Professional Documents
Culture Documents
Data and Formulae Sheet - Periodic Table
Data and Formulae Sheet - Periodic Table
Uploaded by
KHÁNH VÂN DIỆPOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Data and Formulae Sheet - Periodic Table
Data and Formulae Sheet - Periodic Table
Uploaded by
KHÁNH VÂN DIỆPCopyright:
Available Formats
Data and formulae sheet
Important IR stretching frequencies
Those above 1650 cm–1 are most important.
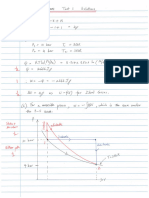
Equations
Kw = 1.0 × 10-14
pKa + pKb = 14
Kw = Ka × Kb
pH = -log[H3O+]
Kw = [H3O+][OH-]
−𝑏𝑏 ± √𝑏𝑏 2 − 4𝑎𝑎𝑎𝑎
𝑥𝑥 =
2𝑎𝑎
Zero First Second
rate = k rate = k[A] rate = k[A]2
[A]t = –kt + [A]o ln[A]t = –kt + ln[A]o
𝒍𝒍𝒍𝒍 𝟐𝟐
𝒕𝒕𝟏𝟏 =
𝟐𝟐 𝒌𝒌
𝑬𝑬𝒂𝒂 𝑬𝑬𝒂𝒂 𝟏𝟏 𝒌𝒌𝟐𝟐 −𝑬𝑬𝒂𝒂 𝟏𝟏 𝟏𝟏
𝒌𝒌 = 𝑨𝑨𝒆𝒆−𝑹𝑹𝑹𝑹 𝒍𝒍𝒍𝒍𝒍𝒍 = − � � + 𝒍𝒍𝒍𝒍𝒍𝒍 𝒍𝒍𝒍𝒍 = ( − )
𝑹𝑹 𝑻𝑻 𝒌𝒌𝟏𝟏 𝑹𝑹 𝑻𝑻𝟐𝟐 𝑻𝑻𝟏𝟏
Constants Conversions
R = 8.314 J K–1 mol–1 p° = 105 Pa
NA = 6.022 × 1023 mol–1 1 bar = 105 Pa
F = 96487 C mol–1 T(K) = T(°C) + 273.15
You might also like
- Dynamics FULL Equation SheetDocument2 pagesDynamics FULL Equation SheetZachNo ratings yet
- Solution Manual for an Introduction to Equilibrium ThermodynamicsFrom EverandSolution Manual for an Introduction to Equilibrium ThermodynamicsNo ratings yet
- CHAPTER 03 - 2nd Part - Properties of Pure Fluids-May20Document49 pagesCHAPTER 03 - 2nd Part - Properties of Pure Fluids-May20Scorpion Royal100% (1)
- CHEM 1252 Exam Information Sheet Fall 2021Document2 pagesCHEM 1252 Exam Information Sheet Fall 2021Joe joeNo ratings yet
- Gabarito Lista3Document6 pagesGabarito Lista3faculdade arquivosNo ratings yet
- Formula Sheet NumericalDocument3 pagesFormula Sheet NumericalZain BhinderNo ratings yet
- Lembar Perhitungan ReagaenDocument8 pagesLembar Perhitungan ReagaenZahra AlifiaNo ratings yet
- Chapter V-Composed Reactions KineticsDocument15 pagesChapter V-Composed Reactions KineticsZeina Abi FarrajNo ratings yet
- All Important Curves Sketching (P1) : Name Shape Shortcut Remark ExampleDocument1 pageAll Important Curves Sketching (P1) : Name Shape Shortcut Remark ExampleNacon ProNo ratings yet
- 3-D Conduction in Cylindrical BodyDocument5 pages3-D Conduction in Cylindrical Bodyvikram adityaNo ratings yet
- CHEM 123 Formula SheetDocument2 pagesCHEM 123 Formula Sheetemmacont2060No ratings yet
- Formulario F1003Document1 pageFormulario F1003adeve19No ratings yet
- Simusoidal Circuits and RLC CircuitsDocument20 pagesSimusoidal Circuits and RLC CircuitsIbrahim GhunaimNo ratings yet
- 09-24-21 Lecture 14 and CEP 13-14Document20 pages09-24-21 Lecture 14 and CEP 13-14Rose ChenNo ratings yet
- S05 - Formulario Comunicaciones SatelitalesDocument7 pagesS05 - Formulario Comunicaciones SatelitalesEu DerbNo ratings yet
