Professional Documents
Culture Documents
187 - Biochemistry Physiology) Enzyme Inhibition
187 - Biochemistry Physiology) Enzyme Inhibition
Uploaded by
kalloliCopyright:
Available Formats
You might also like
- Full Download Book Elements of Chemical Reaction Engineering Global Edition 6Th Edition PDFDocument28 pagesFull Download Book Elements of Chemical Reaction Engineering Global Edition 6Th Edition PDFduane.lundgren266100% (28)
- Kami Export - Caylen Johnson - Kami Export - Counting - Atoms - 2010Document2 pagesKami Export - Caylen Johnson - Kami Export - Counting - Atoms - 2010XxC JayxXNo ratings yet
- Chemsheets A2 1001 Kinetics BookletDocument20 pagesChemsheets A2 1001 Kinetics BookletMarc WilfordNo ratings yet
- 9 Draw StructDocument6 pages9 Draw StructkalloliNo ratings yet
- Assign#1 Enzyme KineticsDocument1 pageAssign#1 Enzyme KineticsCarla Sibal100% (2)
- Outline: Factors Affecting Enzyme ActivityDocument11 pagesOutline: Factors Affecting Enzyme ActivityTee BeeNo ratings yet
- 3 Ezyme InhibitionDocument3 pages3 Ezyme InhibitionkalloliNo ratings yet
- Botany J-Adhikary Enzymology 4Document9 pagesBotany J-Adhikary Enzymology 4Dharmesh R.DNo ratings yet
- Enzyme KineticsDocument23 pagesEnzyme KineticsGOCNo ratings yet
- DocumentDocument7 pagesDocumentANIS NURALYSA AFFENDINo ratings yet
- GROUP 7 Che 4101Document64 pagesGROUP 7 Che 4101nica roseNo ratings yet
- L1 - Drug and Drug Targets - 2023Document116 pagesL1 - Drug and Drug Targets - 2023Nguyễn Minh KhuêNo ratings yet
- Enzyme InhibitorsDocument14 pagesEnzyme InhibitorsMetalica Mira EspeletaNo ratings yet
- Enzyme Inhibitors. Definition, Classification, and Main PropertiesDocument3 pagesEnzyme Inhibitors. Definition, Classification, and Main PropertiesAkmaral TleubaevaNo ratings yet
- Enzymes All NUBDocument9 pagesEnzymes All NUBPIH SHTNo ratings yet
- Week 2 - Enzyme Kinetics All - v2 - 3slide HandoutsDocument14 pagesWeek 2 - Enzyme Kinetics All - v2 - 3slide HandoutsFizza HussainNo ratings yet
- Inhibition: Maria Roceline P. Sandoy 2MT01Document15 pagesInhibition: Maria Roceline P. Sandoy 2MT01Roceline SandoyNo ratings yet
- Lehninger Principles of Biochemistry: Fourth EditionDocument40 pagesLehninger Principles of Biochemistry: Fourth EditionSanchit JainNo ratings yet
- Lecture Note - Enzyme InhibitorDocument10 pagesLecture Note - Enzyme InhibitorBright ChimezieNo ratings yet
- 7 Kinetika Enzim InhibitorDocument69 pages7 Kinetika Enzim InhibitorHernanda WidipasuryaNo ratings yet
- Mikrobiologi Industri: EnzymeDocument39 pagesMikrobiologi Industri: EnzymeEunike RhizaNo ratings yet
- Section 8.5 Enzymes Can Be Inhibited by Specific MoleculesDocument8 pagesSection 8.5 Enzymes Can Be Inhibited by Specific MoleculesMohamed FaizalNo ratings yet
- Enzymes Are Always: Compressed Notes Chapter 4: Biocatalysis Sb025Document7 pagesEnzymes Are Always: Compressed Notes Chapter 4: Biocatalysis Sb025LIM ZHI SHUENNo ratings yet
- Enzymology Part 1-2Document12 pagesEnzymology Part 1-2Anya IgnacioNo ratings yet
- Past Year CreDocument22 pagesPast Year CreNURUL SYAHIRAH BINTI ABDUL HALIMNo ratings yet
- Lecture 6 - Enzymes As Drug TargetsDocument31 pagesLecture 6 - Enzymes As Drug TargetsAlex LesNo ratings yet
- Lesson 1-Enzymes Part IDocument4 pagesLesson 1-Enzymes Part IAbbigail Dela RosaNo ratings yet
- BIOCATALYSIS 20142015 JWPNDocument4 pagesBIOCATALYSIS 20142015 JWPNINTAN ZURINA BINTI AHMAD EDIDIKNo ratings yet
- 04 2020enzyme InhibitionDocument20 pages04 2020enzyme InhibitionDharmesh R.DNo ratings yet
- Enzymes All NUBDocument8 pagesEnzymes All NUBPIH SHTNo ratings yet
- Report BiotechDocument6 pagesReport Biotechnica roseNo ratings yet
- Receptor & Enzymes As Cellular Drug Targets - R.Raju - 2019Document31 pagesReceptor & Enzymes As Cellular Drug Targets - R.Raju - 2019abdulNo ratings yet
- Enz KineticsDocument20 pagesEnz KineticsSirsha PattanayakNo ratings yet
- 4.2 Enzyme Chemistry Part 2Document6 pages4.2 Enzyme Chemistry Part 2Geraldine Marie SalvoNo ratings yet
- Enzyme KineticsDocument5 pagesEnzyme KineticsCarlo Quinsayas SablanNo ratings yet
- A. Competitive Inhibition: Table 3Document1 pageA. Competitive Inhibition: Table 3Tran TuanNo ratings yet
- The Mode of Action of EnzymesDocument11 pagesThe Mode of Action of EnzymesAI NURLAELASARI RNo ratings yet
- Simple Enzyme Kinetics I. Enzyme Inhibition - InhibitorsDocument2 pagesSimple Enzyme Kinetics I. Enzyme Inhibition - InhibitorsfintastellaNo ratings yet
- Enzymes Part 2Document3 pagesEnzymes Part 2Mar LagmayNo ratings yet
- Quiz 1 - Biochemical EngineeringDocument7 pagesQuiz 1 - Biochemical EngineeringAlyssa CariazoNo ratings yet
- Enzyme: Specific Proteins That Catalyze Biochemical: Constituents of Enzyme MoleculeDocument6 pagesEnzyme: Specific Proteins That Catalyze Biochemical: Constituents of Enzyme MoleculeAnya IgnacioNo ratings yet
- Lab #4: Enzymes: BackgroundDocument12 pagesLab #4: Enzymes: BackgroundchristineNo ratings yet
