chemical Repository. 5-Azacytidine. Last updated
August 13, 2001. (http://ntp-server.niehs.nih.gov and search 320-67-2).
Nyce, J., S. Weinhouse, and P.N. Magee. 5-Methylcytosine Depletion During Tumour
Development: An Extension of the Miscoding Concept. Br. J. Cancer Vol. 48, 1983, pp. 463-
475.
Pascale, R.M., M.M. Simile, and F. Feo. Genomic Abnormalities in Hepatocarcinogenesis.
Implications for a Chemopreventive Strategy. Anticancer Res. Vol. 13, 1993, pp. 1341-1356.
Pharmion. http://www.pharmion.com (select therapeutics, then hematology and oncology
products), 2001.
Riggs, A.D., and P.A. Jones. 5-Methylcytosine, Gene Regulation, and Cancer. Adv. Cancer Res.
Vol. 40, 1983, pp. 1-30.
Santini, V., H.M. Kantarjian, and J-P Issa. Changes in DNA Methylation in Neoplasia:
Pathophysiology and Therapeutic Implications. Ann. Intern. Med. Vol. 134, No.7, 2001, pp.573-
586.
Takenaga, K. Modification of the Metastatic Potential of Tumor Cells by Drugs. Cancer
Metastasis Rev. Vol. 5, 1986, pp. 67-75.
Trissel, Lawrence A. Handbook on Injectable Drugs. Washington: American Society of Hospital
Pharmacists September 1996.
Velge, P., B. Kaeffer, E. Bottreau, and N. Van Langendonck. The Loss of Contact Inhibition and
Anchorage-Dependent Growth are Key Steps in the Acquisition of Listeria monocytogenes
Susceptibility Phenotype by Non-Phagocytic Cells. Biol. Cell Vol. 85, No. 1, 1995, pp. 55-66.
REASONABLY ANTICIPATED TO BE A HUMAN CARCINOGEN TENTH REPORT ON CARCINOGENS
You might also like
- Lung Cancer Report Sample High enDocument1 pageLung Cancer Report Sample High enSiddhesh PawarNo ratings yet
- AACR 2022 Proceedings: Part A Online-Only and April 10From EverandAACR 2022 Proceedings: Part A Online-Only and April 10No ratings yet
- Expression of A Breast-Cancer-Associated Protein (Ps2) in Human Neuro-Endocrine TumoursDocument5 pagesExpression of A Breast-Cancer-Associated Protein (Ps2) in Human Neuro-Endocrine TumoursSarly FebrianaNo ratings yet
- Corrections: Neuroscience. Cell BiologyDocument8 pagesCorrections: Neuroscience. Cell BiologySammer BurgosNo ratings yet
- Inhibition of Protein Kinase C-SRC Reduces The Incidence of Breast Cancer Metastases and Increases Survival in Mice: Implications For TherapyDocument12 pagesInhibition of Protein Kinase C-SRC Reduces The Incidence of Breast Cancer Metastases and Increases Survival in Mice: Implications For Therapybiomarinium76No ratings yet
- S 018 AzatDocument4 pagesS 018 Azatrendra boyke2023No ratings yet
- Tong Hop N-Nitroso PropranolonDocument11 pagesTong Hop N-Nitroso PropranolonTrần Sơn HảiNo ratings yet
- 5-Azacytidine Prevents Cisplatin Induced Nephrotoxicity and PotentiatesDocument9 pages5-Azacytidine Prevents Cisplatin Induced Nephrotoxicity and PotentiatesTorres Pineda OsvaldoNo ratings yet
- N-Acetyltransferase 2 Genetic Polymorphism Effects ofDocument21 pagesN-Acetyltransferase 2 Genetic Polymorphism Effects ofIsrael GonzalezNo ratings yet
- Endometriosis As. RetinoatDocument7 pagesEndometriosis As. RetinoatTuti Nursyah PutriNo ratings yet
- Wauson, 2002Document9 pagesWauson, 2002evanconstantine77No ratings yet