- Formulario IngDocument1 pageFormulario IngCopias e ImpresionesNo ratings yet
- Civil Engineering Hydrology (CEHDROL) FormulasDocument2 pagesCivil Engineering Hydrology (CEHDROL) FormulasChamberlaine LopezNo ratings yet
- 01 Time Domain SpecificationDocument26 pages01 Time Domain SpecificationShadowツNo ratings yet
- PEP 2020 Phase 2 Selection Test 5 - SolutionDocument9 pagesPEP 2020 Phase 2 Selection Test 5 - SolutionMarcus PoonNo ratings yet
- Rumus FisikaDocument2 pagesRumus FisikaJatiurNo ratings yet
- Convección Externa Forzada Convección Interna Forzada Qs CCTTDocument3 pagesConvección Externa Forzada Convección Interna Forzada Qs CCTTMaria JaraNo ratings yet
- Unit IiDocument59 pagesUnit IishaibazNo ratings yet
- Formula Sheet Physics 11Document2 pagesFormula Sheet Physics 11melissa.figueroamoralesNo ratings yet
- Formulario FísicaDocument2 pagesFormulario FísicaJorge NaalNo ratings yet
- Lecture 1 Handout - Introduction To Reaction KineticsDocument14 pagesLecture 1 Handout - Introduction To Reaction Kineticsaliggulam16No ratings yet
- Lesson 8 Generalized Thermal Resistance NetworkDocument24 pagesLesson 8 Generalized Thermal Resistance Networksurya kiranNo ratings yet
- Formulario Termo NyNDocument1 pageFormulario Termo NyNElenaNo ratings yet
- FisIII FormularioDocument1 pageFisIII FormularioVázquez Solís Yoshua Dakari 2CNo ratings yet
- General Chemistry Lecturer-2Document34 pagesGeneral Chemistry Lecturer-2Bảo Long Trần LêNo ratings yet
- 3trigonometric FormulaeDocument2 pages3trigonometric FormulaeMahek IrfanNo ratings yet
- Aim-To Simulate Analysis Current and Voltage in Step Response of RLC Circuit Without Any InitiallyDocument4 pagesAim-To Simulate Analysis Current and Voltage in Step Response of RLC Circuit Without Any Initiallysaksham mahajanNo ratings yet
- Physics 141 Equation Sheet-Knight 4 EdDocument1 pagePhysics 141 Equation Sheet-Knight 4 EdSimon SituNo ratings yet
- Equation Sheet SemesterDocument2 pagesEquation Sheet SemesterakNo ratings yet
- ADRIANO, Rhod Christian C. - LM Report No. 3Document9 pagesADRIANO, Rhod Christian C. - LM Report No. 3Rhod AdrianoNo ratings yet
- SOLUTIONDocument3 pagesSOLUTIONPrince QuimnoNo ratings yet
- HE Lecture 5 PDFDocument13 pagesHE Lecture 5 PDFpresidentisc nit-rourkelaNo ratings yet
- T1 - Vectors, Graphs of MotionDocument6 pagesT1 - Vectors, Graphs of MotionGGNo ratings yet
- SumsDocument41 pagesSumsshoyeb haqueNo ratings yet
- Piping NetworksDocument14 pagesPiping NetworksKarel Grace ColotNo ratings yet
- Es 62Document6 pagesEs 62Karl Sternberg ZaldariagaNo ratings yet
- Magnetic Effect of Current - Quick Revision - Classnotes (For Print Out) - (Lakshya JEE 2023)Document8 pagesMagnetic Effect of Current - Quick Revision - Classnotes (For Print Out) - (Lakshya JEE 2023)MeghanshNo ratings yet