- Enzymology Part 1Document2 pagesEnzymology Part 1Ella LobenariaNo ratings yet
- Enymes L2 2019 NC 6thDocument26 pagesEnymes L2 2019 NC 6thChamara ChathurangaNo ratings yet
- Chapter Iv EnzymesDocument6 pagesChapter Iv EnzymesJohn TacordaNo ratings yet
- Enzyme InhibitorDocument15 pagesEnzyme InhibitorNatalia BalbinottNo ratings yet
- Technological Institute of The Philippines: 363 P. Casal St. Quiapo, ManilaDocument8 pagesTechnological Institute of The Philippines: 363 P. Casal St. Quiapo, ManilaKrizzia Anne ShengNo ratings yet
- EnzymesDocument33 pagesEnzymesMary Sutingco100% (1)
- Chapter 2 P-2 Enzyme-Inhibition 1Document39 pagesChapter 2 P-2 Enzyme-Inhibition 1Raihan I. SakibNo ratings yet
- Chemistry in Everyday LifeDocument39 pagesChemistry in Everyday LifeArya StarkNo ratings yet
- Biology Biomolecules Part 2Document14 pagesBiology Biomolecules Part 2Ralph Kenneth LunaNo ratings yet
- The Mechanism of Enzymatic Action: Enzyme Chemical ReactionDocument16 pagesThe Mechanism of Enzymatic Action: Enzyme Chemical Reactionteam TSOTARENo ratings yet
- L-3 Enzyme InhibitorsDocument4 pagesL-3 Enzyme InhibitorsLiyakath AliNo ratings yet
- Dasar Dasar MetabolismeDocument39 pagesDasar Dasar MetabolismeWidychii GadiestchhetyaNo ratings yet
- EnzymesDocument2 pagesEnzymesAljon AniesNo ratings yet
- BIO 1400 Topic 6 Enzymes Structure and Function - 2023Document9 pagesBIO 1400 Topic 6 Enzymes Structure and Function - 2023Nicholas LukondeNo ratings yet
- Ws ch04 e PDFDocument7 pagesWs ch04 e PDF6B - 06 招芷澄 CHIU TSZ CHINGNo ratings yet
- Enzymes: Table 6-1 6-2Document7 pagesEnzymes: Table 6-1 6-2Adnan AliNo ratings yet
- Enzymes-Biology PresentationDocument52 pagesEnzymes-Biology PresentationAdeenNo ratings yet
- 2023-08-18 Enzymes KineticsDocument52 pages2023-08-18 Enzymes KineticsAjay MahalkaNo ratings yet
- Yjugessur E. M. Presentation Competitive Inhibition 25 09 2022Document32 pagesYjugessur E. M. Presentation Competitive Inhibition 25 09 2022Yudhistir JugessurNo ratings yet
- Tutorial 4 SolutionsDocument5 pagesTutorial 4 SolutionsjoelypamelaandersonNo ratings yet
- c1.1 EnzymesDocument36 pagesc1.1 Enzymeshwqhd asjdhuaNo ratings yet
- Lecture 3: Part 3 Enzyme Kinetics and Inhibition: DR Evelyne DeplazesDocument18 pagesLecture 3: Part 3 Enzyme Kinetics and Inhibition: DR Evelyne Deplazeskristal eliasNo ratings yet
- Pages from Physiology Integrated Approach-3Document1 pagePages from Physiology Integrated Approach-3kalloliNo ratings yet
- pectoral chorsiaDocument30 pagespectoral chorsiakalloliNo ratings yet
- 1 Enzymes Why They Are Efficient and SpecificDocument7 pages1 Enzymes Why They Are Efficient and SpecifickalloliNo ratings yet
- protDocument2 pagesprotkalloliNo ratings yet
- jogyallapur revenue mapDocument1 pagejogyallapur revenue mapkalloliNo ratings yet
- tadasinakoppa revenueDocument1 pagetadasinakoppa revenuekalloliNo ratings yet
- 6 Plasma CurvesDocument3 pages6 Plasma CurveskalloliNo ratings yet
- 8 MetabolismDocument3 pages8 MetabolismkalloliNo ratings yet
- 9 EliminationDocument5 pages9 EliminationkalloliNo ratings yet
- 5 PrinciplesDocument2 pages5 PrincipleskalloliNo ratings yet
- 3 Ezyme InhibitionDocument3 pages3 Ezyme InhibitionkalloliNo ratings yet
- Transfer of Building Works To PWOs and PSUsDocument1 pageTransfer of Building Works To PWOs and PSUskalloliNo ratings yet
- 2 Kinetics of Catlyzed ReactionsDocument4 pages2 Kinetics of Catlyzed ReactionskalloliNo ratings yet
- Reso 3Document2 pagesReso 3kalloliNo ratings yet
- 2007 Imso ShortanswerDocument10 pages2007 Imso ShortanswerkalloliNo ratings yet
- DPP-2 Complex-Number Ques @motion LecturesDocument1 pageDPP-2 Complex-Number Ques @motion LectureskalloliNo ratings yet
- 12.3 Structural Representations of Organic Compounds 12.3.1 Complete, Condensed and Bond-Line Structural FormulasDocument3 pages12.3 Structural Representations of Organic Compounds 12.3.1 Complete, Condensed and Bond-Line Structural FormulaskalloliNo ratings yet
- Ashirvad uPVC Casing PipesDocument2 pagesAshirvad uPVC Casing PipeskalloliNo ratings yet
- Pages From David R. Klein - Organic Chemistry As A Second Language - First Semester Topics-Wiley (2017)Document1 pagePages From David R. Klein - Organic Chemistry As A Second Language - First Semester Topics-Wiley (2017)kalloliNo ratings yet
- Pages From TB - 9th EdDocument1 pagePages From TB - 9th EdkalloliNo ratings yet
- DPP-4 Complex-Number Ques @motion LecturesDocument1 pageDPP-4 Complex-Number Ques @motion LectureskalloliNo ratings yet
- DPP-1 Circle Ques @motion LecturesDocument1 pageDPP-1 Circle Ques @motion LectureskalloliNo ratings yet
- Rigidity of Double /triple Bonds: C H H H H H C H H H H C H H H CDocument7 pagesRigidity of Double /triple Bonds: C H H H H H C H H H H C H H H CkalloliNo ratings yet