- Endo Study 2013Document0 pagesEndo Study 2013Agent Orange LegacyNo ratings yet
- A Toxicogenomics Approach To Identify New Plausible Epigenetic Mechanisms of Ochratoxin A Carcinogenicity in RatDocument15 pagesA Toxicogenomics Approach To Identify New Plausible Epigenetic Mechanisms of Ochratoxin A Carcinogenicity in Ratabyssinia2183No ratings yet
- E-Cadherin Expression in Human Epithelial Ovarian Cancer and Normal Ovary (International Journal of Cancer, Vol. 74, Issue 3) (1997)Document6 pagesE-Cadherin Expression in Human Epithelial Ovarian Cancer and Normal Ovary (International Journal of Cancer, Vol. 74, Issue 3) (1997)Ahmed MagzoubNo ratings yet
- Ijhs 12345+8686 8699Document14 pagesIjhs 12345+8686 8699ali ahmedNo ratings yet
- Cancer Prevention and Treatment With ResDocument11 pagesCancer Prevention and Treatment With ResSrishti Agarwal100% (1)
- Helland Radioth Onc 2006 PDFDocument6 pagesHelland Radioth Onc 2006 PDFFernanda RibeiroNo ratings yet
- Estrogen Receptor B Polymorphism Is Associated With Prostate Cancer RiskDocument6 pagesEstrogen Receptor B Polymorphism Is Associated With Prostate Cancer RiskTomaNo ratings yet
- Mutations of KRAS That Have A Profound Impact On Cancer Genomic Medicine Currently Being PursuedDocument4 pagesMutations of KRAS That Have A Profound Impact On Cancer Genomic Medicine Currently Being PursuedResearch ParkNo ratings yet
- Generation and Characterization of A Mouse/human Chimeric Antibody Directed Against Extracellular Matrix Protein TenascinDocument11 pagesGeneration and Characterization of A Mouse/human Chimeric Antibody Directed Against Extracellular Matrix Protein TenascinSayda DhaouadiNo ratings yet
- The Silencing of Long Non-Coding RNA ANRIL Suppresses Invasion, and Promotes Apoptosis of Retinoblastoma Cells Through The ATM-E2F1 Signaling PathwayDocument29 pagesThe Silencing of Long Non-Coding RNA ANRIL Suppresses Invasion, and Promotes Apoptosis of Retinoblastoma Cells Through The ATM-E2F1 Signaling PathwayJosué Cristhian Del Valle HornaNo ratings yet
- Focal Amplification of The AndrogenDocument8 pagesFocal Amplification of The AndrogenalexNo ratings yet
- Grant 1997Document10 pagesGrant 1997Django BoyeeNo ratings yet
- Hialuronidasa Tserrulatus Pesseni 2001Document10 pagesHialuronidasa Tserrulatus Pesseni 2001Michael rosoNo ratings yet
- Chen 2009Document11 pagesChen 2009darksoul4040940No ratings yet
- Cancer Oral 1Document9 pagesCancer Oral 1Maria CorenaNo ratings yet
- Resistance in Human Cervical Tumor Cells: Ercc1 Expression As A Molecular Marker of CisplatinDocument5 pagesResistance in Human Cervical Tumor Cells: Ercc1 Expression As A Molecular Marker of CisplatinAmanda Kelly N. MendonçaNo ratings yet
- Phyllanthus Amarus and Euphorbia Hirta On BenzoDocument11 pagesPhyllanthus Amarus and Euphorbia Hirta On BenzoInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Construction of A Reference Material Panel For Detecting Kras/Nras/Egfr/Braf/Met Mutations in Plasma CtdnaDocument7 pagesConstruction of A Reference Material Panel For Detecting Kras/Nras/Egfr/Braf/Met Mutations in Plasma Ctdnasylvi293No ratings yet