- Quiz No. 1 SolutionDocument20 pagesQuiz No. 1 SolutionCamille SalmasanNo ratings yet
- Integral Part 2Document9 pagesIntegral Part 2Richie James TomarongNo ratings yet
- Electric PotentialDocument10 pagesElectric PotentialSrishti PathakNo ratings yet
- Oxford AQA A Level Physics Unit 3 Insert Jan19Document4 pagesOxford AQA A Level Physics Unit 3 Insert Jan19Pop VNo ratings yet
- Formularium MechanicaDocument2 pagesFormularium MechanicanokeuhNo ratings yet
- ElectronicII Lecture7 Filters ExercicesDocument23 pagesElectronicII Lecture7 Filters ExercicesSlime UNICORNNo ratings yet
- 10 Nov Himanshu TriDocument70 pages10 Nov Himanshu TriroyalNo ratings yet
- Exam Formula Sheet (1) PhysicsDocument3 pagesExam Formula Sheet (1) PhysicsIncksNo ratings yet
- Formulario MecánicaDocument2 pagesFormulario MecánicaNicoll CalvacheNo ratings yet
- Exercice MoufidDocument2 pagesExercice MoufidTEST - TESTNo ratings yet
- Home Assignment 1 - Ersa Berliana - 04211941000041Document6 pagesHome Assignment 1 - Ersa Berliana - 04211941000041roberto luckyNo ratings yet
- The DSB-SCDocument21 pagesThe DSB-SCmhww9jqqpjNo ratings yet
- First Order Circuits - RC and RLDocument22 pagesFirst Order Circuits - RC and RLTyspoNo ratings yet
- FormulaDocument2 pagesFormulaThanh NgânNo ratings yet
- Steel ResitDocument11 pagesSteel ResitDarren XuenNo ratings yet
- Cheat - Sheet - Exam 1Document2 pagesCheat - Sheet - Exam 1LoganNo ratings yet
- Exam3 EquationsheetDocument1 pageExam3 EquationsheetNguyễn Minh AnhNo ratings yet
- Drying Rate and Time: Mass Transfer Operation-2Document9 pagesDrying Rate and Time: Mass Transfer Operation-2Chintan ModiNo ratings yet
- ESYS1000 Lab 1 - Face To FaceDocument13 pagesESYS1000 Lab 1 - Face To FaceKHÁNH VÂN DIỆPNo ratings yet
- ESYS1000 Lab 0 - Face To FaceDocument5 pagesESYS1000 Lab 0 - Face To FaceKHÁNH VÂN DIỆPNo ratings yet
- Additional Questions 2 - DC2Document4 pagesAdditional Questions 2 - DC2KHÁNH VÂN DIỆPNo ratings yet
- ESYS1000 Lab 0 - OnlineDocument3 pagesESYS1000 Lab 0 - OnlineKHÁNH VÂN DIỆPNo ratings yet
- FOP Assignment2 90025224 User DocumentDocument11 pagesFOP Assignment2 90025224 User DocumentKHÁNH VÂN DIỆPNo ratings yet
- Declaration - of - Originality - ONLINE - Fillable FormDocument1 pageDeclaration - of - Originality - ONLINE - Fillable FormKHÁNH VÂN DIỆPNo ratings yet
- Assignment 02 202301 BentleyDocument5 pagesAssignment 02 202301 BentleyKHÁNH VÂN DIỆPNo ratings yet
- Calculus For Engineers FormulaeDocument4 pagesCalculus For Engineers FormulaeKHÁNH VÂN DIỆPNo ratings yet
- Test 1 - 2017 - SolutionDocument4 pagesTest 1 - 2017 - SolutionKHÁNH VÂN DIỆPNo ratings yet