- Func Tional Groups: Heteroatoms-Atoms Other Than Carbon or Hydrogen. Common Heteroatoms Are NitroDocument3 pagesFunc Tional Groups: Heteroatoms-Atoms Other Than Carbon or Hydrogen. Common Heteroatoms Are NitrokalloliNo ratings yet
- 3 Sigma and PaiDocument1 page3 Sigma and PaikalloliNo ratings yet
- Problem 4.11: Which Would You Expect To Be More Stable: (CH) C or (CF) C ? Why?Document2 pagesProblem 4.11: Which Would You Expect To Be More Stable: (CH) C or (CF) C ? Why?kalloliNo ratings yet
- The Bonds in Ethyne: SP P P P SP S P PDocument1 pageThe Bonds in Ethyne: SP P P P SP S P PkalloliNo ratings yet
- Hyperconjugation: The Reactions of Alkenes - The Stereochemistry of Addition ReactionsDocument2 pagesHyperconjugation: The Reactions of Alkenes - The Stereochemistry of Addition ReactionskalloliNo ratings yet
- The Bonds in Ethene: P P P S SP SP P SPDocument1 pageThe Bonds in Ethene: P P P S SP SP P SPkalloliNo ratings yet
- EnzymeKinetics PDFDocument21 pagesEnzymeKinetics PDFaathiraNo ratings yet
- Slide Biop Enz KinDocument19 pagesSlide Biop Enz KinFahmi Ramadhan PutraNo ratings yet
- Assignment 2 Reactor Design Single ReactionDocument4 pagesAssignment 2 Reactor Design Single ReactionNitin MauryaNo ratings yet
- Kinetics Revision Worksheet 2 (Solutions)Document8 pagesKinetics Revision Worksheet 2 (Solutions)Lee Jun HuiNo ratings yet
- CHE244 Lab Report Effect of Residence TiDocument26 pagesCHE244 Lab Report Effect of Residence Tiwan nur mursyidahNo ratings yet
- Act# 10 WORKSHEET On STOICHIOMETRY (LAB)Document3 pagesAct# 10 WORKSHEET On STOICHIOMETRY (LAB)henz assasinNo ratings yet
- A Substance Produced by A Living Organism Which Acts As A Catalyst To Bring About A Specific Biochemical ReactionDocument14 pagesA Substance Produced by A Living Organism Which Acts As A Catalyst To Bring About A Specific Biochemical ReactionPrincely ImmanuelNo ratings yet
- 1.2. Boxes and Arrows To Differential EquationsDocument14 pages1.2. Boxes and Arrows To Differential EquationsOmer GaddouraNo ratings yet
- MIT Lecture 6 5.07 Biochemistry LectureDocument16 pagesMIT Lecture 6 5.07 Biochemistry LectureakiridoNo ratings yet
- Ex0 Questions SolutionsDocument7 pagesEx0 Questions SolutionsBiniyam haileNo ratings yet
- Types: Chemical ReactorDocument3 pagesTypes: Chemical ReactorVeni DwiNo ratings yet
- Bab 2 - Kinetika Reaksi HomogenDocument12 pagesBab 2 - Kinetika Reaksi HomogenDiah Ayu TriatNo ratings yet
- EnzymeDocument15 pagesEnzymeShreyashNo ratings yet
- Michaelis Menten Equation SummaryDocument7 pagesMichaelis Menten Equation SummaryBüşra GültekinNo ratings yet
- Enzymes PresDocument34 pagesEnzymes Presm. uma deviNo ratings yet
- Chemical Kinetics - CBSE Sol File Ex-1Document5 pagesChemical Kinetics - CBSE Sol File Ex-1PriyanshiNo ratings yet
- KINETICSDocument47 pagesKINETICSMarilia BonorinoNo ratings yet
- Schnakenberg 1979Document12 pagesSchnakenberg 1979andres felipe ortizNo ratings yet
- Emailing Chemical Kinetics (Class 12)Document12 pagesEmailing Chemical Kinetics (Class 12)roceniNo ratings yet
- Factors Affecting Enzyme ActivityDocument7 pagesFactors Affecting Enzyme ActivityDahliza KamatNo ratings yet
- Exercises KineticsDocument7 pagesExercises KineticsFahad AlasmiNo ratings yet
- Week 6 - Physical-ScienceDocument2 pagesWeek 6 - Physical-ScienceKayla TiquisNo ratings yet
- Homework Solutions/Kinetics 1Document11 pagesHomework Solutions/Kinetics 1Eduardo Rosado HerreraNo ratings yet
- Prof. K.K.Pant Department of Chemical Engineering IIT DelhiDocument45 pagesProf. K.K.Pant Department of Chemical Engineering IIT DelhiApaar MudgalNo ratings yet
- Elements of Chemical Reaction Engineering Prentice Hall International Series in The Physical and Chemical Engineering Sciences Ebook PDF VersionDocument62 pagesElements of Chemical Reaction Engineering Prentice Hall International Series in The Physical and Chemical Engineering Sciences Ebook PDF Versionadolfo.milliken178100% (57)
187 - Biochemistry Physiology) Enzyme Inhibition
187 - Biochemistry Physiology) Enzyme Inhibition
Uploaded by
kalloliOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
187 - Biochemistry Physiology) Enzyme Inhibition
187 - Biochemistry Physiology) Enzyme Inhibition
Uploaded by
kalloliCopyright:
Available Formats
Last edited: 9/10/2021
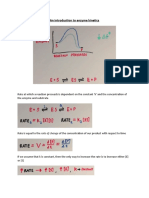
1. ENZYME INHIBITION
Enzyme Inhibition: Reversible and Irreversible Inhibition Medical Editor: Jona Frondoso
OUTLINE Two Types of Enzyme Inhibition
I) INTRODUCTION (i) Irreversible
II) TYPES OF REVERSIBLE INHIBITION Bind to enzymes through covalent bonds [Harvey &
III) IRREVERSIBLE INHIBITION
Ferrier, 2011]
IV) APPENDIX
V) REVIEW QUESTIONS (ii) Reversible
VI) REFERENCES
Bind to enzymes through non-covalent bonds
[Harvey & Ferrier, 2011]
I) INTRODUCTION Dilution of the enzyme-inhibitor complex results in
the dissociation of the reversibly bound inhibitor
Enzyme inhibitors have ways of affecting the normal [Harvey & Ferrier, 2011]
process of reaction (Figure 1).