- Neuronal SIRT1 Activation As A Novel Mechanism Underlying The Prevention of Alzheimer Disease Amyloid Neuropathology by Calorie RestrictionDocument10 pagesNeuronal SIRT1 Activation As A Novel Mechanism Underlying The Prevention of Alzheimer Disease Amyloid Neuropathology by Calorie Restrictionlala_bojaNo ratings yet
- 2538 FullDocument6 pages2538 FullAdelyna AndreiNo ratings yet
- The Evolving Role of The Endocannabinoid System in Gynaecological CancerDocument19 pagesThe Evolving Role of The Endocannabinoid System in Gynaecological CancernusantaramedikasolusindoNo ratings yet
- Mô Tả Kỹ Về Nhuộm Tế Bào ApoptosisDocument16 pagesMô Tả Kỹ Về Nhuộm Tế Bào ApoptosisSang (CNC) Võ ThanhNo ratings yet
- Differential Display RT PCR of Total RNA From Human Foreskin Fibroblasts For Investigation of Androgen-Dependent Gene ExpressionDocument8 pagesDifferential Display RT PCR of Total RNA From Human Foreskin Fibroblasts For Investigation of Androgen-Dependent Gene ExpressionAltaicaNo ratings yet
- Environmental Toxicology and PharmacologyDocument5 pagesEnvironmental Toxicology and PharmacologyDnyanesh AmleNo ratings yet
- Gentian Violet CICDocument3 pagesGentian Violet CICValerie MeikleNo ratings yet
- Let KiDocument7 pagesLet KiNimra Naveed ShaikhNo ratings yet
- Cho PDFDocument8 pagesCho PDFMichelle SarmentoNo ratings yet
- Influence of Environmental and Nutritional Factors On Salivary Gland Tumorigenesis With A Special Reference To Dietary LipidsDocument6 pagesInfluence of Environmental and Nutritional Factors On Salivary Gland Tumorigenesis With A Special Reference To Dietary LipidsestantevirtualdosmeuslivrosNo ratings yet
- 09 26 2006 Cancer As Genetic DiseasesDocument60 pages09 26 2006 Cancer As Genetic Diseasesapi-3696530No ratings yet
- 2013 AapsjDocument8 pages2013 AapsjSCYoungNo ratings yet
- Cytoprotective Effects of Anthocyanin Nanocomplex Against H O - Induced Oxidative Stress in Leydig TM3 CellsDocument28 pagesCytoprotective Effects of Anthocyanin Nanocomplex Against H O - Induced Oxidative Stress in Leydig TM3 CellsAyu RatihNo ratings yet
- Aragon Ching2017Document3 pagesAragon Ching2017nimaelhajjiNo ratings yet
- Comparative Biochemistry and Physiology, Part C: Linda Bingsohn, Eileen Knorr, Andreas VilcinskasDocument8 pagesComparative Biochemistry and Physiology, Part C: Linda Bingsohn, Eileen Knorr, Andreas VilcinskasbiancacuritibaNo ratings yet
- Epigenetic and Cancer Therapy, 2019Document12 pagesEpigenetic and Cancer Therapy, 2019AbsjeyNo ratings yet
- The Occurrence of Ochratoxin A in Coffee NOV 13Document15 pagesThe Occurrence of Ochratoxin A in Coffee NOV 13kenoNo ratings yet
- Piomelli 2000 PDFDocument10 pagesPiomelli 2000 PDFМатиас Себальос ГусманNo ratings yet
- Sex Steroid Hormone Receptors in Human ThymomaDocument9 pagesSex Steroid Hormone Receptors in Human Thymomarima noviaNo ratings yet
- 3307 FullDocument5 pages3307 FullLuiz PauloNo ratings yet
- 1990 - Bousquet Et Al. - Comparison Between RAST and Pharmacia CAP System A New Automated Specific IgE AssayDocument5 pages1990 - Bousquet Et Al. - Comparison Between RAST and Pharmacia CAP System A New Automated Specific IgE Assaypond_1993No ratings yet