Km and Vmax
(i) Km
Figure 1. Reaction model showing an enzyme reversibly
combining with its substrate to form an ES complex that Reflects the affinity of the enzyme for a particular substrate
subsequently yields product, regenerating the free enzyme. Substrate concentration ([S]) at ½ Vmax
Changes when the equilibrium changes
Where (ii) Vmax
o E is the enzyme
o S is the substrate Maximal velocity
o ES is the enzyme-substrate complex Velocity of the reaction when all enzymes are saturated
o P is the product by the substrate
o k1 is the rate constant for the forward reaction of E Changes when the amount of ES complex formation
and S forming ES changes
o k- 1 is the rate constant for the reverse reaction of ES
forming E and S FYI:
o k2 is the rate constant for the forward reaction of ES Km and Affinity
forming E and P
(i) Small Km
Inhibitor ↑ Affinity of the enzyme for a particular substrate
↓ Concentration is needed to achieve ½ Vmax
Any substance that can diminish the velocity of an enzyme-
catalyzed reaction [Harvey & Ferrier, 2011] (ii) Large Km
↓ Affinity of the enzyme for a particular substrate
↑ Concentration is needed to achieve ½ Vmax
ENZYME INHIBITION BIOCHEMISTRY: Note #1. 1 of 6
II) TYPES OF REVERSIBLE INHIBITION
• Can be competitive, noncompetitive, uncompetitive
(A) COMPETITIVE INHIBITION (B) NONCOMPETITIVE INHIBITION
Competitive inhibitor (CI) Noncompetitive inhibitors (NCI) – inhibitors that bind the
o Structural analog of the actual substrate → can also allosteric site of either the free enzyme or the ES
bind to the active site complex
o Forms noncovalent bonds with the active site Substrate can still bind to the active site while NCI is bound
Noncovalent bonds – weak bonds → can still be to the allosteric site → ESI complex formation → no
broken products formed
o CI bound to the active site → actual substrate can no When the inhibitor is released → ES complex formation
longer bind → ↑ Affinity for the CI while ↓ affinity for the → products formed
substrate→ ↓ ES complex formation → reaction shifts
to the right (by Le Chatelier’s Principle) → can be
(1) Effect on Vmax
achieved by ↑ substrate concentration (Figure 1)
↓ Vmax
o Not all enzymes can only be bound to the substrate
(1) Effect on Vmax o Enzymes can form either ES complex or ESI complex
Normal / Unchanged Vmax o Can never be reached Vmax
Effect can be reversed by ↑ substrate concentration Some enzymes participate in the ESI complex formation →
↑ Substrate concentration that can bind to the active site ↓ ES complex formed →↓ products formed
→ overcoming the concentration of competitive inhibitors
→ ↑ ES complex formation → ↑ product formed or the
(2) Effect on Km
amount of products formed when the inhibitor is absent
(Figure 1) Normal / Unchanged Km
o Means Vmax can still be achieved Because equilibrium for the reaction is not changed or not
thrown off
Reaction can still proceed towards the formation of the
(2) Effect on Km products
↑ Km
Equilibrium is changed
o Since reaction shifted to the right
More substrate is needed to achieve the same ½ Vmax
Figure 3. Noncompetitive inhibition.
(3) Examples
(i) Monoamine oxidase inhibitors (MAO-I)
Used in depression
Inhibit monoamine oxidase (MAO)
Figure 2. Competitive inhibition.
o MAO – breaks down epinephrine, norepinephrine, and
dopamine
(3) Examples (ii) Oxamic acid
(i) Statins Inhibits lactate dehydrogenase
Lactate dehydrogenase
Used in hypercholesterolemia and hypertriglyceridemia
o Converts lactate to pyruvate
Inhibits hydroxymethylglutaryl Coenzyme A (HMG-
o Pyruvate will eventually be converted to glucose
CoA) synthase
(ii) ACE inhibitors
Used in hypertension
Inhibits angiotensin-converting enzyme (ACE)
2 of 6 BIOCHEMISTRY: Note #1. ENZYME INHIBITION
(C) UNCOMPETITIVE INHIBITION III) IRREVERSIBLE INHIBITION
Uncompetitive inhibition (UCI) only binds to the allosteric
Also known as suicide inhibitors
site of the enzyme-substrate complex
Similar to competitive inhibitor
o Allosteric site only opens up once the ES complex is
o Also a structural analog of the substrate
formed
o Hence, can also bind to the active site
Enzyme binds to the substrate → allosteric site opens up
Differs from competitive inhibitor in that it forms covalent
→ UCI binds to the allosteric site → ESI complex formation
bonds
→No products formed
o Covalent bonds – very strong bonds → difficult to be
When the inhibitor is released → ES complex formation
broken down → cannot be reversed by increasing the
→ products are formed (Figure 4)
substrate concentration
Inhibitor binds to the active site→ EI complex formation (via
(1) Effect on Vmax covalent bonds) → suicide inhibition (Figure 5)
↓ Vmax
o Not all enzymes can only be bound to the substrate
o Enzymes can form either ES complex formation or ESI
complex formation
o Can nerver reached Vmax
Some enzymes participate in the ESI complex formation →
↓ ES complex formed →↓ products formed Figure 5. Irreversible inhibition.
(2) Effect on Km (1) Examples
↓ Km (i) Sarin
Equilibrium is changed (shifted to the right)
Nerve gas
ESI formation → ↓ ES complex formation → reaction
Used in chemical warfare
shifts to the right (by Le Chatelier’s principle) → ↓
Inhibits acetylcholinesterase
substrate concentration → ↓ Km
o Acetylcholinesterase – breaks down acetylcholine
(ii) Aspirin
Irreversible cyclooxygenase (COX) inhibitor
COX – catalyzes the formation of prostaglandins
and leukotrienes [Harvey & Ferrier, 2011]
Figure 4.Uncompetitive inhibition.
(3) Examples
(i) Arsenic
Poison
Inhibit glyceraldehyde-3-phosphate dehydrogenase
(G3PDH)
o G3PDH – converts glyceraldehyde-3-phosphate to
1,3-bisphosphoglycerate
(ii) Lithium
Inhibits an enzyme in the phosphoinositide pathway
ENZYME INHIBITION BIOCHEMISTRY: Note #1. 3 of 6
IV) APPENDIX
Table 1. Summary of Reversible and Irreversible Inhibitors
Irreversible
Reversible Inhibitors
Parameter Inhibitors
Competitive Noncompetitive Uncompetitive
Binds with the Binds only with the Binds with the active
Binds with the active
allosteric site of either allosteric site of the site via covalent
(i) Mechanism site via noncovalent
the free enzyme or ES complex bonds
bonds
the ES complex
↓ -
(ii) Km ↑ Normal
↓ ↓ -
(iii) Vmax Normal
Statins MAO-I Arsenic Sarin
(iv) Examples ACE-I Oxamic acid Lithium Aspirin
Figure 6. Overview of enzyme inhibition.
4 of 6 BIOCHEMISTRY: Note #1. ENZYME INHIBITION
V) REVIEW QUESTIONS
1) Which of the following statements is correct about
enzyme kinetics?
a) Km is a reflection of affinity
b) Km is the substrate concentration at half-maximal
velocity
c) Vmax is the velocity when all the enzymes are
saturated
d) All of the statements are correct.
2) Which of the following statements is correct about
competitive inhibition?
a) Reduces Km
b) Increases Vmax
c) Forms covalent bonds with the active site
d) Example are statins
3) Which of the following statements is correct about
noncompetitive inhibition?
a) Unchanged Km
b) Increases Vmax
c) Examples are ACE inhibitors
d) Binds only to the allosteric site of the ES complex
4) Which of the following statements is correct about
uncompetitive inhibition?
a) Increases Km
b) Decreases Vmax
c) Examples are MAO-I
d) Binds to the allosteric site of either the free enzyme
or the ES complex
5) Which of the following statements is correct about
irreversible inhibition?
a) Forms covalent bonds with the active site
b) Examples include aspirin and nerve gases
c) Increase in substrate concentration will not reverse
the inhibition
d) All of the choices are correct.
CHECK YOUR ANSWERS
VI) REFERENCES
Harvey, R., & Ferrier, D. (2011). Lippincott's Illustrated
Reviews - Biochemistry (5th ed.). Philadelphia: Lippincott Williams &
WIlkins.
ENZYME INHIBITION BIOCHEMISTRY: Note #1. 5 of 6
6 of 6 BIOCHEMISTRY: Note #1. ENZYME INHIBITION
Last edited: 9/11/2021
1. ENZYME INHIBITION
Enzyme Inhibition: Reversible and Irreversible Inhibition Medical Editor: Jona Frondoso
I) REVIEW QUESTIONS WITH ANSWERS
QUESTIONS ANSWERS
1) Which of the following statements is correct about Correct answer = D.
enzyme kinetics?
a) Km is a reflection of affinity
b) Km is the substrate concentration at half-maximal
velocity
c) Vmax is the velocity when all the enzymes are saturated
d) All of the statements are correct.
2) Which of the following statements is correct about Correct answer = D. Increases Km. Normal Vmax. Forms
competitive inhibition? noncovalent bonds with the active site.
a) Reduces Km
b) Increases Vmax
c) Forms covalent bonds with the active site
d) Example are statins
3) Which of the following statements is correct about Correct answer = A. Decreases Vmax. ACE inhibitors are
noncompetitive inhibition? examples of competitive inhibitors; Examples of
a) Unchanged Km noncompetitive inhibitors are MAO inhibitors and oxamic acid.
b) Increases Vmax Binds to the allosteric site of either the free enzyme or the ES
c) Examples are ACE inhibitors complex.
d) Binds only to the allosteric site of the ES complex
4) Which of the following statements is correct about Correct answer = B. Decreases Km. MAO-I are examples of
uncompetitive inhibition? noncompetitive inhibitors; Examples of uncompetitive
a) Increases Km inhibitors are arsenic and lithium. Binds only to the allosteric
b) Decreases Vmax site of the ES complex.
c) Examples are MAO-I
d) Binds to the allosteric site of either the free enzyme or
the ES complex
5) Which of the following statements is correct about Correct answer = D.
irreversible inhibition?
a) Forms covalent bonds with the active site
b) Examples include aspirin and nerve gases
c) Increase in substrate concentration will not reverse the
inhibition
d) All of the choices are correct.