- вред ПАУDocument9 pagesвред ПАУИван ШиёнокNo ratings yet
- Citosol (Thiamylal Sodium) Triggers Apoptosis and Affects Gene Expressions of Murine Leukemia RAW 264.7 Cells - RS-C Wu, C-S Yu, 2Document1 pageCitosol (Thiamylal Sodium) Triggers Apoptosis and Affects Gene Expressions of Murine Leukemia RAW 264.7 Cells - RS-C Wu, C-S Yu, 2Alondra MaldonadoNo ratings yet
- Barlaskar Et Al - Pre Clinical Targeting of IGFR1 in ACC - 2009Document9 pagesBarlaskar Et Al - Pre Clinical Targeting of IGFR1 in ACC - 2009Camilla SampaoliNo ratings yet
- Martin Zenker-Noonan Syndrome and Related Disorders - A Matter of Deregulated Ras Signaling (Monographs in Human Genetics Vol 17) - S. Karger AG (Switzerland) (2009)Document178 pagesMartin Zenker-Noonan Syndrome and Related Disorders - A Matter of Deregulated Ras Signaling (Monographs in Human Genetics Vol 17) - S. Karger AG (Switzerland) (2009)Lupu AndreeaNo ratings yet
- 15-Li Chen (531-538)Document8 pages15-Li Chen (531-538)Nguyễn Thanh TùngNo ratings yet
- Document PDFDocument10 pagesDocument PDFMichael TanNo ratings yet
- International Journal of Cancer Volume 85 Issue 3 2000Document8 pagesInternational Journal of Cancer Volume 85 Issue 3 2000BenePicarNo ratings yet
- 2022 - Interplay Between The Histone Variant H2A.Z and The Epigenome in Pancreatic CancerDocument19 pages2022 - Interplay Between The Histone Variant H2A.Z and The Epigenome in Pancreatic CancerPedro Antonio Ávila LópezNo ratings yet
- RPL N 5,10 MTHFRDocument7 pagesRPL N 5,10 MTHFRjawaralopangNo ratings yet
- Polymorphisms in The Promoter Regions of The Matrix Metalloproteinases-7, - 9 and The Risk of Endometriosis and AdenomyosisDocument5 pagesPolymorphisms in The Promoter Regions of The Matrix Metalloproteinases-7, - 9 and The Risk of Endometriosis and AdenomyosisAndy WijayaNo ratings yet
- Diseases of The ColonDocument59 pagesDiseases of The ColonElvisNo ratings yet
- A Comparitive and Prognostic Study of Circulating Tumor Cells and Vegfr-2 in Different Histological Grades of Oral Squamous Cell CarcinomaDocument10 pagesA Comparitive and Prognostic Study of Circulating Tumor Cells and Vegfr-2 in Different Histological Grades of Oral Squamous Cell CarcinomaIJAR JOURNALNo ratings yet
- AAOS Oncology (50 Soal)Document18 pagesAAOS Oncology (50 Soal)Fasa RoshadaNo ratings yet
- Multiple Lipomas Linked To An RB1 Gene Mutation in A Large Pedigree With Low Penetrance RetinoblastomaDocument5 pagesMultiple Lipomas Linked To An RB1 Gene Mutation in A Large Pedigree With Low Penetrance RetinoblastomaMaria Burke-ChaussonNo ratings yet
- Thesis Statement For Prostate CancerDocument5 pagesThesis Statement For Prostate Cancerashleycornettneworleans100% (2)
- Oropharingeal Carcinoma Parvathaneni2019Document14 pagesOropharingeal Carcinoma Parvathaneni2019kikicreNo ratings yet
- Neoplasms of The Gastrointestinal Tract: Laura W. Lamps, M.DDocument39 pagesNeoplasms of The Gastrointestinal Tract: Laura W. Lamps, M.DINDRINo ratings yet
- Abbé - EstlanderDocument4 pagesAbbé - EstlanderKoretta Esmeral AtehortúaNo ratings yet
- Breast CancerDocument50 pagesBreast CancerZainab Idrees- F2FNo ratings yet