ENZYME INHIBITION BIOCHEMISTRY: Note #1. 1 of 1
You might also like
- Full Download Book Elements of Chemical Reaction Engineering Global Edition 6Th Edition PDFDocument28 pagesFull Download Book Elements of Chemical Reaction Engineering Global Edition 6Th Edition PDFduane.lundgren266100% (28)
- Kami Export - Caylen Johnson - Kami Export - Counting - Atoms - 2010Document2 pagesKami Export - Caylen Johnson - Kami Export - Counting - Atoms - 2010XxC JayxXNo ratings yet
- Chemsheets A2 1001 Kinetics BookletDocument20 pagesChemsheets A2 1001 Kinetics BookletMarc WilfordNo ratings yet
- 9 Draw StructDocument6 pages9 Draw StructkalloliNo ratings yet
- Assign#1 Enzyme KineticsDocument1 pageAssign#1 Enzyme KineticsCarla Sibal100% (2)
- Outline: Factors Affecting Enzyme ActivityDocument11 pagesOutline: Factors Affecting Enzyme ActivityTee BeeNo ratings yet
- 3 Ezyme InhibitionDocument3 pages3 Ezyme InhibitionkalloliNo ratings yet
- Botany J-Adhikary Enzymology 4Document9 pagesBotany J-Adhikary Enzymology 4Dharmesh R.DNo ratings yet
- Enzyme KineticsDocument23 pagesEnzyme KineticsGOCNo ratings yet
- DocumentDocument7 pagesDocumentANIS NURALYSA AFFENDINo ratings yet
- GROUP 7 Che 4101Document64 pagesGROUP 7 Che 4101nica roseNo ratings yet
- L1 - Drug and Drug Targets - 2023Document116 pagesL1 - Drug and Drug Targets - 2023Nguyễn Minh KhuêNo ratings yet
- Enzyme InhibitorsDocument14 pagesEnzyme InhibitorsMetalica Mira EspeletaNo ratings yet
- Enzyme Inhibitors. Definition, Classification, and Main PropertiesDocument3 pagesEnzyme Inhibitors. Definition, Classification, and Main PropertiesAkmaral TleubaevaNo ratings yet
- Enzymes All NUBDocument9 pagesEnzymes All NUBPIH SHTNo ratings yet
- Week 2 - Enzyme Kinetics All - v2 - 3slide HandoutsDocument14 pagesWeek 2 - Enzyme Kinetics All - v2 - 3slide HandoutsFizza HussainNo ratings yet
- Inhibition: Maria Roceline P. Sandoy 2MT01Document15 pagesInhibition: Maria Roceline P. Sandoy 2MT01Roceline SandoyNo ratings yet
- Lehninger Principles of Biochemistry: Fourth EditionDocument40 pagesLehninger Principles of Biochemistry: Fourth EditionSanchit JainNo ratings yet
- Lecture Note - Enzyme InhibitorDocument10 pagesLecture Note - Enzyme InhibitorBright ChimezieNo ratings yet
- 7 Kinetika Enzim InhibitorDocument69 pages7 Kinetika Enzim InhibitorHernanda WidipasuryaNo ratings yet
- Mikrobiologi Industri: EnzymeDocument39 pagesMikrobiologi Industri: EnzymeEunike RhizaNo ratings yet
- Section 8.5 Enzymes Can Be Inhibited by Specific MoleculesDocument8 pagesSection 8.5 Enzymes Can Be Inhibited by Specific MoleculesMohamed FaizalNo ratings yet
- Enzymes Are Always: Compressed Notes Chapter 4: Biocatalysis Sb025Document7 pagesEnzymes Are Always: Compressed Notes Chapter 4: Biocatalysis Sb025LIM ZHI SHUENNo ratings yet
- Enzymology Part 1-2Document12 pagesEnzymology Part 1-2Anya IgnacioNo ratings yet
- Past Year CreDocument22 pagesPast Year CreNURUL SYAHIRAH BINTI ABDUL HALIMNo ratings yet
- Lecture 6 - Enzymes As Drug TargetsDocument31 pagesLecture 6 - Enzymes As Drug TargetsAlex LesNo ratings yet
- Lesson 1-Enzymes Part IDocument4 pagesLesson 1-Enzymes Part IAbbigail Dela RosaNo ratings yet
- BIOCATALYSIS 20142015 JWPNDocument4 pagesBIOCATALYSIS 20142015 JWPNINTAN ZURINA BINTI AHMAD EDIDIKNo ratings yet
- 04 2020enzyme InhibitionDocument20 pages04 2020enzyme InhibitionDharmesh R.DNo ratings yet
- Enzymes All NUBDocument8 pagesEnzymes All NUBPIH SHTNo ratings yet
- Report BiotechDocument6 pagesReport Biotechnica roseNo ratings yet
- Receptor & Enzymes As Cellular Drug Targets - R.Raju - 2019Document31 pagesReceptor & Enzymes As Cellular Drug Targets - R.Raju - 2019abdulNo ratings yet
- Enz KineticsDocument20 pagesEnz KineticsSirsha PattanayakNo ratings yet
- 4.2 Enzyme Chemistry Part 2Document6 pages4.2 Enzyme Chemistry Part 2Geraldine Marie SalvoNo ratings yet
- Enzyme KineticsDocument5 pagesEnzyme KineticsCarlo Quinsayas SablanNo ratings yet
- A. Competitive Inhibition: Table 3Document1 pageA. Competitive Inhibition: Table 3Tran TuanNo ratings yet
- The Mode of Action of EnzymesDocument11 pagesThe Mode of Action of EnzymesAI NURLAELASARI RNo ratings yet
- Simple Enzyme Kinetics I. Enzyme Inhibition - InhibitorsDocument2 pagesSimple Enzyme Kinetics I. Enzyme Inhibition - InhibitorsfintastellaNo ratings yet
- Enzymes Part 2Document3 pagesEnzymes Part 2Mar LagmayNo ratings yet
- Quiz 1 - Biochemical EngineeringDocument7 pagesQuiz 1 - Biochemical EngineeringAlyssa CariazoNo ratings yet
- Enzyme: Specific Proteins That Catalyze Biochemical: Constituents of Enzyme MoleculeDocument6 pagesEnzyme: Specific Proteins That Catalyze Biochemical: Constituents of Enzyme MoleculeAnya IgnacioNo ratings yet
- Lab #4: Enzymes: BackgroundDocument12 pagesLab #4: Enzymes: BackgroundchristineNo ratings yet
- Enzymology Part 1Document2 pagesEnzymology Part 1Ella LobenariaNo ratings yet
- Enymes L2 2019 NC 6thDocument26 pagesEnymes L2 2019 NC 6thChamara ChathurangaNo ratings yet
- Chapter Iv EnzymesDocument6 pagesChapter Iv EnzymesJohn TacordaNo ratings yet
- Enzyme InhibitorDocument15 pagesEnzyme InhibitorNatalia BalbinottNo ratings yet
- Technological Institute of The Philippines: 363 P. Casal St. Quiapo, ManilaDocument8 pagesTechnological Institute of The Philippines: 363 P. Casal St. Quiapo, ManilaKrizzia Anne ShengNo ratings yet
- EnzymesDocument33 pagesEnzymesMary Sutingco100% (1)
- Chapter 2 P-2 Enzyme-Inhibition 1Document39 pagesChapter 2 P-2 Enzyme-Inhibition 1Raihan I. SakibNo ratings yet
- Chemistry in Everyday LifeDocument39 pagesChemistry in Everyday LifeArya StarkNo ratings yet
- Biology Biomolecules Part 2Document14 pagesBiology Biomolecules Part 2Ralph Kenneth LunaNo ratings yet
- The Mechanism of Enzymatic Action: Enzyme Chemical ReactionDocument16 pagesThe Mechanism of Enzymatic Action: Enzyme Chemical Reactionteam TSOTARENo ratings yet
- L-3 Enzyme InhibitorsDocument4 pagesL-3 Enzyme InhibitorsLiyakath AliNo ratings yet
- Dasar Dasar MetabolismeDocument39 pagesDasar Dasar MetabolismeWidychii GadiestchhetyaNo ratings yet
- EnzymesDocument2 pagesEnzymesAljon AniesNo ratings yet
- BIO 1400 Topic 6 Enzymes Structure and Function - 2023Document9 pagesBIO 1400 Topic 6 Enzymes Structure and Function - 2023Nicholas LukondeNo ratings yet
- Ws ch04 e PDFDocument7 pagesWs ch04 e PDF6B - 06 招芷澄 CHIU TSZ CHINGNo ratings yet
- Enzymes: Table 6-1 6-2Document7 pagesEnzymes: Table 6-1 6-2Adnan AliNo ratings yet
- Enzymes-Biology PresentationDocument52 pagesEnzymes-Biology PresentationAdeenNo ratings yet
- 2023-08-18 Enzymes KineticsDocument52 pages2023-08-18 Enzymes KineticsAjay MahalkaNo ratings yet
- Yjugessur E. M. Presentation Competitive Inhibition 25 09 2022Document32 pagesYjugessur E. M. Presentation Competitive Inhibition 25 09 2022Yudhistir JugessurNo ratings yet
- Tutorial 4 SolutionsDocument5 pagesTutorial 4 SolutionsjoelypamelaandersonNo ratings yet
- c1.1 EnzymesDocument36 pagesc1.1 Enzymeshwqhd asjdhuaNo ratings yet
- Lecture 3: Part 3 Enzyme Kinetics and Inhibition: DR Evelyne DeplazesDocument18 pagesLecture 3: Part 3 Enzyme Kinetics and Inhibition: DR Evelyne Deplazeskristal eliasNo ratings yet
- Pages from Physiology Integrated Approach-3Document1 pagePages from Physiology Integrated Approach-3kalloliNo ratings yet
- pectoral chorsiaDocument30 pagespectoral chorsiakalloliNo ratings yet
- 1 Enzymes Why They Are Efficient and SpecificDocument7 pages1 Enzymes Why They Are Efficient and SpecifickalloliNo ratings yet
- protDocument2 pagesprotkalloliNo ratings yet
- jogyallapur revenue mapDocument1 pagejogyallapur revenue mapkalloliNo ratings yet
- tadasinakoppa revenueDocument1 pagetadasinakoppa revenuekalloliNo ratings yet
- 6 Plasma CurvesDocument3 pages6 Plasma CurveskalloliNo ratings yet
- 8 MetabolismDocument3 pages8 MetabolismkalloliNo ratings yet
- 9 EliminationDocument5 pages9 EliminationkalloliNo ratings yet
- 5 PrinciplesDocument2 pages5 PrincipleskalloliNo ratings yet
- 3 Ezyme InhibitionDocument3 pages3 Ezyme InhibitionkalloliNo ratings yet
- Transfer of Building Works To PWOs and PSUsDocument1 pageTransfer of Building Works To PWOs and PSUskalloliNo ratings yet
- 2 Kinetics of Catlyzed ReactionsDocument4 pages2 Kinetics of Catlyzed ReactionskalloliNo ratings yet
- Reso 3Document2 pagesReso 3kalloliNo ratings yet
- 2007 Imso ShortanswerDocument10 pages2007 Imso ShortanswerkalloliNo ratings yet
- DPP-2 Complex-Number Ques @motion LecturesDocument1 pageDPP-2 Complex-Number Ques @motion LectureskalloliNo ratings yet
- 12.3 Structural Representations of Organic Compounds 12.3.1 Complete, Condensed and Bond-Line Structural FormulasDocument3 pages12.3 Structural Representations of Organic Compounds 12.3.1 Complete, Condensed and Bond-Line Structural FormulaskalloliNo ratings yet
- Ashirvad uPVC Casing PipesDocument2 pagesAshirvad uPVC Casing PipeskalloliNo ratings yet
- Pages From David R. Klein - Organic Chemistry As A Second Language - First Semester Topics-Wiley (2017)Document1 pagePages From David R. Klein - Organic Chemistry As A Second Language - First Semester Topics-Wiley (2017)kalloliNo ratings yet