- Benign Paratesticlar Cyst - A Mysterical FindingDocument2 pagesBenign Paratesticlar Cyst - A Mysterical FindingInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Modified Transverse-Vertical Gross ExaminationDocument26 pagesModified Transverse-Vertical Gross Examinationananda rizkyNo ratings yet
- Ib Style Questions Methylation & Gene Expression: © David Faure, InthinkingDocument3 pagesIb Style Questions Methylation & Gene Expression: © David Faure, InthinkingMichelle Amruth SampigeNo ratings yet
- Nishino Et Al 2012 Revised Recist Guideline Version 1 1 What Oncologists Want To Know and What Radiologists Need To KnowDocument9 pagesNishino Et Al 2012 Revised Recist Guideline Version 1 1 What Oncologists Want To Know and What Radiologists Need To KnowJelena JovanovicNo ratings yet
- Central Nervous System Cancers: NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines)Document178 pagesCentral Nervous System Cancers: NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines)Hermanto YuliusNo ratings yet
- Breast - Dr. HammouriDocument12 pagesBreast - Dr. HammouriMohammad PropensityNo ratings yet
- Karen CharmDocument2 pagesKaren CharmHafni Kumar0% (1)
- Meet 4. Penyiapan Obat SitostatikaDocument47 pagesMeet 4. Penyiapan Obat SitostatikaChoirun NisaNo ratings yet
- Biopsija Sentinel Limfnog Čvora Kod Karcinoma Dojke: Sentinel Lymph Node Biopsy in Breast NeoplasmsDocument5 pagesBiopsija Sentinel Limfnog Čvora Kod Karcinoma Dojke: Sentinel Lymph Node Biopsy in Breast NeoplasmsyellowbieNo ratings yet
- Benefits of Physical ActivityDocument1 pageBenefits of Physical ActivityChristien Mae UnabiaNo ratings yet
- Jurnal Veruka VulgarisDocument3 pagesJurnal Veruka Vulgariscitra annisa fitriNo ratings yet
- Fight Club by Chuck PalahniukDocument149 pagesFight Club by Chuck PalahniukВікторія МисикNo ratings yet
- Ijsrp p11558Document5 pagesIjsrp p11558Zuhri mardiahNo ratings yet
- The Breast Journal - 2021 - Al Naqqash - Trastuzumab Beyond Progression in HER2 Positive Metastatic Breast CancerDocument3 pagesThe Breast Journal - 2021 - Al Naqqash - Trastuzumab Beyond Progression in HER2 Positive Metastatic Breast CancerhamzaNo ratings yet
- Colon CancerDocument17 pagesColon CancerYaska MusaNo ratings yet
- PSA Level and Gleason Score in Predicting The Stage of Newly Diagnosed Prostat CancerDocument6 pagesPSA Level and Gleason Score in Predicting The Stage of Newly Diagnosed Prostat CancergumNo ratings yet
- 2020 WHO Classification of Female Genital TumorsDocument9 pages2020 WHO Classification of Female Genital TumorsLieblingsmensch andipaNo ratings yet
- An Ultrasonogram Reporting System For Thyroid Nodules Stratifying Cancer Risk For Clinical ManagementDocument4 pagesAn Ultrasonogram Reporting System For Thyroid Nodules Stratifying Cancer Risk For Clinical ManagementMuhammad FiqranNo ratings yet
- Premalignant and Malignant Skin DiseasesDocument25 pagesPremalignant and Malignant Skin Diseasesmedical1acc2No ratings yet
- Is A Traditional Colonoscopy or A Virtual Colonoscopy BetterDocument1 pageIs A Traditional Colonoscopy or A Virtual Colonoscopy BetterPaul JensenNo ratings yet