- Pages From TB - 9th EdDocument1 pagePages From TB - 9th EdkalloliNo ratings yet
- DPP-4 Complex-Number Ques @motion LecturesDocument1 pageDPP-4 Complex-Number Ques @motion LectureskalloliNo ratings yet
- DPP-1 Circle Ques @motion LecturesDocument1 pageDPP-1 Circle Ques @motion LectureskalloliNo ratings yet
- Rigidity of Double /triple Bonds: C H H H H H C H H H H C H H H CDocument7 pagesRigidity of Double /triple Bonds: C H H H H H C H H H H C H H H CkalloliNo ratings yet
- Func Tional Groups: Heteroatoms-Atoms Other Than Carbon or Hydrogen. Common Heteroatoms Are NitroDocument3 pagesFunc Tional Groups: Heteroatoms-Atoms Other Than Carbon or Hydrogen. Common Heteroatoms Are NitrokalloliNo ratings yet
- 3 Sigma and PaiDocument1 page3 Sigma and PaikalloliNo ratings yet
- Problem 4.11: Which Would You Expect To Be More Stable: (CH) C or (CF) C ? Why?Document2 pagesProblem 4.11: Which Would You Expect To Be More Stable: (CH) C or (CF) C ? Why?kalloliNo ratings yet
- The Bonds in Ethyne: SP P P P SP S P PDocument1 pageThe Bonds in Ethyne: SP P P P SP S P PkalloliNo ratings yet
- Hyperconjugation: The Reactions of Alkenes - The Stereochemistry of Addition ReactionsDocument2 pagesHyperconjugation: The Reactions of Alkenes - The Stereochemistry of Addition ReactionskalloliNo ratings yet
- The Bonds in Ethene: P P P S SP SP P SPDocument1 pageThe Bonds in Ethene: P P P S SP SP P SPkalloliNo ratings yet
- EnzymeKinetics PDFDocument21 pagesEnzymeKinetics PDFaathiraNo ratings yet
- Slide Biop Enz KinDocument19 pagesSlide Biop Enz KinFahmi Ramadhan PutraNo ratings yet
- Assignment 2 Reactor Design Single ReactionDocument4 pagesAssignment 2 Reactor Design Single ReactionNitin MauryaNo ratings yet
- Kinetics Revision Worksheet 2 (Solutions)Document8 pagesKinetics Revision Worksheet 2 (Solutions)Lee Jun HuiNo ratings yet
- CHE244 Lab Report Effect of Residence TiDocument26 pagesCHE244 Lab Report Effect of Residence Tiwan nur mursyidahNo ratings yet
- Act# 10 WORKSHEET On STOICHIOMETRY (LAB)Document3 pagesAct# 10 WORKSHEET On STOICHIOMETRY (LAB)henz assasinNo ratings yet
- A Substance Produced by A Living Organism Which Acts As A Catalyst To Bring About A Specific Biochemical ReactionDocument14 pagesA Substance Produced by A Living Organism Which Acts As A Catalyst To Bring About A Specific Biochemical ReactionPrincely ImmanuelNo ratings yet
- 1.2. Boxes and Arrows To Differential EquationsDocument14 pages1.2. Boxes and Arrows To Differential EquationsOmer GaddouraNo ratings yet
- MIT Lecture 6 5.07 Biochemistry LectureDocument16 pagesMIT Lecture 6 5.07 Biochemistry LectureakiridoNo ratings yet
- Ex0 Questions SolutionsDocument7 pagesEx0 Questions SolutionsBiniyam haileNo ratings yet
- Types: Chemical ReactorDocument3 pagesTypes: Chemical ReactorVeni DwiNo ratings yet
- Bab 2 - Kinetika Reaksi HomogenDocument12 pagesBab 2 - Kinetika Reaksi HomogenDiah Ayu TriatNo ratings yet
- EnzymeDocument15 pagesEnzymeShreyashNo ratings yet
- Michaelis Menten Equation SummaryDocument7 pagesMichaelis Menten Equation SummaryBüşra GültekinNo ratings yet
- Enzymes PresDocument34 pagesEnzymes Presm. uma deviNo ratings yet
- Chemical Kinetics - CBSE Sol File Ex-1Document5 pagesChemical Kinetics - CBSE Sol File Ex-1PriyanshiNo ratings yet
- KINETICSDocument47 pagesKINETICSMarilia BonorinoNo ratings yet
- Schnakenberg 1979Document12 pagesSchnakenberg 1979andres felipe ortizNo ratings yet
- Emailing Chemical Kinetics (Class 12)Document12 pagesEmailing Chemical Kinetics (Class 12)roceniNo ratings yet
- Factors Affecting Enzyme ActivityDocument7 pagesFactors Affecting Enzyme ActivityDahliza KamatNo ratings yet
- Exercises KineticsDocument7 pagesExercises KineticsFahad AlasmiNo ratings yet
- Week 6 - Physical-ScienceDocument2 pagesWeek 6 - Physical-ScienceKayla TiquisNo ratings yet
- Homework Solutions/Kinetics 1Document11 pagesHomework Solutions/Kinetics 1Eduardo Rosado HerreraNo ratings yet
- Prof. K.K.Pant Department of Chemical Engineering IIT DelhiDocument45 pagesProf. K.K.Pant Department of Chemical Engineering IIT DelhiApaar MudgalNo ratings yet
- Elements of Chemical Reaction Engineering Prentice Hall International Series in The Physical and Chemical Engineering Sciences Ebook PDF VersionDocument62 pagesElements of Chemical Reaction Engineering Prentice Hall International Series in The Physical and Chemical Engineering Sciences Ebook PDF Versionadolfo.milliken178100% (57)